Abstract
Migraine is a common and often disabling neurovascular disorder. Changes in the metabolism and the central processing of serotonin, as well as abnormalities in the modulation of the central and peripheral trigeminal nociceptive pathways, have been shown to play significant roles in migraine pathophysiology. Recent evidence suggests that a low serotonin state facilitates activation of the trigeminal nociceptive pathways. In addition, several pharmacological agents that modulate serotonin are used in the treatment of migraine. Specifically there are seven FDA approved, 5-hydroxytryptamine (5-HT) 1B/1D receptor agonists, used for the acute abortive therapy of migraine. Zolmitriptan is one such triptan. Zolmitriptan is available as a tablet, orally disintegrating tablet and as a nasal spray. It is rapidly absorbed and detectable within the plasma, within 2 to 5 minutes for the nasal spray and within 15 minutes for the tablet. Zolmitriptan reaches peak plasma levels in 2-4 hours, with good levels maintained for up to 6 hours. Although the metabolism of zolmitriptan is predominantly hepatic, only 25% of zolmitriptan is bound to plasma proteins. Thus it is unlikely for drug interactions involving the displacement of highly protein-bound drugs. Zolmitriptan is very well tolerated with less than half of participants in clinical trials reporting adverse events, most of which were mild and transient. Although rare, serious cardiovascular events have been reported with all triptans. However, when patients are appropriately selected, zolmitriptan is both, a safe and effective acute migraine abortive agent. In this article, we will first briefly review the biological role of serotonin and the literature linking serotonin to migraine pathophysiology. This will be followed by a comprehensive review of the pharmacodynamics, pharmacokinetics and efficacy of zolmitriptan. Finally, the clinical application of the use of zolmitriptan in migraine therapy will be discussed.
Introduction
Migraine is a common, often disabling neurovascular disorder. Changes in the metabolism and the central processing of serotonin or 5-hydroxytryptamine (HT), as well as abnormalities in the modulation of the central and peripheral trigeminal nociceptive pathways, have been shown to play significant roles in migraine pathophysiology. Recent evidence suggests that a low serotonin state facilitates activation of the trigeminal nociceptive pathways. 1
Drugs like fenfluramine and reserpine, which increase the availability of 5-HT from neurons and platelets, have been shown to provoke migraine.2,3 And ictally, a 30% decrease in platelet serotonin levels along with a 60% increase in plasma serotonin levels has been demonstrated. 4 In contrast, there is evidence that inter-ictally, (outside of an acute migraine attack), CNS serotonin activity is low.5,6 Specifically, in one functional imaging study of migraineurs, a selective 5-HT transporter ligand and single photon emission computed tomography were used to evaluate serotonin transporter protein [SERT] interictally. 5 An increased availability in the brainstem 5-HT transporter was demonstrated, a finding which supports decreased 5-HT levels interictally in migraineurs; and this finding suggests either a decreased synthesis and/or release of serotonin or increased catabolism of serotonin, Figure 1.5,8 This is further supported by a second functional imaging study of migraineurs, who underwent magnetic resonance imaging and positron emission tomography using a radioligand antagonist of 5-HT1A receptors in which an increase of the ligand binding potential in the posterior cortical and limbic areas of the brain was found, a finding also compatible with a decrease in central serotonin in migraineurs. 6
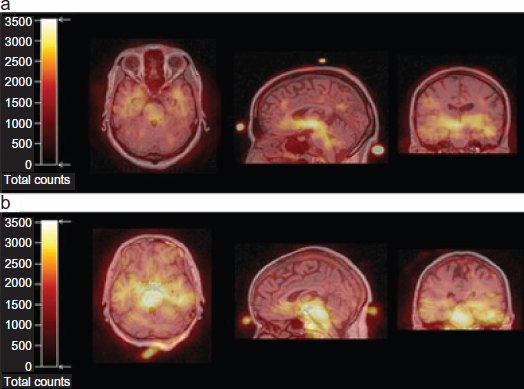
Transverse (left row), coronary (middle row) and sagittal (right row) co-registered MRI/123I-ADAM-SPECT scans are shown. The superimposed MRI/SPECT-markers are visible at nasion, vertex and inion. While a low 123I-ADAM-uptake is seen in the mesopontine brainstem of a healthy subject (a), an intense signal is found in mesencephalon and pons of a migraineur (b). 5
The therapy selection in migraine depends on headache severity, frequency, pattern of associated symptoms, comorbid illnesses, and the patient's treatment response profile. Accordingly, the treatment can be abortive for acute headaches or preventive. 7 This review will focus on zolmitriptan, one of the seven triptans available in the United States which are used in the abortive therapy of acute migraine attacks. We will first briefly discuss the role of serotonin in migraine pathophysiology. This will then be followed by an in-depth review of the pharmacodynamics and pharmacokinetics of zolmitriptan and an examination of its efficacy and utility for acute migraine abortive therapy.
