Abstract
Due to the growing rate of multi-drug resistant bacteria in complicated infections, the need for new broad-spectrum antimicrobials is paramount. Doripenem, a new addition to the intravenous carbapenem class, has recently been approved for the treatment of complicated lower urinary tract infections and/or pyelonephritis (cUTI) and complicated intra-abdominal infections (cIAI) in adult patients. Doripenem exhibits potent
Introduction
In the last decade, a global rise of multi-drug resistant organisms has dramatically reduced the number of available antibiotic treatment options.1–5 Patients infected with multidrug resistant organisms commonly have poor clinical outcomes, prolonged hospital stays, and greater hospital costs when compared with antibiotic susceptible strains,6,7 thus posing a serious health care concern in critically ill patients. This scenario is evermore present in difficult to treat infections, such as complicated intra-abdominal (cIAI) and complicated urinary tract infections (cUTI). When such infections are not treated appropriately within a short timeframe (i.e. ≤24 h), they have often led to further complications that may increase morbidity and mortality.
Complicated IAI are best described as infections that initiate at a foci and disseminate to the peritoneal space. Consequently, this leads to peritonitis and abdominal abscesses.8,9 Moreover, the pathogens responsible for this occurrence of cIAI depend on the location at which it was acquired, such as the community or hospital, and upon the location within the body in which the infection originated. For the most part, Enterobacteriaceae (i.e.
The need for broad-spectrum agents has also been important in treating cUTIs10–12 because the tendency for patients to be infected with resistant organisms is higher than in uncomplicated urinary tract infection.
10
Patients with cUTIs are those with a structural or functional abnormality of the genitourinary tract.11,12 The most common pathogen is
In spite of this growing epidemiologic and microbiologic problem, the carbapenem class has retained their activity to most bacterial pathogens and their resistance mechanisms.4,5,14 Recently, a newly developed 1-β-methyl carbapenem, doripenem, received United States Food and Drug Administration (US FDA) approval for the treatment of cUTI and cIAI. 15 This paper will review doripenem characteristics as they pertain to the pharmacology, microbiology, and treatment of cUTI and cIAI.
Pharmacology and Mechanism of Action
Doripenem is a parenteral, broad-spectrum antibiotic with a methyl group at the 1-position that allows for stability when it undergoes renal excretion. Moreover, doripenem also possesses a unique 2-position side chain that provides stability against β-lactamases and added affinity to the binding site.16–18 As depicted in Figure 1, doripenem has a similar structure to other carbapenems. It has a 4-member lactam ring that is bound to a 5-member thiazolidinic ring; however, it is unique with the addition of a sulfamoylaminoethyl-pyrrolidnylthio group side chain on position 2.16,19 It is thought that this addition increases the acidity of doripenem, which in turn provides for its enhanced
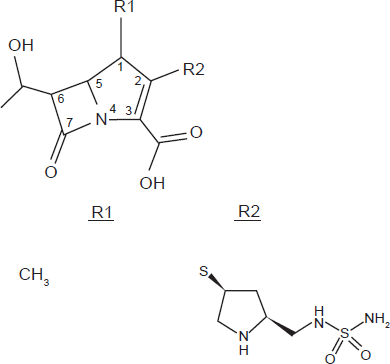
Doripenem chemical structure. R1) Methyl-group: Increases stability toward hydrolysis by dehydropeptidase-I (DHP-I). R2) Side Chain: Confers high level of
Similar to other class members, doripenem achieves bactericidal activity by inhibiting the penicillin binding protein (PBP).
18
The PBP is responsible for maintaining the cell shape of the organism by elongating and cross-linking the peptidoglycan within the bacterial cell wall. When the cell wall is disrupted, it breaks down, which leads to cell lyses and ultimately cell death. Based on the
Microbiology
Analogous to other carbapenems, doripenem possesses
Susceptibility/resistance breakpoints for anti-pseudomonal carbapenems as defined by CLSI, FDA, and EUCAST.
