Abstract
Several commercial Giardia immunoassays were evaluated in baboons for sensitivity and specificity as well as ease of use in a large specific pathogen–free (SPF) colony. An additional objective was to identify the assemblage(s) of Giardia duodenalis present in this baboon colony. A direct immunofluorescent antibody test (IFAT) was used as the reference test. Tests evaluated were a patient-side rapid test for dogs and cats, a human rapid test, and a well-plate ELISA designed for use with humans. Test sensitivities and specificities were compared using the McNemar paired t-test and were further evaluated for agreement using an unweighted Cohen kappa statistic. When compared to the IFAT reference, both human tests were more sensitive than the veterinary test. Based on PCR and sequencing of the G. duodenalis small-subunit ribosomal RNA and glutamate dehydrogenase loci, assemblage AI was present in this baboon colony. We found that 10 of the 110 (9%) baboons in this SPF colony were infected with a zoonotic strain of G. duodenalis.
Giardia duodenalis is an enteric protozoan parasite with a broad host range that includes humans and nonhuman primates (NHPs). The parasite is commonly found in NHPs, where it is of both veterinary and public health importance.8,12 G. duodenalis infections in NHPs can result in diarrhea and ill thrift, particularly in young animals. 6 The prevalence of G. duodenalis infection in NHPs varies greatly depending on species surveyed, geographic location, and type of housing (free range, zoo, or laboratory animals), with estimates of 2.2–57%.10,11,19 In captive baboons, prevalences of 10% were reported from Belgium and 17% from China.12,14 In wild baboons, prevalences of 26% in Saudi Arabia 10 and as high as of 58% in Uganda 11 have been reported.
G. duodenalis has been divided into 8 assemblages (A–H) that are genetically distinct but morphologically identical.4,8 Assemblages A and B infect both humans and a wide range of mammals; assemblages C and D are typically found in canids, E in cloven-hoofed livestock, F in felids, G in rodents, and H in marine mammals.4,8 In addition, there are subtypes within assemblages A and B, although there is not complete agreement among researchers as to how many subtypes exist. Assemblage A currently consists of subtypes AI–AIV, with human isolates belonging to AI and AII, and animal isolates belonging to AI, AIII, and AIV. 18 Assemblage B is considered to have many subtypes that do not form genetically authentic sub-assemblages. 18 Both assemblages A and B have been reported in NHPs, with assemblage B being the predominant assemblage reported.12,24 Specifically in olive baboons (Papio cynocephalus anubis), only assemblage B has been reported in the literature. 16 Therefore, NHPs are capable of serving as reservoirs for zoonotic transmission of G. duodenalis.
Detection of G. duodenalis infections in NHPs by standard morphologic techniques is difficult. Cysts are shed intermittently in feces, and are difficult to recognize without training. In nonhuman primates, endoparasites are often diagnosed using preserved feces and variations of ether sedimentation techniques, direct smears, or fecal flotations.10,11 If researchers are specifically concerned with finding G. duodenalis, an immunofluorescent antibody test (IFAT) is sometimes used, 19 but such tests require specialized equipment and training. There are rapid tests available to detect Giardia in humans, dogs, and cats. However, there are no rapid detection tests that are designed and/or optimized for use in NHPs.
Previously, G. duodenalis cysts were detected on routine centrifugal zinc sulfate fecal flotations in our specific pathogen–free (SPF) baboon colony. Fecal samples were then screened with a commercial rapid veterinary fecal antigen test (VFAT; SNAP Giardia antigen test, IDEXX Laboratories, Westbrook, ME) validated for use in dogs and cats. There were discrepant results between the fecal flotations and the VFAT, which prompted us to seek a reliable immunoassay that could be used for screening NHPs, and led to the consideration of human detection tests for use in NHPs. We evaluated the VFAT for use in dogs and cats and 2 tests for humans—a rapid test (RT; Giardia/Cryptosporidium Quik Chek, TECHLAB, Blacksburg, VA) and a well-plate ELISA (WPE; Giardia II ELISA, TECHLAB)—and compared each immunoassay to a direct IFAT (Merifluor Cryptosporidium/Giardia, Meridian Bioscience, Cincinnati, OH), which is widely recognized as the most sensitive and specific procedure for detection of G. duodenalis.2,9
All housing and husbandry procedures for the baboon colony were in compliance with the Guide for the Care and Use of Laboratory Animals 7 and the Association for Assessment and Accreditation of Laboratory Animal Care International. The University of Oklahoma Health Sciences Center Institutional Animal Care and Use Committee approved protocols for the maintenance of the baboon colony. The baboon colony was derived and maintained using procedures outlined previously. 23 Baboons were fed a high-protein monkey diet (5045, LabDiet, St. Louis, MO). Their diet was enriched with a forage mix of grains on a daily basis, and they were fed a mix of fruits and vegetables several times per week.
