Abstract
Cefiderocol is a unique siderophore cephalosporin antimicrobial agent that has shown promise in treating complicated urinary tract infections (cUTI). Urinary tract infections are commonly diagnosed infections with risk increasing with age and prevalence more common in women. cUTI poses a risk of recurrence and is more likely to be associated with antibiotic-resistant bacteria. The Food and Drug Administration approved cefiderocol for use as a last-line option in the treatment of cUTI including pyelonephritis. Cefiderocol has activity against all forms of carbapenemases due to its ability to overcome the mechanisms of carbapenemase resistance. Because of this, resistance to cefiderocol is unlikely to occur. Studies show cefiderocol is well tolerated among younger patients and patients greater than 65 years of age, the latter making up most of the study population. Renal dose adjustments are recommended. Dose adjustment in the presence of hepatic impairment is not recommended, as hepatic clearance represents a minor elimination pathway for cefiderocol. The ability of cefiderocol to overcome multiple resistance mechanisms makes it a novel choice in combating multidrug-resistant bacteria in the treatment of cUTI.
Introduction
Urinary tract infections (UTIs) are one of the most common infections treated by clinicians in the United States.1,2 Site of infection and the presence of risk factors can further classify UTIs as uncomplicated or complicated. 3 Complicated UTIs burden the health care system due to frequent reason for hospitalization. Specifically, the morbidity and mortality are higher in patients 65 years of age and older. 4
There is growing concern regarding the prevalence of antimicrobial resistance, and the development of antimicrobial agents to treat infections caused by multidrug-resistant (MDR) pathogens is of priority. Urinary tract infections are commonly caused by
Cefiderocol is currently available on the market under the trade name, Fetroja®. 10 The Food and Drug Administration (FDA) granted approval for cefiderocol in the treatment of cUTI caused by highly antibiotics resistant uropathogens based on clinical trials evaluating efficacy and safety of cefiderocol in cUTI.11,12 The purpose of this article is to provide a review of cefiderocol’s pharmacology, efficacy, safety, and potential clinical application.
Chemistry
Cefiderocol is a siderophore cephalosporin, making it a novel antimicrobial agent. It can be administered as an intravenous infusion after reconstitution. Cefiderocol is available in a vial containing 1 gram of the sterile, lyophilized powder that is white to off-white in appearance. 10
Clinical pharmacology
Cefiderocol has a unique chemical structure, containing both a cephalosporin core and a catechol siderophore side chain.10,13 These features enhance the ability of cefiderocol to overcome several resistance mechanisms. It has activity against all forms of carbapenemases due to its ability to overcome the three mechanisms of carbapenemase resistance (shown in Figure 1): enzyme-facilitated hydrolysis, mutation of porin channels, and efflux pumps.14,15 The catechol 2-chloro-3,4 dihydroxybenzoic acid group at the third position of the R2 side chain acts as the siderophore. It is this portion of the drug that chelates extracellular iron. Normally, bacteria secrete their own siderophores that scavenge iron from their host or environment to create complexes.
15
During times of infection, the innate immune response of the host causes sequestration of intracellularly iron so that bacteria are unable to utilize it. This leads to upregulation of the bacterial iron transport system, enhancing uptake of extracellular iron, or in this case, the siderophore-iron complex of cefiderocol.16,17 The newly formed siderophore-iron complex of cefiderocol penetrates the GNB outer membrane

Cefiderocol mechanism of action.
Pharmacokinetics/pharmacodynamics (PK/PD)
Clinical trials were used to determine the pharmacokinetic profile of cefiderocol in healthy adult subjects.18–20 Both single and multiple dosing intravenous infusions were evaluated; the pharmacokinetic parameters were found to be similar in both groups. As the dose increases, the concentration maximum (Cmax) and area under the curve (AUC) of cefiderocol increase proportionally. In healthy patients, cefiderocol has a Cmax of 89.7 mg/L and an AUC of 386 mg·hr/L, compared with that of cUTI patients with a Cmax and AUC of 138 mg/L and 394.7 mg·hr/L, respectively. The average volume of distribution is 18 L, and plasma protein binding ranges from 40% to 60%, primarily to albumin. The elimination half-life is 2–3 h, and the clearance is 5.18 L/hr. About 90% of cefiderocol is excreted unchanged renally; hepatic metabolism and fecal excretion play a minor role in excretion, 7% and 2.9%, respectively. Renal impairment is shown to impact the AUC, t1/2, and total clearance.
