Abstract
Nitazoxanide is a broad-spectrum agent active against several protozoa, helminths, and bacteria, including C. difficile and H. pylori. It is available as an oral tablet and suspension, both with adequate bioavailability. Nitazoxanide is associated with minimal side effects, has an acceptable safety profile, and has been classified as a pregnancy category B agent. It is 99% protein bound, which could result in drug interactions. It is the preferred agent for the treatment of Cryptospordiosis and Giardiasis in immunocompetent patients and has shown promise for the treatment of rotavirus, mild to moderate initial C. difficile infection, refractory C. difficile infection, Amoebiasis, Blastocystosis, and Taenia saginata.
Introduction
Diarrheal illness is a significant worldwide cause of morbidity and mortality. Those resulting in gastrointestinal illness are greatest in developing countries due to the various routes of transmission specifically tied to hygiene (Table 1). The World Health Organization estimates that each child <4 years old will have an average of 3.2 diarrheal episodes per year. While mortality rates attributed to diarrheal illness have declined, the associated mortality rate is still 3.8 deaths per 1,000 children <4 years old. Diarrheal illness can lead to malnutrition, a significant mortality cause in developing countries especially in those patients infected with Human Immunodeficiency Virus (HIV).
Causative organisms and route of transmission.
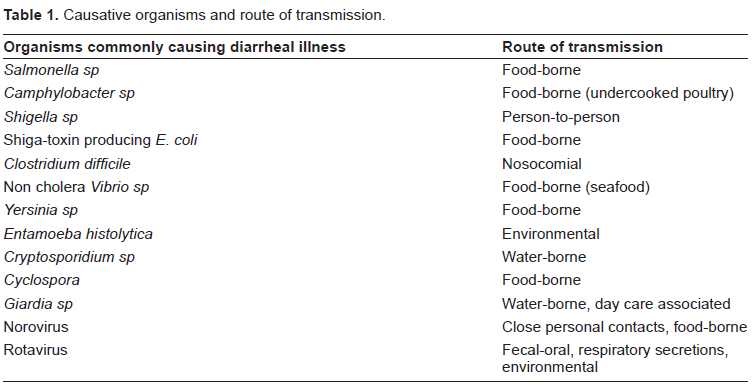
Overall, according to the World Health Organization, protozoal and helminth infections result in gastrointestinal illness for over 450 million patients. Both immunocompetent and immunosuppressed patients are at risk. While traditional therapies continue to be moderately efficacious, new therapies must be developed due to increasing resistance.
Evaluation and Diagnosis of Diarrheal Illness
Diarrheal illness can be classified as acute (≤ 14 days in duration), persistent (>14 days) and chronic (>30 days). Most cases of acute diarrhea are self-limiting and require only supportive care. Primary causes of acute diarrhea include viruses, bacteria and rarely protozoa. When a patient presents with diarrhea, a thorough physical exam and careful history should be taken to determine:
Diarrhea duration, frequency, and consistency (including whether blood is present)
Number of vomiting episodes
Medications patient is currently taking including recent antibiotic use
Comorbidities
Factors that could have exposed the patient to enteric pathogens including:
Residence
Travel history
Presence of pets or other animals
Food history
Other current infectious processes
Dehydration severity
Malnutrition (if present) severity
Immunization history
Exposure to individuals with similar illness
A thorough history can help providers determine the likely causative organism (Table 2).
Quality of diarrheal illness and likely causative organisms.

Stool cultures can be performed to determine the causative organism or to exclude presence of an organism in those patients with inflammatory bowel disease. Testing is indicated when specific pathogens are suspected and their identity needs to be confirmed to determine treatment. Some diagnostics are organism-specific, including assays performed to detect Clostridium difficile toxins A and B and antigen detection assays for various organisms including Entamoeba histolytica. Serologic testing can also be performed when available, but often cannot differentiate between active and past infections. Fecal leukocytes can also be measured as a predictor for bacterial illness; however not all diagnostics are readily available in some areas and other fecal leukocyte assays are dependent on operator skill to achieve accurate results. Molecular diagnostics have been and continue to be developed for the detection of specific pathogens. Unfortunately, while these assays tend to be very sensitive, cost and availability can prevent their use.
Supportive Care and Adjunctive Therapies
All patients with diarrheal illness should receive supportive care. Fluid replacement depends on the patient age, dehydration severity and available resources. 1 The World Health Organization (WHO) recommends a reduced osmolarity oral rehydration solution (Table 3) and has guidelines to estimate the fluid deficit. Intravenous rehydration can be done in severe cases. Recommended intravenous solutions to use for replacement include Ringer's lactate, Ringer's lactate with 5% dextrose, Dhaka solution, half-strength Darrow with 5% dextrose and normal saline. Several studies have also demonstrated the impact of zinc supplementation on acute diarrhea management. The WHO also recommends zinc supplementation during the diarrheal illness until it resolves. The recommended supplement dose is 10 mg to 20 mg of elemental zinc daily for 10 to 14 days until and after resolution of diarrheal illness. This has been shown to lower the incidence of diarrhea in the two to three months following.2,3 Infants with diarrhea should continue to breastfeed, and increased breastfeeding should be encouraged. Antimotility agents do not benefit children with acute or persistent diarrhea and are contraindicated in patients <5 years old. 4 In adult patients, infectious diarrhea is best managed without antimotility agents and their use is contraindicated in patients with Clostridium difficile infection. 5
Composition of reduced (low) osmolarity oral rehydration solution as recommended by WHO and UNICEF.

