Abstract
Several clinical studies demonstrate a beneficial role of angiotensin-converting enzyme (ACE) inhibitors in patients with myocardial infarction, hypertension and diabetes mellitus. This review focuses on the effects of ramipril, a weak inhibitor of ACE that is rapidly hydrolyzed to ramiprilat, an active metabolite. The Heart Outcome Prevention Evaluation (HOPE) study evaluated the effects of ramipril in patients with a high risk for cardiovascular events without pre-existing left ventricular dysfunction or heart failure. In this review, we summarized the effects of ramipril on myocardial infarction, death, diabetes mellitus, and stroke.
Introduction
Coronary artery disease is the leading cause of death, morbidity and mortality in developed countries. In many instances, activation of the renin-angiotensin-aldo sterone axis increases the risk of cardiovascular events 1 and inhibition of this system by angiotensin-converting enzyme inhibitors (ACEIs) lowers cardiovascular diseases. The benefits of ACEIs have been well established in several disorders including hypertension,2,3 myocardial infarction,4–5 congestive heart failure,2,5,7 diabetes mellitus3,8 and renal disease 9 (Fig. 1).
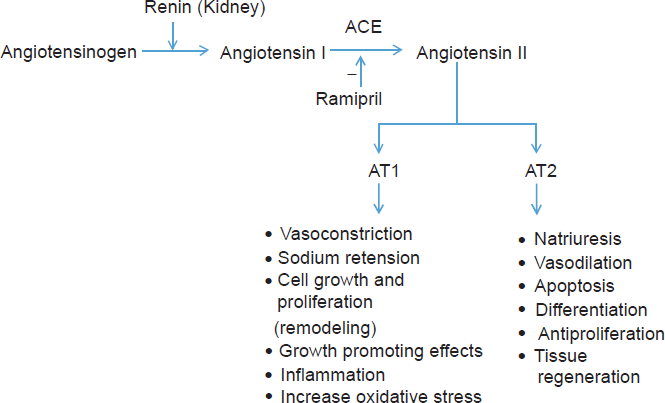
Renin-angiotensin system and the roles of angiotensin II.
Using ACEIs to treat patients having a low ejection fraction (with or without myocardial infarction/heart failure) significantly reduces the risk of ischemic events.10,11 Related to this are other studies showing that ACEIs reduce the risk of cardiovascular and ischemic events in patients without left ventricular dysfunction. 12 The Heart Outcome Prevention Evaluation (HOPE) study was designed to evaluate the effects of ramipril, an ACEI, in preventing ischemic events. 13 The HOPE study represents an important landmark study on the usefulness of ramipril in patients with high risk cardiovascular events without left ventricular dysfunction or heart failure. 5 The HOPE study was a randomized controlled double-blind trial of 9297 patients without left ventricular dysfunction and heart failure who were more than 55 years old. All patients had a history of coronary artery disease, peripheral vascular disease, diabetes mellitus or stroke and one additional risk factor such as hypertension, hypercholesterolemia, smoking and microalbuminuria. Patients received 10 mg/day ramipril or placebo for nearly 4.5 years. The HOPE trial demonstrated that ramipril reduced myocardial infarction, stroke and cardiovascular death. 5 This is discussed in greater detail below.