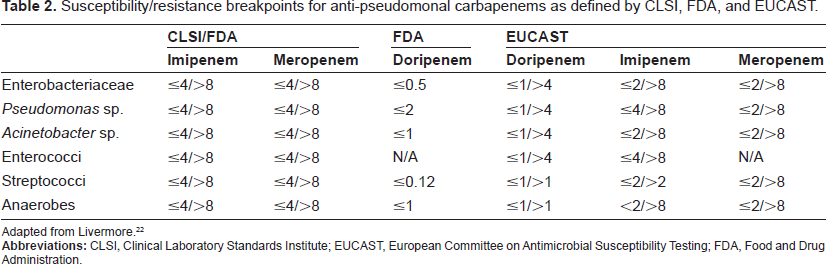
Adapted from Livermore. 22
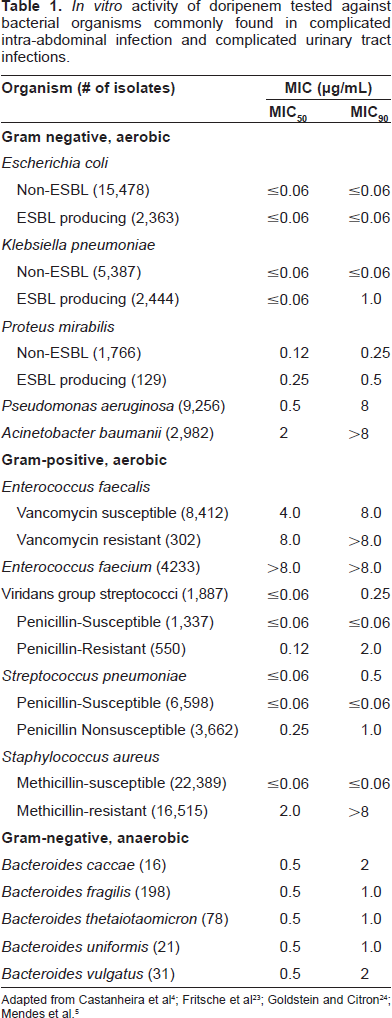
When observing the activity of doripenem against the US derived non-lactose fermenting Gram-negative organisms, doripenem inhibited 84 and 88% of
The activity of doripenem against global Enterobacteriaceae isolates was more than ≥99.5% at both the US FDA and EUCAST MIC breakpoints of ≤0.5 μg/mL.
14
This activity was comparable with meropenem in each Enterobacteriaceae organism tested but was decidedly better than imipenem against the majority of organisms except
In a global surveillance study that evaluated the
When doripenem was evaluated against a variety of anaerobic organisms, it demonstrated activity to all except
Mechanisms of resistance
Similar to other carbapenems, doripenem has a stable structure against most β-lactamase enzymes (e.g. AmpC and ESBL). However, doripenem's ability to resist hydrolysis were not shared against β-lactamases of Ambler class A (KPC and SME), C (VIM), and D (OXA).3,5,14,25,26 The organisms commonly in possession of these carbapenemases are the non-lactose fermenting Gram-negative bacilli (i.e.
Other mechanisms (e.g. loss of porin channels and efflux overexpression) have also had a similar result on doripenem's susceptibility profile to that of meropenem.27,28 For instance, Queenan and colleagues observed carbapenem resistance in
Pharmacokinetics and Pharmacodynamics
Pharmacokinetics
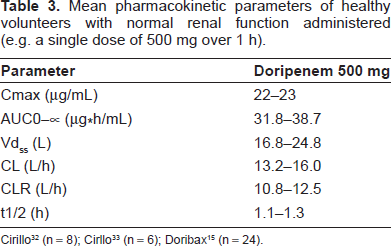
Doripenem displays linear and dose proportional pharmacokinetics when dosed at a range of 250 mg to 1000 mg given over a 0.5, 1, or 4 h infusions. 31 During phase I studies, single or multiple doses (2 or 3 times daily) were used to total a maximum dose of 3000 mg per day (1000 mg every 8 h). The mean plasma pharmacokinetic parameters of a single 500 mg dose (1-h intravenous infusion) are displayed in Table 3. In the studies, the time in order to achieve maximum concentration (Tmax) immediately followed the end of infusion.15,32,33 When compared to the mean Cmax after a 1-h infusion, the 4 h infusion was nearly one-third lower (~8 μg/mL) using the same 500 mg dose; 34 this led to a mean AUC from time 0 to infinity for both infusions at ~34 μg*h/mL. 34
Based on a variety of different sources,34–36 doripenem concentration-time profiles are best described by a 2-compartment model with zero order input and 1 st order elimination. When doripenem was dosed over a course of 7-10 days, no accumulation was observed within each of the cohorts. 31 At steady state, doripenem 500 mg has displayed a median volume of distribution of 16.8 L in healthy volunteers; this is similar to the extracellular fluid volume of ~18 L.15,33 Though research is limited to a few areas of the body (i.e. retroperitoneal fluid, peritoneal exudate, gall bladder, bile, and urine), doripenem has exhibited good tissue distribution with concentrations at or above the MICs of susceptible organisms.15,33,37,38 Moreover, these concentrations appear to be mostly free drug, based on the 8.5% plasma protein binding exhibited by doripenem. 35 In a study by Ikawa et al, doripenem dosed at 500 mg over 0.5 h, had concentrations extensively and rapidly reach the peritoneal fluid in non-infected patients undergoing abdominal surgery. The results showed that 82% of doripenem (based on an AUC0-∞) reached the peritoneal fluid. 37
