Abstract
Background:
Hypertension (HTN) is one of the major risk factors for cardiovascular disease in Western countries, and disease control is of major relevance in order to reduce cardiovascular morbidity and mortality. Different approaches have shown efficacy, and one of the proven therapies for HTN control is the blockade of the renin–angiotensin–aldosterone system, which may be accomplished by means of various drugs with different modes of action. Aliskiren is a novel direct renin inhibitor that reduces both angiotensin I and II blood levels. Different randomized clinical trials (phase II and III) have shown its safety and efficacy either alone or in combination with hydrochlorothiazide. However, although aliskiren has been on the market for some years, reports on the post-marketing experience with aliskiren in the real-world setting are lacking.
Methods:
The Aliskiren in Latin America Study (ALAS) was designed with the aim of describing the effectiveness of aliskiren at reducing blood pressure (BP) values by prospectively assessing BP control in outpatient clinics in different countries in Latin America. A total of 435 sites in 5 Latin American countries (Mexico, Ecuador, Colombia, Argentina, and Venezuela) enrolled 4588 patients who had just been initiated on aliskiren (either alone or in combination with hydrochlorothiazide) based on their treating physicians’ discretion, and they were followed for a 6-month period. Prior antihypertensive drugs could be continued if their doses were not modified along the study.
Results:
At the end of the follow-up period, a statistically significant reduction in BP values was observed, with a mean systolic BP reduction of 29.2 mmHg and a mean diastolic BP reduction of 13.78 mmHg from baseline at the 6-month visit.
Conclusion:
The BP reduction levels and the low adverse event rate demonstrate the adequate efficacy and safety profile of aliskiren (alone or with hydrochlorothiazide).
Introduction
Hypertension (HTN) is one of the major risk factors for cardiovascular disease in Western countries. According to the National Health and Nutrition Examination Survey, the prevalence of HTN in the United States is approximately 30% of the adult population and it revealed a rising trend [Fields et al. 2004]. Several reports from Latin America showed an adjusted HTN prevalence rate ranging from 26% to 42% [Latin American Expert Group, 2009] and it is estimated that HTN accounts for 13% of deaths and 5.1% of the quality-adjusted life years (QALYs) lost in this region [Lawes et al. 2008]. Since the uncontrolled HTN rate in the region is 12–41% [Hernández-Hernández et al. 2010], there is a clear need to enhance pharmacological and nonpharmacological strategies in order to improve blood pressure (BP) control. The availability of new drugs to treat HTN is one of the options to manage this situation. Aliskiren, as the only oral direct renin inhibitor on the market since 2007, has demonstrated to be a safe and effective drug administered alone or in combination with hydrochlorothiazide [Fogari and Zoppi, 2010; Morgado et al. 2011]. However, these safety and efficacy data were derived from randomized controlled trials. Information about how aliskiren works in a real-world setting, including in Latin America, is lacking. Therefore, the main goal of this study was to assess the safety and efficacy of aliskiren (alone or in combination with hydrochlorothiazide) in a real-world sample of hypertensive patients in Latin American countries.
Methods
The Aliskiren in Latin America Study (ALAS) is a prospective, observational, 6-month follow-up cohort study in hypertensive patients treated with aliskiren.
Patients were selected from nonreferral outpatient clinics in Mexico, Ecuador, Argentina, Colombia, and Venezuela. Study patients were eligible if they were 21 years of age or older, but less than 75, and if they had been previously diagnosed with JNC-7 stage 1 or 2 HTN (according to the Seventh Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure [National Heart, Lung, and Blood Institute Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure and National High Blood Pressure Education Program Coordinating Committee, 2003] and the Latin American Guidelines on Hypertension [Latin American Expert Group, 2009]). Patients were required to have been initiated on aliskiren at their physicians’ discretion within 1 week prior to enrollment, due to uncontrolled BP (values above 140/90 mmHg) with any prior nonaliskiren therapy. Owing to the observational nature of the study, all of the patients were required to be on aliskiren before study entry, and the decision to use aliskiren was made by the treating physicians based on their regular practice. Since this study assessed the effect of aliskiren effect on BP in a real-world setting, there was no washout period. In order to have reliable information on a real-world sample of patients treated with an aliskiren-based therapy, there were no restrictions regarding the use of concomitant drugs (cardiovascular or noncardiovascular), except that patients should remain on a stable dose for the duration of the study. After enrollment, patients were followed for 6 months. Patients were not considered eligible for the study if they had proven secondary HTN; were pregnant or breast-feeding; had any serious medical condition that, according to their treating physician’s judgment, could preclude their participation in the study; or if they were unable to follow the study doctor’s directives.