Mechanism of Action, Metabolism and Pharmacokinetic Profile
Mechanisms of ACE inhibition on cardiovascular protection
The role of the renin angiotensin system in regulating blood pressure and electrolyte balance is well established. Angiotensin II also contributes to cardiovascular abnormalities such as atherosclerosis and vascular remodeling. Thus, inhibition of renin-angiotensin-aldosterone represents an important approach to managing several cardiovascular diseases and hypertension. Angiotensin II is produced from a renin substrate, angiotensinogen, which is converted to angiotensin I and this decapeptide is then converted to angiotensin II by ACE. Angiotensin II causes vasoconstriction by activation of AT1 receptors in vascular smooth muscle cells–-thus inhibiting angiotensin II production is a commonly used strategy to treat hypertension.2,14 Angiotensin II promotes oxidative stress, stimulates inflammatory responses, and adversely alters the balance between the thrombotic and fibrinolytic state through AT1 receptors. 14 Angiotensin II stimulates vascular inflammation and increases thrombosis through increased generation of metalloproteins and expression of adhesion molecules by endothelial cells. 15 Angiotensin II also promotes superoxide production through activation of a potent membrane oxidase 16 –-this resultant oxidative stress reduces nitric oxide bioavailability. 15 At a cellular level, angiotensin II enhances smooth muscle cell migration and proliferation, remodeling the left ventricle and endothelial dysfunction. 17
It has also been suggested that angiotensin II is important in the development of atherosclerosis since tissue ACE activity is increased in coronary artery lesions and long-term ACEI reduces atherosclerotic lesion areas in the aorta, carotid, and coronary arteries. 18 Moreover, AT1 receptors are upregulated in both experimental models and in patients with hypercholesterolemia. 19 Thus, the antiatherogenic properties of ACEIs may be due to decreased accumulation and activation of inflammatory cells, reduced proliferation and migration of smooth muscle cells, decreased superoxide generation, and increased endothelial nitric oxide availability. 15
A homologue of ACE has recently been identified and labeled ACE II. 20 This ACE related enzyme is highly expressed in cardiac and renal vascular endothelial cells and is released from the cell surface in a manner analogous to ACE secretion. 21 ACEII hydrolyzes angiotensin II to a vasodilator peptide angiotensin (1-7), which functionally antagonizes the constrictor effects of angiotensin II. Classical ACEIs such as captopril and lisinopril do not inhibit ACEII, 21 implying that the influence of ACEII would be greater during inhibition of ACE with agents such as captopril. 22
Profile of clinical uses of Ramipril
Ramipril is one of the long-acting ACEIs. The ACEIs act by inhibiting the enzymatic activity of ACE; there is no association between the drug's action and renin levels (renin is synthesized and released from the kidneys), despite the fact that ACEIs are most effective in conditions associated with high plasma renin activity. Renin released into the circulation acts on its substrate angiotensinogen and produces angiotensin I, which is then converted to angiotensin II by ACE, a peptidyl dipeptidase (plasma kininase), which at the same time also inactivates bradykinin, a potent vasodilator that acts at least in part by stimulating the release of nitric oxide and prostacyclin from endothelial cells.
ACE inhibitors have several effects in the body: they alter the balance between the vasoconstrictive, salt-retentive and hypertrophic properties of angiotensin II, and they interfere with the vasodilatory and natriuretic effects of bradykinin and with the metabolism of other vasoactive substances (Fig. 1). ACEIs also inhibit angiotensin II generation at vascular and renal sites in addition to reducing catecholamine release from adrenergic nerve endings. Increased prostaglandin synthesis may also play a role in the antihypertensive action of ACEI.
Angiotensin II, the main effector of the renin-angiotensin-aldosterone system, is an important mediator of vascular remodeling in hypertension.22,23 Besides being a potent vasoactive peptide, Ang II also exerts proinflammatory effects on the vasculature by inducing the formation of integrin and adhesion molecules, increasing cytokine release, stimulating smooth muscle growth and causing an increased release of profibrotic mediators through activation of redox-sensitive pathways and transcription factors. Clinical findings suggest that inflammation participates in the mechanisms involved in the pathophysiology of hypertension and its complications.22–24 Vascular inflammation also plays a major role in the progression as well as clinical manifestation of atherosclerosis, and so can also contribute to vascular stiffness of arteries and increase the risk of myocardial infarction and stroke. Thus, therapies that reduce vascular inflammation are likely to reduce cardiovascular events. Ramipril therapy reduces cardiovascular events in patients with coronary artery disease, potentially by reducing vascular oxidative stress and elevation of gene and protein expression of inflammatory mediators.5,22,23,25
Endothelial dysfunction is a key element in most cardiovascular diseases. 25 In patients with cardiovascular risk, chronic ACEI therapy reduces endothelial dysfunction; this may explain why patients treated with ACEIs experience a greater cardiovascular benefit than is attributable to the decrease in blood pressure only. Treatment with ramipril leads to improved endothelial function in normotensive patients with successfully repaired coarctation of the aorta independently of the blood pressure lowering effects of the ACEI. Furthermore, ramipril reduces the expression of proatherogenic inflammatory cytokines (such as IL-6), adhesion molecules (sVCAM-1) and sCD40L in this population. 27
The ACEIs were initially developed for the treatment of hypertension. Subsequently these therapeutic agents found uses in the treatment of other cardiovascular and renal conditions. ACE inhibitors are extremely useful in the treatment of heart failure and post myocardial infarction management. 28 These drugs reduce the severity of heart failure and also improve the maximal exercise tolerance in patients suffering from heart failure. In these patients, ACEIs significantly decrease systemic vascular resistance (afterload), pulmonary capillary wedge pressure (preload), pulmonary vascular resistance and heart size while at the same time increasing cardiac output and exercise tolerance.