Animal handlers collected fecal samples from 110 clinically normal baboons in the SPF colony within a few minutes after each baboon was observed defecating. Fecal samples were placed in coolers with ice packs during transportation and were stored at 4°C until tested. ELISAs were performed on each sample within 1 wk of collection. At the time of testing, a portion of each sample was fixed in 10% phosphate-buffered formalin for the IFAT, which was performed at a later date.
The WPE is an enzyme immunoassay for the qualitative detection of Giardia lamblia (duodenalis) cyst antigen, and the RT is a membrane enzyme immunoassay for the simultaneous qualitative detection of Giardia cyst antigen and Cryptosporidium oocyst antigen in a single test device; both tests are intended for use with human fecal specimens. 15 The VFAT is a rapid patient-side enzyme immunoassay for the detection of G. duodenalis antigen in canine and feline feces. 5 We used the direct IFAT as our reference test to identify G. duodenalis cysts in feces. Samples were run in batches, with positive and negative controls each time the test was run. We performed all tests following the manufacturers’ instructions.
To determine the assemblage(s) of G. duodenalis present, a fragment of the small-subunit ribosomal (r)RNA and glutamate dehydrogenase genes were amplified from a representative cyst-positive sample using primers and protocols previously described in the literature.3,16 We isolated cysts for DNA extraction from a cyst-positive sample using sugar density gradient separation as described previously, 21 and we extracted DNA from the cysts (DNeasy tissue kit, Qiagen, Valencia, CA). Reagents from the kit were used, but with modification as outlined in the literature. 20 Samples positive by PCR underwent gel extraction (QIAquick gel extraction kit, Qiagen) following the manufacturer’s instructions. The resulting product was sequenced at the Biocomplexity Institute of Virginia Tech (Blacksburg, VA) utilizing the same PCR primers. The sample was sequenced in both directions and analyzed using a sequence alignment editor.
We calculated the prevalence of G. duodenalis infection along with sensitivity, specificity, and predictive values for each immunologic test by comparison to the IFAT, which was used as the reference standard.9,17 The McNemar test for significance of changes was used to compare differences in the sensitivities and specificities between the immunologic assays. 1 We analyzed agreement between immunoassays using an unweighted Cohen kappa statistic, 22 with common interpretations of κ from the literature. 13
The prevalence of G. duodenalis infection as determined by the IFAT was 9% (10 of 110). The test with the highest sensitivity when compared to the reference IFAT was the WPE (90%), followed by the RT (80%) and the VFAT (50%; Table 1). The VFAT had the highest specificity (99%) when compared to the reference IFAT followed by the human WPE (97%) and the human RT (95%). The negative predictive value (NPV) for each test was >95% for the immunoassays; none of the tests demonstrated a positive predictive value (PPV) >84%. There were some discrepant results among the samples. Of the 10 samples that were positive by the reference IFAT, 5 were positive on all other immunoassays, 3 were only positive on the human WPE and the human RT, 1 was only positive on the human WPE, and 1 was negative on all other immunoassays (Table 2). Of the 100 samples that were negative on the reference IFAT, 94 were negative on all other immunoassays, 3 were positive on the human WPE and the human RT, 2 were positive on just the human RT, and 1 was positive on the VFAT (Table 2). The only comparison of the 3 tests that showed a statistically significant difference in sensitivities was between the human WPE and the VFAT. There was no statistically significant difference in specificities between any of the test comparisons. Agreement between immunoassays ranged from moderate to nearly perfect agreement (Table 3). There was nearly perfect agreement between the human WPE and human RT. There was also substantial agreement between the IFAT and the human RT, and between the human WPE and the IFAT. Agreement between the other immunoassays was only moderate: VFAT and IFAT, human WPE and VFAT, and VFAT and human RT (Table 3).