Pharmacodynamics of Serotonin
Serotonin is an indole ethylamine neurotransmitter which is actively taken up and stored in platelets. 4 The synthesis of serotonin depends on the availability of tryptophan, an essential amino acid derived from dietary intake. Its synthesis also depends on the activity of the rate-limiting enzyme, tryptophan hydroxylase (TPH). 1 TPH exists in two isoforms identified as TPH1 and TPH2. 1 The TPH2 isoform is brain specific and is exclusively expressed in neurons located within the brainstem raphe nucleus that send widely distributed projections throughout the brain.1,8 Similarly, the majority of serotonin cell bodies are located in the midline brainstem with extensive axonal projection to nearly all regions of the CNS. Although the highest concentrations of 5-HT is found in platelets and in the enterochromaffin cells of the gastrointestinal tract, it is also present in the brain and retina in lesser concentrations.4,9,10
Serotonin is released from vesicles by a calcium dependent exocytotic mechanism. Most of the released 5-HT is recaptured via the serotonin transporter (SERT), an active reuptake mechanism found on serotonergic neurons. Serotonin is degraded by monoamine oxidase (MAO), and preferentially by the MAO-A isoform. 1 Serotonin is then converted to 5-hydroxyindole acetaldehyde and then metabolized by aldehyde dehydroxygenase to produce 5-hydroxyindole acetic acid (5-HIAA), the major 5-HT metabolite excreted.9,10
There are at least fourteen 5-HT receptors. 4 The 5-HT1 receptor class, (as well as all other serotonin receptors except the 5-HT3 receptors) belongs to the G protein coupled receptor super family. The 5-HT1 receptor class is composed of 5 subtype of receptors, including 5-HT1A, 5-HT1B, 5-HT1D, 5-HT1E, and 5-HT1F. All triptans, including zolmitriptan, most significantly activate the 5-HT1B/1D receptors.9,10
The 5- HT1B receptors are located in a variety of areas of the cerebral cortex including the central grey, hippocampus, dorsal raphe and hypothalamus as well as in the smooth muscle of blood vessels.4,11 In contrast to 5-HT1A receptor agonists, (which decrease the firing rate of serotonergic cell bodies, thus decreasing the release of 5-HT), 5-HT1B receptor agonists do not decrease the serotonergic firing rate; they activate serotonergic autoreceptors in terminal fields and thus also decrease the synthesis and release of serotonin. In addition, 5-HT1B receptor activation of the vascular smooth muscle results in intracranial, coronary and peripheral vasoconstriction of vessels.9,13 The 5-HT1D receptor sites are found on nerve terminals predominantly in the extrapyramidal pathways but also in the hippocampus, dorsal nucleus raphe and the cerebral cortex. 12 Similar to the 5-HT1B receptors, 5-HT1D receptors mediate inhibition of the release of 5-HT. However, the activation of 5-HT1D receptors results in inhibition of 5-HT release from cortical nerve terminals. 9
Pharmacodynamics of Zolmitriptan
Mechanism of action
Zolmitriptan is primarily a 5-HT1B/1D receptor agonist. As a result of the 5-HT1B/1D receptor agonism, triptans such as zolmitriptan have been suggested to act on the trigeminovascular system via both peripheral and central nervous system mechanisms. Peripherally triptans act to: 1) block neurogenic inflammation at the trigeminal vascular junction, 2) cause vasoconstriction of intracranial vessels via 5-HT1B activation of vascular smooth muscle and 3) inhibit neuronal depolarization at peripheral intracranial sites via 5-HT1B agonism. Centrally, triptans inhibit excitability of cells in the trigeminal nucleus caudalis via 5-HT1D agonism.13,17
Specifically, zolmitriptan has also been shown to decrease the neuropeptides, calcitonin gene-related peptide (CGRP) and vasoactive intestinal polypeptide (VIP) following stimulation of the trigeminal ganglion in both animal and human models.15,19 This mechanism is consistent with an inhibition of pre-junctional receptors on perivascular afferent fibers of the trigeminal nerve. 20 Zolmitriptan has also shown a dose-dependent inhibition of trigeminal evoked extravasation of radiolabelled-albumin in the dura mater in animal studies. 15 This inhibition of neurogenic plasma protein extravasation reflects the ability of zolmitriptan to act at pre-junctional receptors and thus inhibit trigeminal nerve excitability.21,22 Finally zolmitriptan has also been shown to act centrally in the nervous system. In vitro studies have shown the highest densities of zolmitriptan binding in the trigeminal nucleus caudalis, the nucleus tractus solitarius and the dorsal horns of the C1 and C2 cervical spinal cord. 23 In addition, immunochemical studies have shown that zolmitriptan significantly reduced expression of the immediate-early gene c-fos in the dorsal horns of the medulla and the C1 and C2 cervical cords of cats after electrical stimulation of the superior sagittal sinus. 24 Taken together these mechanisms account for the efficacy of zolmitriptan to successfully treat acute migraine attacks.
