Abstract
The renin-angiotensin system (RAS) activity is a key factor in the pathophysiology and development of hypertension, atherosclerosis, heart failure, and renal disease. It is unclear whether angiotensin-converting-enzyme (ACE) inhibitors and angiotensin-receptor blockers (ARBs) have fully delivered the expected reductions in cardiovascular risk. In fact, the optimized RAS suppression is difficult to achieve with these agents, partly because ACE inhibitors and ARBs both activate compensatory feedback mechanisms that result in renin release and increase plasma renin activity (PRA). Molecular modeling was used to develop aliskiren, a potent, low-molecular-weight, nonpeptide, direct renin inhibitor with sufficient bioavailability to produce sustained suppression of PRA after oral administration. This report discusses the mechanisms of action of oral renin inhibitors and their pharmacokinetic properties. In addition, the report also evaluates the available data regarding the effects of the renin inhibitor aliskiren in the treatment of hypertension and related cardiovascular disorders.
Introduction
The renin-angiotensin system (RAS) activity is a key factor in the pathophysiology and development of hypertension, atherosclerosis, heart failure, and renal disease.1,2 Angiotensin-converting-enzyme (ACE) inhibitors block the conversion of angiotensin I (Ang I) to angiotensin II (Ang II), and angiotensin-receptor blockers (ARBs) block the angiotensin type 1 receptor. However, it is unclear whether ACE inhibitors and ARBs have fully delivered the expected reductions in cardiovascular risk. In fact, optimized RAS suppression is difficult to achieve with these agents, partly because ACE inhibitors and ARBs both activate compensatory feedback mechanisms which result in renin release and increase plasma renin activity (PRA).3–5
Although the predictive value of PRA remains debatable, it has been shown to predict myocardial infarction in stroke survivors enrolled in the Perindopril Protection Against Recruitment Stroke Study (PROGRESS). 6 As such, if PRA is a predictive risk in a population of patients with vascular disease, this may represent residual risk and serve as a novel target of therapy by agents that inhibit renin and neutralize the compensatory rise in PRA. In addition to the presumed benefits of limiting PRA on Ang II bioactivity, renin inhibitors may limit the direct deleterious effects of renin to activate the (pro)renin receptor. Indeed, Nguyen et al 7 demonstrated the existence of a rennin/prorenin receptor. The rennin receptor can trigger intracellular signals by activating the extracellular signal-regulated kinase 1/2 pathway, and it also acts as a cofactor by increasing the efficiency of angiotensinogen cleavage by receptor-bound renin, thereby facilitating Ang II generation and action on the cell surface.
This review will discuss the mechanisms of action of oral renin inhibitors and their pharmacokinetic properties. In addition, the available data regarding the effects of the renin inhibitor aliskiren in the treatment of hypertension and related cardiovascular disorders will be discussed.
Aliskiren
Mechanism of action
Renin inhibition prevents the formation of Ang I and Ang II, as well as all the angiotensin peptides that are subsequently derived from Ang I and Ang II (Fig. 1). The addition of a renin inhibitor to ACE inhibitor or ARB therapy would therefore neutralize the compensatory rise in plasma renin activity that these agents induce, thus potentially enhancing suppression of RAS (Fig. 1). Moreover, because the renin enzyme is such a specific angiotensinogen, namely it is the only known natural substrate, renin inhibition would thus be expected to prove these additional benefits without any additional side effects.
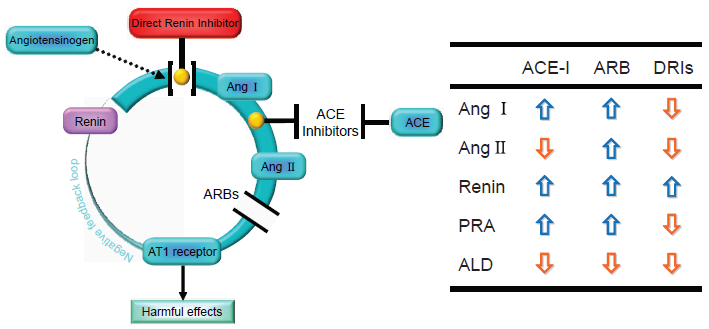
The pharmacological modulations of the renin-angiotensin system. Cleavage of angiotensinogen by the protease renin produces the decapeptide angiotensin I (Ang I). Ang I is then further transformed by the angiotensin-converting enzyme (ACE), producing the octapeptide angiotensin II (Ang II). Ang II is the end product of the cascade and binds to Ang II subtype 1 receptors (AT1), thereby leading to harmful effects on the heart, vasculature, and kidneys. The inhibition of the production or action of Ang II by ACE inhibitors or angiotensin receptor blockers (ARBs) prevents stimulation of the AT1 receptor and disturbs the negative feedback loop through which Ang II normally inhibits renin release by the kidney. ACE inhibitors and ARBs thus stimulate a compensatory increase in renin release from the kidney. A direct renin inhibitor could inhibit the reactive rise in renin activity that occurs with ACE inhibitors or ARBs.