Performance of immunoassays when compared to the direct immunofluorescent antibody test (IFAT) as the reference test.
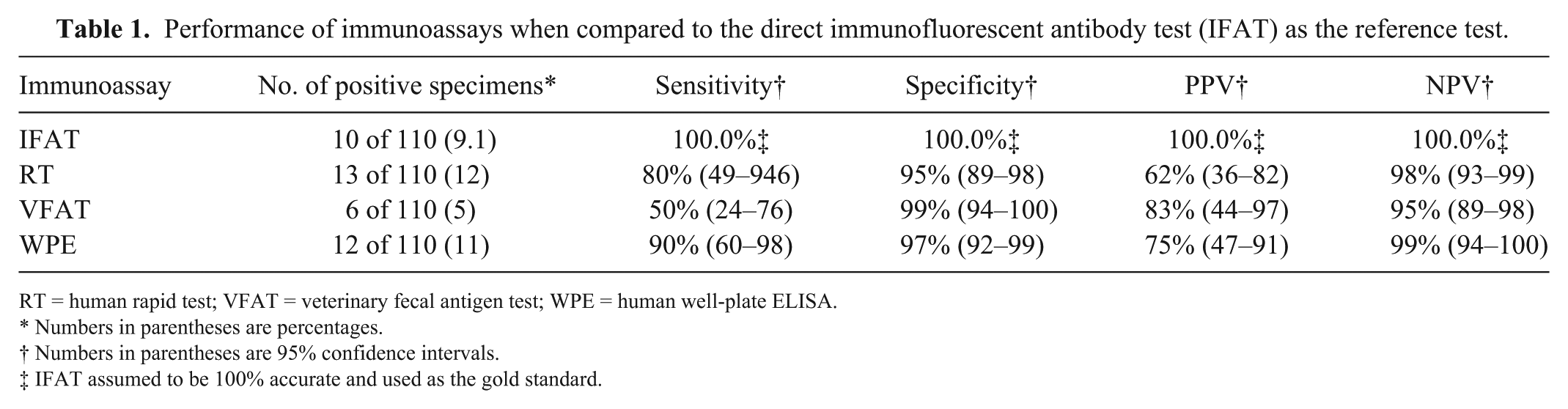
RT = human rapid test; VFAT = veterinary fecal antigen test; WPE = human well-plate ELISA.
Numbers in parentheses are percentages.
Numbers in parentheses are 95% confidence intervals.
IFAT assumed to be 100% accurate and used as the gold standard.
Discrepant results among direct immunofluorescent antibody test (IFAT)-positive and IFAT-negative samples.

RT = human rapid test; VFAT = veterinary fecal antigen test; WPE = human well-plate ELISA.
Agreement between tests as determined by an unweighted Cohen kappa statistic.
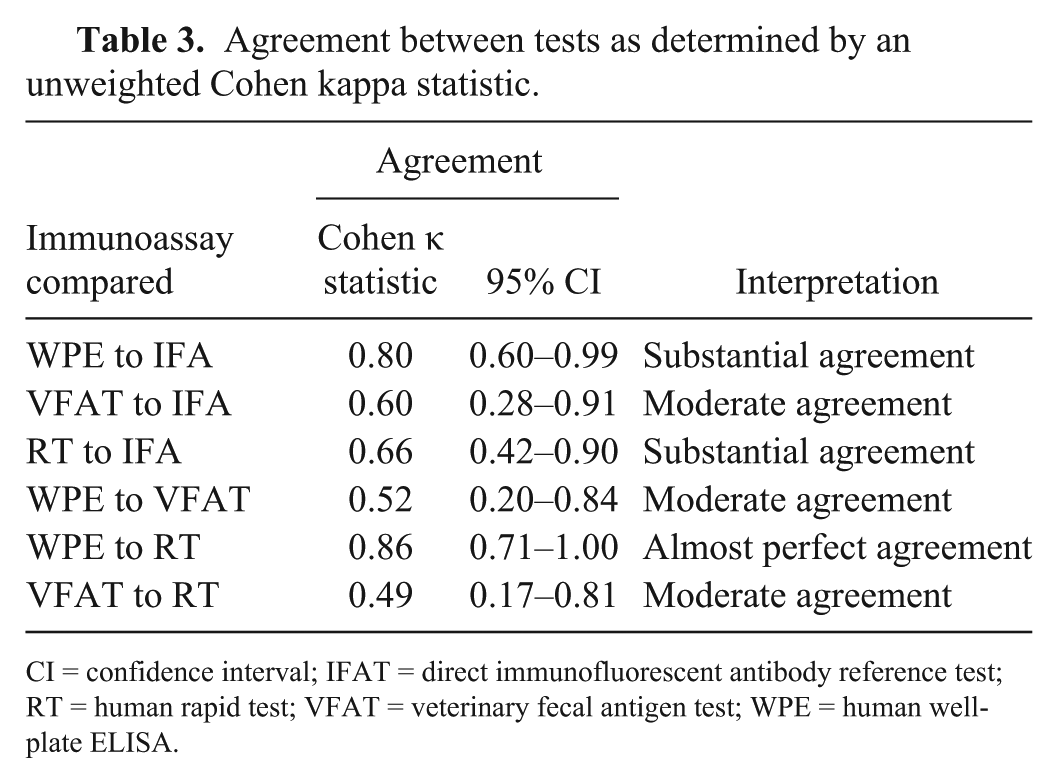
CI = confidence interval; IFAT = direct immunofluorescent antibody reference test; RT = human rapid test; VFAT = veterinary fecal antigen test; WPE = human well-plate ELISA.
We sequenced the parasite DNA to assemblage A at the small-subunit rRNA locus and further subtyped to assemblage AI at the glutamate dehydrogenase locus. The isolate had 100% nucleotide sequence identity to the assemblage AI sequence referenced in the literature (GenBank accession DQ414242).
The prevalence of G. duodenalis infection found in this SPF baboon colony (9%; based on the reference IFAT) is similar to previous reports in the literature. 12 Although infection with G. duodenalis can cause diarrhea and ill thrift, 6 and some fecal samples in our study were soft or diarrheic, the baboons in the colony were considered healthy by husbandry staff and caretakers. However, even when all animals are clinically normal, reliable detection of Giardia is important in establishing and maintaining SPF status of the colony.
The IFAT is widely recognized as the most sensitive and specific procedure for G. duodenalis detection. 9 However, it is time-consuming, requires specialized equipment, and, in large colonies where rapid and simple screening procedures are desired, use of an antigen test may be appropriate even though it may lack the sensitivity and specificity of the IFAT. Because some of the immunoassays only detect cyst antigen and not the presence of cysts, it is possible that some of these discrepant results (Table 2) were actually true positives. The discrepant results seen in our study highlight the difficulty in determining which test is most accurate in the absence of a true gold standard for the detection of G. duodenalis.