The protocol followed the Helsinki Declaration and the local regulations in each country and received approval from the respective Independent Ethics Committees. Patient’s data were collected in Web-based electronic case report forms provided by the sponsor. Monitoring of all clinical data and quality control (QC) reports was performed by a third-party vendor (Research & Data Processing Co.) and the study sponsor.
After signing the informed consent form, patients’ information regarding demographics, medical history, and cardiovascular (including aliskiren) and noncardiovascular medication were recorded. Data were provided by the patient or obtained from patients’ clinical records (i.e. cardiovascular disease history, such as myocardial infarction or stroke). HTN severity and target organ damage were defined according to the JNC-7 criteria. A physical examination including resting BP was performed at baseline (visit 1) and data on BP values were recorded. BP was measured twice, about 2 minutes apart, in the right arm after a 5-minute resting period, following a standardized procedure. All personnel were trained on this procedure. All patients were followed for 6 months. Safety information was recorded at every visit. All adverse events (AEs) were recorded on the appropriate forms and the terminology used was based on the Medical Dictionary for Regulatory Activities (MedDRA), Spanish version [MedDRA MSSO, 2012].
Patients were free to withdraw from the study at any time and for any reason. Also, they could be withdrawn by the study doctor if they experienced any adverse reaction that the investigator considered possibly related to aliskiren, if doses of antihypertensive drugs had to be modified, or if the use of any additional antihypertensive drug [calcium channel blocker (CCB), beta blocker, angiotensin-converting enzyme inhibitor (ACEI), diuretics other than hydrochlorothiazide] was required due to uncontrolled HTN.
The primary objectives of the study were to compare the BP levels measured at baseline (day 1) with those measured at day 180 in patients with JNC-7 stage 1 and 2 HTN, and to assess the incidence of AEs in aliskiren-treated patients. The secondary objectives were to describe the clinical characteristics of the aliskiren-treated population, and to assess the proportion of patients that reach JNC-7 predefined BP targets according to HTN severity.
All analyses were intent-to-treat. Categorical variables were reported as proportions. After testing for normality, continuous variables were reported as mean [± standard deviation (SD)] or median (interquartile range). Differences between stage 1 HTN and stage 2 HTN groups were tested using chi-square, Fisher exact, Student or Wilcoxon tests.
Results
A total of 4588 patients were included in the study from 435 sites (Mexico 274, Ecuador 70, Colombia 70, Argentina 23, and Venezuela 23 sites). The mean time from HTN diagnosis to study enrolment was 6.97 (± 8.8) years. Table 1 shows the demographic and clinical characteristics for the overall sample and for both groups (stage 1 and stage 2). Mean age was 57.8 (± 11.5) years, 42.6% [95% confidence interval (CI) 0.94–1.20] were men and 57.4% (95% CI 0.84–1.06) were women. There were 261 patients [5.7% of the patients; coronary heart disease 3.7% (95% CI 0.47–0.89) and stroke 2.0% (95% CI 0.42–1.02)] with prior cardiovascular or cerebrovascular disease. At baseline (visit 1), mean systolic BP (SBP) was 157.37 (± 15.83) mmHg and mean diastolic BP (DBP) was 92.9 (± 8.36) mmHg, and these values were similar to those obtained before the initiation of aliskiren. Many patients had other HTN-associated risk factors. Overall, 35.8% (95% CI 0.84–1.07) of our population had dyslipidemia and 0.5% (95% CI 0.30–1.66) were obese, 21% (95% CI 0.84–1.12) had hypertriglyceridemia and 22.6% (95% CI 0.67–0.89) had diabetes. HTN-related target organ damage was observed in 1244 patients (27.1%; 95% CI 1.41–2.16) of the sample. Regarding the use of concomitant medication (Table 2), 1273 patients (27.7%; 95% CI 0.83–1.08) were on statins, only 832 patients (18.1%; 95% CI 0.70–0.95) were on oral antidiabetic agents and 1152 patients (25.1%; 95% CI 0.74–0.97) received antiplatelet drugs. Before adding or switching to aliskiren, 1559 patients (34.0%; 95% CI 0.63–0.81) had received angiotensin receptor blockers (ARBs) and 1310 patients (28.6%; 95% CI 0.68–0.89) had received ACEIs. The use of CCBs was observed in 26.7% (95% CI 0.51–0.66) of patients. Diuretics (other than hydrochlorothiazide) and beta blockers were less common.