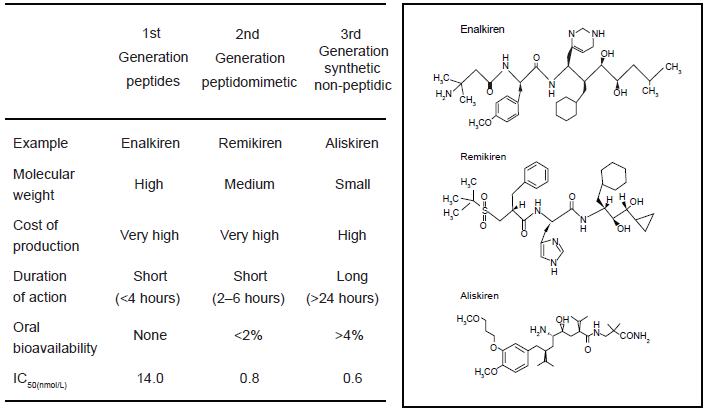
The chemical development of renin inhibitors, leading to the discovery of aliskiren, can be divided into three generations of compounds (Fig. 2): first, peptide analogues of angiotensinogen to block the enzymatic action of renin; 8 second, peptidomimetic compounds that are dipeptide transition-state analogue inhibitors of the active site;9–12 and third, non-peptide-like compounds, of which aliskiren is the most successful example.13,14 The first-generation renin inhibitors are not very potent and they also tend to metabolically unstable. The second-generation compounds were potent (with activity in the nanomolar range) and, when administered by the parenteral route, lowered BP in both animals and humans. These studies thus provide insight into the endocrine and hemodynamic effects of renin inhibitors in animal models. Further development of the second-generation molecules led to longer durations of the action and oral activity, although very high doses were needed in humans. The structural changes in the molecules to improve metabolic stability have been successful, but the oral bioavailability and the associated lowering of BP remained low. Their clinical use was thus limited, not only by their lack of oral activity, but also due to their short duration of action. The third generation of compounds benefited from advances in crystallography and a structure-based approach to drug design, and led to the discovery of aliskiren. Aliskiren has the chemical structure 2(S),4(S), 5(S),7(S)-N-(2-carbamoyl-2-methylpropyl)-5-amino-4-hydroxy-2,7-diisopropyl-8-(4-methoxy-3-[3-methoxypropoxy-]phenyl)-octanamide and a molecular mass of 551.8 g/mol. 14 Aliskiren is an extremely potent competitive inhibitor of renin with an IC50 (concentration inhibiting 50% of activity) of 0.6 nmol/L for both purified human renin and human plasma rennin (Fig. 2). 14 It has a high specificity for primate renin, and it also shows a 10,000-fold lower affinity for related aspartic peptidases. This high specificity for renin makes it unlikely to produce adverse effects through interaction with other enzymes. In comparison to earlier renin inhibitors, aliskiren has favorable physiochemical properties with high aqueous solubility and lower lipophilicity, rendering it more resistant to degradation. This leads to improved bioavailability after oral administration (Fig. 2). Aliskiren is already bound to renin in the plasma prior to uptake by the tissues and binding to the (pro)renin receptor (Fig. 3). Aliskiren may also inhibit receptor-bound renin and prorenin in the tissues (Fig. 3).
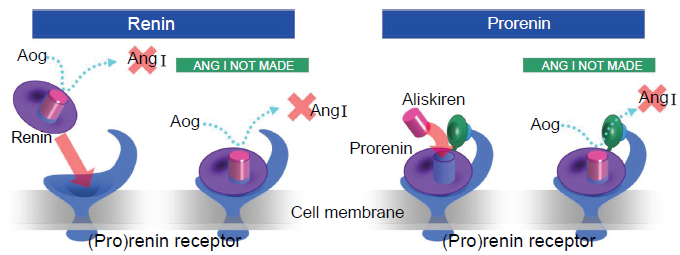
Inhibitory effect of aliskiren on tissue production of Ang II by renin and (pro)renin. Diagrammatic representation of the different conformation of renin and prorenin, and their binding to aliskiren.