Chemistry
Zolmitriptan was developed with the goal of creating a more lipophilic, centrally active and more rapidly absorbed oral tablet than sumatriptan, the first of the triptans to be developed.25,26 Chemically, zolmitriptan is designated as (S)-4[[3-[2-(dimethylamino)ethyl]-1H-indol-5-yl]methyl]-2-oxazolidinone. It has has an empirical formula of C16H21N3O2, with a molecular weight of 287.36 g/mol, Figure 2.27,28
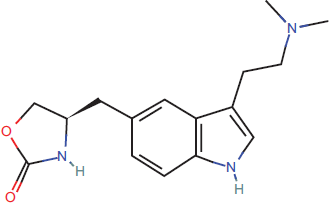
Zolmitriptan: chemical structure. 28
Metabolism and Pharmacokinetics
Receptor binding and activity
Studies using radioligand binding on animal and human blood vessels as well as binding of zolmitriptan on human recombinant serotonin receptors have shown that zolmitriptan acts selectively on the 5-HT1B and 5-HT1D receptor with an affinity of 10:1, respectively.14,29 In addition zolmitriptan has a modest affinity for 5-HT1A and 5-HT1F receptors. 29 The N-desmethyl zolmitriptan (183C91) metabolite of zolmitriptan has similar receptor binding profile to that of the parent compound. However, it is approximately 2 to 8 fold more potent than zolmitriptan at the 5-HT1B/1D receptors. Thus this metabolite of zolmitriptan may contribute to the therapeutic efficacy of the drug. 30 The remaining two metabolites, (N-oxide and indole acetic acid), are pharmacologically inactive. 31
Absorption and distribution
Zolmitriptan is available in three different formulations, a tablet, an orally disintegrating tablet (ODT) and a nasal spray (NS). 4 Although the tablet and ODT have essentially equivalent pharmacokinetics, there are significant differences between the tablet and nasal spray formulations as discussed further in the article. Herein we will review the overall pharmacokinetics for both tablet formulations and the nasal spray formulations, highlighting the similarities and differences in these formulations. Both oral formulations are detectable in the plasma within 15 minutes, with 75% to 80% of the maximum plasma concentration (Cmax) achieved within 1 hour of drug administration.14,32–34 It is important to note that while the presence of migraine associated symptoms, (such as nausea and vomiting), as well as the gastric paresis which occurs in migraineurs may limit ingestion and digestion of oral zolmitriptan, food does not significantly affect the absorption of zolmitriptan tablets. 18
With the NS formulation, zolmitriptan is detectable in the plasma within 2 to 5 minutes.32–34 The early uptake of zolmitriptan NS through the nasopharynx accounts for its rapid absorption. It has been shown that almost 100% of the radioactivity of zolmitriptan NS is detectable in the nasopharynx immediately after dosing, while at even 15 minutes after dosing, less than 20% is detectable in the upper abdomen.4,32–35 And although within the first hour after administration, only 29 to 30% of the total exposure to zolmitriptan NS has been shown to be due directly to intranasal absorption, intranasal exposure to zolmitriptan has been shown to contribute to approximately 71% of the exposure.4,36
The time to peak concentration (tmax) is approximately 2 hours for the tablet, 3 hours for the orally disintegrating table (ODT) and between 3-4 hours for the NS formulation.32–36 In addition, the appearance of zolmitriptan's active metabolite in plasma following intranasal administration occurs at about 30 minutes after nasal administration. 37 Thus although first-pass metabolism in the liver is avoided with the nasal spray formulation, it is likely that the active metabolite of zolmitriptan contributes to the lasting efficacy of the intranasal formulation as with the oral formulations.
Approximately 75%-80% of the Cmax is attained within one hour post dose. Bioavailability is approximately 40%-48% for both oral formulations and 42% for the NS. Good plasma levels are maintained for up to six hours, with lower levels lasting for about 16 hours.14,33
Metabolism and elimination
Approximately one third of zolmitriptan is metabolized renally, while two thirds of zolmitriptan's clearance is non-renal. It is presumed that the non-renal metabolism is metabolic. 35 Specifically a high hepatic extraction is indicated by a non-renal clearance of 520 ml/min. 38 The majority of zolmitriptan is metabolized through the cytochrome P450 (CYP1A2) system; and there are three major metabolites, N-oxide, an indole acetic acid and N-desmethyl-zolmitriptan.4,31 The N-oxide and indole acetic acid metabolites are inactive, while the N-desmethyl zolmitriptan is active. 36 At a dose of 15 mg/day, monoamine oxidase metabolism is also involved in the metabolism of zolmitriptan. However, this dose is not clinically available or used in practice.4,40 Finally, for all formulations, Zolmitriptan's elimination half-life is approximately 3 hours.4,14,33
Pharmacokinetic differences in the ictal and interictal migraine periods
The gastric stasis which may occur in migraineurs may affect the pharmacokinetics of triptans.44,73 One study evaluated the Cmax and area under curve (AUC) values for the 10 mg zolmitriptan tablet in 20 patients with moderate to severe migraine at both ictal and interictal time-points. 44 The median AUC was lower ictally for the migraineurs (18.4 ng/ml • h) as compared to interictally (33.4 ng/ml • h); likewise, the Cmax values were lower ictally (7.9 mg/L) then interictally as well (12.6 mg/L).20,44 Similarly to zolmitriptan, the Cmax and AUC values were lower for the 3 metabolites during a migraine attack.20,44