10
Renal dose adjustments are required with renal impairment, creatinine clearance (CrCl) less than 60 mL/min. In patients with moderate, severe, and end-stage renal disease (ESRD) disease without hemodialysis, cefiderocol accumulation increases. Cefiderocol is dialyzable, with 60% of the dose removed during a 3- to 4-h hemodialysis session. The effects that hepatic impairment has on the pharmacokinetic parameters have not been studied. Dose adjustment in the presence of hepatic impairment is not recommended as hepatic clearance represents a minor elimination pathway for cefiderocol.
10
Cefiderocol exhibits time-dependent killing similar to that of other β-lactam antibiotics. Extending the infusion interval enhances the efficacy of the drug. Cefiderocol has potent activity, both
Efficacy
The Study of Efficacy and Safety of Intravenous Cefiderocol Versus Imipenem/Cilastatin in Complicated Urinary Tract Infections (APEKS-cUTI) trial was a phase II randomized, double-blind, parallel noninferiority study comparing cefiderocol 2 g every 8 h and imipenem/cilastatin 1 g every 8 h in the treatment of cUTI, including uncomplicated pyelonephritis (Table 1).
12
A total of 452 patients were randomized in a 2:1 ratio to receive either cefiderocol or imipenem/cilastatin, and 371 patients were included in the modified intention-to-treat population. The standard duration of therapy was 7–14 days. Participants had to remain inpatient for treatment, and oral antibiotic treatment was not permitted. Twenty-four percent of patients were 75 years or older, with 66 years being the median age. Fifty-five percent of patients were female. The composite primary endpoint was clinical and microbial response at test of cure (TOC), defined as 7 (±2) days after treatment course completion. Clinical outcome was based on the investigator’s evaluation of patient’s clinical signs and symptoms, with response defined as resolution or improvement of complicated urinary tract infection symptoms present at study entry and the absence of new symptoms. Physician assessment was supported by an iterative structured patient questionnaire. Microbiological outcome was based on quantitative microbiological urine cultures. A central reference laboratory (JMI Laboratories, North Liberty, IA, USA) confirmed the isolate identification and did antimicrobial susceptibility testing against a panel of antibiotics, including cefiderocol. Microbial response was defined as 1 × 104 colony-forming units (CFU)/mL or less of GNB. The most prevalent pathogens were
Cefiderocol clinical efficacy trial study designs for cUTI patients.

APEKS,
Test of cure (TOC), defined as 7 (±2) days after treatment course completion.
The treatment difference and 95% CI were based on the Cochran–Mantel–Haenszel method.
Primary endpoint for cUTI patients. Microbiologic eradication was defined as a urine culture that showed that the Gram-negative uropathogen identified at Baseline at ⩾105 colony-forming units (CFU)/mL was reduced to <10³ CFU/mL. Test of cure, defined as 7 days after end of treatment.
The CREDIBLE-CR trial was a phase III randomized, open-label, multicentre, pathogen-focused study comparing cefiderocol 2 g every 8 h and best available treatment (BAT) in the treatment of patients with evidence of a carbapenem-resistant GNB (Table 1).
11
The patients were adult (⩾18 years) who admitted to hospital with nosocomial pneumonia, bloodstream infections or sepsis, or cUTI. Patients were allowed to have adjunctive therapies, and those therapies could be de-escalated in the setting of local susceptibility results. Escalation of therapy, however, was not permitted. The overall demographics were compared by those who had received cefiderocol
Safety
Clinical trials have shown that the side-effect profile for cefiderocol is well tolerated. In the APEKS-cUTI trial, safety was assessed through the identification of adverse events, vital sign measurements, body temperature, clinical laboratory tests (blood chemistry, hematology, and urinalysis), and electrocardiographically assessment. Plasma concentration of hepcidin and iron and iron binding were assessed as prespecified exploratory safety outcomes in patients with acute infection. Adverse events were monitored daily because patients remained in hospital during treatment administration. The commonly reported adverse events in patients receiving cefiderocol were as follows: diarrhea, hypertension, infusion site reactions, and nausea/vomiting (each in 4% of patients), constipation and rash (each in 3% of patients), headache, hypokalemia, candidiasis, cough, and elevations in liver tests (each in 2% of patients) (Table 2).