Pharmacokinetic profiles
The absorbed fraction of orally administered aliskiren is estimated to be about 5% with 90% of the drug excreted unchanged by the fecal route. 15 In contrast to other renin inhibitors (ex. remikiren or enalkiren) with similar affinity for renin, 16 aliskiren has a long plasma half life, namely it is reported to range from 24 to 70 hours.16–19 The long half life of aliskiren may be advantageous for patients occasionally missing doses of the drugs. Steady-state plasma aliskiren levels are achieved after 5 to 8 days of daily dosing. A long plasma half-life and very low urinary excretion (<1%) are observed, thus suggesting that binding of the drug to plasma proteins may be higher than the 49.5% reported for human plasma. 20 Aliskiren is not metabolized by cytochrome P450, it does not interfere with the action of warfarin, 21 and it also shows no clinically relevant pharmacokinetic interactions with lovastatin, atenolol, celecoxib, or cimetidine. 22
Experimental Studies
End organ protection
The specificity of aliskiren for primate renin precludes the use of most animal models in which end organ effects of antihypertensive agents are commonly evaluated. The development of double transgenic rats (dTGR), which express human genes for both renin and angiotensinogen, has provided a suitable animal model in which to investigate the tissue protective effects of renin inhibitors.23,24
Pilz et al 25 compared aliskiren and valsartan in preventing target organ damage in dTGR. Matched 6-week-old dTGR received no treatment, low-dose or high-dose aliskiren, or low-dose or high-dose valsartan. Untreated dTGR showed severe hypertension, albuminuria, and increased serum creatinine by week 7, and 100% mortality rate by week 9. In contrast, high-dose valsartan and both doses of aliskiren lowered BP, reduced the albuminuria and creatinine levels, and resulted in 100% survival at week 9. Treatment with aliskiren and high-dose valsartan also reduced left ventricular hypertrophy (LVH); the magnitude of this effect was somewhat greater with high-dose aliskiren. In other renal protection studies using the double transgenic rat model, aliskiren reduced renal inflammation and fibrosis as well as albuminuria. 26 In dTGR rats with diabetic nephropathy, aliskiren reduced albuminuria and other markers of renal damage, including the expression of TGF-β and collagens III and IV. 27 When aliskiren was compared with either ACE inhibitors or ARBs, the renal protective effects were approximately equal.25,27
Effect of aliskiren on endothelial function
An important question is whether direct renin inhibition improves endothelial function and nitric oxide (NO) bioavailability. Aliskiren augments both basal and acetylcholine-stimulated NO production in Watanabe heritable hyperlipidemic (WHHL) rabbits and improves endothelium-dependent vasorelaxation in thoracic aortic segements. 28 Endothelial NO synthase (eNOS) bioavailability is regulated by different mechanisms, including transcriptional upregulation of eNOS, posttranscriptional activation of eNOS, and reduction of reactive oxygen species-mediated breakdown of NO. Aliskiren improves the NO bioavailability via at least two pathways. Aliskiren affects eNOS by increasing eNOS mRNA stability and posttranscriptional stimulation of eNOS mRNA. The phosphorylation of Ser1177 within eNOS by Akt is critical for the activation of eNOS, thus the effects of aliskiren were investigated in this regard. Aliskiren activates protein kinase Akt, which leads to posttranscriptional activation of eNOS via phosphorylation. Aliskiren may restore eNOS uncoupling via augmenting tetrahydrobiopterin (BH4) levels, an essential cofactor for eNOS. 29 This multidomain enzyme contains 2 functional regions: a flavin-containing reductase domains and a heme-containing oxygenase domain, which has binding sites for the triad of heme, L-arginine, and BH4. Between these 2 domains, there is a regulatory calmodulin-binding sequence. Electron transfer from reduced nicotinamide-adenine dinucleotide phosphate to heme occurs in the presence of calcium/calmodulin, and with L-arginine present, electrons can flow to the heme moiety to reduce oxygen, which, in turn, is used to oxidize L-arginine to NO. BH4 functioning as both an allosteric and redox cofactor for eNOS, stabilizes eNOS and improves the binding affinity of L-arginine and the heme ligand for eNOS. Under physiological conditions, eNOS, in a homodimeric configuration, functions normally and become catalytically active in the presence of adequate amounts of BH4. Diminished levels of BH4 “uncouple” eNOS and prevent stable formation of its homodimeric configuration. This uncoupled enzyme now serves to produce potent free radicals, such as superoxide anions.