Several randomized clinical trials suggest that inhibition of the renin-angiotensin-aldosterone system reduces the risk of new type II diabetes in patients with arterial hypertension or with congestive heart failure. ACEIs also have a particularly useful role in the treatment of patients with diabetic nephropathy because they diminish proteinuria and stabilize renal function (even in the absence of reduction in blood pressure). This beneficial effect may result from an improvement in intrarenal hemodynamic vascular resistance, with decreased glomerular efferent resistance leading to a reduction of intraglomerular capillary pressure.
Pharmacokinetics
Ramipril is a prodrug which in itself is a poor inhibitor of ACE, but after absorption, is rapidly hydrolyzed in vivo to ramiprilat, an active metabolite29–31 with a long elimination half-life, thus permitting once-daily administration. Approximately 56% of an administered oral dose of ramipril is absorbed and converted to ramiprilat. Ramiprilat has a greater affinity for ACE than does captopril or enalaprilat.29,30 The peak plasma concentration of ramipril and ramiprilat appear to be linearly related to dose30,31 and peak levels are found within 1 to 3 hours of administration. Using a single oral dose of 5 mg ramipril, the bioavailability values of ramipril and ramiprilat were 28% and 44%, respectively. 32 Thus, patients with liver cirrhosis have enough capacity to metabolize and excrete ramipril, but are unable to form ramiprilat, although enough ramiprilat was formed for sufficient ACE inhibition. 32 The percentage of protein binding of ramipril and ramiprilat is 73% and 56%, respectively, and the volume of distribution for ramipril is approximately 90 lit. 33
During its first-pass metabolism in the liver, ramipril is rapidly and completely hydrolyzed to its active metabolite, ramiprilat. Ramipril and ramiprilat are both conjugated and then excreted in the urine. 31 A fraction of ramipril is also metabolized to an inactive metabolite (diketopiperazine ester) which is then excreted by the kidneys.
The half-life of ramipril is about 1.1 to 4.5 hours. After absorption of ramipril, peak plasma concentrations of ramipril rapidly decrease because of distribution in body fluids, and binding to plasma proteins and tissue ACE.31,32 Ramiprilat also binds to tissue ACE. The half-life of unbound ramiprilat is about 9-18 hours while the half-life of ramiprilat that is bound to tissue ACE has is more than 50 hours and perhaps even as long as 110 hours. 33
Alterations of pharmacokinetics during disease
Cirrhosis
The plasma concentration of ramipril is elevated in patients with severely impaired liver function. Pidlich et al studied the pharmacokinetics of ramipril and ramiprilat in 12 cirrhotic patients. 34 There was a delay in the time needed to obtain peak plasma levels of ramipril and ramiprilat following a single oral dose of 5 mg ramipril. However, overall ACE inhibition was still at 92%, leading to the conclusion that although patients with liver cirrhosis still had enough capacity to metabolize and excrete the parent compound ramipril, they are unable form ramiprilat.
Renal disease
Renal dysfunction affects the clearance and elimination of ramiprilat but not that of ramipril. 35 The duration of ramipril induced ACE inhibition is prolonged according to the degree of renal impairment. 35 Thus, the dosage of ramipril should be reduced according to the extent of renal impairment, especially in patients having creatinine clearances lower than 40 ml/min/1.73 m2. 32 In such patients, it is suggested that treatment should start with a dosage of 1.25 mg/day and gradually increased to a maximum 5 mg/day to adequately control blood pressure. 32
The pharmacokinetics of ramiprilat (2.5 mg after each dialysis over 28 days) was studied in 12 hypertensive patients receiving regular hemodialysis. 36 There was a consistently high degree of ACE inhibition throughout the study and the patients had well controlled blood pressures. Ramipril did not modify the time to peak for ramiprilat concentrations, but increased the peak plasma concentration significantly compared to healthy controls. The ramiprilat hemodialysis clearance was 31.7 ml/min on the first day and 21 ml/min on day 29. However, steady state levels of ramiprilat were reached despite an increase in ramiprilat concentrations.
