Abstract
Introduction
Tigecycline is the first member of a new class of antibiotics, the glycylcyclins. 1 It is derived from minocycline by adding a glycyclamide moiety at position 9 of the D ring. This modification extended tigecycline antibacterial activity by conquering the acquired efflux pumps and ribosomal protection, the main mechanisms of tetracycline resistance. 1
Tigecycline is currently approved by the US Food and Drug Administration (FDA) for treating complicated intra-abdominal infections, complicated skin and skin structure infections, and community-acquired pneumonia.2,3 This approval was based on large randomized controlled trials (RCTs) that showed tigecycline efficacy compared to standard antimicrobials.4 -6 In these RCTs, nausea and vomiting were the most frequent adverse events, leading to tigecycline discontinuation in 5% of patients. 2 Less frequently reported adverse events were photosensitivity, skin rash, and pancreatitis, likely inherited by tigecycline from tetracyclines due to structural similarity.
Hypoglycemia is an uncommon yet severe adverse effect of tigecycline therapy. 3 In hospitals, due to various causes, hypoglycemia is associated with unfavorable outcomes, including prolonged hospitalization and increased mortality.7,8 Therefore, healthcare facilities continuously implement and update protocols to prevent hypoglycemia and identify its risk factors. 9 Like other antibiotics, the expansion of tigecycline use to treat infections caused by multi-drug resistant (MDR) bacteria has increased the rate of adverse events, including hypoglycemia.10,11 This review discusses the characteristics, risk factors, and possible mechanisms of hypoglycemia that develop during tigecycline therapy.
Methods
Search Strategy
A search of the literature was conducted in 3 independent phases. In the first phase, in collaboration with a medical librarian, the OVID interface was used to search the MEDLINE, Embase, and Cochrane Central Register of Controlled Trials (CENTRAL) electronic database from inception until August 1, 2023. To identify relevant citations, a sensitive search strategy comprised the following search terms (using combinations of subject headings [ie, MeSH in MEDLINE] and keywords): “tigecycline,” “tygacil,” “hypoglycemia,” “hypoglycaemia,” “hypoglycemic coma,” “hypoglycaemic coma,” “decreased blood glucose,” and “low blood glucose level.” The search strategy was modified for each database using appropriate thesaurus terms and fields. In the second phase, 2 investigators searched the US FDA Adverse Event Reporting System (FAERS) Public Dashboard on July 1, 2023, for reports of tigecycline and hypoglycemia from June 1, 2005, until July 1, 2023. The FAERS data contain drug information, including name, active ingredient, route of administration, and the drug’s role in the adverse event. Each report has a primary suspected drug with one or more adverse reactions and may include other drugs used by the patient. In the third phase of the search, 2 investigators searched MEDLINE for RCTs with the search term “tigecycline.” Then they manually screened the results for the following terms: “hypoglycemia,” “hypoglycaemia,” and “low blood glucose.”
Inclusion and Exclusion Criteria
Case reports and studies conducted in humans and published in English were included. The exclusion criteria included pediatrics if reported (age <18 years). To avoid duplications, pooled and pharmacokinetic data analyses from tigecycline phase II, III, IV studies were also excluded.
Hypoglycemia is defined as a blood glucose level < 3.9 mmol/L (<70 mg/dL) in patients with diabetes mellitus (DM) 12 or < 3.0 mmol/L ( <55 mg/dL) in patients without DM. 13 Severe hypoglycemia is defined as hypoglycemia with symptoms, according to recent guidelines from the American Diabetes Association. 12 Citations reporting hypoglycemia inconsistent with these definitions were excluded from further consideration in the present review.
Results
The literature search initially identified 17 unique citations, including 9 case reports, 7 retrospective studies, and 1 systematic review of retrospective studies.
Case Reports
Three of the 9 identified case reports were excluded from further consideration, 2 for nonrelevance and one as a duplicate publication.14 -19 Two of the 6 included case reports were published in full manuscripts, and the other 4 were published as conference abstracts. All patients described in the 6 case reports developed hypoglycemia while receiving the FDA-approved dose of tigecycline (100 mg loading followed by 50 mg every 12 hours). 3 A summary of the case reports is presented in Table 1. None of the patients described in the case reports received tigecycline in combination with fluoroquinolone, a class of antibiotics known for their potential to induce dysglycemia, including hypoglycemia. 20
Patient Characteristics and Features of Hypoglycemia During Tigecycline Therapy.