Effect of aliskiren on atherosclerosis
Nussberger et al 30 demonstrated that aliskiren treatment reduced the atherosclerosis volume and vulnerability in 2-kidney, 1-clip apolipoprotein E-/- mice, and this effect was at least partially independent of any change in the blood pressure. Likewise, Lu et al 31 demonstrated that aliskiren profoundly reduced atherosclerosis in the LDLLr-/-mice fed a fat-enriched diet. The reduction in atherosclerosis noted in the LDLLr-/- mice with aliskiren appears to be quantitatively greater than that reported with ARBs or ACEs in similar models, across different studies. Furthermore, aliskiren has protective effects on atherosclerotic changes in Watanabe heritable hyperlipidemic (WHHL) rabbits. 28 Therefore, at least in animal models, aliskiren exhibits an anti-atherosclerotic effect. Importantly, it appears that renin deficiency in bone marrow-derived cells decreases atherosclerosis via the inhibition of monocyte adhesion to endothelial cells in the vascular wall, involving a prominent role for macrophage-derived renin in atherosclerosis. 31
Combination therapy with aliskiren and valsartan is also beneficial for WHHL rabbits. 28 Combination therapy has an additive effect on plaque volume. These data suggest that combination therapy of an ACEI/ARB with aliskiren may be a superior strategy with respect to vascular protection. Whereas aliskiren may be similar to conventional RAS blockers in monotherapy situations, it offers unique benefits in combination therapy that are superior to the addition of an ACEI to ARB or vice versa. Recent data suggest that the addition of an ARB to an ACEI does not promote an additional clinical benefits in patients with atherosclerosis. 32 The ability of aliskiren to neutralize the rise in PRA in AECI- or ARB-treated patients translates into a reduction in Ang II production and action that exceeds what can be achieved by combination therapy with either agent alone. This may explain in part the hemodynamic superiority of combining valsartan and aliskiren in hypertension, 33 the beyond blood pressure-mediated renal benefits noted in patients with diabetic nephropathy (Aliskiren in the Evaluation of Proteinuria in Diabetes [AVOID] Trial), 34 and the profound reductions in brain natriuretic peptide noted in ACEI/ARB-treated patients with heart failure. 35
Clinical Studies
Effects on the RAS
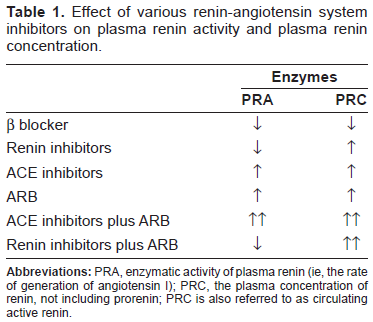
In a crossover study in normotensive volunteers receiving a low-sodium diet, Nussberger et al 36 evaluated the effects of 4 oral doses of aliskiren (40, 80, 160, 640 mg) in comparison to placebo and the ACE inhibitor enalapril (20 mg) on components of the RAS. After single oral doses and 8 days of repeated once-daily dosing, aliskiren reduced PRA in a dose-dependent manner. In comparison to a placebo, the highest doses of aliskiren reduced Ang II levels by a maximum of 89% and 75% on days 1 and 8, respectively. At doses of aliskiren >80 mg/day, there was a 40% to 50% decrease in both plasma and urinary aldosterone levels. Higher doses of aliskiren enhanced natriuresis. Although enalapril dramatically reduced the Ang II levels to a comparable degree as aliskiren, its administration was associated with a > 15-fold increase in PRA. Both aliskiren and enalapril increased the plasma renin concentration (PRC). Because normal feedback inhibition is interrupted by Ang II, renin inhibition consistently elicits a rise in the level of circulating active renin (Table 1). This escape process, which also occurs during treatment with ACE inhibitors and ARBs, explains why these drug classes behave as incomplete blockers of the renin system (Table 1). If increased renin levels are deleterious, then it is doubtful that these drugs would be as effective as they are in reducing long-term cardiovascular end points. Nevertheless, the PRC levels after the administration of aliskiren, particularly when combined with other drugs, are significantly higher than have been seen with other antihypertensive regimens (Table 1). Azizi et al 18 examined the mechanistic aspects of dual RAS blockade after single oral doses of aliskiren and the AT1 receptor blocker valsartan. In a crossover study, placebo, aliskiren (300 mg), valsartan (160 mg), and a low-dose combination of the 2 drugs (aliskiren 150 mg + valsartan 80 mg) were given to 12 sodium-depleted normotensive men. Monotherapy with aliskiren decreased PRA. Valsartan increased PRA as well as circulating levels of Ang I and Ang II. In the combination, PRA, Ang I, and Ang II levels were approximately equal to that seen with a placebo. Therefore, the addition of aliskiren eliminated the compensatory increase in PRA and its downstream products that occurred after ARB administration.
Effect of various renin-angiotensin system inhibitors on plasma renin activity and plasma renin concentration.