Demographic and clinical characteristics of patients included in the study.

CI, confidence interval; CKD, chronic kidney disease; JNC, Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure; OR, odds ratio; SD, standard deviation.
Use of concomitant medication in the ALAS.

ACEI, angiotensin-converting enzyme inhibitor; CCB, calcium channel blocker; CI, confidence interval; JNC, Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure; OR, odds ratio.
Regarding the use of aliskiren, in most patients it was used as monotherapy (4073 patients, 88.8%; 95% CI 1.56–2.29). As shown in Table 3, the proportion of patients using 150 or 300 mg doses was similar, 40.3% (95% CI 3.18–4.11) and 48.4% (95% CI 0.34–0.44), respectively. However, in patients with target organ damage, the most common dose was 300 mg, which was used as monotherapy.
Patterns of aliskiren treatment according to severity of hypertension.

ACEI, angiotensin-converting enzyme inhibitor; CI, confidence interval; HCT, hydrochlorothiazide; JNC, Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure; OR, odds ratio; TOD, target-organ damage.
A total of 2140 patients (46.6%) and 2178 patients (43.4%) had stage 1 and stage 2 HTN, respectively. Baseline mean SBP and DBP were significantly lower in stage 1 patients compared with stage 2 patients (148.29 versus 167.6 mmHg, p = 0.01; 90.55 versus 95.73 mmHg, p = 0.01, respectively; see Figure 1). As seen in Table 1, patients with stage 1 HTN were younger, and there were fewer diabetic and obese patients, with less previous coronary heart disease, peripheral vascular disease, and stroke compared with stage 2 HTN patients. Stage 1 patients also had less use of additional medications, such as antiplatelet agents and antidiabetic drugs. The use of statins was similar between stage 1 and stage 2 groups.

Baseline systolic and diastolic blood pressure (mean values) in JNC1 and JNC2 patients.
Of the 4588 patients initially enrolled, 3147 (68.6%) patients completed the 6-month follow-up period. The 31.4% drop-out rate was due to AEs in 17 cases (0.37%), and loss to follow-up in the remaining cases. For the entire cohort, a statistically significant reduction in BP values was observed (Figure 2), with a mean SBP reduction of 29.2 mmHg and a mean DBP reduction of 13.78 mmHg from baseline at the 6-month visit. BP control was different in stage 1 and stage 2 patients. In stage 1 patients (Figure 3), mean SBP was significantly higher at baseline compared with 6-month visit values (148.29 versus 126.34 mmHg, p = 0.001, respectively) with a 21.95 mmHg mean absolute reduction. For DBP, comparable results were found (90.52 versus 78.95 mmHg, p = 0.001, baseline versus 6-month visit, respectively, with a mean absolute DBP reduction of 11.57 mmHg). Regarding the stage 2 group, for SBP, the values were 167.41 and 130.35 mmHg at baseline and 6-month visits, respectively (p = 0.001). The mean absolute SBP reduction was 37.06 mmHg. For DBP, the values were 95.73 and 79.52 mmHg at baseline and 6-month visits, respectively (p = 0.001). The mean absolute DBP reduction was 16.21 mmHg.

Systolic and diastolic blood pressure (mean values) at baseline versus 6-month visit (all population). BL, baseline; 6m, 6 months.