Therapeutic Efficacy
Ramipril in stroke
Stroke is the second leading cause of death and disability in developed countries.37,38 It is estimated that approximately 550000 new stroke cases occur yearly in North America, accounting for a tremendous increase in health-related chronic disabilities. 37 Although high blood pressure is a risk factor for stroke, ACEIs can decrease the risk of stroke by other mechanisms independent of lowering blood pressure including inhibition of vascular smooth muscle cell proliferation, decreased atherosclerotic processes and plaque rupture and enhanced fibrinolysis.1,15 Thus, ACEIs have the potential to lower the risk of stroke.
Clinical and experimental studies confirm a beneficial effect of ACEIs in reducing vascular events including stroke. The Perindopril Protection against Recurrent Stroke Study (PROGRESS) evaluated the effects of a blood-pressure-lowering regimen in hypertensive and non-hypertensive patients with a history of stroke or transient ischaemic attack over 4 years of follow-up. 39 This study reported that perindopril and indapamide reduced the risk of stroke by 28%. Experimental studies also report that ACEIs reduce stroke incidence and stimulate neuronal regeneration. 40 Ramipril treatment prevents the vascular changes (loss of vascular density, collagen intensity) seen in hypertensive rats. 41
The HOPE study utilized a double blind randomized trial in which participants aged over 55 with a high risk of cardiovascular events received either ramipril (10 mg) or placebo. 37 Strokes and transient ischemic attacks were lower in the ramipril group compared to the placebo group (3.4% vs. 4.9% and 4.1% vs. 4.9%, respectively) and 0.4% of patients in the ramipril group had fatal strokes compared to a 1% incidence in the placebo group. Only 2.2% of the ramipril group had ischemic strokes compared to 3.45% in the placebo group. In addition, fewer patients in the ramipril-treated group had multiple strokes and functional and cognitive impairment (1.8% vs. 2.3%). The HOPE trial also reported that the relative risk of any stroke was reduced by 32% in patients receiving ramipril and the risk of a fatal stroke was reduced by 61%. These benefits occurred regardless of baseline blood pressure and were independent of the modest blood pressure lowering effects in the ramipril group. The study also reported that the beneficial effects of ramipril was not confined to patients with high blood pressure as these benefits were also observed in patients receiving aspirin and other blood pressure lowering drugs. Ramipril (10 mg) reduced stroke by 33% in diabetic patients.8,42 Thus, based on the HOPE trial it is suggested that patients at high risk for cardiovascular events be treated with ACEIs such as ramipril regardless of blood pressure or other treatments. These and other studies suggest that long-term treatment with ramipril has beneficial effects in reducing fatal and non-fatal stokes and transient ischemic attacks.