Doripenem is eliminated renally by glomerular filtration and active tubular secretion with no indication of hepatic metabolism (i.e. CYP 450 enzymes).15,33 When healthy males were administered a single 500 mg dose over 1-h, ~94% of doripenem was recovered in the urine over 24 h; the majority of doripenem was unchanged drug (~75%) with the remainder as the inactive metabolite, doripenem M-1 (~19%). Feces were also determined to be a route of elimination for active doripenem; however, it was very minimal at ≤ 1%. 33
In studies that observed patients with various renal functions,32,39 it was determined that doripenem dose regimens should be adjusted based on renal function below normal creatinine clearance (CrCL). This was due to the increased exposure observed in subjects with declining renal function due to decreased glomerular filtration. In two trials, single dosages of doripenem 500 mg (0.5 or 1h infusion) were used in subjects with various rates of renal function based on CrCL. Subjects with impaired renal function experienced a ~2 and ~8-fold increase in AUC0-∞ and ~2 to ~7-fold increased half-life when compared with the normal controls (CrCL ≥ 80 ml/min). When pre-dialysis and post-dialysis were compared to each other, pre-dialysis (doripenem dosed 2 h prior to 4 h dialysis) removed ~90% of doripenem, while, post-dialysis (doripenem dosed 1 h after dialysis) removed ~52% of doripenem. To our knowledge, the above mentioned hemodialysis studies are the only pharmacokinetic information on dialysis available. As a result, more information is needed in this area, specifically looking at dose effects on peritoneal dialysis and continuous renal replacement therapy (i.e. continuous veno-venous hemodialysis and continuous veno-venous hemodialysis-filtration).
When doripenem was evaluated in a variety of healthy subjects with varying characteristics (i.e. age, sex, and race), it was determined that dosage adjustments were not required. 15 However, among the various populations observed there were subtle differences in pharmacokinetic parameters. For instance, among the various races and age groups administered doripenem, only the Hispanic and geriatric populations was dissimilar when the mean CL was shown to be increased by >14% and mean AUC0-∞ was 49% higher, respectively. 15 Incidentally, the differences in the geriatric population were regarded to be attributable to age-related CrCL changes.
Pharmacodynamics
Based on various murine thigh infection models40–42 and a time-kill study,
43
doripenem has demonstrated rapid bactericidal time-dependent activity against Gram-positive and Gram-negative organisms. The
In animal studies, free drug concentrations above the MIC (
In various published in silico modeling studies, optimal dosing schemes to maximize doripenem exposures in blood,35,36 blood + urine,
44
and blood + peritoneal fluid
38
were generated (Table 4). However, in two of the studies,35,38 the population pharmacokinetic parameters were taken from healthy subjects with normal renal function (mean CrCL in both studies ≥ 80 ml/min) and it was thought that these simulated dosing regimens may not reflect populations with impaired renal function or infected patients. To correct for these limitations, two studies used subjects with varying renal function with and without concomitant infection.36,44 These results were similar to healthy subject data; a dose at 500 mg every 8 h administered as 1 and 4 h infusions had a ≥90% probability of achieving a
A comparison of pharmacokinetic/pharmacodynamic breakpoints for doripenem regimens in 4 different patient population studies.