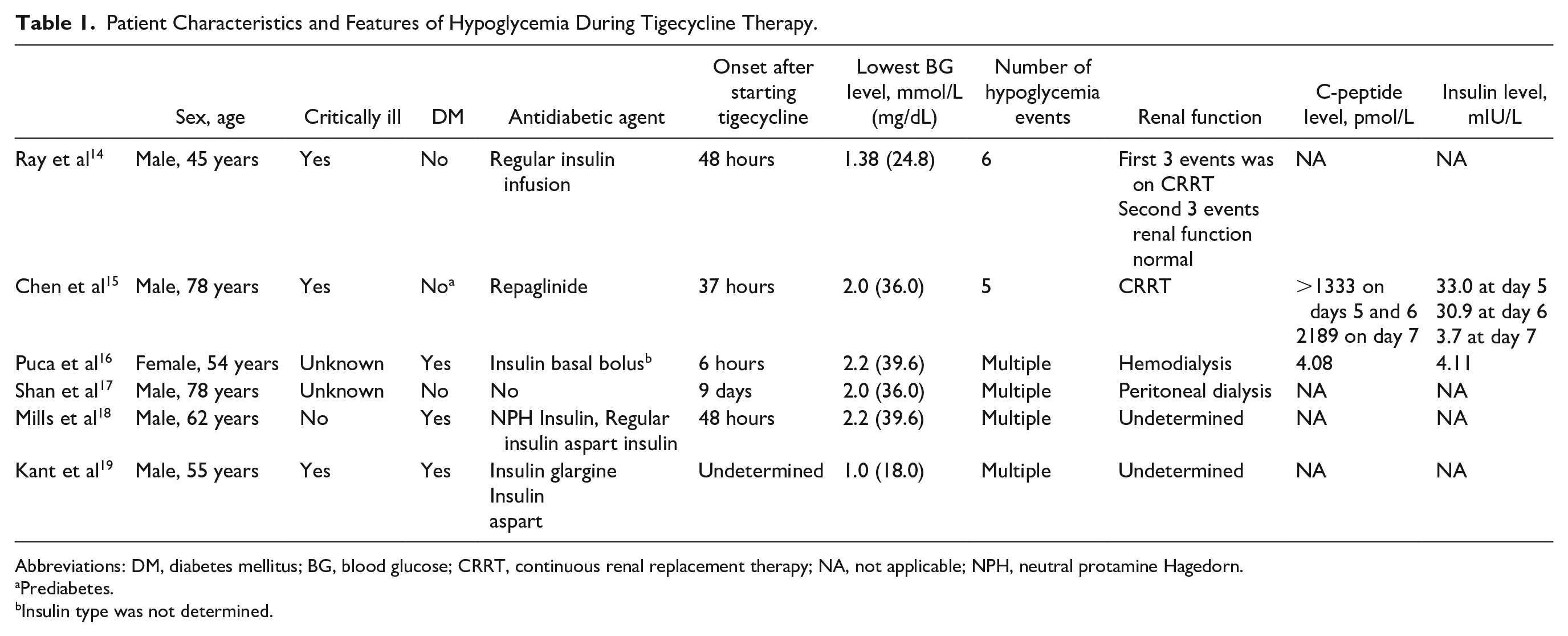
Abbreviations: DM, diabetes mellitus; BG, blood glucose; CRRT, continuous renal replacement therapy; NA, not applicable; NPH, neutral protamine Hagedorn.
Prediabetes.
Insulin type was not determined.