Nitazoxanide for Treatment of Diarrheal Illness
Nitazoxanide (NTZ) was first studied as a potential treatment for helminth infections in animals. 6 It has since been studied in humans and, based on broad spectrum antiparasitic activity in humans, marketed in Latin America since 1996 due to the great incidence of intestinal parasitic infections in that region. In 2004, NTZ was approved by the FDA for the treatment of diarrhea caused by Cryptosporidium parvum and Giardia lamblia in children 1-11 years of age. It is the treatment of choice for these infections. Additional in vitro and clinical trial data have been collected to determine the activity and efficacy of NTZ against other organisms causing gastrointestinal illness, such as Clostridium difficile, rotavirus and Helicobacter pylori.
Pharmacology
NTZ, 2-acetyloxyl-N-(5-nitro-2-thiazolyl), is a nitrothiazole derivative prodrug, structurally related to metronidazole, with broad antiparasitic and anaerobic antimicrobial activity.7,8 Antiprotozoal activity results from interference with energy production via the pyruvate-ferredoxin oxidoreductase (POFR) enzyme-dependent electron transfer reaction. Activity against Giardia lamblia has been postulated to be due to inhibition of a nitroreductase homologue, GINR1 based on affinity chromatography data. 9 Other mechanisms of action are likely, but have not yet been elucidated. (Alinia package insert). 10
Several in vitro activity studies have been conducted demonstrating broad-spectrum NTZ activity against both prokaryotic and eukaryotic organisms including Giardia intestinalis, Clostridium difficile, Bacteroides species, Prevotella species, Fusobacterium species, Clostridium perfringens, Peptostreptococcus species, Cryptosporidium parvum, Giardia lamblia, Trichomonas vaginalis, Entamoeba histolytica, Echinococcus granulosus, Helicobacter pylori and rotavirus.7,11–18
NTZ is available as a 500 mg oral tablet and a 100 mg/5 mL oral suspension. The oral suspension contains 1.48 g of sucrose per 5 mL which should be considered when administering to diabetic patients. (Alinia package insert).
Pharmacokinetics
NTZ oral tablet bioavailability data are not currently available. Relative bioavailability studies have been conducted comparing the tablet to the oral suspension. The oral suspension is 30% less bioavailable than the tablet formulation. Similar cure rates for patients receiving tablets or oral suspension while being treated for C. parvum infections have been achieved (96% vs. 87%). (Alinia package insert) After oral administration, NTZ is rapidly converted at the gut wall and liver by carboxyl esterases and in the plasma by esterases to tizoxanide (desacetylnitazoxanide) (TZ). TZ has an elimination half-life of 1.5 hours. TZ is then glucuronidated to create tizoxanide glucuronide (TZG) primarily, and minor urinary metabolites salicyluric acid and aminonitrothiazole. Food increases absorption of both the tablet and oral suspension. The area under the curve (AUC) of the two active metabolites, TZ and TZG, increased almost 2-fold when administered with food. The Cmax for both metabolites, when administered with food, increased by 50%. Peak times and half-lives increase in proportion to dose and are likely due to saturable transport and elimination mechanisms. 19
TZ is excreted into the urine (33%), bile and feces (67%), with plasma and urinary half-lives of 1 to 1.6 hours and 7.3 hours, respectively. TZG is excreted into the urine and bile and is eliminated at a slower rate than TZ8,20 The pharmacokinetics of TZ and TZG have not been studied in patients with renal or hepatic impairment, geriatric patients, or pediatric patients (<12 years). Specific dosing guidelines for patients with renal and hepatic impairment are unavailable. Prescribers should take into account renal and hepatic function when dosing patients.
Drug interactions could occur due to TZ being highly protein bound (99%). (Alinia package insert) Patients receiving warfarin and nitazoxanide concomitantly should have their INR monitored more frequently to determine if warfarin dose reductions must be made.
Safety/Tolerability
NTZ is a well tolerated agent as demonstrated by initial trials. Thirty-two healthy male volunteers received up to 4 g doses of NTZ with minor adverse effects. Adverse effects were mostly limited to gastrointestinal side effects that increased as dose increased. 19 In addition, sixteen healthy male volunteers were randomly assigned to receive placebo or NTZ 500 mg orally × 1, then twice daily for 7 days or 1 g orally × 1 followed by twice daily for 7 days. TZ and TZG significantly accumulated with 1 g twice daily dosing. One gram doses were also associated with a higher rate of abdominal discomfort and diarrhea. 21
The most common adverse events reported by immunocompetent subjects >12 years receiving tablets in controlled and uncontrolled studies were abdominal pain (6.6%), diarrhea (4.2%), headache (3.1%) and nausea (3%). Pediatric patients receiving oral suspension also reported abdominal pain (7.8%), diarrhea (2.1%), vomiting (1.1%) and headache (1.1%) as the most common adverse events. Adverse event incidence in immunocompetent adult or pediatric patients did not differ significantly from those receiving placebo in placebo-controlled trials. Yellow sclerae due to drug deposition has been reported, but this resolves after drug discontinuation. (Alinia package insert). NTZ has been classified as pregnancy category B.