Ramipril in cardiac hypertrophy and heart failure
Activation of the renin-angiotensin-aldosterone system also plays a key role in the pathogenesis of heart failure 43 since angiotenisn II is a vasconstrictor, increases sympathetic tone and produces sodium and fluid retention. 17 Angiotensin II mediates remodeling of the left ventricle and alterations of vascular structure. 17 Angiotensin II increases cardiac preload and afterload through AT1 receptors and causes cardiac muscle hypertrophy. 44 It is possible that ACEIs also have direct anti-hypertrophic effects on cardiac muscle. 1 Long-term treatment with ACEIs increases cardiac output, reduces left ventricular filling pressure and heart rate in patients with heart failure and left ventricular systolic dysfunction.45,46 Other studies suggest that treatment with ACEIs prevents heart failure and hospitalization of patients with a low ejection fraction after myocardial infarction.11,47,48 Enalapril treatment also abolishes the ECG changes that occur in the hypertrophied left ventricle. 49
The results of the HOPE study also suggests similar reports similar beneficial effects of ramipril in patients either with or those at risk of heart failure irrespective of left ventricular dysfunction. 5 The HOPE study demonstrated that ramipril was able to prevent the development/persistence of left ventricular hypertrophy in high risk patients without known left ventricular dysfunction regardless of blood pressure and presence or lack of documented coronary artery disease. 12 In fact, ramipril treatment appears to cause a greater regression/prevention of left ventricular hypertrophy in normotensive or controlled hypertensive patients either with or without coronary artery disease, suggesting that the effect of ramipril on left ventricular hypertrophy was independent of blood pressure reduction. This direct impact of ramipril on left ventricular hypertrophy was also reported in other studies. 50 The Large-Scale Acute Infarction Ramipril Efficacy (AIRE) study concluded that ramipril therapy (5 or 10 mg/day) decreases mortality by 27% in patients with heart failure after myocardial infraction. 51
The effects of ACEIs on left ventricular remodeling may represent an important mechanism for their beneficial effects in patients with heart failure.52,53 A recent study examined the effect of ramipril on postinfarction ventricular remodeling and suggested that ramipril attenuated left ventricular remodeling through antioxidative mechanisms. 53 ACE also catalyzes the degradation of bradykinin, leading to the possibility that the benefits of ACEIs are related to increasing bradykinin levels. A recent experimental study of the effects of ramipril on left ventricular dysfunction after myocardial infarction in a kininogen-deficient rat model demonstrated that ramipril attenuated the left ventricular dysfunction. 54 The role of bradykinin is supported in another experimental study where the effect of ACEIs on cardiac remodeling in the canine myocardium was attenuated by administration of a bradykinin antagonist, leading to the suggestion that the beneficial effects of ACEIs in the treatment of heart failure may be related to increased kinin levels. 55 Additional mechanisms thought to be important in the use of ACE inhibitors in vascular protection in patients with or without heart failure include the antiproliferative, antithrombotic, antiatherosclerotic and anti-inflammatory effects of ACEIs such as ramipril. 56
Ramipril in diabetes mellitus
The benefits of ACEIs in diabetes mellitus and diabetic complications are well established since ACEIs may exert favorable metabolic effects by preventing diabetes. The Captopril Prevention Project represents a clinical trial showing that a reduction in newly diagnosed diabetes in captopril treated hypertensive patients compared to those receiving either beta blockers or diuretics. 57 In addition, treatment with ACEIs such as ramipril reduces urinary albumin excretion and slows the progression of diabetic complications such as diabetic nephropathy and retinopathy.58,59
The effect of ramipril in diabetic subjects is summarized in Table 1. In the HOPE and MICROHOPE studies, 8 treatment of diabetic patients with 10 mg ramipril reduced the primary outcomes of myocardial infarction (22%), stroke (33%) or cardiovascular death (37%). Secondary outcomes of these patients were reduced by 24% and other outcomes such as heart failure, worsening angina pectoris and transient ischemic attack were also reduced in the ramipril group. These trials also demonstrated that 5.4% of the patients receiving placebo developed diabetes compared to 3.6% in ramipril-treated patients. The risk of developing nephropathy, renal failure or the need for laser treatment was also decreased in the ramipril-treated group. These beneficial effects occur even in patients using other drugs such as aspirin, lipid-lowering agents or beta-blockers.
Some clinical trials examining the effect of ramipril in diabetic subjects.
