Abstract
The sirolimus-eluting stent (SES) Cypher was the first commercially available drug-eluting stent. The use of this stent has resulted in significantly lower rates of restenosis and lesion revascularization compared to bare metal stents and balloon angioplasty. In this review, angiographic and clinical outcomes in patients treated with SES are compared to those treated with bare metal stents and other drug-eluting stents. Furthermore, efficacy and safety outcomes of SES in complex lesions (left main stenosis, bifurcation lesions, chronic total occlusions, long lesions and small vessels) and high risk populations (diabetes and acute myocardial infarction) are presented.
Introduction
With the introduction of drug-eluting stents (DES), restenosis after percutaneous coronary intervention (PCI) has declined substantially with repeat revascularization rates of 5% to 10%.1–7 In achieving single-digit clinical and angiographic restenosis rates, utilization of DES has facilitated a change in practice patterns by reducing the need for repeat PCI and coronary artery bypass graft (CABG) procedures. We review the SES Cypher (Cordis, Miami, Florida) in terms of mechanism of neointimal tissue inhibition, short and long-term clinical outcomes, and efficacy in specific patient and lesion subtypes.
Restenosis
Pathophysiology
Restenosis of the arterial lumen has been the “Achilles’ heel” for the long-term success of PCI with plain balloon angioplasty and then with bare metal stents (BMS). During balloon angioplasty, arterial recoil and intimal/medial dissection are a response to stretch injury and laceration from balloon expansion of the arterial lumen. 8 While endovascular stenting addresses recoil and dissection, it leads to smooth muscle cell migration to the site of injury and build-up of extracellular matrix resulting in neointimal proliferation. 9 Though all PCI procedures induce neointimal formation, in only some is there enough to develop restenosis. BMS use leads to greater neointimal formation than balloon angioplasty alone (Fig. 1), thus eliminating one restenotic mechanism but exacerbating another.8–10
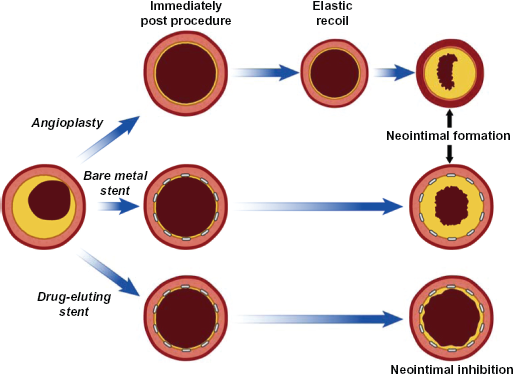
Mechanisms of restenosis with balloon angioplasty, bare metal stents, and drug-eluting stents. Post-procedure, stents prevent elastic recoil. Note how drug-eluting stents prevent neointimal formation.
Inhibition of neointimal proliferation
Initial attempts to limit neointimal hyperplasia formation after BMS implantation included multiple systemically administered oral medications11,12 and intravascular brachytherapy. 13 However, when these modalities were unsuccessful it appeared that inhibition of neointimal formation resulting from the inflammatory and smooth muscle proliferative response might require localized pharmacologic delivery.
Local, stent-based delivery of anti-inflammatory and antiproliferative agents inhibits neointimal proliferation by modulating the inflammatory response after balloon and stent injury to vascular intima and/or media. Sirolimus,14–20 its analogues,21–24 and paclitaxel25–30 inhibit neointimal thickening in animal models of vascular injury, and human coronary arteries in clinical studies. These hydrophobic pharmacologic agents interfere with the early phases of the cell cycle of replication. Sirolimus (rapamycin, rapamune®, Wyeth, Philadelphia, PA) was approved by the Food and Drug Administration (FDA) in 1999 as an immunosuppressive agent for the prevention of renal transplant rejection. A macrolide antibiotic, sirolimus and its analogues (everolimus, zotarolimus, tacrolimus, and biolimus A9) bind to the cytosolic immunophilin FK506-binding protein (FKBP12), inhibiting the activation of mammalian target of rapamycin (mTOR), thereby blocking cellular transition from G1 to S phase of the cell cycle (Fig. 2), resulting in inhibition of cell proliferation.10,14 The substitution of the native hydroxyl group at position 42 in all sirolimus analogues allows for modification of chemical properties while retaining biological activity. These changes in chemical properties affect drug incorporation and release from the polymer and lead to some differences in degree of neointimal inhibition. 31
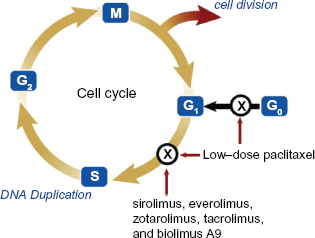
Effects of sirolimus, sirolimus analogues, and paclitaxel on the cell cycle. Sirolimus and its analogues cause cell cycle arrest between G1 to S phase of the cell cycle whereas paclitaxel at low doses acts at both G1/S and G0.