Gender differences
Women show a higher mean Cmax and AUC values than men, (25.2 mg/L and 173.8 mg/L • h vs. 16 mg/L and 88.9 mg/L • h respectively) an observation, which is significant only after a 5 mg dose and more pronounced with a 10 mg. 39 At doses greater than or equal to 5 mg, oral bioavailability of zolmitriptan has been shown to be greater in women than men.39,41,42 The half-life (t1/2) was unchanged between the genders. These differences are thought to be due to the reduced first-pass metabolism of the drug in women. In addition higher plasma concentrations, higher values of area under the concentration curve (AUC) and lower total clearance have been observed in women as compared to men.27,41 However, clinically, these gender differences have not been demonstrated to influence the efficacy of zolmitriptan.18,43
Pharmacokinetics in pediatric and adolescent populations
The pharmacokinetics of zolmitriptan are largely similar in pediatric and adolescent populations as compared to adults, with minor differences. An open-label, parallel-group study, involving 21 healthy adolescent (mean age 14.5 years) and 18 healthy adults (mean age 39.1 years) assessed the pharmacokinetics and tolerability of zolmitriptan and its active metabolite for the acute treatment of migraine. 45 All subjects received a single oral 5 mg dose of zolmitriptan. The AUC and Cmax of zolmitriptan were similar in both age groups, with the T1/2 found to be 3.01 hours in adolescents, as compared to 3.75 hours in adults (p ≤ 0.05). As compared to adults, adolescents had an AUC that was 36% higher, while the Cmax was found to be increased by 39%. The half-life was similar in both groups. Adverse events were also similar in both groups in regards to the nature, intensity, and frequency of events. However, elimination of zolmitriptan was found to be faster in adolescents than adults, as suggested by the shorter T1/2. 45
The pharmacokinetics of zolmitriptan in the elderly
Although the Cmax and AUC values for zolmitriptan, (5 to 15 mg) and the active N-desmethyl metabolite were similar in those older than 65 years of age to young adults, the renal clearance was reduced in the elderly. In addition, the reduced renal clearance was associated with significantly increased Cmax values of the N-oxide metabolite and the Cmax and AUC values of the indole acetic acid metabolite. 46 However as mentioned previously the N-oxide metabolite and the indole acetic acid are inactive and of limited, if any, clinical significance.
Patients with hepatic impairment
In patients with severe hepatic impairment, the mean Cmax, Tmax and AUC increase by 1.5, 2 and 3 fold respectively. In addition, following a large dose of zolmitriptan (10 mg), a 20-80 mmHg increase in blood pressure has been shown to occur in approximately 26% of patients.4,49,50 In one open-label, parallel-group study of 37 participants, the pharmacokinetics of a single 10 mg dose of zolmitriptan was compared between 10 healthy volunteers, 11 patients with moderate hepatic impairment, and 10 patients with severe hepatic impairment without ascites, and 6 patients with severe hepatic impairment with ascites. The metabolism of zolmitriptan was reduced in patients with severe hepatic impairment as compared with healthy subjects. In addition, in patients with severe hepatic impairment, the peak plasma concentrations were 47% higher and half-life was prolonged by 157% as compared with healthy subjects. (The changes were similar in the presence and absence of ascites.) Plasma concentrations of the three major metabolites of zolmitriptan were also reduced in the patients with hepatic impairment. Thus although no dose adjustment is necessary in those with moderate hepatic impairment, in migraineurs with severe hepatic impairment zolmitriptan dosage should be reduced. 48
Patients with renal impairment
A parallel group study evaluated the pharmacokinetics of a single, 10 mg dose of zolmitriptan in patients with moderate to severe renal impairment (creatinine clearance ≤40 ml/min) as compared to age- and sex-matched healthy volunteers (creatinine clearance ≥60 ml/min). 35 Mean peak concentrations of zolmitriptan and its active N-desmethyl metabolite were similar in both groups although AUC 0-∞ for the active metabolite was increased by 35% in the renally impaired patients. Baseline blood pressures were higher in the renally impaired group; and after zolmitriptan administration, there was a transient, small increase in blood pressure in both groups. The rise in systolic blood pressure was not significantly greater in the renally impaired group (23 mmHg) as compared to the controls (16 mmHg). In addition, there were no substantial changes in the plasma concentrations of zolmitriptan or its active metabolite. Thus, the dose of zolmitriptan in patients with renal impairment does not require adjustment, unless patients are on hemodialysis. 35
Patients with hypertension