Reproduction studies performed in rats and rabbits using doses of 3200 mg/kg/day and 100 mg/kg/day, respectively, have not been associated with impaired fertility or fetal harm. There are no trials in pregnant women. Trials have not been conducted to determine if NTZ is secreted into breast milk. (Alinia package insert).
Efficacy
Cryptosporidiosis
Human cryptosporidiosis can be caused by several species of the genus Cryptosporidium, including C. hominis and C. parvum. Most patients experience a self-limited diarrheal illness, however immunosuppressed and pediatric patients can experience a more severe, prolonged illness. Cryptosporidium sp. are transmitted from animals to humans, through contaminated water and via the fecal-oral route. Most infected patients live in developing countries where sanitary conditions are poor. Cryptosporidium sp. are highly resistant to treatment and produce large amounts of hearty oocysts that persist outside the body. 22 Currently, NTZ is the treatment of choice for immunocompetent patients with cryptosporidiosis. Limited data are available for treatment of immunosuppressed patients with cryptosporidiosis. The agents studied include NTZ, paromomycin and azithromycin. 23 Further information regarding the treatment of cryptosporidiosis with NTZ in immunosuppressed patients is outlined below.
Cryptosporidiosis in immunocompetent patients
A prospective, randomized, double-blind, placebo-controlled study was conducted in pediatric and adult patients infected by C. parvum presenting with diarrhea. Fifty immunocompetent patients aged > 12 years were randomized to receive NTZ 500 mg, or a matching placebo, twice daily with food for 3 consecutive days. Forty-nine (total) immunocompetent pediatric patients aged 1 to 3 years received 100 mg NTZ suspension, while those aged 4 to 11 years received 200 mg NTZ suspension, or a matching placebo, twice daily with food for three consecutive days. Eighteen patients, 11 pediatric, and 7 adolescent patients, were positive for another protozoan parasite, including G. lamblia, E. histolytica and B. hominis, at study enrollment and were included in randomization. NTZ treatment resulted in a statistically significant clinical response in the overall patient population compared with placebo (39/49 vs. 20/49, p < 0.0001) and in pediatric patients (21/24 vs. 9/24, p = 0.0004), but not in patients > 12 years old alone. Parasitologic response occurred in all NTZ patients (33/49 vs. 11/50, p < 0.0001), patients aged >12 years (15/25 vs. 6/25, p = 0.0209) and pediatric patients (18/24 vs. 5/25, p = 0.0002). Study results for patients infected only with C. parvum were similar to the overall reported results. 24
A randomized, double-blind, placebo-controlled, modified intent-to-treat study was conducted in 90 Egyptian immunocompetent patients >12 years of age to determine if NTZ reduced Cryptosporidium-associated diarrhea. Patients with diarrhea containing Cryptosporidium oocysts were randomized to one of three treatment arms. Patients received NTZ 500 mg tablet orally twice daily or 500 mg oral suspension or matching placebo tablets twice daily with food. Patients receiving NTZ tablets (27/28, p < 0.0001) and suspension (27/31, p = 0.0003) had statistically significantly higher clinical response rates at day 7 than those receiving placebo (11/27). Statistically significant differences in microbiological response were present when comparing NTZ oral tablet therapy (26/28, p = <0.0001) and NTZ oral suspension therapy (28/31, p < 0.0001) to placebo (10/27). Of the original 90 patients, 40 patients were reevaluated at day 14. Those patients who attained a “well” clinical response at day 7 maintained that response. For those patients who had not attained a well response at day 7, 12 of 12 of patients who had received NTZ tablet therapy and 15 of 17 of those who had received NTZ oral suspension therapy experienced a “well” clinical response compared with 4 of 11 receiving placebo (p < 0.0001 for both groups respectively). 17
Cryptosporidiosis in immunosuppressed patients
Fifty HIV-seropositive and 50 HIV-seronegative pediatric patients infected with C. parvum and experiencing diarrhea were randomized to receive NTZ 100 mg oral suspension or matching placebo twice daily with food for 3 consecutive days. Patients were evaluated on day 7 for clinical response and on days 7 and 8 for parsitologic reponse. In the HIV-seronegative patients, statistically significant differences were detected in clinical response (14/25 vs. 5/22, p = 0.037), parasitological response (13/25 vs. 3/22, p=0.007), and mortality rate (0/25 vs. 4/22, p=0.041). No statistically significant differences were detected in HIV-seropositive patients.
