Abstract
The renin-angiotensin system can be inhibited through inhibition of angiotensin I generation from angiotensinogen by direct renin inhibitors, inhibition of angiotensin II generation from angiotensin I by angiotensin-converting enzyme inhibitors and by direct inhibition of the action of angiotensin II receptor level. Aliskiren, the first direct renin inhibitor to reach the market, is a low molecular weight, orally active, hydrophilic nonpeptide. It blocks angiotensin I generation, while plasma renin concentration increases because the drugs blocks the negative feed-back exerted by angiotensin II on renin synthesis. Aliskiren is suitable for once-daily administration because of its long pharmacological half-life. Because of its mechanism of action, aliskiren may provide the additional opportunity to inhibit progression of atherosclerosis at tissue level. Hypertension is an approved indication for aliskiren, which is also promising for the treatment of heart failure and diabetic nephropathy. The efficacy of this drug on major clinical events is being tested in large ongoing clinical trials.
Keywords
In the present issue of the Journal, Imanishi and coworkers 1 provide an excellent status-of-the-art review on aliskiren, a novel orally active direct renin inhibitor, the first to successfully reach the market. Although the advent of a new class of drugs is always an interesting scientific occurrence, from a clinical standpoint one may wonder whether there is a place for a drug like aliskiren in the already prosperous cardiovascular armamentarium.
Pharmacological Novelty
The pharmacological properties of aliskiren are intriguing. Aliskiren works by linking to active renin on a deep cleft of its molecular structure. This cleft is the site of hydrolysis of the Leu10-Val11 bond of angiotensinogen, which leads to the generation of the decapeptide fragment angiotensin I, the initial and rate-limiting step of the renin-antensin-aldosterone system. 2 Therefore, treatment with aliskiren is associated with a reduced, not increased, generation of angiotensin I (‘plasma renin activity’). This is a unique feature (Table 1) that differentiates aliskiren not only from traditional antihypertensive drugs which activate the renin angiotensin system (such as diuretics and calcium channel blockers), but also from angiotensin converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARBs). 3 There is evidence that increased levels of plasma renin activity are associated with a higher risk of cardiovascular complications.4,5 Another promising feature of aliskiren is the potential for inhibiting renin and activated prorenin when they are bound to the (pro)renin receptors.2,3 This might offer the additional opportunity to inhibit progression of atherosclerosis at tissue level. Of course, these effects require extensive experimental and clinical testing.
Effects of different classes of antihypertensive drug on plasma renin activity and concentration.
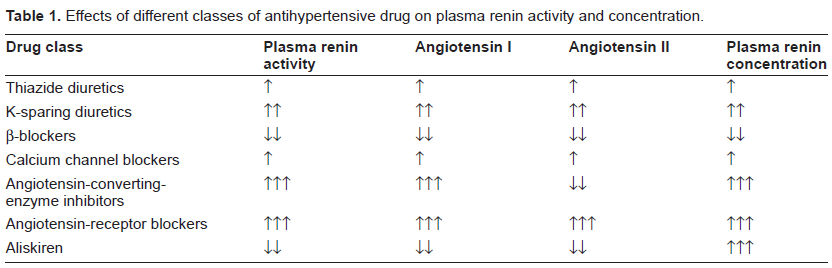
On the other hand, plasma renin concentration increases with aliskiren because the drug blocks the negative feedback exerted by angiotensin II on renin synthesis. Because such an increase has been reported to be up to 34-fold (i.e. superior than that induced by ACE inhibitors or ARBs), 3 it will be important to investigate whether these high levels of renin may exert biological effects through stimulation of (pro)renin receptors. It will be also important to clarify whether the levels of angiotensin I and angiotensin II will remain suppressed during chronic therapy, in the absence of biologically relevant ACE-escape like phenomena.
Clinical Effects
The clinical properties of aliskiren are being clarified in the context of an impressive program (ASPIRE HIGHER) 6 of ongoing and recently concluded clinical studies. The antihypertensive effects of aliskiren monotherapy are comparable to those of ACE inhibitors, ARBs, and other antihypertensive agents at equipotent doses. However, its antihypertensive potency is increased by combination with drugs that elicit a reactive increase in the plasma renin activity such as diuretics, ACE inhibitors and angiotensin receptor blockers.7–13 Aliskiren is particularly indicated in patients who are intolerant to ACE inhibitors, as well as in those with elevated activity of the renin angiotensin system, like the white subjects and young people.