Pharmacokinetic differences between subjects with and without hypertension were not clinically significant. 41 Specifically, the pharmacokinetics and pharmacodynamics of zolmitriptan (5, 10, and 20 mg) in patients with hypertension were evaluated in a randomized, placebo-controlled, doubleblind, four-period crossover study. Sixteen patients with mild to moderate hypertension and 17 healthy, age- and sex-matched control subjects were evaluated. The pharmacokinetics of zolmitriptan and its metabolites were found to be dose proportional. The AUC0-∞ and Cmax were slightly higher in patients with hypertension at all doses. However, this was only statistically significant for the AUC at a 20 mg dose. Although zolmitriptan produced a small increase in blood pressure in all subjects, both those with and those without hypertension) it was not significantly different between groups. This indicates that zolmitriptan can be administered for treatment of migraine in patients with controlled hypertension without dose adjustment. 41
Differences in pregnancy/nursing mothers
Currently, zolmitriptan is pregnancy category C, as are all triptans. The use of triptans during pregnancy is controversial, as there are no well controlled studies of triptans in pregnant women. However, no adverse effects with triptan use during pregnancy, including malformations, have been demonstrated in the voluntary registries collected by pharmaceutical companies, although early delivery and lower birth weight have been reported in triptan users.51–53
Although levels of zolmitriptan in human milk have not been compared to serum levels, concentrations of two other triptans have been shown to be lower in human breast milk than in serum. 54 Studies with zolmitriptan in lactating rats demonstrated equivalent milk and plasma levels at one hour after dosing but four times higher milk levels than plasma at four hours after administration of zolmitriptan. 60
Ethnic differences
There are no known ethnicity related effects on the pharmacokinetics of zolmitriptan. Uemura et al reported that following a single-dose (2.5 or 5 mg) oral or intranasal preparation, the pharmacokinetic profile of zolmitriptan and its active metabolite in healthy Japanese subjects was consistent with Caucasian subjects. 18
Similarly, Yates et al reported that the pharmacokinetic parameters of zolmitriptan 2.5 mg tablets were similar between Caucasian and Japanese volunteers. 55
Drug-drug Interactions
Only one-fourth of zolmitriptan is bound to plasma proteins, suggesting that significant displacement of other protein bound drugs is unlikely. 56 No clinically significant interactions with other commonly prescribed medications in headache management such as metoclopramide, dihydroergotamine, fluoxetine, and propranolol, or monoamine oxidase-B inhibitors such as selegiline, have been demonstrated.56,57 However, it is contraindicated to give zolmitriptan to a patient on MAO-A inhibitor or within 24 hours of use of an ergot or another triptan.59,60 In addition, it has been demonstrated that cimetidine inhibits the metabolism of zolmitriptan and its active metabolite, increasing the mean Cmax by 16 and 50% and the mean AUC by 48 and 105%, respectively. Mean T1/2 was prolonged by 2.2 hours for zolmitriptan and by 4.2 hours for its active metabolite. Thus the dosage of zolmitriptan may need to be decreased or started at a lower in patients regularly using cimetidine. 112 Finally, it has been shown that the absorption of zolmitriptan when administered intranasally is not affected by reduction in local nasal blood flow with nasal decongestants. 58
Therapeutic Use
Zolmitriptan is approved for the treatment of migraine with and without aura in Europe and the United States (U.S). Multiple studies have demonstrated the clinical efficacy of zolmitriptan in the use of acute migraine as described below. Like all triptans, zolmitriptan is not indicated for hemiplegic and basilar migraine.27,61
Dosing
Zolmitriptan tablets and oral disintegrating tablets (ODT) are available in 2.5 mg and 5 mg strengths. 62 The tablets are recommended to be given as a single dose which can be repeated within 2 hours if needed to a maximum of 10 mg in a 24-hour period. The nasal spray (NS) is available in a 5 mg strength and is recommended as a single spray in one nostril, which can be repeated within 2 hours if needed, to a maximum of 10 mg in a 24-hour period. 62
Clinical Efficacy
Efficacy in migraine
Regardless of formulation, zolmitriptan has been shown to have superior efficacy to placebo. The efficacy of zolmitriptan tablets has been demonstrated in multiple double-blind placebo controlled trials.43,63–65 At 2 hours, the mean headache response (HR) rate is approximately 64% for the 2.5 mg zolmitriptan tablet as compared to 35% for placebo; similarly the 2 hr HR rate is 65% for the zolmitriptan 5 mg tablets as compared to 27% for placebo. In addition the mean 2 hour pain free (PF) rate was 24.5% for the 2.5 mg zolmitriptan tablet and 28.6% for the 5 mg tablet, as compared to the placebo 2 hr PF rates of 8.5% and 4.3% respectively. Finally, the mean 24 hour headache recurrence rate is 30% for the 2.5 mg tablet and 31% for and the zolmitriptan 5 mg tablet as compared to 38% and 48% for the placebo, Figure 3 and 4.43,63–65
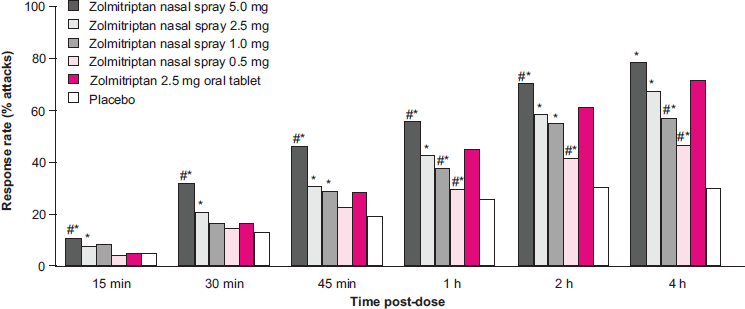
Headache response rates at all time points (for attacks 1 and 2) following treatment of moderate or severe migraine with zolmitriptan nasal spray, zolmitriptan oral tablet or placebo.