Platform for drug delivery
The primary focus for evaluating the efficacy and safety of DES has been on the anti-inflammatory and/ or antiproliferative agents, with little attention paid to the various stent platforms. Stent configuration and strut thickness were important predictors of short and long-term outcomes with BMS. Coil stents and hybrid coil tubular stents are more flexible and trackable than their tubular or multicellular counterparts, but have lower radial strength and result in greater recoil. However, in randomized clinical trials, use of meshwire and flexible coil BMS designs resulted in significantly higher restenosis rates compared with the relatively rigid, slotted tube or multicellular design stents.32–33
While thicker stent struts (140 microns) increase visibility, radial strength, and arterial wall support, these stents are associated with greater restenosis compared with thinner stent struts (50 microns), 34 presumably by causing greater vascular injury, which in turn triggers more neointimal formation. 32 While these characteristics are applicable to BMS, they may not apply to DES. The goal of achieving uniform drug elution may require more stent surface area contact with the vessel wall, which is achieved with high metal-to-surface ratio and symmetrical expansion. On the other hand, a low-profile, flexible, conformable platform may be more deliverable for the more challenging lesions in calcified and tortuous vessels but may produce non-uniform drug distribution.
Drug delivery for coronary arteries requires both a stent as a platform to transport the drug and a polymer for uniform, controlled drug delivery at the site of stent implantation. Polymer coatings are used to adhere sirolimus on stainless steel stents. The SES Cypher uses an erodible topcoat that is biologically inert but various investigators have raised concerns regarding the inflammatory response potentially induced by polymers at the site of DES implantation. This in turn can manifest as a local hypersensitivity reaction, lead to delayed restenosis, or of greater concern–-late stent thrombosis. 35
Predictors of restenosis
Predictors of restenosis with balloon angioplasty and stent placement can be categorized as clinical or angiographic. Clinical predictors include diabetes, hypertension, and presenting with an acute coronary syndrome. 36 Angiographic predictors include smaller reference vessel size, longer lesion length, final diameter stenosis and saphenous vein graft treatment. In this review, the impact of SES on these categories will be presented.
Clinical Studies Evaluating SES
Key definitions
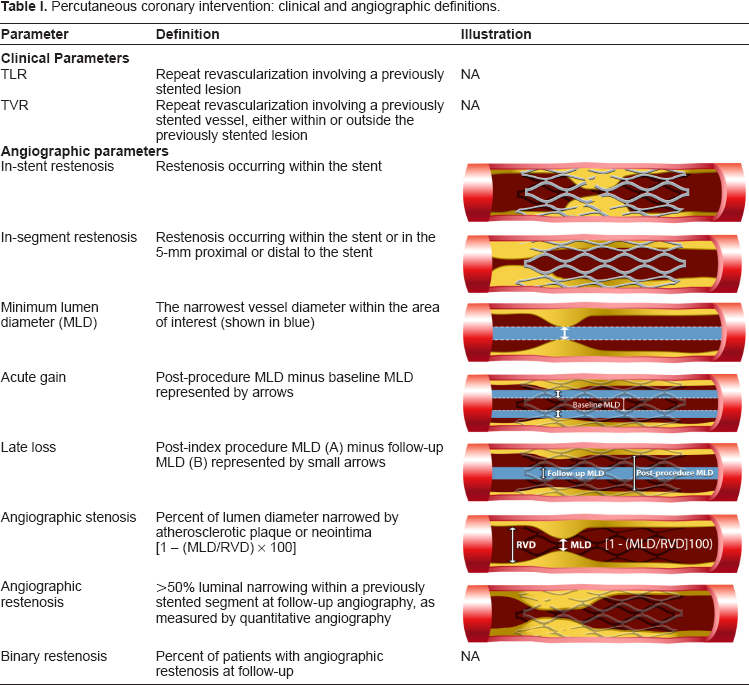
To ensure systematic and consistent evaluation of data regarding DES, it is important to review some key definitions (Table 1). As balloon overhang and diminution in antiproliferative drug dose effect can occur beyond stent edges, DES efficacy is better measured by in-segment (includes stented segment and 5 mm proximal/distal to stent edge) rather than in-stent evaluation. Both late loss and angiographic restenosis are terms typically reported in clinical trials, but revascularization based on late loss or angiographic restenosis is not necessarily ischemia driven, and the clinical relevance of these terms has been difficult to establish. In fact, Weintraub et al demonstrated that repeat revascularization rates are higher in clinical trials with angiographic follow-up, 37 possibly due to the “oculostenotic reflex”, while, target lesion revascularization (TLR) in clinical practice and in registries without angiographic follow-up is usually ischemia-driven and subsequently lower. Using data from BMS and DES trials, Mauri et al demonstrated that while late loss is strongly related to angiographic restenosis (r 2 = 0.73), 38 it correlates poorly with clinical restenosis (TLR) (r 2 = 0.22). 39 Furthermore, Ellis et al have also observed a threshold with the paclitaxel-eluting stent (PES) Taxus (Boston Scientific, Natick, MA) where there is adequate patency to accommodate 0.5 to 0.75 mm of late lumen loss before the TLR rate exceeds 5%. 40 Major adverse cardiovascular events (MACE) are defined in most studies as the total number of patients who died, had a myocardial infarction or required lesion (or vessel) revascularization.
Percutaneous coronary intervention: clinical and angiographic definitions.