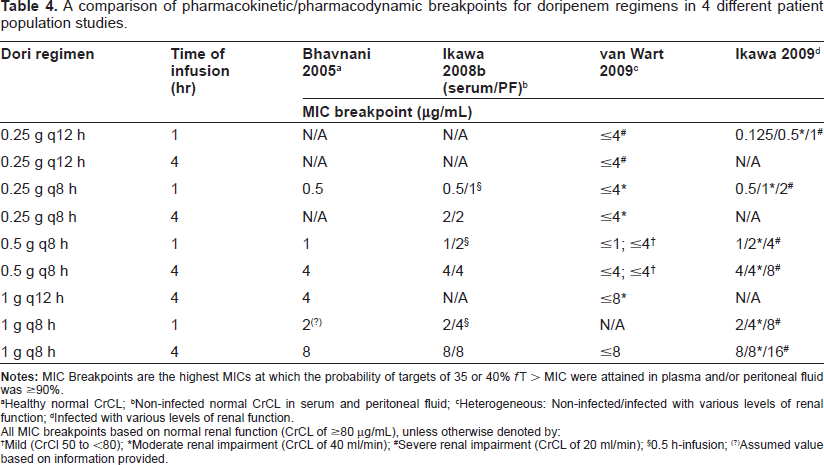
Healthy normal CrCL;
Non-infected normal CrCL in serum and peritoneal fluid;
Heterogeneous: Non-infected/infected with various levels of renal function;
Infected with various levels of renal function.
All MIC breakpoints based on normal renal function (CrCL of ≥80 μg/mL), unless otherwise denoted by:
Mild (CrCl 50 to <80);
Moderate renal impairment (CrCL of 40 ml/min);
Severe renal impairment (CrCL of 20 ml/min);
0.5 h-infusion;
Assumed value based on information provided.
In another pharmacodynamic study, urinary bactericidal titers (UBT) and area under the 24 h urinary bactericidal titers curve (AUBT) were used to determine the pharmacodynamic parameters of doripenem in urine. 45 Wagenlehner and colleagues used data and clinical isolates from Naber and colleagues’ phase III clinical trial to determine and compare the UBT and AUBTs of doripenem and levofloxacin. Median results showed that doripenem had significantly higher UBT and AUBT than levofloxacin for 6 of 7 isolates at a range of 1.5 to 65,536 and 224 to 909,312 compared with 0 to 128 and 0 to 2,048. UBTs and AUBTs have correlated well with microbiological failures in the case of levofloxacin, suggesting a target attainment rate of UBTs of at least 100/24 hours. There was however no correlation of microbiological failures and UBTs and AUBTs in the case of doripenem. However in the majority of those cases non-resolution of the complicating urological factors could be attributed to failures.
Clinical Studies
Based on the available clinical trials data conducted in patients with cIAI46,47 and cIAI (DORI-06),11,48 doripenem received US FDA approval for the treatment of the aforementioned infections. Other clinical trials have been published for the treatment of ventilator associated pneumonia and nosocomial pneumonia; however, doripenem has not yet received approval for the treatment of either indication, although the results were promising.49,50 In Europe, doripenem is approved for UTI, IAI, as well as hospital-acquired pneumonia.
Complicated intra-abdominal infections
Doripenem's efficacy and safety for the treatment of complicated intra-abdominal infections was determined in an international, multicenter, double-blinded, randomized controlled Phase III trial. In this study,
46
476 adult patients (≥18 years of age) were randomized to receive doripenem 500 mg IV every 8 h as a 1 h infusion (n = 242) or meropenem 1000 mg every 8 h as a 3 to 5 min bolus (n = 233). In patients with a CrCl < 50 ml/min, dose adjustments were made in both treatment arms. In line with the difference in drug administration durations, patients also received a dummy placebo for control either before administering meropenem or after doripenem. Once a minimum of 9 doses were received and clinical improvements were observed, patients were allowed to switch from study drug to oral amoxicillin-clavulanate 875/125 mg bid for the remainder of the 5 to 14 day treatment course. The co-primary efficacy end points were the clinical cure rates of patients infected with ≥1 bacterial organism that were susceptible to both study drugs at the test of cure (TOC) visit (21-60 days post-completion of study drug therapy) and those patients identified with an infection regardless of susceptibility. The other endpoints included clinical cure rates in clinically (CE) and microbiologically evaluable (ME) patients at the end of study drug therapy, early follow-up (1 to 2 weeks after treatment), and TOC visit. Patients were stratified by region and within each region, by primary site of infection and APACHE II score (≤10 vs. > 10). Clinical cure rates were 85.9% and 85.3% (0.6% difference; 95% confidence interval, -7.7% to 9.0%), respectively. In the microbiological modified intent to treat (mMITT) population, the clinical cure rates were at 77.9% and 78.9% (-1.0% difference; 95% CI, -9.7% to 7.7%) for doripenem and meropenem, respectively. In each of the outcomes observed, doripenem met non-inferiority (lower limit of the 2-sided 95% CI for the difference in the clinical cure rates was ≥-15%) to meropenem. Both study drugs were administered for an average of 6.6-6.8 days, while the oral formulation was continued for approximately 10.3-10.4 days. Among the three most common organisms found at baseline (
In another cIAI trial, Solomkin and colleagues expanded the number of subjects by pooling the phase III study by Lucasti and colleagues with another similar clinical trial in cIAI by Malafaia.