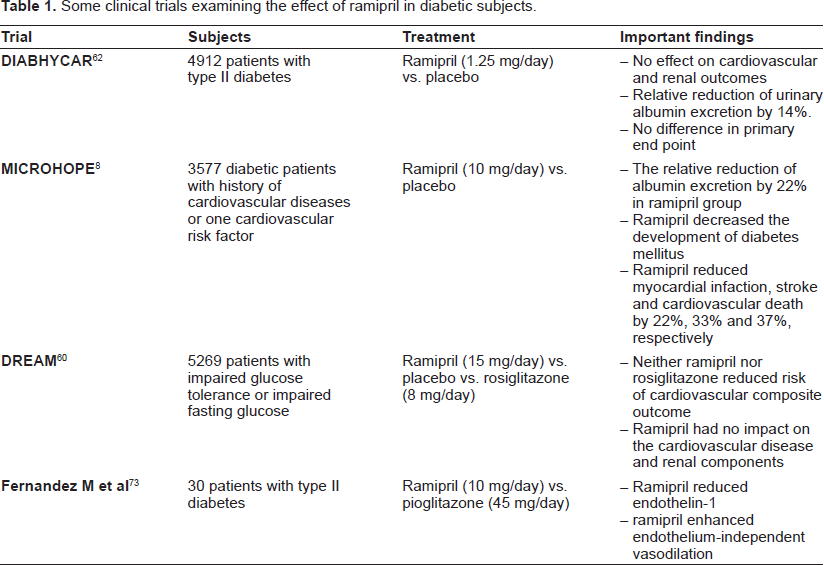
The recent DREAM Trial Study compared the effects of ramipril and rosiglitazone on cardiovascular and renal outcomes in 5269 people with impaired glucose tolerance and/or impaired fasting glucose. 60 Subjects were randomized to receive either 15 mg/day ramipril or placebo for 3 years. While ramipril did not reduce the risk of cardio-renal composite outcome or it's components and did not affect cardiovascular events, it modestly reduced postload glucose levels and increased the regression of impaired glucose tolerance or impaired fasting glucose. 60 The absence of benefits of ramipril in the DREAM trial study (compared to the HOPE study) may be due to a short follow up and lower incidence of cardiovascular diseases in trial subjects. Furthermore, inhibition of the renin-angitensin-aldosterone system significantly reduces the incidence of type II diabetes in patients with hypertension or congestive heart failure. 61
The DIABHYCAR study investigated the effects of a low dose (1.25 mg) of ramipril on cardiovascular and renal outcomes of patients with type II diabetes and showed that this dose of ramipril, which is eightfold lower than the daily dose used in the HOPE and micro-HOPE studies,5,8 had no beneficial effects. 62 The DIABHYCAR study concluded that a marked inhibition of the renin-angiotensin-aldosterone system was required for cardiovascular protection in patients with type II diabetes, and this was likely to occur with higher doses ramipril.
Several mechanisms have been suggested for the beneficial effects of ACEIs on diabetes including improved insulin sensitivity, decreased levels of free fatty acids, improved pancreatic blood flow and decreases in oxidative stress and hepatic clearance of insulin.15,63,64
Effect of ramipril on blood vessels
Endothelial cells have a key role in maintaining vascular homeostasis by regulating vascular tone, cell proliferation and homeostasis. 26 Endothelial dysfunction is an early step in the pathogenesis of cardiovascular dysfunction associated with hypertension, diabetes and atherosclerosis.65,66 Studies in animal models of atherosclerosis in rabbits and monkeys demonstrate that long-term treatment with ACE inhibitors reduced atherosclerotic lesion areas in aorta, coronary and carotid arteries.67,68 The atherosclerotic processes is reversed or significantly inhibited by treating Apo-E knockout with ACEIs. 69
There is increased accumulation of tissue ACE in human coronary atherosclerotic arteries, 18 which is thought to contribute to plaque instability and myocardialinfarction. Thus, inhibitionofangiotensin II production via ACE inhibitors can disrupt the events causing development of cardiovascular diseases. The Study to Evaluate Carotid Ultrasound Changes in Patients Treated with Ramipril and Vitamin E (SECURE) evaluated the effects of ramipril on the progression of atherosclerosis. 70 A total of 732 patients underwent duplicate B-mode carotid ultrasound examinations and the results demonstrated that a 37% reduction in carotid artery intima/media ratio following 4.5 years ramipril treatment at a dose of 10 mg. The results of this trial also suggest that the reduction in atherosclerosis due to ramipril treatment was not fully explained by the blood pressure lowering effects of ramipril and could well result from other direct effects on vascular smooth muscle. The United Kingdom Prospective Diabetes Study (UKPDS) also indicates that the beneficial effects of ACEIs are larger than those predicted from only lowering blood pressure. 71