Hypertension
Comparison with placebo
Placebo-controlled trials have assessed the antihypertensive effects of aliskiren monotherapy.33,37–42 Short-term treatment (4 to 8 weeks) with once-daily aliskiren at doses >75 mg/day consistently produced significant BP reduction in comparison to a placebo (Fig. 4). 37 A dose-response relationship was documented up to 300 mg/day; little or no additional BP reduction was observed with a higher dose (600 mg). When given for 8 weeks, three doses of aliskiren (150, 300, and 600 mg/day) reduced PRA by 70% to 73% from baseline without a measurable dose-dependent effect on this parameter. Increases in PRC were dose-related in patients receiving 150 mg (+128%), 300 mg (180%), and 600 mg (386%). Most of the antihypertensive effects of aliskiren were achieved during the first 2 weeks, and a near-maximum BP reduction occurred by week 4. Monitoring the 24-hr BP showed that aliskiren significantly reduced the mean ambulatory systolic and diastolic BP over 24-h.
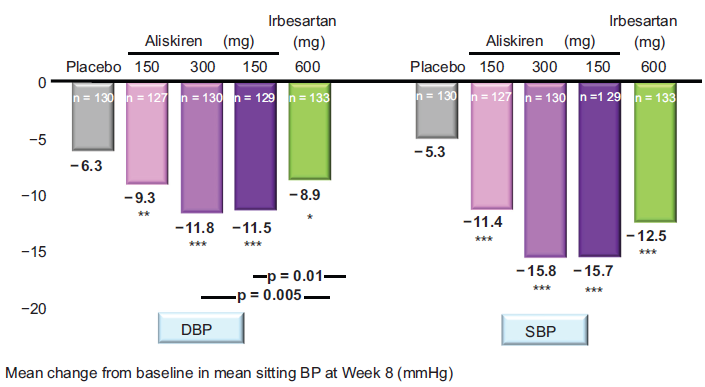
Aliskiren monotherapy provides dose-dependent reductions in DBP and SBP. The mean change from baseline in mean sitting diastolic and systolic blood pressure (BP) (DBP and SBP, respectively) after 8 weeks of treatment with 150, 300, 600 mg of aliskiren, or 150 mg of irbesartan.
Combination therapy
Aliskiren has been compared with commonly used antihypertensive agents, including hydrochlorothiazide (HCTZ), ramipril, and several ARBs. In general, the efficacy of aliskiren is similar to that of other antihypertensive drugs. Eight weeks of treatment with aliskiren at doses of 150 and 300 mg/day resulted in a BP reduction comparable to that of standard therapy with low-dose HCTZ (12.5 mg/day, 25 mg/day). Monotherapy with HCTZ activated the RAS. The PRA increased by 44.7% at a dose of 12.5 mg/day and 71.9% with 25 mg/day; corresponding increases in PRC were 26.1% and 108.4%, respectively. 38 Because of their complimentary pharmacologic actions, diuretics are natural combination partners with drugs such as aliskiren that block the RAS. In a large factorial design study, 2776 patients with stage I and II hypertension received a placebo, aliskiren (75, 150, or 300 mg), HCTZ (6.25, 12.5, or 25 mg), or various combinations of aliskiren + HCTZ over an 8-week period. 35 The BP reduction with combination of aliskiren and HCTZ seemed to be fully additive (i.e. the BP-reducing effect observed with combinations was approximately equal to the sum of BP reduction obtained with each component) less the placebo response. An average BP reduction of 21.2/14.3 mmHg was seen at the highest combination dose (aliskiren 300 mg + HCTZ 25 mg). In obese hypertensive subjects, aliskiren 300 mg/HCTZ 25 mg was equipotent in comparison to irbesartan 300 mg/HCTZ 25 mg indicating that the combination of a direct renin inhibitor and diuretic is as effective as commonly used diuretic/ARB. 42 Combination with aliskiren blocked the increases in PRA seen with HCTZ monotherapy while PRC levels increased markedly. The administration of aliskiren/HCTZ 150/12.5 mg reduced PRA(-49.6%) and increased PRC (+305%).