Randomized clinical trials
The First-in-Man (FIM) study of the SES in Brazil provided proof of concept that local delivery of an anti-inflammatory agent can inhibit neointimal formation in simple de-novo coronary lesions. 20 Sirolimus-eluting Bx Velocity stents were implanted in 30 patients with either slow release (SR, >= 28 day) or fast release (<15 day) formulations. Long-term angiographic and intravascular ultrasound (IVUS) follow up at four years in 26 patients showed no stent thrombosis, target-lesion revascularization, death or myocardial infarction in the slow release group (current SES formulation). 41
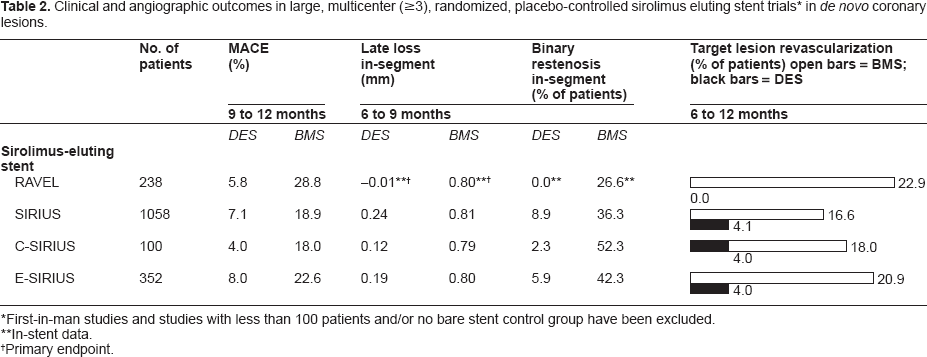
After the initial FIM results, a series of small randomized trials of SES1,5,6 (Table 2) demonstrated the efficacy of sirolimus in reducing restenosis. The Randomized Comparison of a Sirolimus-Eluting Stent with a Standard Stent for Coronary Revascularization (RAVEL) study was one of the first randomized, double blinded, clinical trials that demonstrated superiority of SES over BMS (n = 238) in preventing neointimal proliferation and restenosis. The initial benefit with the use of SES persisted at 5 year follow-up. 42
Clinical and angiographic outcomes in large, multicenter (≥3), randomized, placebo-controlled sirolimus eluting stent trials * in de novo coronary lesions.
First-in-man studies and studies with less than 100 patients and/or no bare stent control group have been excluded.
In-stent data.
Primary endpoint.
These studies were followed by the large-scale, pivotal, randomized clinical trial Sirolimus-Eluting Stent in De-Novo Native Coronary Lesions (SIRIUS) comparing SES vs. BMS. 2 Superior angiographic and clinical outcomes were observed with the SES Cypher at 9-month follow-up (TLR: SES 4.1%, BMS 16.6%; P = <0.001 and MACE: SES 7.1%, BMS 18.9%; P = <0.001). At long-term follow-up of 5 years, these benefits persisted (TLR: SES 9.4%, BMS 24.2%; P = <0.001 and MACE: SES 20.3%, BMS 33.5%; P = <0.001). 43 No differences in death, MI, or non target lesion revascularization were observed between the two groups. Thus in multiple studies the Cypher SES leads to significant risk reduction in angiographic and clinical restenosis compared to its BMS control. 44
Head-to-head comparisons
SES vs. PES
Previously, six randomized, unblinded, comparisons of the SES versus the PES have been reported.45–50 The trials differed in sample sizes (range 202 to 1386), number of study sites (range 1 to 89), and patient populations enrolled (3 unselected,45,47,49 1 diabetic, 46 1 in-stent restenosis, 48 and 1 complex lesion). 50 Four of the trials were powered for angiographic (late lumen loss or restenosis) rather than clinical (MACE) endpoints. Of the 6 head-to-head trials, in the two-center Randomized Comparison of a Sirolimus With a Paclitaxel Eluting Stent For Coronary Revascularization (SIRTAX) trial, a significant difference in clinical restenosis was observed between the two DES (TLR rate: SES 4.8%, PES 8.3%; P = 0.03). 49 A meta-analysis of these 6 trials found that implantation of the SES was associated with a lower rate of TLR (5.1% SES vs. 7.8% PES; P = 0.001) and angiographic binary restenosis (9.3% SES vs. 13.1% PES; P = 0.001) (Table 3). 51
Head-to-head trials comparing Cypher sirolimus-eluting (SES) and Taxus paclitaxel-eluting (PES) stents.
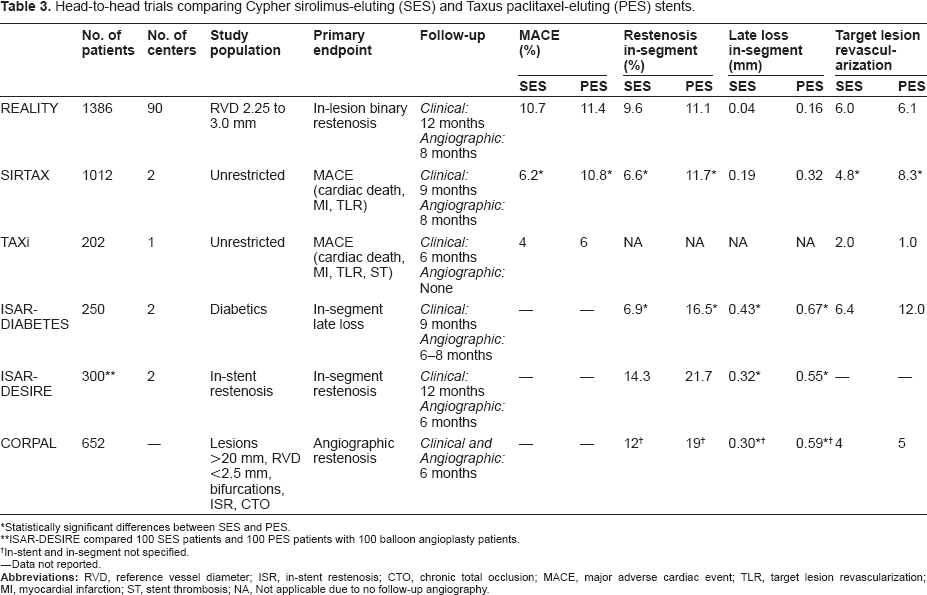
Statistically significant differences between SES and PES.