51
In this study,
47
doripenem again demonstrated non-inferiority to meropenem at 84.6% versus 84.1% (+0.5% difference; 95% CI -5.5% to 6.4%) in the ME population, respectively. In the mMITT population, the clinical cure rates were 76.2% versus 77.3% (-1.1% difference; 95% CI -7.4% to 5.1%), respectively. Doripenem demonstrated numerically higher clinical cure rates compared with meropenem in patients with higher APACHE II scores, but this group was too small for a statistical assessment. Microbiological cure rates in the ME population were again similar between doripenem and meropenem at 84.3% versus 84.5%, respectively. Microbiologial cure rates were also similar in the by-isolate assessment for
Lower complicated urinary tract infections and pyelonephritis
During 2 phase III multi-center, clinical trials, doripenem 500 mg every 8 h infused over 1 h was administered to patients for at least 9 doses before the option of a treatment switch to oral levofloxacin 250 mg daily was offered for the remainder of the 10 day treatment course in clinically improving patients. Between the 2 trials, one study was a non-comparative single armed trial (DORI-06); 48 meanwhile, the trial by Naber et al 11 was double dummy designed comparing doripenem and levofloxacin 250 mg IV daily at a 1 h infusion. In both studies, non-inferiority was defined if the lower limit of the 2-sided 95% CI for the treatment difference (doripenem minus levofloxacin) with respect to microbiological cure (eradication of the baseline pathogen) at TOC (5-11 days in comparative study or 6-9 days in non-comparative study) in the ME and mMITT was ≥-10%. Among the two studies, a total of 795 patients were given ≥1 dose of doripenem (n = 423 in non-comparative and n = 372 in comparative), while 376 patients were given ≥1 dose of levofloxacin.
Demographic data of all cohorts in the ME at TOC population were similar with the majority of patients being Caucasian females at ~52 years of age. In the comparative study, an equal amount of patients had either pyelonephritis or cUTI, with ~8% of the ME population having bacteremia. Doripenem was non-inferior to levofloxacin in both studies, as shown by the respective agents microbiological cure rates in ME at 82.1% vs. 83.4% (-1.3% difference; 95% CI -8.0% to 5.5%) for the comparative study and 83.6% vs. 83.4% (+0.2% difference; 95% CI -6.6% to 7.0%) for non-comparative study. Doripenem also demonstrated non-inferiority to levofloxacin in the mMITT population for both studies with the microbiological cure rates at 79.2% vs. 78.2% (+1.0% difference; 95% CI -5.6% to 7.6%) for the comparative study and 82.5% vs. 78.2% (+4.3% difference; 95% CI -2.1% to 10.7%) for the non-comparative study, respectively. Clinical cure rates at TOC within each of the doripenem groups in comparative and non-comparative studies were 95.1% and 93.0%, respectively. Within the ME patients at TOC, the eradication rates of
Safety and Tolerability
Doripenem was well tolerated in phase I-III clinical studies. As shown in Figure 2, the adverse events reported in phase III trials were generally mild with most subjects commonly experiencing: headache, diarrhea, phlebitis, elevated hepatic enzymes, rash, nausea, and anemia.11,15,48–50
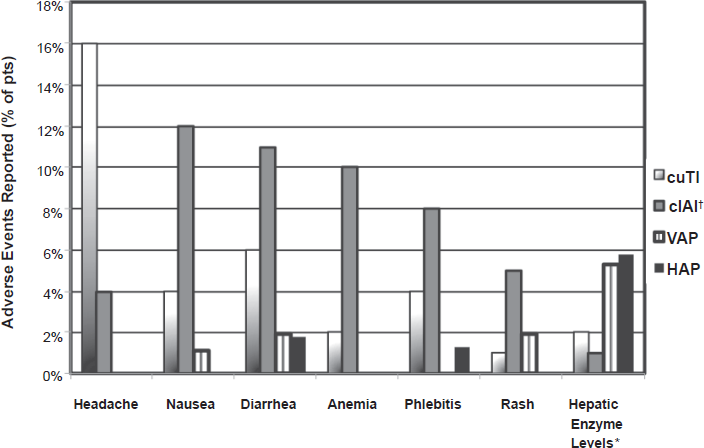
Bar graph representing the% patients reporting a drug-related adverse event to doripenem from five phase III clinical trials.