The clinical effectiveness and safety of a high-dose combination of aliskiren and the ARB valsartan was tested in 1,797 patients with a mean baseline BP 154/100 mmHg (Fig. 5). 33 Patients were randomly assigned to receive a placebo, aliskiren, valsartan, or the combination of aliskiren/valsartan. The dosage in the active treatment arms was titrated to the maximum recommended doses of each agent (aliskiren 300 mg, valsartan 320 mg); the combination dose was aliskiren/valsartan 300/320 mg. The high-dose combination reduced BP by a mean of 17.2/12.2 mmHg, significantly greater than was observed with either component (aliskiren 13.0/9.0 mmHg, valsartan 12.8/9.7 mmHg). The PRA increased with valsartan (+160%), decreased with aliskiren (-73%), and was also suppressed by the combination (-44%). The PRC increased with all active treatments; the increase in the combination therapy arm (+912%) was statistically greater than that seen with aliskiren (+468%) or valsartan (+138%) alone. The highdose combination of aliskiren and valsartan was well tolerated. There was an increased incidence of hyperkalemia (> 5.5 mmol/l) in patients receiving the combination with those receiving monotherapy. In 13 of 18 patients with this finding, potassium returned to the normal range at the end of the study without any dosage adjustment. 33
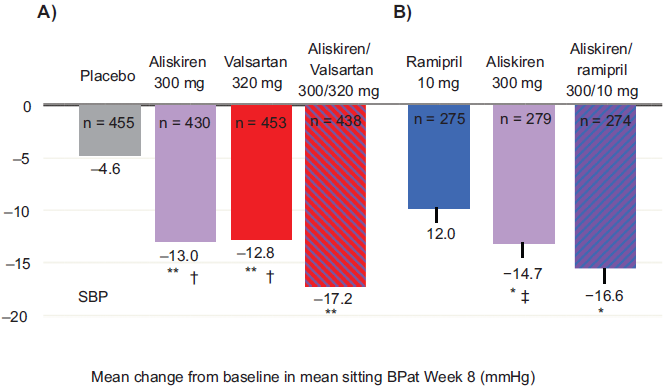
Aliskiren combination therapy provides significantly greater BP reductions than either component monotherapy. A) Effects of aliskiren alone and in combination with valsartan on the mean change from baseline in the mean sitting systolic BP (SBP). **p < 0.0001 vs placebo. †p < 0.0001 vs. aliskiren/valsartan combination therapy. B) Effects of aliskiren alone and in combination with ramipril on the mean change from baseline in the mean sitting systolic BP (SBP).
The effects of dual RAS blockade with aliskiren and the ACE inhibitor ramipril were studied in 837 patients with diabetes and hypertension (Fig. 5). 43 The doses used in this study were the most commonly used dose of ramipril (10 mg), the maximum recommended dose of aliskiren (300 mg), and the combination of these drugs (aliskiren/ramipril 300/10 mg). The BP was reduced by 16.6/12.8 mmHg with combination therapy, which was statistically greater than the BP reduction seen with aliskiren (14.7/11.3 mmHg) or ramipril (12.0/10.7 mmHg) alone.
End-organ Protection
A combination strategy is now being utilized in studies examining the effects of aliskiren on intermediate markers of end organ damage. The AVOID (Aliskiren in evaluation of prOteinuria in Diabetes) study 34 assessed whether dual renin system intervention with aliskiren added to current optimal treatment with losartan could provide additional renal protection compared to the addition of a placebo in hypertensive patients with type 2 diabetes and macroalbuminuria (Fig. 6). Male and female patients aged 18-85 years with hypertension (mean sSBP > 140 mmHg nad/or mean sDBP > 90 mmHg), proteinuria (urinary albumin creatinine ratio (UACR) ≥200 mg per g to ≤3,500 mg per g) and type 2 diabetes mellitus were included in the trial. All patients were started on 100 mg of losartan for 12 to 14 weeks, in addition to the optimal hypertension therapy, to reach a target BP of 130/80 mmHg. Following the runin period, eligible patients with BPs below 150/95 mmHg and UACRs ≥ 100 mg per g were randomized or a placebo in addition to losartan and optimal anti-hypertensive therapy. After 12 weeks, the patients receiving aliskiren were force titrated to 300 mg aliskiren once-daily and treatment continued for all patients for a further 12 weeks. The primary efficacy criterion was the change in UACR with the secondary criterion of BP control. Six hundred patients were evaluated at the end of the treatment period. Aliskiren provided an 18% reduction in UACR in comparison to the placebo at week 24 (p = 0.0009). The BP control was similar between the two treatment groups and remained controlled throughout the study. Treatment with aliskiren was well tolerated; the incidence of adverse effects and serious adverse events was similar between the aliskiren and placebo groups. The most commonly reported adverse events were headache, nasopharyngitis and dizziness. The effect of aliskiren on decreasing albuminuria was observed within 3 days and reached a maximal effect within 28 days of treatment with doses of 300 mg once-daily.