ISAR-DESIRE compared 100 SES patients and 100 PES patients with 100 balloon angioplasty patients.
In-stent and in-segment not specified.
–-Data not reported.
Another more recent meta-analysis of 16 randomized trials inclusive of the prior 6 trials also concluded that the pooled data suggested superiority of the SES Cypher in reduction of reintervention and stent thrombosis (Fig. 3). 52 The trials differed in sample sizes (range 70 to 2098), number of study sites, and patient populations enrolled (6 unselected, 1 diabetic, 1 in-stent restenosis, 3 complex lesion, 2 acute myocardial infarction, 1 multivessel disease, 1 small vessel, 1 long lesion). Six of these trials were powered for angiographic (late lumen loss or restenosis), eight for clinical (MACE) endpoints, and two were follow up IVUS assessments. No differences in death or MI were observed between the two stents. However, interpretation of the above findings is limited by the heterogeneity of patient/lesion population across studies and the lack of multicenter (≥3 sites) design and independent core angiographic laboratory in many of the studies.
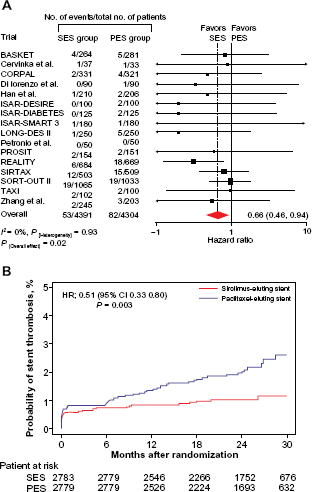
Analysis of reintervention according to the drug-eluting stent type (A) Absolute numbers of patients requiring reintervention and hazard ratios (HRs) for this end point with sirolimus-eluting stent (SES) versus paclitaxel-eluting stent (PES) for individual trials and pooled population. The HRs are shown on a logarithmic scale. The size of the square is proportional to the weight of the individual studies, measured as the inverse of the estimated variance of the log HR. (B) Kaplan-Meier curves of reintervention in each of the stent groups for the pooled population. Hazard ratio indicates the HR associated with the SES. Reprinted with permission from Schomig et al. J Am Coll Cardiol 2007;50:1373–80.
While meta-analyses are widely used to guide and support clinical strategies, it is important to consider that meta-analyses have been found to inaccurately predict the outcomes of large randomized trials 35% of the time. 53 In the 16 randomized trials, the second largest, multicenter clinical trial, the REALITY trial, was conducted with an independent angiographic core laboratory comparing the SES Cypher against the PES Taxus while the largest one, SORT OUT II, had an independent end points committee but no independent angiographic core laboratory. In the REALITY trial, comparable rates of TLR, MACE, and binary restenosis were noted with both stents in a complex patient population (28% diabetics, mean reference vessel diameter 2.4 mm, mean lesion length 17 mm, 86% American Heart Association/American College of Cardiology (AHA/ACC) type B2/C lesions). 45 The largest study was a randomized, blinded trial in Denmark (n = 2098) by the Danish Organization on Randomized Trials with Clinical Outcome (SORT OUT II), that was conducted as a collaboration between 5 Danish university PCI hospitals. 54 Results of this study showed no significant differences in outcomes with SES and PES at 18 months (TLR: SES 4.5% vs. PES 5.9%; P = 0.14 and MACE: 10.0% vs. 11.6%; P = 0.21).
SES vs. ZES
Limited head-to-head comparative data with newer DES including the zotarolimus-eluting stent (ZES) Endeavor (Medtronic, Santa Rosa, CA) and everolimus-eluting stent (EES) Promus/Xience V (Boston Scientific, Natick, MA/Abbott Laboratories, Abbott Park, IL) exist. A Randomized Trial to Confirm the Safety and Efficacy of the Zotarolimus Eluting Coronary Stent in De Novo Native Coronary Artery Lesions (ENDEAVOR III) compared the ZES Endeavor (n = 323) with the SES Cypher (n = 113). Though the 8-month primary endpoint of in-segment late loss, with a non-inferiority margin of 0.20 mm, was not met (ZES 0.34 ± 0.44 mm vs. SES 0.13 ± 0.32 mm, P < 0.001), 9-month clinically driven TLR (ZES 6.3% vs. SES 3.5%, P = 0.34), MACE (ZES 7.6% vs. SES 7.1%, P = 1.0) and target vessel failure (ZES 12.0% vs. SES 11.5%) were similar between the two stents. 55
SES vs. EES
A multicenter, randomized, prospective study, the Efficacy of Xience/Promus versus Cypher in rEducing Late Loss after stENTing (EXCELLENT) is ongoing in Korea and will enroll 1400 patients to compare EES vs. SES for angiographic late loss, and for target vessel failure. 56
Registries
The SES registries (Table 4) were designed to evaluate clinical outcomes with the use of SES in higher-risk and more heterogeneous patient populations than those enrolled in clinical trials. Important considerations for evaluating DES registry data include the number of centers (range 1 to >100), the proportion of specific high-risk patients and lesions, the limited and highly variable data monitoring practices, angiographic versus clinical follow-up and the variable definitions of endpoints across registries.