Case 1
A 45-year-old man was admitted to an intensive care unit for cellulitis and sepsis secondary to a snake bite. 14 After admission to the intensive care unit, the patient developed an acute kidney injury requiring continuous renal replacement therapy (CRRT) and experienced multiple hypoglycemia events during 2 courses of tigecycline therapy. Hypoglycemia started to develop on day 2 of the first 7-day course of tigecycline, wherein the patient experienced 3 hypoglycemia events. During the second course of tigecycline, the patient developed successive severe hypoglycemic events (blood glucose level: 1.3 mmol/L [23.4 mg/dL]), and the tigecycline therapy was discontinued. Blood glucose levels remained low for approximately 40 hours after the final discontinuation of tigecycline therapy.
Case 2
A 78-year-old man with prediabetes was admitted to the hospital with pneumonia. 15 During hospitalizations, the patient developed an acute kidney injury that required CRRT. The first hypoglycemia event (blood glucose level: 2.8 mmol/L) occurred 4 days after the start of tigecycline therapy and was resolved by oral glucose administration. On day 6 of tigecycline therapy, the patient suffered severe and sustained hypoglycemia (blood glucose level: 2.0 mmol/L [36.0 mg/dL]). As a result of this event, the patient was switched from tigecycline to imipenem-cilastatin and polymyxin B and administered intravenous 50% dextrose by infusion for 48 hours. Notably, the patient received repaglinide for 5 days concomitantly with tigecycline, which was discontinued 1 day before the cessation of tigecycline therapy. The measured c-peptide and insulin levels were elevated on days 5 and 6 of tigecycline therapy. One day after stopping tigecycline (2 days after the last dose of repaglinide), the insulin level was within the normal range, while the c-peptide level continued to be elevated. The patient died after 3 days of cessation of tigecycline therapy.
Case 3
A 54-year-old woman with Type 2 DM and end-stage kidney disease on hemodialysis was admitted to the hospital for Staphylococcus epidermidis bacteremia. 16 The patient experienced a rapid onset of severe hypoglycemia 6 hours after the first dose of tigecycline, followed by multiple hypoglycemia events (blood glucose levels 2.2-2.6 mmol/L [39.6-46.8 mg/dL]) over a period of 16 hours. No information was provided regarding concomitant insulin administration or other antidiabetic agents before hypoglycemia events. The measured c-peptide and insulin levels were within the normal range during the hypoglycemia events.
Case 4
A 78-year-old man on peritoneal dialysis was admitted to the hospital with diffuse peritonitis. 17 After 9 days of tigecycline therapy, the patient developed symptomatic hypoglycemia (blood glucose level: 2.0 mmol/L [36.0 mg/dL]). Tigecycline was immediately stopped, and a bolus of 50% dextrose was administrated to restore euglycemia. Blood glucose level remained low after 3 days of tigecycline therapy cessation, which required the administration of intravenous dextrose infusion in addition to oral glucose for 10 days after the hypoglycemia event.
Case 5
A 62-year-old man with type 1 DM developed a nonhealing foot wound requiring a prolonged antibiotic course. 18 Initially, the patient received a combination of vancomycin and ertapenem, which was stopped due to skin rash after 30 days. The regimen was changed to tigecycline to complete therapy for 20 more days. Within 48 hours of starting tigecycline, the patient developed repeated episodes of hypoglycemia (blood glucose: 2.2-3.3 mmol/L [39.6-59.4 mg/dL]), measured by home glucometer, with accompanying hypoglycemia symptoms. The patient’s pre-antibiotic insulin regimen (stable in dose for >1 year) consisted of 20 units of neutral protamine Hagedorn (NPH) insulin with breakfast and another 10 units at bedtime, 10 units of regular insulin before breakfast and 5 units before dinner, and 5 units of insulin aspart with lunch (0.67 units/kg). By the end of the tigecycline treatment course, the insulin regimen was reduced to 20 units of NPH insulin before breakfast, another 4 units at bedtime, and 5 units of insulin aspart before dinner (0.37 units/kg). The patient denied increased activity levels, anorexia, nausea, and emesis. His post-antibiotic total daily insulin dose was increased back to 40 units (0.54 units/kg). Hypoglycemia resolved with the discontinuation of tigecycline therapy.