Forty-three patients that did not respond to the initial NTZ course or placebo (19 HIV-seronegative and 24 HIV-seropositive) received 100 mg twice daily for three consecutive days and were evaluated for response. Of the HIV-seronegative patients, 92% who initially received NTZ and 86% who initially received placebo experienced clinical response and 50% and 43% achieved parasitologic response, respectively. Of the HIV-seropositive patients, 77% of patients who initially received NTZ and 55% of patients who initially received placebo experienced clinical response and 25% and 33% experienced parasitologic response, respectively. Many of the patients included in the trials described by Amadi et al. were not only infected with HIV, but were also significantly malnourished secondary to the diarrheal illness. Treatment of this patient population becomes extremely important since the status of these patients can be quite critical. This trial demonstrated a significant decline in mortality with the NTZ use and should be considered for treatment of similar patients. 25
Another randomized, double-blind, placebo-controlled crossover study was conducted in 66 patients infected with HIV and C. parvum. Patients were randomized to three arms, NTZ 500 mg, 1 g, or a matching placebo twice daily for 14 days. Those who initially received placebo were reassigned to receive NTZ after completing the initial placebo course. Statistically significant parasitological cure rates, as evaluated on days 15, 22, and 29, existed between the NTZ groups and placebo (p = 0.016 and p = 0.013, respectively). Parasitological cure rates for each NTZ regimen were similar. Symptomatic resolution occurred in 86% of patients receiving NTZ. 26
Twelve hospitalized patients with stage 4 AIDS and Cryptosporidiosis were given NTZ 500 mg tablets twice daily for 7 consecutive days. Seven of the 12 patients experienced a 95%, or greater, reduction in fecal microbiological load upon examination 7 and 14 days following treatment initiation. Four of the 7 patients with microbiological response also experienced symptom resolution. 27
NTZ was successful in resolving cryptosporidiosis in a 31-year old man with acquired immune deficiency syndrome (AIDS). The patient had previously received paromomycin 2 g daily inhalation therapy to treat pulmonary cryptosporidiosis. One month after pulmonary resolution, the patient presented with cholestasis and cholangiolitis and was given NTZ 500 mg orally twice daily for 12 weeks. Upon NTZ treatment cessation, serum alkaline phosphatase had decreased from 10 times the normal range at treatment initiation, to twice the normal range. The patient redeveloped cholestasis with elevated alkaline phosphatase one year later and was retreated with NTZ causing similar clinical resolution. 28
Three hundred and sixty five patients >/= 3 years old from the United States with AIDS received 500 to 1500 mg of NTZ twice daily for an unpredetermined amount of time as part of a compassionate use program. Of the 365 patients, 357 were included in the intent-to-treat analysis. Fifty-nine percent of patients achieved a sustained clinical response during treatment durations varying from 1 to 1528 days. 29
While significant clinical and parasitologic responses for immunosuppressed human patients have not been achieved in randomized, placebo-controlled trials, animal data suggests NTZ could be efficacious for biliary C. parvum infection treatment. Gerbils were immunosuppressed with dexamethasone and challenged orally with 100,000 C. parvum oocytes. From day 0 to 12, the gerbils were given 200 mg/kg/day NTZ orally, paromomycin or no agent. Compared with the control animals, statistically significant differences in intestinal oocyte shedding at day 12 (p = 0.01) and ileal and gallbladder involvement (p < 0.03). 30
Giardiasis
Giardiasis is an infectious disease caused by Giardia lamblia, a flagellate protozoan. Several pathogenic G. lamblia strains exist. G. lamblia is transmitted via the fecal-oral route and is often transmitted through the ingestion of water and food contaminated with cysts, or through contact with colonized animals or humans. 31 Both epidemic outbreaks and sporadic infections have been known to occur. 32 Highest infection rates occur in pediatric patients <5 years old. Therapies of choice for adult and pediatric giardiasis patients include metronidazole, tinidazole and NTZ. 33
Giardiasis in immunocompetent patients
A prospective randomized, double-blind, placebo-controlled study was conducted in 89 patients 12 to 65 years old experiencing diarrhea and positive for G. intestinalis and/or E. histolytica and/or E. dispar comparing NTZ 500 mg oral tablet or matched placebo twice daily for 3 consecutive days with food. Patients receiving NTZ had an 81% clinical response rate compared with 40% for those receiving placebo (p = 0.0002). Statistically significant differences in clinical response rate were present in subgroup analyses of patients infected with G. intestinalis only and Entamoeba sp. only compared with placebo (p = 0.0237 and 0.0256 respectively). Patients infected with G. intestinalis and E. histolytica or E. dispar did not have a statistically significant difference in clinical response rate compared with placebo (p = 0.2774). Parasitologic response rate for patients with G. intestinalis infection was 71% compared with 0% in the placebo group (p < 0.0001), while response rate for patients infected with either E. histolysitica and/or E. dispar receiving NTZ was 69% compared with 39% in the placebo group (p = 0.0148). 34
Giardiasis in immunosuppressed patients
Abboud et al. describe a case report in which a 31-year old male with AIDS was treated with NTZ after he failed treatment for G. duodenalis infection with metronidazole and albendazole. In April 1997, the patient was diagnosed with giardiasis and was treated unsuccessfully five times with metronidazole and albendazole. In April 1998, the patient received combination therapy with metronidazole and albendazole which was unsuccessful. In October 1998, NTZ 500 mg twice daily for 10 days was initiated, followed by 1 g twice daily for 15 days. The patient did not experience clinical improvement although TZ and TZG blood levels (3 and 7 mcg/mL on day 8 and 3 and 10 mcg/mL on day 15, respectively) were consistent with those found in healthy patients receiving the same dose. In January 1999, NTZ therapy was reinitiated at 1.5 g twice daily for 30 days, which resulted in symptomatic and microbiologic resolution in February 1999 and at follow up in April 1999. 35
Comparative trials for giardiasis treatment