12
The CREDIBLE-CR trial found that cefiderocol was associated with a higher rate of all-cause mortality in patients with carbapenem-resistant Enterobacteriaceae (CRE) infections when compared with BAT with activity against GNB. All-cause mortality occurred in 34/101 (33.7%) receiving cefiderocol as compared with 9/49 (18.4%) with BAT through day 49.
11
In general, the higher mortality rate was associated with nosocomial pneumonia or bloodstream infections or sepsis with
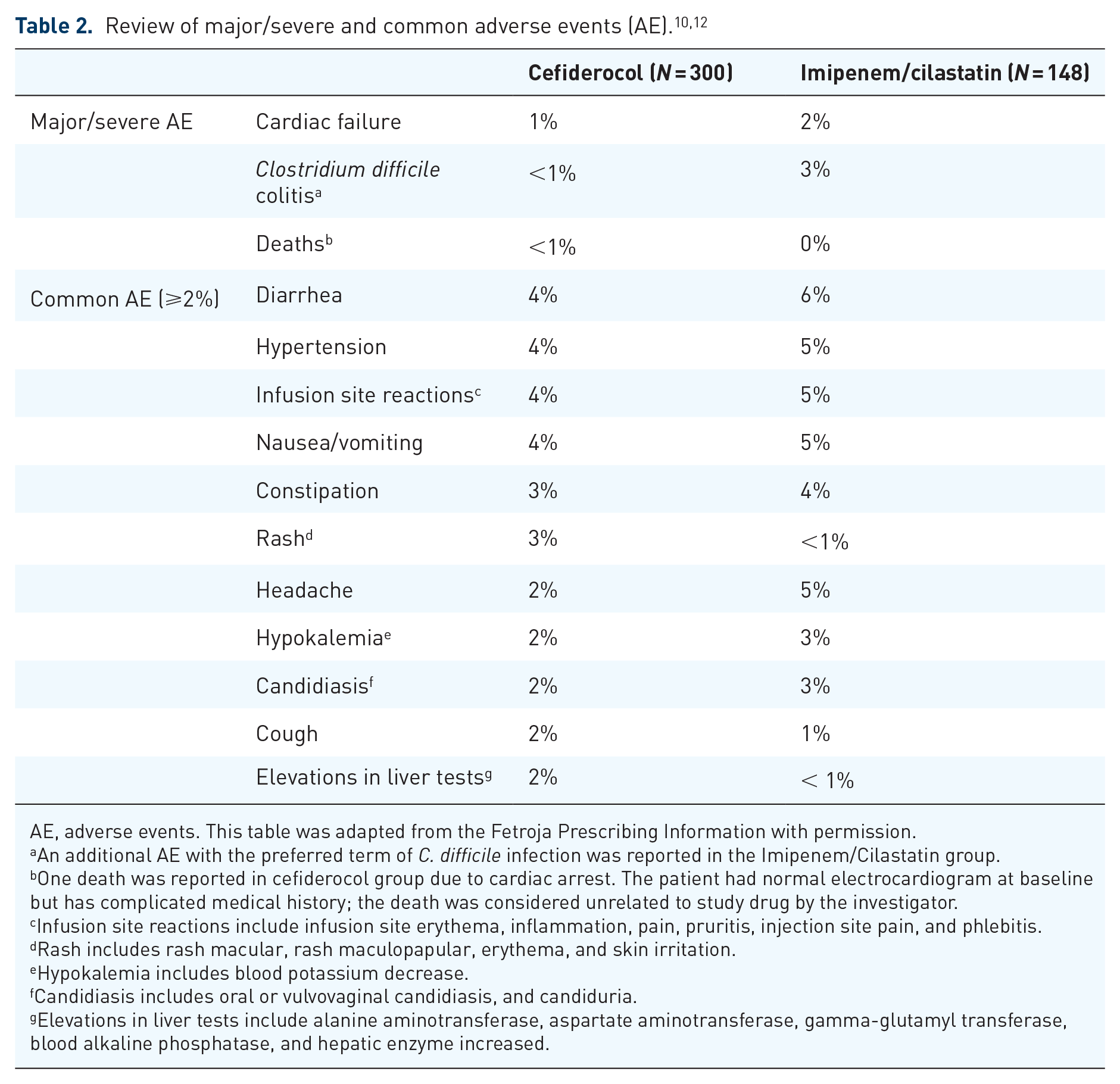
AE, adverse events. This table was adapted from the Fetroja Prescribing Information with permission.