One-year clinical outcomes in SES registries–-use of SES in routine interventional cardiology practice.
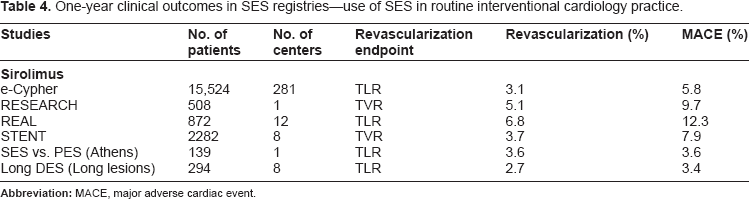
Some of the largest and most published registries include the international e-Cypher SES registry (n = 15,524), 57 the Strategic Transcatheter Evaluation of New Therapies (STENT) registry (n = 2282), 58 the Rapamycin-Eluting Stent Evaluated at Rotterdam Cardiology Hospital (RESEARCH) registry (n = 508), 59 German Cypher stent registry (n = 1726), 60 and the Italian Cypher Registro Regionale Angioplastiche (REAL) registry (n = 872). 61 Most of these registries include patients with diabetes, multivessel disease, in-stent restenosis, acute myocardial infarction (MI), and chronic total occlusions. 57
One-year follow-up from these and other DES registries has demonstrated the efficacy of SES in limiting the need for repeat target vessel revascularization (TVR) to less than 10%, with 1-year MACE rates ranging from 3.1% to 12.3% (Table 4). Among these registries, the STENT registry is one of the larger prospective, multicenter, concurrent, consecutive-enrolling, registry of patients treated with either the SES or PES. The results from this registry show comparable rates of 9-month TVR (SES 3.7% vs. PES 2.9%, P = 0.13) and MACE (SES 7.9% vs. PES 6.8%, P = 0.2) 58 with both stents.
High-risk Populations
The presence of diabetes, longer lesion length, and narrower luminal diameter are associated with higher restenosis with BMS. 36 Data regarding the effectiveness of DES in such patients and lesion types are available from randomized clinical trials specifically enrolling such patients, substudies of larger randomized trials, and real-world registries. In addition, patients presenting with ST elevation MI (STEMI) comprise a cohort of patients in whom the efficacy and safety of utilizing DES had been questioned until multiple recently completed clinical trials enrolling such patients. Investigation into the efficacy of SES in saphenous vein graft, left main, multivessel, and bifurcation stenting is ongoing.
Diabetes
Diabetic patients undergoing PCI with BMS have higher restenosis rates than nondiabetics.62–64 Most of the evidence for DES efficacy in diabetic patients comes from subgroup analyses of pivotal trials and registries. As we review these data, it is important to note that the antimigratory and antiproliferative effects of paclitaxel on smooth muscle cells have been shown in an in vitro model of insulin resistance, while this effect appears to be attenuated with sirolimus in the presence of hyperglycemia. 65 This differential response may lead to variable antirestenotic effect of these drugs in diabetics.
The TLR rate among DES-treated diabetic patients in the pivotal trials of SES (6.9%) and PES (5.1%) were only marginally higher than the TLR rates for the overall study population (4.1% SES and 3.1% PES).2,4 In general, the relative risk reduction in TLR for diabetics treated with DES was similar to the overall study population, though with higher absolute TLR rates.2,4 In an indirect comparison of these two DES from 10 trials, clinical outcomes with the two DES were comparable in diabetic patients. 66 Similarly, among 2830 diabetic patients in the STENT Registry, TVR rates were comparable with both stents (4.3% SES vs. 4.1% PES, P = 0.76).67,58
A meta-analysis of clinical efficacy of DES in diabetic patients analyzed all randomized controlled trials (RCT) and 16 registries (from 1/2002–2/2007) to determine MACE and revascularization outcomes in diabetic patients with SES or PES. 68 Pooled analysis of RCTs (n = 2422) showed similar TLR and MACE for SES vs. PES [TLR: 7.6% (95% CI 5.8% to 9.9%) vs. 8.6% (95% CI 6.5% to 11.3%); MACE: 12.9% (95% CI 8.5% to 19.2%) vs. 15.4% (95% CI 12.4% to 19.1%)] (Fig. 4). Pooled analysis of 16 registries (n = 10,156) included in this analysis showed similar TVR and MACE for SES vs. PES [TVR 7.2% (95% CI 4.5% to 11.2%) vs. 5.8% (95% CI 3.9% to 8.5%); MACE: 11.9% (95% CI 8.5% to 16.4%) vs. 10.1% (95% CI 7.3% to 13.8%)] (Fig. 5). The Swedish Coronary Angiography and Angioplasty Registry (SCAAR) evaluated the rate of clinical/ischemia driven TLR of different DES in patients with and without diabetes (n = 19,004). 69 Clinically driven TLR was 3.3% with SES Cypher at one-year and 4.9% at 2 years. The SES Cypher had a 30% higher risk of restenosis in diabetics than in non-diabetics. However, SES Cypher and PES Taxus Liberte had similar and lowest TLR (4.6% Cypher, 4.1% Taxus Liberte). These data suggest that the use of SES Cypher and PES Taxus in diabetics results in similar clinical outcomes.