Case 6
A 55-year-old man with a poorly controlled type 1 DM, with no prior history of hypoglycemia, was started on tigecycline for the treatment of osteomylitis. 19 After 3 weeks of tiegcycline theraopy, the patients was admitted to an intensive care unit for loss of consciousness due to severe hypoglycemia. At the time of admission, the patient reported experiencing several symptomatic episodes of hypoglycemia at home since the start of tigecycline therapy, which required 2 visits to the emergency department (blood glucose level: 1.1-1.5 mmol/L [19.8-27.0 mg/dL]). The patient confirmed there was no change in diet or physical activity during the course of tigecycline therapy. On admission to the hospital, the patient had a blood glucose level of 1.0 mmol/L and was treated successfully with intravenous dextrose. During hospitalization, glycemic control was achieved with reduced insulin requirements of 8 units of insulin glargine every morning and 1-2 units of premeal insulin aspart. After completing a 6-week course of tigecycline, his blood glucose levels gradually increased, and insulin requirements increased to 38 total units daily.
Retrospective Studies
Four retrospective studies and a systematic review of retrospective studies were initially identified in the literature search. Three of the retrospective studies and the systematic review were subsequently excluded for not meeting the hypoglycemia definition and nonrelevance.
A retrospective study published as a conference abstract described 10 patients with cystic fibrosis who received various antimicrobials, including tigecycline, moxifloxacin, linezolid, and amikacin, for the treatment of nontuberculous mycobacteria lung infection. 21 The age of the patients was undetermined. Three (30%) patients developed hypoglycemia during 1-2 weeks of treatment with tigecycline. The measured blood glucose levels during the hypoglycemia events ranged from 1.6 mmol/L (28.8 mg/dL) to 2.7 mmol/L (48.6 mg/dL). None of the 3 patients had DM or pancreatitis. Upon cessation of tigecycline therapy, the hypoglycemia resolved without recurrence.
Epidemiology Studies
Three epidemiological studies were initially identified in the literature search. One study that assessed FAERS data in 2012 was excluded because a more recent study evaluated the same data domain in 2019 and was included. 22 The latter study appeared in 2 citations: a conference abstract, which was excluded, and a full manuscript, which was included.
An epidemiologic surveillance study extracted data from the FAERS and assessed the association of various antimicrobials with hypoglycemia from January 2004 until December 2017. 22 The reporting odds ratio (ROR) was calculated as the ratio of the odds of reporting hypoglycemia versus all other adverse reactions for a given drug compared with the odds of reporting hypoglycemia versus all other adverse reactions for all other drugs present in the FAERS. Tigecycline was associated with hypoglycemia in 14 patients, resulting in a ROR of 3.32 (95% confidence interval [CI]: 1.95-5.65). After adjusting for sulfonylurea and meglitinide use, tigecycline continued to be associated with hypoglycemia (ROR: 3.34, CI: 1.96-5.68). No information was provided regarding patient’s demographics and hypoglycemia characteristics in this study.
FDA Adverse Event Reporting System (FAERS)
Forty-six cases of hypoglycemia related to tigecycline use were identified in the FAERS. Three of these cases were excluded because the patients were younger than 18 years of age. The remaining 43 patients included 25 men, 14 women, and 3 patients of undetermined sex. The calculated median age of the included patients was 65 (interquartile range [IQR]: 55-83). A concomitant antidiabetic agent was administered with tigecycline to 9 patients (21.4%), 5 of whom received insulin (11.9%) and 4 of whom received an oral antidiabetic agent (9.5%; glimepiride and repaglinide each in 2 patients). There was no reported concomitant administration of quinolone antibiotics with tigecycline in any of the patients. All the reported events were described as serious, except one report, and 4 (9.5%) patients died.
Randomized Clinical Trials
The search of MEDLINE for RCTs assessing tigecycline initially yielded 42 studies. Of these, 5 were excluded for nonrandomized designs, and 12 were excluded for pooled analyses of RCTs. Two of the remaining RCTs reported hypoglycemia.23,24 These studies provided no details regarding the severity of the hypoglycemia events or blood glucose levels.