The tolerability profile of aliskiren is placebolike at the licensed doses of 150 mg and 300 mg. 14 Diarrhea is more frequent than with placebo only at the dose of 600 mg. 15 The hepatic route of elimination makes it suitable in patients with renal failure. The lack of interference with several agents including warfarin and statins further enhance the tolerability profile le of the drug. Pregnancy and bilateral renal artery stenosis are among the few contraindications to the use of aliskiren.
On the basis of the above considerations, hypertension, diabetic nephropathy and heart failure are clinical conditions for which aliskiren appears particularly attractive. In the Aliskiren in the Evaluation of Proteinuria in Diabetes (AVOID) Trial 16 reduced by 18% the urinary albumin creatinine ratio as compared with placebo, on top of standard treatment, in hypertensive patients with type 2 diabetes and macroalbuminuria. In the aliskiren observation of heart failure treatment (ALOFT) study, 17 aliskiren significantly reduced, compared to placebo and on top of standard treatment, the plasma NT-pro BNP, BNP and urinary aldosterone. In the Aliskiren in Left Ventricular Hypertrophy (ALLAY) study, 18 aliskiren was as effective as losartan in inducing left ventricular hypertrophy regression in hypertensive patients.
Ongoing Studies
Several important studies with aliskiren are ongoing. For example, the ALTITUDE (ALiskiren Trial In Type 2 diabetes Using cardio-renal Disease Endpoint), study is a placebo-controlled, randomized, double-blind study in 8600 patients with type 2 diabetes associated with albuminuria or a history of cardiovascular disease or reduced renal function. The study, which is expected to finish in 2012, will be concluded when the primary end-point (composite of cardiovascular and renal events) will occur in 1628 patients. The ATMOSPHERE (Aliskiren Trial to Mediate Outcome Prevention in Heart FailuRE) will be conducted in heart failure patients not dissimilar from those investigated in ALOFT and the primary end-point will be a composite of cardiovascular death and re-hospitalization for heart failure. The AVANT-GARDE (Aliskiren and VAlsartan to Reduce NT-proBNP via Renin-AnGiotensin-AldosteRone System BlockaDE) study is conducted in 1100 patients who are stabilised after an acute coronary syndrome and who present a normal left ventricular systolic function. The primary end-point in AVANTGARDE is the change in NT-proBNP levels. The placebo-controlled APOLLO (Aliskiren in Prevention Of Later Life Outcomes) trial will be conducted in elderly subjects with normal BP, no overt cardiovascular disease, and a high cardiovascular risk profile. The AQUARIUS (Aliskiren QUantitative Atherosclerosis Regression Intravascular Ultrasound Study) study, still in a planning stage, will address the change in atherosclerotic burden under treatment with aliskiren compared with placebo in patients with coronary artery disease. 6
In the setting of the ASPIRE HIGHER program, aliskiren is also investigated in head-to-head comparisons with different classes of BP-lowering drugs. For example, the TARGET HIGHER study will investigate the association of aliskiren with valsartan in hypertensive patients with diabetes and/ or microalbuminuria. The ACCELERATE (Aliskiren and the Calcium ChannEL BlockER Amlodipine Combination as Initial TreatmEnt Strategy in Stage I and II Hypertension) study will test the hypothesis that BP levels are lower over 32 weeks of treatment in patients receiving the combination of aliskiren with amlodipine simultaneously from the beginning of the study when compared with patients who receive this drugs sequentially. 6
Conclusions
Apart from the remarkable novelty associated with its mechanism of action, good news from aliskiren comes from the encouraging results of available studies in patients with hypertension, diabetes and congestive heart failure. In these patients, aliskiren was extremely well tolerated and effective in lowering blood pressure and improving intermediate measures of outcome such as proteinuria, left ventricular hypertrophy and biochemical markers of heart failure.6,15
Notably, the efficacy of aliskiren in the above clinical conditions was evident on top of standard therapy, thus remarking the concept that a complete blockade of the renin-angiotensin-system at its first rate-limiting step may be beneficial in specific categories of patients. The long duration of action, which makes this drug suitable for once-daily administration, and the hepatic site of metabolism, which makes the drug indicated also in patients with renal failure, are further good news from this promising drug.
Disclosures
The authors report no conflicts of interest.
Footnotes
Acknowledgment
This work has been supported in part by the no-profit Foundation Fondazione Umbra Cuore e Ipertensione-ONLUS, Perugia, Italy.