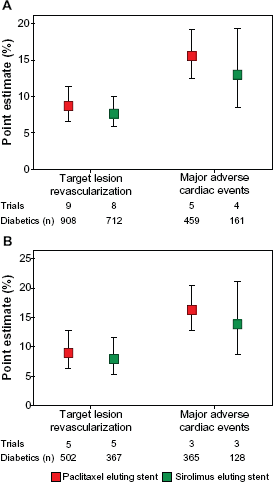
Clinical event rates for diabetic patients treated with DES in RCTs. Point estimates for target lesion revascularization and major adverse cardiac events for diabetic patients treated with drug-eluting stents (DES) in (A) all randomized controlled trials (RCTs) and (B) RCTs with Silber score > 5. Bars indicate 95% confidence intervals. Reprinted with permission from Mahmud et al. J Am Coll Cardiol 2008;51:2385–95.
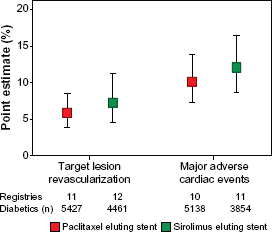
Clinical event rates for diabetic patients treated with DES in registries. Point estimates for target vessel revascularization and major adverse cardiac events for diabetic patients treated with DES in all registries. Bars indicate 95% confidence intervals. Reprinted with permission from Mahmud et al. J Am Coll Cardiol 2008; 51:2385–95.
Long lesions
Though increased lesion length is associated with higher restenosis rates, there is no agreed upon definition of a long lesion. Lesion length >20 mm has been used as a criterion for a long lesion in studies specifically evaluating the use of DES in such long lesions. In the Korean Long DES trial, a prospective, non randomized trial of 527 patients with SES, PES or BMS, with average lesion length greater than 32 mm, SES had lower TLR (SES: 3.8% PES: 6.0% BMS 19.2%, P = < 0.001). 70 The same investigators corroborated the SES TLR results with Long DES-II, a randomized trial of 500 patients with either SES or PES with average lesion length greater than 33.9 mm (TLR for SES: 2.4%, PES: 7.2%; P = 0.012). 71
Current limited evidence suggests that in lesions longer than 20 mm, SES is superior to PES. A few caveats include (1): most of the studies showing SES superiority were performed in Korea and have not been confirmed in other countries, and (2) PES has hitherto been Taxus Express in most studies, and only now are more data being accumulated on high risk lesions with the second generation Taxus Liberte stent.
Small vessels
Neointimal tissue generated after stent implantation accounts for a larger proportion of the lumen area in smaller diameter vessels. Evidence regarding DES efficacy in small coronary vessels (<2.75 mm) comes from small, randomized SES trials,5,6,72–74 one randomized trial comparing SES and PES in small vessels (<2.80 mm), 75 and subgroup analyses from randomized trials and registries of SES.1–4,7,76,77 Revascularization rate in small vessels ranged from 1.3% to 17.7%, with the highest rates occurring in the PES treated group in a subgroup analysis (n = 370) of the SIRTAX trial, (2 year TLR: SES: 6% vs. PES: 17.7%; P < 0.001). 77 No differences in death or MI were reported.
Acute myocardial infarction
A recent meta-analysis analyzed 13 randomized trials and 18 registry studies from 2004 to 2008 comparing DES to BMS in patients with STEMI. 78 Of the RCTs, 6 studies were for SES alone (n = 1192), 3 studies were for both SES and PES (n = 635), and 4 trials were for PES alone (n = 2688). In 4 registries data were reported for SES alone (n = 650), while the other 14 registries reported outcomes for both SES and PES (n = 11,216). Data from the pooled RCTs and registries showed no difference in the rates of death, repeat MI, and stent thrombosis up to 36 months. Importantly, significant reductions in TVR were reported in RCTs (DES: 5.3%; BMS: 11.5%; P < 0.001) and registries (1 year: RR: 0.54; 95% CI 0.40 to 0.70 and 2 year: RR:0.71; 95% CI 0.61 to 0.83).
Peripheral arterial disease
The role of DES in the peripheral vasculature remains largely undefined. In Europe, the SES Cypher has been approved for treatment of infrapopliteal lesions in severe claudication and critical limb ischemia, and initial clinical and angiographic data seem promising.79,80 In the Sirolimus-Coated Cordis Self-expandable Stent (SIROCCOI/II) trials, 47 patients received sirolimus-eluting self-expanding nitinol stents (SES SMART) and 46 patients received uncoated self-expanding nitinol stents (bare SMART) in the superficial femoral artery.81,82 At 24 months, restenosis and TLR were equivalent (restenosis: SES SMART: 22.9% vs. bare SMART: 21.1%; P = 1.0 and TLR: SES SMART: 6% vs. bare SMART: 13%; P = 0.30).
Other high-risk populations
Treatment of saphenous vein grafts (SVG), left main coronary artery (LMCA) disease, chronic total occlusions (CTO), in-stent restenosis and bifurcation lesions continue to pose substantial challenges.