The HOPE study demonstrated that ramipril therapy reduced the incidence of acute coronary syndrome in patients with vascular diseases (9.9% vs. 12.3% in placebo). 5 Treatment with ramipril for three weeks (5 mg/day) results in significantly improved renal vasodilatation to L-Arg in uncomplicated type 1 diabetic patients. 72 Hypertensive patients treated with ramipril (2.5 mg/day for 12 weeks) decreased carotid artery compliance and distensibility; these beneficial effects were reversed when treatment was stopped. A recent study reported that the addition of ramipril to intensive insulin therapy of type II diabetic patients improved vascular dysfunction and inflammatory markers independently of glycemic control. 73 This treatment regimen (ramipril and insulin) also enhanced endothelium-independent vasodilation while at the same time reducing endothelin-1 levels. 97
Ramipril: myocardial infarction and cardiovascular events
Activation of the renin-angiotensin-aldosterone system can increase the risk of ischemic heart disease. 74 Several studies have reported that ACEIs reduced the risk of cardiovascular death, myocardial infarction, stroke, hospitalization for heart failure while also reducing the need for revascularization in such patients.7,11,47,48,75 The HOPE study evaluated the effects of ramipril treatment of high-risk patients without heart failure or left ventricular dysfunction.5,76 In this trial, 9247 participants at high risk for cardiovascular events were studied during a mean 4.5 years of follow-up. The data of this trial showed that patients in the ramipril-treated group had a reduced relative risk of non-fatal myocardial infarction, either Q-wave or non-Q-wave MI, no additional worsening of angina and lowered cardiovascular related deaths compared to the placebo group. While 14% of patients died from cardiovascular causes or had myocardial infarction in the ramipril group, the rate was significantly higher in the placebo treated group (17.8%). Ramipril treatment significantly lowered the rate of coronary revascularization procedures and also reduced death from any cause (from 12.2% to 10.4%). The reductions in MI and stroke (by 19% and 31%, respectively) due to ramipril treatment exceed those that would be expected from modest changes in blood pressure alone, and the benefits of ramipril also occurred in patients taking other effective therapies such as antiplatelet agents, beta-blockers, and lipid lowering drug, suggesting that the benefits of ramipril are additive to these agents, although in a defined substudy of HOPE, it is suggested that the effects of ramipril on cardiovascular morbidity and mortality may be related to the effects on blood pressure patterns over a 24-hour period.
The beneficial effects of ramipril has also been examined in sudden cardiac death and nonfatal cardiac arrest in high-risk patients without heart failure or left ventricular dysfunction where the ACE inhibitor reduced the composite outcome of sudden death (arrhythmic death and cardiac arrest) by 21%. 77 These results are similar to studies using other ACE inhibitors (trandolapril, 47 zofenopril, 78 enalapril 79 and captopril). 11
The MITRA PLUS study enrolled 14608 patients with ST-elevation myocardial infarction and reported that ramipril treatment significantly reduced hospital mortality and non-fatal coronary and cerebrovascular events compared to patients not receiving ACEIs. 80 As shown in Table 2, ramipril significantly reduced hospital mortality and nonfatal coronary and cerebrovascular events (but not in the rate of heart failure or discharge) compared to other ACEIs. Importantly, ramipril treatment does not alter the diurnal periodicity of myocardial infarction (peak event rates between 6 pm and noon) 81 although it non the less reduces the incidence of acute myocardial infarction. 82
Some clinical trials and experimental studies on the effect of ramipril on cardiovascular disease and outcome.