A randomized active-control clinical trial was conducted comparing NTZ to metronidazole for the treatment of symptomatic giardiasis in 110 pediatric patients from northern Peru. Patients 2 to 3 years old in the NTZ treatment arm received 100 mg oral suspension twice daily for three consecutive days, while those 4 to 11 years old received 200 mg oral suspension twice daily for three days. Patients randomized to the metronidazole treatment arm and 2 to 5 years old received 125 mg oral suspension twice daily for five days, while those 6 to 11 years old received 250 mg oral suspension twice daily for five days. Using an intention-to-treat analysis, 85% of patients receiving NTZ and 80% of patients receiving metronidazole attained a “well” clinical response before the day 7 follow up examination, a nonstatistically significant difference. Upon microbiological analysis, 71% of NTZ and 75% of metronidazole patients were negative for G. intestinalis cysts in either of two stool samples collected 7 to 10 days following treatment initiation, a nonstatistically significant difference. 36
An open, randomized trial was conducted in 166 pediatric patients to compare the efficacy and safety of NTZ 7.5 mg/kg twice daily for 3 days and one dose of tinidazole 50 mg/kg for G. lamblia treatment. Of the 137 pediatric patients who completed the study, the parasitological cure rate was statistically significantly higher with single dose tinidazole therapy compared with multidose NTZ (90.5% and 78.4%, p = 0.05). 37
In vitro metronidazole, NTZ and TZ activities were assessed for several G. intestinalis, E. histolytica and T. vaginalis isolates. TZ was 8 times more active than metronidazole in metronidazole sensitive G. intestinalis isolates and twice as active in the metronidazole-resistant G. intestinalis isolate. In 10 E. histolytica isolates, TZ was twice as active as metronidazole. In 14 T. vaginalis isolates, TZ was 1.5 times as active compared with metronidazole. 12
Amoebiasis
Amoebiasis occurs after inactive pathogenic Entamoeba histolytica cysts are ingested. After cyst ingestion, the trophozoite is released and colonizes the intestinal tract. Infections can lead to amoebic dysentery, liver abscesses, or more serious manifestations. Not all E. histolytica strains are pathogenic. In fact, 90% of E. histolytica related infections are asymptomatic. Areas without appropriate sanitation and those with low socioeconomic status have a higher prevalence of pathogenic infection. Infection risk is inversely related to age. 38 First line therapies for symptomatic Entamoeba histolytica infections are metronidazole and tinidazole. 33
A randomized, double-blind, placebo-controlled trial was conducted in 64 Egyptian outpatients with intestinal or hepatic amoebiasis secondary to Entamoeba histolytica. Patients aged >/= 12 years old received NTZ 500 mg, patients aged 4 to 11 years old received 200 mg and patients 1 to 3 years old received 100 mg, or matched placebo, twice daily for three days. Statistically significant differences existed in symptom resolution on day 7 and microbiologic resolution, based on two post-treatment stool samples between those patients receiving NTZ and placebo affected by intestinal amoebiasis (94% vs. 50%, p < 0.001 and 94% vs. 43%, p < 0.0001, respectively). Seventeen adult patients hospitalized for hepatic amoebiasis were not randomized, and instead were given NTZ 500 mg twice daily for 10 days. All patients with hepatic amoebiasis responded to NTZ therapy. 29
Blastocystosis
Blastocystosis is caused by one of nine related protozoon, although human infection was originally thought to be due to one species, Blastocystis hominis. These species possess morphological variation and exist as one of four forms: vacuolar, granular, amoeboid or cystic. Cultures from those individuals presenting with gastrointestinal illness are positive for the amoeboid form. It has been theorized that infections are due to the amoeboid form collecting on the host's intestinal wall since the amoeboid form is highly adhesive. 39 The mode of transmission is unknown, but is suspected to be fecal-oral, or through ingestion of contaminated water. Because so much of the pathogenic mechanism for B. hominis is unknown, some controversy still surrounds its classification as a pathogen.40–42 Accepted therapies for blastocystosis include metronidazole, iodoquinol, trimethoprim-sulfamethoxazole, and NTZ.43,44 Some B. hominis strains have demonstrated resistance to metronidazole, necessitating investigation of other agents. 45
Two double-blind, placebo-controlled studies were carried out in 84 patients to measure NTZ efficacy in Blastocystis hominis treatment. Patients >/= 12 years old received NTZ 500 mg tablet, 4 to 11 years old 200 mg NTZ suspension, and <4 years old 100 mg NTZ suspension or a matching placebo twice daily for three consecutive days. On day 7, patients were evaluated for clinical response and stool samples were collected between days 7-10 for microbiologic evaluation. Response rates were published as pooled results, limited data were published for individual age groups. Researchers stated that response rate was similar for all age groups. Overall clinical response rate was 86% for patients receiving NTZ compared with 38% for those receiving placebo (p < 0.0001). Overall microbiologic response rate was 86% for patients receiving NTZ compared with 12% for those receiving placebo (p < 0.0001). 46
Cyclosporiasis
Cyclosporiasis is an infection caused by Cyclospora cayetanensis. Infection results in watery diarrhea, gastrointestinal distress and myalgias. The organism is transmitted via the fecal-oral route or by ingesting contaminated water. Outbreaks in the United States from 1995-2000 were attributed to contaminated raspberries imported from Guatemala.47–49
First-line therapy for C. cayetanensis treatment is trimethoprim-sulfamethoxazole. In a documented case report, NTZ therapy was administered to a sulfa-allergic patient infected with C. cayetanensis who failed ciprofloxacin therapy (dose not published). After receiving 7 days of NTZ therapy (dose not published), the 40 year-old woman experienced symptomatic improvement and parasitologic resolution. 50
Enterocytozoon bieneusi
E. bieneusi is a microsporidial pathogen that causes opportunistic infections resulting in severe diarrheal illness in immunocompromised patients, particularly HIV-positive patients. Currently, there is no preferred or established treatment for infected patients. Albendazole has been shown to be partially effective in treatment, but more effective agents must be found.