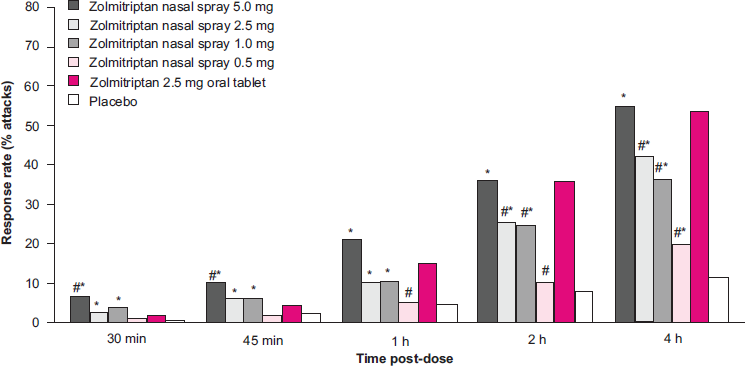
Pain-free rates from 30 minutes (for attacks 1 and 2) after treatment of moderate or severe migraine with zolmitriptan nasal spray, zolmitriptan oral tablet or placebo.
There is also evidence of a consistent response across multiple attacks in individual patients for both, the 2.5 mg and 5 mg tablets of zolmitriptan. Tuchman et al showed that the 2-hour headache response across attacks was 85% for the 2.5 mg tablet and 79% for the 5 mg zolmitriptan tablet. In addition 64% of zolmitriptan users reported a headache response in greater than 75% of their acute migraine attacks. In a few patients, when a second dose was required, the response rates were also consistent across attacks. Thus zolmitriptan is effective in the treatment of multiple migraine attacks in the same patient. 66
Zolmitriptan oral tablets have been directly compared to sumatriptan, rizatriptan and eletriptan tablets in double-blind, placebo controlled studies.26,67,70,71,75 Large randomized, double-blinded studies have shown that zolmitriptan and sumatriptan have no significant differences in the primary endpoints for efficacy, including the 2 hour headache response rate.26,67,70 In an analysis by Becker et al Zolmitriptan 2.5 mg tablets were compared to sumatriptan 50 mg and found no significant difference for the 2 hour headache response between the drugs. 70
Similarly, in a study comparing rizatriptan 10 mg tablet to the zolmitriptan 2.5 mg tablet, no statistically significant difference in the 2 hour headache response rate was found. Specifically, 71% of those using rizatriptan 10 mg, 67% of zolmitriptan 2.5 mg users and 30% of those who received placebo reported significant headache relief at 2-hours. However, rizatriptan 10 mg tablet (43%) was marginally more significant than zolmitriptan 2.5 mg tablet (36%) in regards to the 2 hour pain free response, (p ≤ 0.05). 71
Steiner et al compared zolmitriptan 2.5 mg to eletriptan 80 mg, (a dose not available in the United States), and to eletriptan 40 mg in a double-blind, placebo controlled study. Although eletriptan 80 mg (74%) was superior to zolmitriptan 2.5 mg (60%) for the 2 headache response rate, there was no difference in the 2 hour headache response between eletriptan 40 mg, (the largest dose of eletriptan available in the United States), and zolmitriptan 2.5 mg. Nor were there any significant differences in the other endpoints between both drugs. 75
Zolmitriptan orally disintegrating tablet (ODT) has been shown to have superior efficacy against placebo in three double-blind, placebo controlled trials in the treatment of acute migraine.72–74 Zolmitriptan 2.5 mg and 5 mg ODT showed a 63% and 59% 2 hour headache response rate as compared to 22% and 31% for placebo. The 2 hour pain free rate was 27%-47% and 31% for zolmitriptan 2.5 mg and zolmitriptan 5 mg ODT respectively versus 7%-20% and 11% for placebo. The mean 24 hour sustained pain free response for the 2.5 mg ODT is 29% (versus11% for placebo) and for the 5 mg ODT was 43% (versus 16% for placebo).72–74
As with the tablet formulations, zolmitriptan nasal spray has been shown in several studies to be significantly more effective for pain relief at 2 hours as compared to placebo for acute migraine therapy.58,77,78 In the first of these studies, the 2 hour headache response rate for the 2.5 mg and 5 mg nasal spray doses, were 59% and 70% respectively, as compared to 31% for placebo; and the headache response was significantly greater for both doses as compared to placebo as early as 15 minutes. 77 A similar response was shown by Rapoport et al for the 2.5 mg and 5 mg zolmitriptan tablets, but beginning at 30 minutes. 63 Furthermore, the headache response to the 5 mg NS dose was also significantly greater than the 2.5 mg NS dose beginning at 15 minutes. In the third study evaluating the NS formulation of zolmitriptan, the two hour headache response rate of zolmitriptan 5 mg NS (70%) was superior to both placebo (31%) and the 2.5 mg oral tablet (61%). 58 In addition the 24-hour pain-free rate for the 5 mg NS dose was significantly greater than placebo at 15 mins; and at 30 mins both the 5 mg and 2.5 mg NS doses were superior to placebo. This was also confirmed by Dodick et al who showed that the 5 mg NS exhibited a significant headache response response as compared to placebo as early as 15 minutes. 78