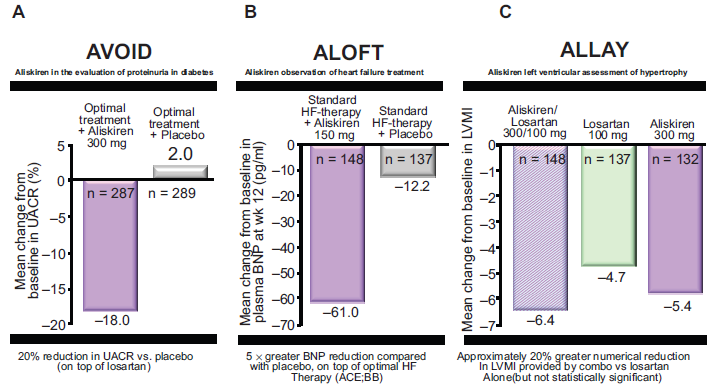
Aliskiren and end-organ protection. A) The aliskiren in the evaluation of proteinuria in diabetes (AVOID) study showed that treatment with 300 mg of aliskiren daily reduced the mean urinary albumin-to-creatinine ratio (UACR) by 20% in comparison to a placebo. B) The aliskiren observation of heart failure treatment (ALOFT) study showed that BNP decreased by a mean ± SD of 61.0 ± 257 pg/mL in the aliskiren group and by 12.2 ± 243 pg/mL in the placebo group (P = 0.0160). C) The aliskiren in left ventricular hypertrophy (ALLAY) trial demonstrated highly significant reductions in LVMI from baseline in all treatment groups, with 4.9-g/m2 (5.4%), 4.8-g/m2 (4.7%), and 5.8-g/m2 (6.4%) reduction in the aliskiren, losartan, and combination arms, respectively (P < 0.0001 for all treatment groups). Panel A was adapted from Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK. Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med. 2008;358:2433-46. 34 Panel B was adapted from McMurray J, Pitt B, Latini R, et al. Effects of the oral direct renin inhibitor aliskiren in patients with sympathetic heart failure. Circulation: Hear Failure. 2008;1:17-24. 35 Panel C was adapted from Parving HH, Persson F, Lewis JB, Lewis EJ, Hollenberg NK. Aliskiren combined with losartan in type 2 diabetes and nephropathy. N Engl J Med. 2008;358:2433-46. 34
The ALOFT (aliskiren observation of heart failure treatment) study 35 was designed to assess the safety of adding 150 mg aliskiren to standard therapy in hypertensive patients with stable heart failure (Fig. 6). Efficacy evaluations were also included, such as B-type natriuretic peptide (BNP) and N-terminal (NT)-pro BNP levels, in PRA and in echocardiographic measurements. A total of 302 hypertensive patients with a mean age 68 years who had had stable New York Heart Association (NYHA) heart failure class II–IV for 1 month or longer were recruited. The patients were randomized to 12 weeks of treatment with 150 mg aliskiren or placebo, in addition to the stable doses of ACE inhibitor, ARB, aldosterone antagonist or β-blockers that they were already taking. The mean plasma BNP levels in these patients were above 100 pg per ml (28.9 pmol per L) at baseline. In Comparison to a placebo, aliskiren reduced plasma NT-pro BNP by 25% (p = 0.0106); plasma BNP by 25% (p = 0.016) and urinary aldosterone by 21% (p = 0.015). There was also a favorable change in a Doppler-echocardiographic measurement of left ventricular filling pressure. Aliskiren was well tolerated and there was no significant excess of hypotension or renal dysfunction. These improvements in key indicators of heart-failure severity, namely BNP, NT-pro BNP and urinary aldosterone, warrant further investigation in larger clinical trials.
The Aliskiren in Left Ventricular Hypertrophy (ALLAY) study 44 was designed to assess primarily whether the combination of aliskiren and losartan alone in reducing LV mass (LVM) index (LVMI), secondarily whether aliskiren was comparable to losartan in reducing LVMI, and finally whether aliskiren, alone or in combination with losartan, was safe and as well tolerated in patients with hypertension (Fig. 6). A total of 465 patients with hypertension, increased their ventricular wall thickness, and body mass index >25 kg/m2 were recruited. The patients were randomized into groups receiving 9 months of treatment with aliskiren 300 mg, losartan 100 mg, or their combinations. The systolic and diastolic blood pressures decreased similarly in all of the treatment groups (6.5 ± 14.9/3.8 ± 10.1 mmHg in the aliskiren group; 5.5 ± 15.6/3.7 ± 10.7 mmHg in the losartan group; 6.6 ± 16.6/4.6 ± 10.5 mmHg in the combination arm; P < 0.0001 within groups, P = 0.81 between groups). LVMI was reduced significantly from baseline in all of the treatment groups (4.9-, 4.8-, and 5.8 g/m2 reductions in the aliskiren, losartan, and combination arms, respectively; P < 0.0001 for all treatment groups). The reduction in LVMI in the combination group was not significantly different from that with losartan alone (P = 0.52). Aliskiren was as effective as losartan in reducing LVMI (P < 0.0001 for non-inferiority). In addition, the safety and tolerability were similar across all treatment groups. Taken together, aliskiren was as effective as losartan in promoting LV mass regression. The reduction in LV mass with the combination of aliskiren plus losartan was not significantly different from that with losartan monotherapy, independent of blood pressure lowering. These findings suggest that aliskiren was as effective as an ARB in attenuating this measure of myocardial end-organ damage in hypertensive patients with LV hypertrophy.