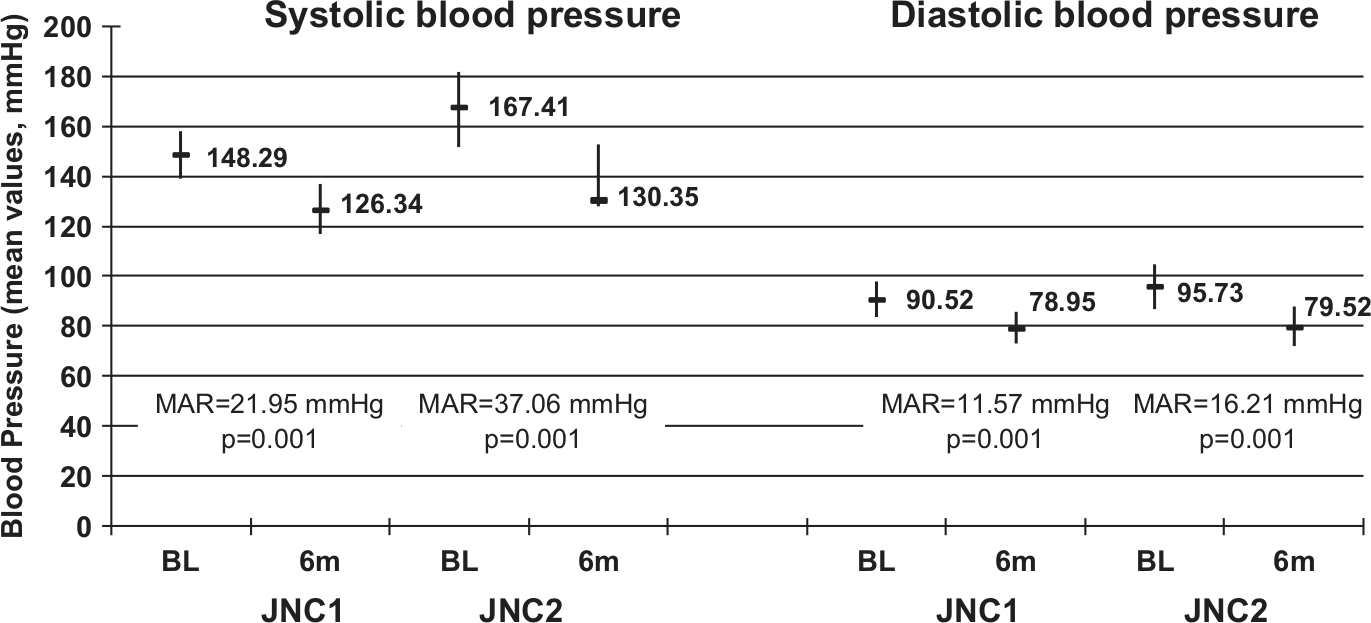
Blood pressure (mean systolic and diastolic values) in JNC1 and JNC2 patients at baseline versus 6-month visit. BL, baseline; 6m, 6 months; MAR, mean absolute reduction.
In terms of aliskiren effectiveness, when the study population was assessed per protocol (i.e. all of the patients that completed the study), the proportion of patients who achieved BP control based on their target BP was 85.4% in the stage 1 group and 80.4% in the stage 2 group. However, when the intent-to-treat population was considered (i.e. the entire population that received at least one dose of aliskiren), these percentages fell to 33.8% in the stage 1 group and 24.7% in the stage 2 group. When only patients with target organ damage were considered, these percentages were 28.8% and 26.7% for stage 1 and stage 2 groups, respectively. These figures are similar to the median control rates reported recently in the literature for different countries in Latin America (20–30%) [López-Jaramillo et al. 2013]. The prescribed doses of Aliskiren remained basically the same throughout the study. Only 676 patients (14.73%) required dosing adjustments.
Regarding safety, during the 6-month follow-up 177 patients (3.85%) reported AEs. Each of these patients reported only 1 AE; therefore, 177 AEs were reported for the 4588 patients enrolled. Of these, 54 events were considered aliskiren related (1.17%) and only 2 were severe AEs. The most frequently reported AEs were headache in 24 patients (0.52%), dizziness in 13 patients (0.28%), nausea in 8 patients (0.17%), and diarrhea in 7 patients (0.15%). No cases of symptomatic hypotension were observed. Eight patients required hospitalization for non-aliskiren-related causes, and no deaths were reported.