A published case report describes a 37 year old HIV and hepatitis C positive male infected with E. bieneusi, an opportunistic infection resulting in malabsorption and persistent diarrhea. The patient's antiretroviral therapy was discontinued and he was started on albendazole 1200 mg per day for 15 consecutive days. Clinical improvement did not occur with albendazole therapy, and he was subsequently started on NTZ 1 g twice daily for 60 consecutive days. By the end of the 60 day treatment, the patient was having normal bowel movements and his stool was negative for E. bieneusi spores on days 11, 19, and 60 of treatment. Three posttreatment microbiological examinations over the following two months continued to be negative. 51
Helminth infections
Ascaris lumbricoides is the most frequently soil-transmitted helminth in Mexico and accounts for 42% of all helminth infections in Colima, Mexico. Infection occurs most often in areas with poor sanitation, where human fecal material is used as fertilizer, and is associated with malnutrition and iron deficiency anemia. 52 Preferred therapies use albendazole, mebendazole and ivermectin. 33
Eighty pediatric patients and 200 patients >12 years old diagnosed with parasitic infections, including A. lumbricoides, were given NTZ 200 mg oral suspension or 500 mg oral tablet twice daily with food for three consecutive days. Many patients were infected with multiple parasites including E. histolytica, G. lamblia and B. hominis. NTZ resulted in the microbiological resolution of 88% of Ascariasis cases overall. Patients also experienced clinical symptom resolution 81%-100% of the time. 53
Comparative trials for helminth infections
Three randomized clinical trials were conducted in children from Peru to compare the safety and efficacy of NTZ to albendazole for the treatment of A. lumbricoides and T. trichiura infections and NTZ to praziquantel in the treatment of H. nana infection. Several patients had secondary concomitant infections. Patients were randomized to treatment based on the predominating organism egg count. Overall, 210 patients were randomized to receive standard approved single dose regimens of aldendazole and praziquantel as appropriate (200 mg and 25 mg/kg, respectively) or NTZ oral suspension. Patients 2 to 3 years of age received NTZ 100 mg and patients aged 4 to 11 years of age received NTZ 200 mg. NTZ was given twice daily for three days. It should be noted that 23% of the patients randomized were below normal weight or height, anorexic or malnourished. A patient was considered “cured” if they no longer excreted eggs. Similar “cure” rates were present between the standard agents and NTZ for A. lumbricoides and H. nana infections, but not in T. trichiura infections. In these patients, 89% of patients receiving NTZ were cured compared to 58% of patients receiving aldendazole (p = 0.022). 54
Mixed parasitic infections in immunocompetent patients
Two hundred and forty six adult and pediatric patients from Mexico with mixed parasitic infections were given NTZ to determine if NTZ was active against infections with multiple causative organisms. Adult patients received NTZ 500 mg every 12 hours for three consecutive days while pediatric patients received NTZ oral suspension 200 mg every 12 hours for three consecutive days. Stool samples were examined on days 7, 8, 13, 14, and 15 after treatment initiation. NTZ displayed variable efficacy (71%-100%) against mixed infections, demonstrating its broad-spectrum activity. 55
Mixed parasitic infections in immunosuppressed patients
Two hundred and seven adult Zambian patients with AIDS and diarrhea for one month or longer while infected with unspecified parasites were randomized to received NTZ 1 g or matched placebo twice daily for two weeks. A statistically significant difference in clinical response was demonstrated favoring NTZ treatment (75% vs. 58% p = 0.03). This difference was greater in patients with CD4 counts less that 50 cells/microliter (p = 0.007). NTZ treatment did not translate into better parasitologic clearance or reduced mortality. 56
Comparative trials for mixed parasitic infections
A double-blind, placebo-controlled study was carried out in 275 pediatric patients from Mexico with helminthic or intestinal protozoan infections to compare the effectiveness of NTZ, quinfamide, mebendazole, or both quinfamide and mebenda-zole. Patients were randomized to receive NTZ 200 mg for three days, one dose of quinfamide 100 mg, mebendazole 200 mg for three days, or both quinfamide and mebendazole. Eradication rates were determined on day 14 after treatment initiation. Statistically significant differences in eradication rates did not exist between the groups. The study did not report whether there were differences in symptom improvement between the treatment groups. 57
Taenia saginata (beef tapeworm)
Taenia sagniata are hermaphroditic tapeworms that can live as adults in the human gastrointestinal tract. Infection is more common in areas where undercooked beef is consumed. After the larval cysts are consumed, protoscolices are released and attach to the intestinal wall. Each protoscloex can give rise to an adult tapeworm. Current treatments of choice are praziquantel and niclosamide. Resistance is developing to these agents and other options must be explored.