Based on our results, it appears that the human WPE could be used as a more convenient and yet still reliable test for detection of Giardia in baboons than the IFAT. This conclusion is based on its high sensitivity and specificity when compared to the reference test as well as its substantial agreement with the reference IFAT. Although the sensitivity and specificity of the human RT were lower, this test also had substantial agreement with the IFAT and nearly perfect agreement with the human WPE, making the human RT also suitable for the detection of Giardia in baboons. Selecting which test to use depends on the purpose of testing, either general screening of an entire colony or testing of individual baboons in which Giardia infection is suspected. For large-scale screening, the human WPE might be preferred given that it allows for testing multiple samples at the same time. If only a few baboons need to be tested, then the human RT could be a useful patient-side test. Not surprisingly, the results of this comparison also illustrate that the immunoassays developed for humans (RT and WPE) performed better in NHP testing than the VFAT. The human immunoassays also had higher sensitivity and better agreement with the IFAT than the VFAT.
To our knowledge, assemblage AI Giardia has not been previously reported in olive baboons. Assemblage B has been reported in olive baboons, and is the more common assemblage reported in NHPs in the literature. 12 Our finding of assemblage AI Giardia provides additional evidence that baboons can serve as reservoirs of zoonotic G. duodenalis assemblages.
Footnotes
Declaration of conflicting interests
One of the authors (AZ) has received research support or honorarium from TECHLAB Inc. (Blacksburg, VA) and IDEXX Laboratories (Westbrook, ME).
Funding
This study was supported by NIH/OD P40 OD010431 (RW) and TECHLAB Inc. (Blacksburg, VA).