The Real Life Intranasal Zolmitriptan Experience (REALIZE) study was a randomized, double-blinded, placebo controlled study, which evaluated 912 patients to assess the efficacy of zolmitriptan 5 mg NS as compared to placebo. 79 The primary endpoint was freedom from pain and migraine associated symptoms (MAS) at 1 hour after the first dose. The total symptom relief rate, (freedom from pain and MAS), at 1 hour post-dose was higher in the zolmitriptan 5 mg nasal spray group (14.5%) than in the placebo group (5.1%), P < 0.0001. And this difference was significant from 30 minutes post-dose. Treatment with zolmitriptan also produced a higher headache response rate from 10 minutes post-dose (15.1% vs. 9.1%; p = 0.0079) and a higher pain-free rate from 30 minutes post-dose (7.7% vs. 3.2%; p = 0.0039). Zolmitriptan NS was also significantly superior to placebo in terms of sustained pain-free status and patients’ ability to perform normal activities. 79
Efficacy for relief of migraine associated symptoms and disability
At doses greater than or equal to 2.5 mg, zolmitriptan reduces the migraine associated symptoms of nausea, photophobia and phonophobia. Specifically zolmitriptan 2.5 mg reduced the rate of nausea reported before treatment, from 47% to 28% at two hours after treatment and to 19% 4 hours after drug administration as compared with placebo.63,65 Zolmitriptan 5 and 10 mg showed similar reductions. 63 Zolmitriptan 2.5 mg was also associated with pronounced reductions in the incidences of associated photophobia and phonophobia.63,65 The rates of photophobia dropped from 85% pretreatment to 42% at 2 hours and 30% at 4 hours. Similarly, the rates of phonophobia decreased from 73% (pretreatment), to 32% at 2 hours after treatment and to 24% at 4 hours after treatment. This reduction was also observed with the 5 mg and 10 mg dose. 63
In addition to improvement in the migraine-associated symptoms, patients who reported some impairment in normal activity during a migraine had improved functioning with zolmitriptan 2.5 and 5 mg treatment. In one study, 35% of patients treated with zolmitriptan (2.5 mg and 5 mg) had mild or no impairment in activity at 2 hours; compared to only 6% of placebo recipients. 63 Another large study examined patients with moderate to severe impairment in activity at baseline and found that 63% of zolmitriptan 2.5 mg users had improvement as compared to 38% of placebo recipients. 65
Prevention and treatment of recurrent or persistent headache
Zolmitriptan is effective in the treatment of persistent and/or recurrent migraine headache.63,84 However, a repeat dose given prophylactically in the absence of headache at 8 hours after the initial treatment of a migraine attack did not have a significant effect on the incidence of or time to recurrence.20,84 In those patients who received a second dose of zolmitriptan (1-10 mg) at 4 to 24 hours hours after the initial treatment, the response rate at 2 hours was 54% for zolmitriptan group versus 35% for placebo in patients treating a single attack. The dose of zolmitriptan was determined at time of randomization of subjects. 63
Efficacy in menstrually-related migraine
Although the FDA has not specifically approved any triptan, including zolmitriptan, for menstrually-related migraine (MRM) or the short-term prophylaxis of MRM, zolmitriptan has been shown to be efficacious in women with migraine either associated or not associated with menses and for short-term prophylaxis of MRM.63,65,85 In a study by Loder et al 579 women with menstrually-related migraine treated mild migraine with 2.5 mg zolmitriptan tablets and moderate to severe attacks with 5 mg zolmitriptan tablets. Zolmitriptan was more effective than placebo in achieving headache response as early as 30 minutes in the zolmitriptan group. At 2-hours, headaches were still responsive in 48% of zolmitriptan treated attacks as compared with 27% with placebo. 86
Although there are no direct comparator studies between any triptans for short-term prophylaxis of MRM, frovatriptan, sumatriptan, naratriptan and zolmitriptan have shown to be effective short-term prophylactic agents for MRM.87–90 Tuchman et al evaluated the efficacy of zolmitriptan for the short-term prophylaxis of MRM. Over 200 women were treated perimenstrually (starting 2 days before the onset of menstruation and continued for 7 days total) with 2.5 mg zolmitriptan TID or BID as compared to placebo for 3 menstrual cycles. Both doses showed a significant decrease in MRM as compared to placebo. No dose dependant response was seen, suggesting that the 2.5 mg dose BID dosage is sufficient for short-term prophylaxis of MRM. 90
Efficacy of pre-emptive treatment of migraine before pain onset
The use of zolmitriptan prior to the development of head pain is likely not effective. One small pilot study of 16 migraine with aura patients who received, in crossover fashion during the aura phase of 2 migraine attacks, zolmitriptan 20 mg or placebo within 10 minutes of the onset of aura and when no headache was present. 93 Three of 16 patients (19%) responded to zolmitriptan while none responded to placebo. All 16 patients developed a migraine headache.