A phase III trial compared tigecycline versus ertapenem for treating diabetic foot infections with or without osteomyelitis. 23 In this double-blind study, patients with diabetic foot without osteomyelitis (primary study, n = 944) and with osteomyelitis (substudy, n = 117) were randomized to receive 150 mg daily of tigecycline or ertapenem 1 g daily ± vancomycin. Hypoglycemia occurred in 34 (7.1%) patients in the tigecycline group and 24 (5.1%) in the ertapenem group in the primary study. In the substudy, 16 (21.1%) of the patients who received tigecycline experienced hypoglycemia, whereas none in the comparator group experienced hypoglycemia (P < 0.001).
The other RCT was a phase II, double-blind trial investigating the efficacy and safety of 2 doses of delafloxacin versus tigecycline in patients with skin and skin structure infections. 24 Fifty patients received tigecycline, and 100 patients received either 300 mg or 450 mg delafloxacin. The median duration of tigecycline therapy was 6.8 days. Blood glucose levels below the normal range were observed in 9 (17.6%), 2 (4.08%), and 1 (2%) of the patients who received 450 mg delafloxacin, 300 mg delafloxacin, and tigecycline, respectively. One patient (1.9%) who received 450 mg of delafloxacin developed hypoglycemia, whereas none who received tigecycline developed hypoglycemia.
Discussion
Hypoglycemia is an uncommon adverse event of tigecycline therapy, developing at a rate of <2%. 3 To our knowledge, this review is the first to discuss and summarize the available evidence regarding this metabolic complication that may tax tigecycline therapy.
There is no specific onset time for hypoglycemia occurrence during tigecycline therapy. According to the available reports, it may develop within 6 hours after the initial dose of tigecycline or any time during extended courses of weeks of therapy. Therefore, clinicians need to be vigilant for the possibility of hypoglycemia during the entire course of tigecycline therapy. In the reports identified in our literature search, patients were symptomatic during hypoglycemia events, and the measured blood glucose levels were <3.0 mmol/L, consistent with the definitions of severe hypoglycemia in patients with or without DM.12,13 On the contrary, mild hypoglycemia may have been developing during tigecycline therapy; however, it could be overlooked due to underreporting or difficulty in recognizing. 25 Studies are needed to assess the impact of tigecycline on glucose homeostasis precisely.
Persistence is another property of tigecycline-related hypoglycemia, where the hypoglycemia is sustained for days after tigecycline cessation. Tigecycline has a relatively long half-life, which would explain the persistence of low blood glucose levels after therapy discontinuation. After administering multiple doses of tigecycline and achieving a steady state, the reported elimination half-life exceeds 42 hours. 3 In many instances, patients with long-lasting hypoglycemia after tigecycline cessation require extended intravenous infusion of dextrose solutions. In some cases, to achieve euglycemia after the failure of 5% dextrose solution to restore euglycemia, more invasive interventions might be necessary, such as insertion of a central catheter to intravenously infuse hypertonic 50% dextrose solution. 15
Four of the 6 reported cases in this review described patients with severe renal dysfunction and required a form of dialysis. Tigecycline is predominantly excreted in bile as an unchanged drug and conjugated metabolites. 3 Less than 33% of tigecycline undergoes renal excretion, and there is no recommendation in the FDA prescribing information for dose adjustment in patients with various stages of renal dysfunction. Recent data demonstrated that the area under the concentration-time curve for tigecycline is increased by more than 30% in patients with severe renal dysfunction and that removal by dialysis is inefficient. 26 Furthermore, a pharmacokinetic study of patients with septic shock showed that the area under the concentration-time curve for tigecycline, calculated based on trough levels, was higher in patients on CRRT than in other patients. 27 Several studies have demonstrated renal dysfunction as a risk factor for various adverse tigecycline-related events, such as hypofibrinogenemia and pancreatitis.28,29 Possibly, renal dysfunction also predisposes patients to hypoglycemia during tigecycline therapy by causing drug overexposure.
It is difficult to use c-peptide levels to derive conclusions regarding changes in endogenous insulin secretion during tigecycline therapy, according to the available literature. In this review, c-peptide levels during hypoglycemia were tested in 2 case reports and were within the normal range in one patient and elevated in the other.15,16 It is worth mentioning that the patient with elevated c-peptide level was receiving repaglinide (half-life of 1-2 hours), which was stopped 24 hours before the onset of hypoglycemia.