Safety
In a pooled analysis of data including 2,316 patients who received aliskiren monotherapy, the tolerability profile of aliskiren was similar to that of either a placebo or ARBs at doses up to 300 mg daily (Table 2). 41 An increased incidence of diarrhea (9.5%) was observed at a dose of 600 mg in comparison to a placebo (1.2%). The total number of reported adverse events was similar to those with a placebo. Serious adverse events occurred in 11 patients (0.5%) who received aliskiren in comparison to 5 patients (0.6%) who received a placebo.
Safety of aliskiren.
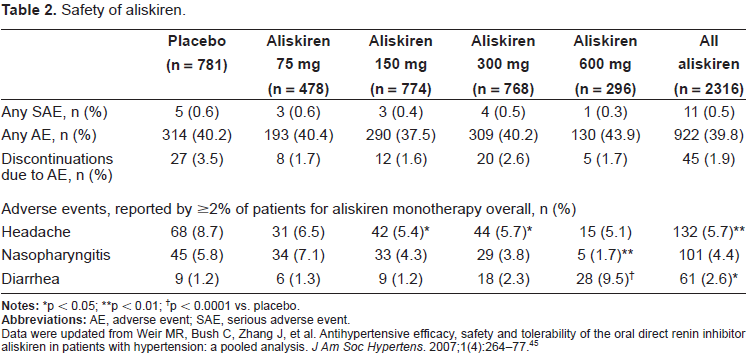
p < 0.05;
p < 0.01;
p < 0.0001 vs. placebo.
Data were updated from Weir MR, Bush C, Zhang J, et al. Antihypertensive efficacy, safety and tolerability of the oral direct renin inhibitor aliskiren in patients with hypertension: a pooled analysis. J Am Soc Hypertens. 2007;1(4):264-77. 45
Place in Therapy
Aliskiren might be clinically indicated in combination therapy with drugs that lead to a reactive increase in the PRA, such as diuretics, ACE inhibitors and ARBs. Aliskiren might be useful in younger patients, who have a more active RAS than older people. Aliskiren might also be useful in patients intolerant of ACE inhibitors, for the treatment of disorders in which Ang II contributes to the pathogenesis and for secondary prevention of cardiovascular diseases. Moreover, aliskiren offers additional safety for patients with cardiovascular disease and concomitant renal dysfunction, because it is preferentially eliminated via the liver without much interference with other drugs. Like ACE inhibitors, aliskiren behaves as a vasodilator with the potential to improve the elasticity of the large arterioles. Conversely, they are likely to be subject to the same contraindications as ACE inhibitors and ARBs, such as pregnancy and bilateral renal-artery stenoses.
Conclusions
Aliskiren produces a dose-dependent BP reduction and placebo-like tolerability up to the plateau in the dose-response curve that occurs at approximately 300 mg/day. Its antihypertensive potency is equivalent to those of ARBs, ACE inhibitors, and diuretics. Furthermore, combining aliskiren with other antihypertensive drugs provides a greater therapeutic efficacy, without any adverse drug interactions being observed. The blockade of the RAS obtained with ACE inhibitors or ARBs can be further enhanced by adding aliskiren to these agents.
Experimental and clinical studies show that aliskiren therefore has a considerable potential for organ protection beyond BP reduction. These important cardiovascular effects occur without any limiting adverse events, and the long half-life of the compound also justifies once-daily administration. Although critical questions regarding the effectiveness of aliskiren in blocking the effects of renin/pro-renin at the site of the (pro)renin receptor remain to be answered, renin inhibition opens up a new therapeutic potential for a more complete blockade of the RAS and further decreases in both cardiovascular morbidity and mortality. Forthcoming studies (ATMOSPHERE, and APOLLO, etc.) will address issues related to end organ protection and reduction in long-term cardiovascular end-points.
Disclosure
The authors report no conflicts of interest.