Researchers evaluated 52 patients with treatment resistant T. saginata, with or without additional parasites, for clinical resolution with NTZ therapy. Enrolled patients failed multiple courses of traditional therapy with niclosamide, praziquantel or both. Patients >14 years of age received 500 mg NTZ twice daily for three consecutive days while those < 14 years of age received 20 mg/kg/day. Fifty-one of 52 patients experienced clinical response to NTZ therapy. Of those who provided stool specimens, 92.7% were negative for eggs and proglottides. Patients received follow up examinations at 2, 4, 8, and 12 weeks. Upon follow up, 98.1% were negative for eggs and proglottides. 58
Fascioliasis
Fasiciolosis is caused by two trematodes, Fasciola hepatica and Fasciola gigantica. Both humans and animals, particularly sheep and cattle, can be infected and transmit the flukes. Infections are most common in developing countries, although France has an especially high infection rate. Transmission typically occurs when humans ingest watercress grown in sheep-raising areas or contaminated water lettuce, mint, alfalfa, parsley or other raw freshwater greens. F. hepatica often locates to the liver after infection and colonizes the biliary ducts causing fibrosis of the proximal biliary tree.59–61 Drugs of choice for treatment of fasiciolosis caused by F. hepatica include triclabendazole, bithionol or NTZ. 33
In a randomized, placebo-controlled study, 38 adult and 43 pediatric Peruvian patients were evaluated to determine the relative safety and efficacy of NTZ for the treatment of human fascioliasis. Patients > 12 years received NTZ 500 mg tablet, patients 4-11 years received NTZ 200 mg oral suspension, and patients <4 years received NTZ 100 mg oral suspension, or a matching placebo, twice daily for 7 consecutive days. Patients returned on the seventh day for clinical symptom evaluation. Two stool samples were obtained 24 hours apart, 30 to 60 days after study enrollment, to determine if microbiologic resolution occurred. Of the adult patients who received NTZ, 60% experienced clinical and micro-biologic resolution compared with 13% of patients receiving placebo (p = 0.0422). Of the pediatric patients who received NTZ, 40% experienced clinical and microbiologic resolution compared with 0% of patients receiving placebo (p = 0.0387). There was no statistically significant difference in the rate or type of adverse events experienced by the adult or pediatric patients. One patient receiving NTZ had to discontinue therapy secondary to a rash that developed 5 hours after her first dose. 62
Helicobacter pylori
Helicobacter pylori is a gram-negative, microaerophilic, flagellate bacterium present in the stomach and duodenum of humans. While approximately 80% of carriers are asymptomatic, others develop stomach inflammation, ulceration and cancer. H. pylori disrupts the stomach mucosa resulting in exposure of the stomach lining to acidic secretions. Current preferred treatment involves the use of multiple antibiotics with a proton-pump inhibitor (PPI). While this therapy is effective, treatment resistance is an issue warranting the investigation of other agents. 63
H. pylori strains isolated from patients with duodenal ulcer and nonulcer dyspepsia were subject to in vitro sensitivity analysis to NTZ and TZ. Some strains demonstrated metronidazole resistance. NTZ and TZ had similar minimum inhibitory concentrations (MICs) and those MICs were consistent for all strains, including those resistant to metronidazole (MIC50s = 1, 1, 2 mcg/mL and MIC90s = 4, 4, >32 mcg/mL respectively). This data suggested that NTZ might be an effective agent for H. pylori treatment. 64
Twenty dyspeptic patients positive for H. pylori infection who previously failed at least one course of eradication therapy were given NTZ 1 g twice daily for 10 days. Patients did not receive histamine-2 receptor antagonists (H2RA) or PPIs during NTZ therapy. NTZ monotherapy was unable to achieve clinical cure in any patient. Pharmacokinetic studies performed on biopsied gastric mucosal tissue did not show adequate TZ levels. Patients reported numerous side effects, including diarrhea, nausea, and abdominal pain, at a higher rate than those normally reported with PPI-based triple therapy. 14
Clostridium difficile
Clostridium difficile is an anerobic gram-positive rod infecting both immunocompetent and immunosuppressed hosts. C. difficile most often infects patients who have experienced disruption of the normal flora of the gastrointestinal tract usually due to prior antibiotic use. Transmission occurs via the fecal-oral route upon ingestion of spores. Spore activation and microbial growth result in toxin production leading to colitis. Metronidazole and oral vancomycin are mainstays of therapy, but a percentage of patients continue to relapse after receiving multiple treatment courses. Alternatives to metronidazole and oral vancomycin must be explored for the treatment of relapses and recurrences. 5
Comparative trials in C. difficile treatment
Recent literature has explored the role of NTZ in primary C. difficile colitis treatment. In a double-dummy, noninferiority study, 44 adult patients were randomized to receive metronidazole 250 mg every six hours for 10 days, while 49 patients received NTZ 500 mg every 12 hours for 7 days and 49 patients received NTZ 500 mg every 12 hours for 10 days. Patients with signs of toxic megacolon were excluded from the study. A statistically significant difference was not present between the three arms when measuring time to symptom resolution (p = 0.2). Statistically significant differences in clinical response (82.4%, 90% and 88.9% respectively) were not present. Recurrence, mortality, and adverse event rates for each arm were also similar. 65
Forty-nine adult patients were evaluated in a prospective, double-blind, randomized, double-dummy, controlled trial comparing vancomycin and NTZ for the treatment of C. difficile infection. Patients who experienced more than one occurrence of C. difficile infection during the six months preceding enrollment were excluded from the trial. Patients were randomized to receive NTZ 500 mg orally every 12 hours or vancomycin 125 mg orally every 6 hours for 10 days. Patients characteristics were analyzed based on a previously published severity scale to determine whether their illness was severe or not severe. End-of-treatment response (resolution of all signs and symptoms attributable to C. difficile infection), relapse rate and sustained response rate were evaluated for all patients. Statistically significant differences were not present amongst the treatment or severity groups. Although the trial was not sufficiently powered to demonstrate noninferiority, this trial suggests NTZ as an alternative treatment to vancomycin to treat C. difficile infection. 66
NTZ for treatment resistant C. difficile infection
Thirty-five patients who had failed metronidazole for C. difficile colitis were given NTZ 500 mg twice daily for 10 days. Twenty eight of these patients had failed one >/= 14 day metronidazole course while the other 7 had initially responded to metronidazole treatment, then experienced >/= 2 recurrences. Of the 28 patients initially failing metronidazole, 14 were cured while 6 relapsed and 8 failed NTZ treatment. Of the 7 patients with multiple recurrences, 5 were cured, while one relapsed and another failed NTZ treatment. 67
Rotavirus
Rotavirus is a double-stranded RNA virus. It is the leading cause of severe diarrhea among infants and young children worldwide. Rotavirus is transmitted via the fecal-oral route. Patients can be infected more than once, but each subsequent infection is less severe. Rotavirus primarily affects pediatric patients, particularly those 6 months to 2 years of age, and accounts for approximately 50% of all pediatric hospitalizations. A vaccine is currently available which could significantly decrease infection rate.68–71 Current treatment is restricted to supportive care. No antiviral agents are currently approved for rotavirus treatment.