In 2 case reports, patients with type 1 DM continued to receive tigecycline despite the continuous occurrence of symptomatic hypoglycemia events, which required admission to the intensive care unit in one case. Due to its uncommon occurrence, healthcare providers may be unaware of hypoglycemia occurrence with tigecycline use. Therefore, making prescribers especially vigilant for this potentially serious adverse event in patients with DM is crucial.
In the RCT of patients with diabetic foot, hypoglycemia was more frequent in patients on tigecycline than in patients on ertapenem, which is consistent with tigecycline having an impact on blood glucose levels. 23 In this trial, all the enrolled patients were known for DM and presumably received a blood glucose-lowering agent to control blood glucose levels during the study. However, there are no descriptions of medications and associated doses or data on blood glucose levels. In the other RCT, one hypoglycemia event was described with delafloxacin, whereas none were described with tigecycline. Generally, phase II RCTs have limited validity for assessing uncommon and severe adverse events because of their relatively small sample size. In addition, fluoroquinolone antimicrobials are well-known for their potential to induce hypoglycemia, particularly in patients with severe infections failing to maintain physiological response to regulate blood glucose level.20,22
The mechanism of tigecycline-induced hypoglycemia is unknown. However, many mechanisms have been proposed, including stimulation of insulin release and/or increased liver sensitivity to insulin. 14
Clinically, tigecycline lacks significant drug interactions and is not known to potentiate the blood glucose-lowering activities of insulin or antidiabetic agents. An epidemiology study showed an association between tigecycline and hypoglycemia that was independent of the use of oral antidiabetic agents. 22 In our assessment of the US FDA FEARS data on hypoglycemia in patients undergoing tigecycline therapy, less than 12% of the patients received a concomitant insulin therapy. However, patients receiving insulin to control blood glucose may need more than a 50% reduction in total daily insulin dose when developing the first episode of hypoglycemia while receiving tigecycline therapy.
None of the 3 patients with cystic fibrosis described in a retrospective study received tigecycline had DM, indicating an antidiabetic agent. 21 However, tigecycline was combined with other antimicrobials, including fluoroquinolone, which could induce hypoglycemia. 30 This study provided no details regarding blood glucose control, as cystic fibrosis patients frequently develop hyperglycemia.
There are several limitations in the description of tigecycline-related hypoglycemia in this review. It is not clear whether the hypoglycemia events in this review were iatrogenic (ie, caused by tigecycline) or spontaneous (ie, a marker of disease severity and poor prognosis). 31 Many patients in the case reports and retrospective studies were severely ill and were admitted to an intensive care unit, where hypoglycemia is a common marker of severe illness. Guidelines recommend tigecycline to treat infections caused by MDR bacteria, nonsusceptible to other antimicrobials. 32 Patients with MDR bacterial infections are often severely ill and, therefore, at risk for hypoglycemia. 33 Studies are needed to primarily assess the risks of developing hypoglycemia during tigecycline therapy. We could not perform a statistical analysis to evaluate the risks of hypoglycemia in patients undergoing tigecycline therapy because of the limited quantitative data available in the literature. Furthermore, many of the reports in this review were not available in full text, which hindered us from looking more deeply into the case data.
In conclusion, hypoglycemia is an uncommon adverse event associated with tigecycline therapy and may develop at any time during treatment. Hypoglycemia can be severe and persists for more than 24 hours after tigecycline discontinuation. Renal dysfunction or hemodialysis may predispose to severe hypoglycemia during tigecycline therapy. Further research is needed to investigate risk factors and potential mechanisms of hypoglycemia development during tigecycline therapy.
Footnotes
Author Contributions
Hakeam A. Hakeam: Conceptualization of the review, methodology, formal analysis, investigation, data curation, writing original draft.
Khadija A. Sarkhi: Conceptualization of the review, acquisition, analysis, and interpretation of data, and editing the manuscript.
Alla Iansavichene: Data interpration and analysis, review, and critically editing the manucsript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