Discussion
There is a lack of post-marketing information about medicinal products throughout Latin America. Many post-marketing studies report safety and efficacy rates based on the analysis of administrative data, e.g. claims for drug prescriptions [Walker, 2010; Strom, 2006]. This observational cohort study provides a unique set of data from real-world patients treated at their physicians’ discretion with the novel oral direct renin inhibitor aliskiren, a drug that decreases the circulating levels of both angiotensin I and II, thus blocking the renin–angiotensin–aldosterone system from its very beginning [Nussberger et al. 2002]. Patients were recruited from five Latin American countries, representative of the diversity in clinical management and health systems across the region.
The efficacy of aliskiren has been demonstrated in several randomized controlled trials. Aliskiren treatment (alone or combined with hydrochlorothiazide) led to SBP reductions ranging from 14.7 to 22 mmHg and reductions in DBP of 4.8–16 mmHg [Villamil et al. 2007; Geiger et al. 2009; Schmieder et al. 2009; Uresin et al. 2007]. In our study, slightly higher reductions in both SBP and DBP were observed. This is a remarkable finding, as this is a study in real-world patients managed in a real-world scenario with less strict clinical control than in a randomized controlled trial. We observed that BP reductions were different in patients with stage 1 and stage 2 HTN. These observations are consistent with the aggressiveness needed in the management of patients with a high cardiovascular risk [Latin American Expert Group, 2009; National Heart, Lung, and Blood Institute Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure and National High Blood Pressure Education Program Coordinating Committee, 2003]. Although per-protocol baseline measurements were taken up to 7 days after aliskiren initiation, these values were similar to those obtained from medical records before the initiation of aliskiren. This supposed lack of effect during the first week of aliskiren use is in line with the known pharmacological properties of the drug, which requires 2–3 weeks of use to achieve its maximal effect.
Regarding aliskiren safety, an overall adverse event rate of 3.85% was observed. Compared with published data from clinical studies and safety reports sent to the Food and Drug Administration, the incidence of aliskiren-related adverse events in our observational study is rather low [Siragy, 2011].
From the original sample of 4588 patients, only 3147 (68.6%) completed the 6-month follow-up period. The drop-out rate is consistent with the adherence rate seen in ACEI- and ARB-treated patients [Dezii, 2000; Giorgi et al. 2011], which showed that only 55–60% of patients remain on the prescribed treatment at 6 months. In the case of aliskiren, a survey of prescription claims data reported that the 6-month adherence was 67% [Chang et al. 2011].
Limitations
Our study has several limitations. First, this is an observational study and there is no control group. Second, since the study was intended to show the results of the use of aliskiren in a real-life setting in patients with previously uncontrolled HTN, the patients did not undergo a wash-out period. Third, we did not collect detailed information on concurrent medication, except that we confirmed that concurrent drugs were not modified and the results show the effects of aliskiren as a monotherapy or combined with other antihypertensive drugs (aliskiren-based therapy). Finally, compliance was not measured as it was not among the study objectives.
Conclusion
In our cohort of nonselected patients who were managed in a real-world setting, aliskiren was found to be an effective and safe option to treat mild to moderate HTN, either as monotherapy or combined with hydrochlorothiazide. The difficulties in achieving in real life a task that, at first glance, seems to be simple, are of clinical relevance. The results seem to help foster in physicians the appropriate dosing of aliskiren as monotherapy, and the timely indication of combinations for the treatment of HTN and the reduction of avoidable cardiovascular events.
Footnotes
Acknowledgements
The authors want to acknowledge Dr Jorge Abate, from RDP, Caracas, Venezuela, for the statistical analysis and Dr Hugo Silva, Argentina, for their help in the preparation of the manuscript.
Funding
This work was supported by Novartis (Latin America).
Conflict of interest statement
Dr Volman is the HEOR Cardiovascular Manager, at Novartis Argentina. The remaining authors have no conflicts of interest to declare.