Serious adverse events, although limited were: hypersensitivity reactions,
In pregnant women, doripenem has been given a rating of category B. 15 This is in response to doripenem not showing teratogenicity and not producing any adverse effects following its administration in pregnant animals. Unfortunately, there are no clinical trials with doripenem's use in pregnant woman, so it has been suggested that doripenem only be used when necessary. Among other specific populations, the use of doripenem in nursing mothers or pediatrics is not advised, mainly because doripenem has never been tested for safety in either population. 15 When doripenem was compared between subjects with varying degrees of renal function, no serious adverse event was reported. 32 However, based on the fact that only 6-8 patients were used in each cohort and only 1-2 doses were administered, careful monitoring should still be taken in renal impaired patients.
Drug Interactions, Compatibility, Stability
When Horiuchi and colleagues 54 examined the concurrent use of doripenem with valproic acid, it was found that doripenem had no effects on valproic acid anti-convulsive activity. However, this observation contrast those found in other studies that noticed reduction in the plasma concentration of valproic acid when coadministered with doripenem. 56 Therefore, despite their documented findings, Horiuchi and colleagues 54 suggested that doripenem, like other carbapenems, not be coadministered with valproic acid in epileptic patients. Another agent suggested to not be coadministered with doripenem is probencid. 15 Probencid has been documented to interfere with active tubule secretion of doripenem. Consequentially, this interaction reduces the renal elimination of doripenem thus reducing the CL, which in turn would increase the systemic exposure of doripenem (i.e. Cmax and AUC).
When doripenem (diluted in normal saline and dextrose injection) during Y-site administration was tested against 82 other agents for physical compatibility, it was found that doripenem was compatible with 75 agents. Among these agents, 3 combined with doripenem in dextrose 5% and 7 drugs in normal saline resulted in precipitation after at least 4 hours of testing. These agents include: diazepam, potassium phosphate, and propofol in dextrose and normal saline and 4 different amphotericin B containing drugs (i.e. amphotericin B, cholesteryl sulfate complex, lipid complex, and liposomal) in normal saline. 57
Unlike other carbapenems, doripenem can remain stable for as long as 12 h at room temperature conditions in normal saline. 21 This provides sufficient time to allow doripenem to be administered as an extended infusion, thus permitting greater exposures against organisms with higher MICs.
Place in Therapy
Within the last decade there have been higher occurrences reported of multi-drug resistant bacterial infections in various institutions both nationally,14,58,59 and globally.4,5,23,60 For instance, in certain regions of the United States, the percentage of
Doripenem is a potent carbapenem with many potential benefits and possible utilizations. However, because clinical trials are designed to determine non-inferiority, doripenem's role maybe best suited as a secondary agent in directed therapy for cUTI and cIAI, unless an institution's microbiological surveillance suggests otherwise. That being said, doripenem does possess the ability to inhibit mutant selection and often requires the need for two resistance mechanisms to result in clinical resistance. Other attributes that allow for doripenem to be a potential important agent for both cIAI and cUTI include its broad-spectrum activity, stability at room temperature, low protein binding, good tissue penetration, and mild adverse effects. As a result, doripenem is well suited, particularly as a prolonged infusion, for serious infections (e.g. nosocomial pneumonia) where multidrug resistant Gram-negatives might be suspected.
Conclusion
Doripenem is a carbapenem with potent
Disclosure
The authors have received no financial support for this review; however, Dr. Kuti is a member of the speaker's bureau for Ortho-McNeil. Meanwhile, Dr. Nicolau has received research grants, is a consultant for and is on the speaker's bureau for Ortho-McNeil.