An additional AE with the preferred term of
One death was reported in cefiderocol group due to cardiac arrest. The patient had normal electrocardiogram at baseline but has complicated medical history; the death was considered unrelated to study drug by the investigator.
Infusion site reactions include infusion site erythema, inflammation, pain, pruritis, injection site pain, and phlebitis.
Rash includes rash macular, rash maculopapular, erythema, and skin irritation.
Hypokalemia includes blood potassium decrease.
Candidiasis includes oral or vulvovaginal candidiasis, and candiduria.
Elevations in liver tests include alanine aminotransferase, aspartate aminotransferase, gamma-glutamyl transferase, blood alkaline phosphatase, and hepatic enzyme increased.
The use of cefiderocol is reserved for the treatment of cUTI with limited or no alternative treatment options. There is currently no data available on the effects of cefiderocol in pregnant and lactating women. The cUTI trial study population included adults ⩾18 years of age. The safety and efficacy in patients <18 years of age are unknown. The safety in subjects ⩾65 years of age (52.7% in the cUTI trial) is comparable to that of younger subjects. 10 Renal impairment is common in elderly patients; monitoring and dose adjustments should be made accordingly.
Drug interactions
No clinically significant drug interactions have been identified. False-positive results in dipstick tests have been reported and alternative methods for testing are recommended. Cefiderocol is not an inhibitor or inducer for cytochrome P450 enzymes or transporter systems. 5
Dosing and administration
Cefiderocol is available in a single-dose vial containing 1 g sterile, lyophilized powder, which should be reconstituted with 10 mL of 0.9% sodium chloride or 10 mL of 5% dextrose solution.
10
Reconstituted vials may be stored at room temperature for up to 1 h. The reconstituted solutions can then be added to 100 mL infusion bag of normal saline or 5% dextrose. The infusion bag is stable at room temperature and refrigerated for 6 h and 24 h, respectively. Cefiderocol is administered
Pharmacoeconomics
The wholesale acquisition cost (WAC) for a carton of 10, 1 g vials of cefiderocol is $1,897.50.
24
The recommended dose of 2 g
Discussion and recommendations
Antimicrobial resistance remains an urgent public health issue. The Centers for Disease Control and Prevention (CDC) reports that each year 2.8 million people contract an antimicrobial-resistant infection; of those, 35,000 infections result in death.
26
The sensible use and prescribing of antimicrobial agents and the development of novel antimicrobials are essential to slow the progression of antimicrobial resistance.
26
In the United States and Europe, nonfermenting gram-negative bacteria have posed a greater risk for drug resistance due to multiple mechanisms such as porin loss or efflux pumps.
9
In clinical practice, it is still lack of treatment options against MBL producers.
27
The most efficacious therapy in the treatment of MDR GNB infections is still unclear despite the newer approved antibiotics. The unique broad spectrum of cefiderocol against a large range of MDR GNB makes it a novel choice in the treatment of cUTI.
28
FDA approved cefiderocol for the treatment of cUTI including pyelonephritis. It may be considered as an empirical regimen for the cUTI patients with risk of MBL- or OXA-48 like carbapenemase producing strains or concomitant risk of carbapenemase-resistant
Cefiderocol is administered intravenously and thus must be given in the hospital setting. For this reason, it is important to consider patient-specific factors and the necessity of MDR GNB coverage prior to selecting cefiderocol. Cefiderocol is not a first-line agent and should only be used when first-line options have either failed, or MDR GNB are suspected or confirmed. Costs associated with cefiderocol administration may be offset by NTAP through CMS due to its QIDP designation. 10
Conclusion
Cefiderocol is a novel siderophore cephalosporin antibiotic recently approved for the treatment of cUTI and acute pyelonephritis. In addition to cUTI, cefiderocol is now also indicated for hospital-acquired pneumonia and ventilator-associated pneumonia. It has shown clinically significant antimicrobial coverage against MDR GNB. More post-market studies are needed to determine its role in treating cUTI and other infections in clinical practice.
Footnotes
Acknowledgements
We thank Ivy Poon, PharmD, BCPS, Brenea M. Webb, and Yuri J. Amons for the medical writing and editorial support.
Author contributions
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The preparation in this publication was supported, in part, by the National Institute on Minority Health and Health Disparities of the National Institute of Health under Award Number 2U54MD007605-27A1. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Health.