Safety and Tolerability
The adverse effects profile and tolerability of 10 mg ramipril were evaluated in several studies. Similar to the findings with other ACEIs, dry cough is more frequent in patients treated with ramipril therapy than with placebo. The most common side effects of ramipril are: headache, dizziness and fatigue. For example, the adverse effects of ramipril profile in one study were reported to be dry cough (15.1%), dizziness (6%), and hypotension (2.4%) with 13% of patients discontinuing treatment because of the side effects. In the ONTARGET study, the effect of telmisartan (an angiotensin receptor blocker) was compared with ramipril in patients with vascular disease or high-risk diabetes. 6 Telmisartan had a higher rate of hypotensive symptoms and syncope compared to ramipril, however, the rate of cough and angioedema was higher in the ramipril-treated group. The VALIANT study (Valsartan in Acute Myocardial Infarction Trial), reported similar findings when comparing captopril and valsartan. 83 Only 3.3% of ramipril group stopped taking ramipril because of coughing in the DIABHYCAR study of low dose ramipril (1.25 mg/day) in patients with type II diabetes. 62
Analysis of the results of the HOPE study in women indicated that cough and angioedema were the most common causes for discontinuing ramipril therapy and more women than men required discontinuation of ramipril treatment because of cough alone (11.3% women in ramipril group vs. 5.7% in men received ramipril). 84 Discontinuation of ramipril therapy due to dizziness or hypotension wasn't different between ramipril and placebo groups in the HOPE study, however, uncontrolled hypotension leading to treatment stoppage was lower in the ramipril group. 5 In addition, severe angioedema leading to treatment discontinuation was higher in the ramipril group compared to the placebo treated group (0.4% vs. 0.2%). 5
Ramipril prescription
The HOPE study advised that ramipril be started at 2.5 mg once a day, then increased to 5 mg for three weeks and finally increased to 10 mg daily as tolerated. 32 After 2-4 weeks, plasma urea, creatinine and plasma levels should be measured and if urea rises by 50% or more or creatinine rises by more than 20%, the drug should be discontinued. Thus, in patients with renal insufficiency (creatinine clearance less than 40 ml/min/1.73 m2) and/or severe hepatic disease, the dosage of ramipril should be adjusted. 32 It has been reported that patients with liver cirrhosis had enough capacity to metabolize and excrete ramipril, but did not have enough capacity to form ramiprilat, leading to the conclusion that if required, the dose of ramipril should start with 5 mg once daily, which is enough for sufficient ACE inhibition. The extent, not the rate of absorption of ramipril, is not affected by the presence of food. In patients with hypertension or recent myocardial infarction, the dose of ramipril may need to be divided and prescribed twice a day. 32
Cost effectiveness of ramipril
Several studies evaluated the cost effectiveness of ramipril in reducing cardiovascular events based on the findings of the HOPE study. A Swiss study of the cost effectiveness of ramipril in patients at high-risk for cardiovascular disease concluded that ramipril was the most favorable therapy in type II diabetic patients. 85 Similar studies in Italy, Germany, Australia and Spain also confirmed the favorable cost-effectiveness of ramipril.86–89 Ramipril is also a cost effective strategy in the secondary prevention of cardiovascular events including stroke.87,90 The retrospective analysis of the AIRE study (The Acute Infarction Ramipril Efficacy Study) concluded that treatment of heart failure with ramipril was cost-effective in reducing the mortality and hospitalization in patients with heart failure. 75
ACE inhibitors vs. angiotensin receptor blockers (ARBs)
During chronic ACEI therapy, plasma and tissue angiotensin II levels are unchanged or elevated, a phenomenon labeled by some as the “angiotensin escape phenomenon”.91,92 Several explanations have been suggested, such as an inadequate dose of ACEI, 93 reactive hyper-reninemia, 94 ACE up regulation 91 and the presence of alternative enzymatic pathways capable of converting angiotensin I into angiotensin II e.g. chymase, which produces angiotensin II independent of ACE.95,96 While AT1 receptors are responsible for many deleterious consequences of renin-angiotensin-aldosterone, AT2 receptors, in contrast, appear to be involved in the control of cell proliferation, cell differentiation and development, angiogenesis, wound healing, tissue regeneration and apoptosis; these biological processes counteract the trophic responses mediated through the AT1 receptors. Additional effects mediated by AT2 receptors include vasodilation and some antifibrotic and antiinflammatory effects.97,98 As a result, there is a strong rationale for blockade of renin-angiotensin-aldosterone by inhibiting AT1 receptors rather than relying on ACE inhibition. Such a more specific approach leaves AT2 receptors unopposed and open for stimulation by alternatively formed angiotensin II, and theoretically, more pronounced AT2 receptor mediated effects following application of AT1 antagonists. 99 However, there are relatively few direct clinical comparisons between these two drug categories in terms of their uses in cardiovascular diseases. Available data comparing the clinical effects of ACEIs with an ARB are summarized in Table 3.
A summary of clinical trials comparing the cardiovascular benefits of angiotensin converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs).

Conclusions
In conclusion, ramipril (10 mg/day) can significantly reduce the risk of myocardial infarction, death and stroke in patients with a history of stroke, diabetes mellitus or peripheral vascular disease. The beneficial effects of ramipril are maintained in patients having cardiovascular disease with or without heart failure or LV dysfunction and should be considered in addition to using other drugs and life style modifications.
Disclosure
The authors report no conflicts of interest.