Saphenous vein graft
Treatment of SVGs has been excluded from randomized DES trials, but preliminary evidence from four small, uncontrolled studies of DES to treat SVG disease is encouraging with revascularization rates of 2.0% to 8.6% at 6 to 12 months follow-up.83–86,60 In one small randomized Reduction of Restenosis. In Saphenous vein grafts with Cypher (RRISC) trial (n = 75) long-term (3-yr) mortality was higher in patients with SES (n = 11, 29%) than BMS (n = 0) implanted in vein grafts. 87 However, this association has not been seen in any of the registry data as well as single center retrospective data. 88
Left main coronary artery
Three small, single-center registries of DES implantation for LMCA disease have demonstrated relatively low MACE (14% to 24%) and TLR (2% to 14%) rates at 6–12 months, including an evaluation that found SES and PES to be comparable in this high-risk lesion subset.89–92 A recent registry study from Korea (MAIN-COMPARE) demonstrated that PCI of left main is noninferior to CABG in terms of death, Q-wave MI and stroke, although TVR was higher in PCI than in CABG. 93 In the PCI group 71.1% (n = 784) received DES of which 77.4% (n = 607) were SES and 22.6% (n = 177) were PES.
The Synergy between PCI with Taxus and Cardiac Surgery (SYNTAX) study was a prospective, randomized study that provided further insight into the relative efficacy of PES relative to CABG for LMCA disease. One year results show that rates of death and MI were similar between the two groups. However, stroke was significantly higher with CABG (2.2% vs. 0.6%, P = 0.003) whereas repeat revascularization was higher with PCI (13.5% vs. 5.9%, P < 0.001). 94 Although SES was not included in SYNTAX, registry data such as the German Cypher Registry suggest similar outcomes. In the German Cypher Registry, 82 patients were treated for unprotected LMCA vs. 118 patients for protected LMCA with SES. Cumulative composite all-cause death, non-fatal MI, and TVR was similar at 6 months (unprotected: 14.1%, protected: 13.1%) and 3 years (unprotected: 11.8%, protected: 20.2%). 95 Another recent randomized trial, the Unprotected Coronary Left Main Lesions Study Investigators (ISAR-LEFT-MAIN), reported similar MACE (SES: 15.8%, PES: 13.6%; P = 0.44) with both Cypher and Taxus stents. 96
Multivessel stenting
The first study to show a possible benefit for the SES Cypher vs. CABG was the Arterial Revascularization Therapies Study-II (ARTS-II). 97 ARTS-II was a multicenter, non-randomized trial that compared multivessel SES Cypher (n = 607) to a historical CABG control cohort (n = 605) from the prior ARTS I trial (multivessel bare metal stent vs. CABG). 98 Major adverse cardiac or cerebrovascular events were favorable for SES vs. CABG (6.4% vs. 9%, P < 0.05) at 6 months. The Future Revascularization Evaluation in Patients With Diabetes Mellitus: Optimal Management of Multivessel Disease (FREEDOM) trial has enrolled patients since 2004, and will complete enrollment in December of 2011. This is a prospective, randomized, superiority trial enrolling 2400 diabetic patients with multivessel coronary disease randomized to either CABG or PCI (SES or PES). Evaluating separate and composite MACCE endpoints will be informative.
Bifurcation lesion
Although small studies of DES implantation in bifurcation lesions have demonstrated lower clinical and angiographic restenosis rates than BMS, the issues of optimal technique (e.g. crush, T-stenting, or final kissing balloon angioplasty) and side branch strategy (stenting or plain balloon angioplasty) remain controversial.99–103 Currently, most investigators favor a provisional side branch stenting strategy for bifurcation lesions. 104 In the Coronary Bifurcation Application of the Crush Technique Using Sirolimu-Eluting Stents (CACTUS) study, where 350 patients with bifurcation lesions were prospectively enrolled and randomized to provisional versus crush technique, Colombo et al, suggest provisional stenting outcomes (MACE:15% vs. 15.8%; P = NS) are similar to crush technique outcomes. 105
Chronic total occlusions
For chronic occlusions, several small trials of DES in recanalized CTOs have demonstrated substantial reduction in clinical and angiographic restenosis compared with BMS.106–108
In most of these studies evaluating the use of SES in highly complex lesions, SES use has led to lower restenosis compared to BMS without a higher risk of death, MI, or stent thrombosis, except for the “crush” technique in bifurcation lesions, which is associated with a stent thrombosis rate of 3.5%. 99
Acute and Long-term Safety
Short-term safety
The safety of DES can be evaluated in terms of cardiac death, MI, and stent thrombosis. Randomized trials, observational registries, and meta-analyses have demonstrated similar and low rates of death, MI, and stent thrombosis between DES and BMS at 9 to 12-month follow-up.1,2,57,109–112
Long-term mortality
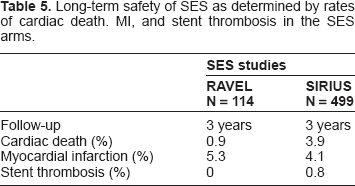
The three-year rates of cardiac death, MI and stent thrombosis are also comparable with both SES and PES treated patients (Table 5).113–116 At three years, in RAVEL (n = 114), cardiac death was 0.9% and in SIRIUS (n = 499), cardiac death was 3.9%. In one study using SES (n = 233), independent long-term predictors of death included: depressed LV function, proximal LAD lesions, age, female gender, use of glycoprotein 2b3a inhibitors, and subsequent acute myocardial infarction. 117 The concern that very late stent thrombosis may also increase long-term mortality is discussed below.