After demonstrating in vitro activity of TZ against rotavirus, researchers performed a randomized, double-blind, placebo-controlled clinical trial evaluating 38 pediatric, hospitalized patients with severe rotavirus gastroenteritis. Patients 4-11 years received 200 mg NTZ oral suspension twice daily or matched placebo for three consecutive days, while patients <4 years received 100 mg NTZ suspension or matched placebo for three consecutive days. NTZ treatment significantly reduced gastrointestinal symptom duration with a median time from first dose to symptom resolution of 31 hours for NTZ patients compared with 75 hours for those receiving placebo (p = 0.0137). Subgroup analyses were performed since 36 of the 38 patients were < 24 months old. A statistically significant difference comparing median time from first dose to symptom resolution was present in patients <24 months old receiving NTZ compared with those receiving placebo (p = 0.0251). Virologic resolution was not measured in this trial. 17 Based on the above data, NTZ is an option to decrease time to symptom resolution, although a cost-benefit analysis must be performed since rotavirus gastroenteritis is a short-lived infection. The decision to treat should also be based on geographic location of patients. Patients in developing countries and areas with poor sanitation are at increased risk. According to the World Health Organization, 527,000 deaths in 2004 of children <5 years of age were attributed to rotavirus infections.
Fifty outpatients > 12 years old presenting with diarrhea secondary to adenovirus, norovirus or rotavirus were randomized to receive NTZ 500 mg or a matched placebo twice daily for three consecutive days. The median time to symptom resolution was 1.5 days for those receiving NTZ and 2.5 days for those receiving placebo (p < 0.0001) without regard to specific pathogen. Statistically significant differences in symptom resolution favoring NTZ treatment were present in those infected with rotavirus and norovirus (p = 0.0052 and p = 0.0295, respectively), but not in those individuals infected with adenovirus. 72
The comparative efficacy of NTZ and probiotics were evaluated for acute rotavirus infection treatment in a randomized, single-blind controlled trial in Bolivian children. Seventy-five patients 28 days to 24 months old with rotavirus diarrhea were randomized to treatment with oral NTZ (15 mg/kg/day), a combination of oral probiotics, or supportive care with rehydration solutions. The primary outcome of mean hospitalization duration was significantly reduced in patients who received NTZ or probiotics compared with rehydration therapy alone (81 h, 72 h and 108 h, respectively; p = 0.017). Patients receiving NTZ or probiotics also had significant reductions in diarrhea duration compared with rehydration therapy alone (54 h, 48 h, and 79 h, respectively; p = 0.009). While significant differences were not present when comparing the NTZ or probiotic therapy to placebo treatment groups, both agents were effective in decreasing length of stay and diarrhea duration. Cost-benefit analyses should be performed when deciding to treat acute rotavirus infections, especially when using antimicrobial agents. 73
Conclusion
NTZ has broad-spectrum activity against a number of parasites, bacteria, and even viruses. Its in vitro activity against several protozoa, helminths, C. difficile, and H. pylori should encourage researchers to perform additional trials to further evaluate its efficacy. It is the drug of choice for cryptosporidiosis and giardiasis in immunocompetent patients. Recent trials suggest that NTZ is noninferior to metronidazole and vancomycin for the treatment of C. difficile infection without colitis. NTZ has also shown promise in decreasing illness duration in patients with rotavirus infection. Limited data exists regarding its use in immunosuppressed patients and there is no literature evaluating its use for diarrheal illness prophylaxis. Further trials must be conducted in this patient population, perhaps with longer treatment durations due to the increased illness complexity, to determine if NTZ is efficacious. Currently, several trials are being conducted to evaluate if NTZ has a role in hepatitis C or Crohn's disease treatment and to further elucidate its role in the treatment of immunosuppressed patients with cryptosporidiosis. Other trials being conducted are further evaluating NTZ role in C. difficile, rotavirus, and amebiasis treatment.
Disclosure
The authors report no conflicts of interest.