Long-term safety of SES as determined by rates of cardiac death. MI, and stent thrombosis in the SES arms.
Very late stent thrombosis
A small but significant increase in death and MI 18 months after DES implantation was noted in the Basel Stent Cost-effectiveness Trial-Late Thrombotic Events (BASKET LATE) Trial that first raised concern about very late stent thrombosis as an underlying mechanism for these events. 118 While this difference in death or MI was not significant 24 month after stent implantation, the concern over DES safety persisted. Assessment of the long-term risk of stent thrombosis with DES has been challenged by the lack of a consistent definition of stent thrombosis, the lack of adequately powered trials to assess this endpoint, and the multifactorial etiology of stent thrombosis. Randomized trials with SES and PES have used different definitions of stent thrombosis with respect to the adjudication of events such as sudden unexplained cardiac death and MI in the territory of the target vessel. In the PES trials such late events in the absence of angiographic confirmation have been considered to be stent thrombosis while in most SES trials angiographic confirmation of stent thrombosis has been required.1–4,7 In 2006, the Academic Research Consortium (represented by interventional cardiologists, FDA, academic, and industry representatives) aimed to standardize the definition of stent thrombosis, and increase the consistency and reporting of this end-point (Table 6). 119
Academic research consortium definition of stent thrombosis.

Redefining stent thrombosis in pivotal trials
Based on the ARC definitions, stent thrombosis rates of the 4 pivotal SES trials, RAVEL, SIRIUS, E-SIRIUS and C-SIRIUS in addition to the 4 pivotal trials of PES, have been reported. 120 Based on these ARC definitions for the 4 SES trials, definite or probable stent thrombosis is similar between SES and BMS at early, late, and very late time points (Table 7).
Cumulative incidence stent thrombosis at 4 years.

Predictors of stent thrombosis with DES are similar to BMS, including premature cessation of antiplatelet therapy, chronic renal failure, diabetes, bifurcation lesion, prior brachytherapy, stent under-expansion, overlapping stents, longer total stent length, and potentially aspirin or clopidogrel resistance.121–125 Delayed endothelialization and local hypersensitivity reaction have also been described in autopsy studies of DES in humans. 126 Given the importance of stent thrombosis and its potential life-threatening ramifications, stent thrombosis registries have recently been established which should yield valuable insight.
Procedural and Long-term Pharmacology
The ACC/AHA guidelines for PCI identify anti-platelet and antithrombotic therapies as essential adjunctive therapies for minimizing periprocedural adverse cardiac events. The evidence supporting pre-procedure antiplatelet therapy with aspirin (75 to 325 mg) and a loading dose of clopidogrel (300–600 mg) is substantial. 127 Adjunctive antithrombin therapy with heparin (unfractionated or low-molecular weight), or bivalirudin is also required. Use of the glycoprotein IIb/IIIa inhibitors appear to be most beneficial for patients not pre-treated with clopidogrel or those presenting with acute coronary syndromes. 127 The optimal duration of dual antiplatelet therapy after DES implantation is not established. The 2007 focused ACC/AHA guidelines for PCI recommend 12 months of clopidogrel after DES implantation for all DES (Level of evidence, Class Ib). 128 Beyond 12 months, benefit of long term dual antiplatelet therapy with aspirin and clopidogrel must be weighed against the risk of increased major bleeding. 129 The Dual Antiplatelet Therapy (DAPT) study is currently enrolling patients to compare outcomes of patients on 12-months versus 30-months of dual antiplatelet therapy after DES (trial: NCT00977938).
Premature cessation of antiplatelet therapy may contribute to stent thrombosis and increased mortality. Among 500 DES-treated MI patients in the Prospective Registry Evaluating Myocardial Infarction: Events and Recovery (PREMIER) study, 68 patients (13.6%) had discontinued thienopyridine therapy within 30 days of stent implantation, subsequently associated with increased mortality. 130
Additionally, a marked variability in platelet inhibition response to clopidogrel therapy has been shown to exist. 131 The presence of clopidogrel hypore-sponsiveness and its role in the development of stent thrombosis or late ischemic events is currently being examined.132,133 Additional prognostic markers such as fibrinogen in diabetics, higher BMI, CYP2C19 polymorphisms, may also better predict responders versus non-responders to clopidogrel.134,135 Medications that effect the CYP 450 metabolism of the clopidogrel prodrug such as omeprazole may also need to be discontinued. 136 However, recent analyses of data from TRITON-TIMI 38 suggest no association between PPI use and adverse clinical outcomes in patients treated with stents and dual antiplatelet therapy. 137 In addition, genetic predisposition to clopidogrel hypo-responsiveness may be present with CYP 2C19 polymorphisms which can lead to inadequate conversion of the prodrug to its active metabolite. 135 Prasugrel, a second generation thienopyridine leads to a more consistent and predictable inhibition of platelet reactivity and might be a better alternative to clopidogrel after DES. 138
Conclusions
In summary, this comprehensive review of the SES Cypher data suggest that use of this stent leads to acceptably low rates of repeat revascularization and MACE in a wide variety of patient populations and complex lesion subtypes. Given how well the SES Cypher has performed thus far, we look forward to using next generation SES Cypher with even better outcomes.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