Efficacy of zolmitriptan in migraineurs with allodynia
During a migraine attack central sensitization may occur and is clinically manifested by cutaneous allodynia (CA). Clinically CA is exhibited migraineurs with such things a pain when brushing their hair or wearing glasses etc. Burstein et al showed that the response to triptans depends on the stage of sensitization reached at the time of administration of triptans and that this phenomenon is exhibited in two distinct phases. First, a developmental phase, during which the activity of the central neurons that mediate allodynia depends on a continuous flow of incoming pain signals from the meninges; and second, an established phase, during which the activity of the central neurons is independent of the incoming pain signals. 104 Triptans are less efficacious when central sensitization as exhibited by CA is fully established. 105
Zolmitriptan has been shown to be effective in migraineurs with CA. 102 Lampl et al evaluated 36 episodic migraineurs who were previously unresponsive to sumatriptan 100 mg and who exhibited signs of allodynia. Participants were treated with either zolmitriptan NS or Sumatriptan 100 mg. Both the migraine pain and allodynic symptoms were shown to be reduced within one hour with zolmitriptan 5 mg nasal spray instead of sumatriptan when attacks were treated with 30 minutes of onset of headache. There was also a favorable tendency to decrease pain and allodynia after 2 and 4 h by using zolmitriptan 5 mg nasal spray within 30 min after migraine onset. 102 Based on this finding, it can be recommended that allodynic patients take triptans as soon as possible and preferably within 30 minutes after the onset of migraine.102,103
Long term efficacy
Zolmitriptan appears to maintain efficacy even after repeated administration for multiple attacks of migraine over a year's duration, with high headache response rates reported over all attacks. 100 In the INtranasal 5 mg Dose EXposure (INDEX) study, Dowson et al evaluated 538 patients treating 20,717 migraine attacks with zolmitriptan 5 mg nasal spray over 1 year. Almost 54% of the migraine attacks were rendered pain free at 2 hours, and these results were consistent over the study period. 100 Similarly the International Zolmitriptan Study group, (in an open label study of 2,058 patients treating 31,579 migraine attacks) also reported that zolmitriptan 5 mg tablet remained effective and well tolerated over the duration of a year. 91
Efficacy in nonresponders to previous therapy
Zolmitriptan has also been evaluated for use in non-responders to prior treatment of acute migraine attack. Mathew et al studied 48 patients who were non-responders to sumatriptan 50 mg tablets over a total of 120 migraine attacks. The two hour headache response (moderate/severe to mild or no pain) was seen in 73% of the attacks. 107
In addition 67% of participants treated with zolmitriptan, reported the preference of zolmitriptan over their previous therapy; and 64% of patients considered their migraine control as good.20,92
Efficacy in pediatric and adolescent populations
No triptans are FDA approved for use in pediatric or adolescent migraine; however both the tablet and NS formulations have data suggesting efficacy over placebo. 4 First, in an open-label study Linder and Dowson evaluated 32 adolescents across 276 migraine attacks, and found an overall headache response of 88% and 70% with oral zolmitriptan 2.5 mg and 5 mg, respectively at 2 hours. 68 This study was followed by a double-blind, placebo controlled study of 29 children and adolescents between 6-18 years of age evaluating zolmitriptan 2.5 mg and ibuprofen 200 to 400 mg to placebo. The zolmitriptan 2.5 mg tablet and ibuprofen 200 to 400 mg were equally effective for migraine pain relief in the children and adolescents. 69 The 2 hour pain relief rates were 62% for 2.5 mg of zolmitriptan and 69% for ibuprofen 200 to 400 mg, as compared to only 28% for placebo. Only mild side effects were reported and good tolerability was exhibited for both drugs. 69
The nasal spray formulation of zolmitriptan has also been assessed in adolescents. In a multicenter, randomized, double-blind, placebo-controlled, 2-way, 2-attack, crossover design study, Lewis et al reported that zolmitriptan NS provided fast and significantly effective relief of migraine symptoms and was well tolerated in the acute treatment of adolescent migraine. 47 A total of 248 adolescent patients (12-17 years of age) with a prior diagnosis of migraine with or without aura were studied. The primary efficacy variable was headache response at 1 hour after treatment with either zolmitriptan 5 mg NS or placebo. Results showed that onset of significant pain relief was apparent 15 minutes after treatment with zolmitriptan NS, (37.2% versus 22.8% for placebo, p < 0.05). At 1 hour after the dose, zolmitriptan NS produced a significant headache response rate than did placebo (58.1% vs. 43.3%, p < 0.05). Zolmitriptan NS was also found to be significantly superior to placebo in improvement in pain intensity, pain-free rates, sustained resolution of headache, and resolution of associated migraine symptoms in adolescents. Return to normal activities was consistently faster and less use of escape medication reported with zolmitriptan NS than with the placebo in the adolescent migraineurs. Finally zolmitriptan NS was well tolerated. 47 Although zolmitriptan is not FDA approved for use in adolescents, these studies suggest that zolmitriptan does have efficacy for treatment of acute migraine in adolescents; and in pediatric patients who do not respond to NSAIDS, triptans such as zolmitriptan may be effective.108–110
Efficacy in cluster headache
Three multi-center placebo-controlled studies have shown that zolmitriptan is effective in the acute treatment of cluster headache. Bahra et al evaluated 124 cluster patients and showed that 47% of those patients taking zolmitriptan 10 mg showed a significant headache response as compared to 29% of those given placebo when zolmitriptan was taken within 10 minutes of onset of cluster attacks. A similar result could not be seen with the 5 mg dose. 81 In a second study, Cittadini et al evaluated 5 mg of zolmitriptan NS and 10 mg of zolmitriptan NS as compared to placebo in 69 cluster headache sufferers across three attacks per participant. The 30 minute headache relief rates were as follows: 21% for placebo, 40% for the 5 mg NS and 62% for the 10 mg NS. Both doses were significantly more effective than placebo, Figure 5. 82 In a third study, Rapoport et al evaluated both zolmitriptan 5 mg NS and zolmitriptan 10 mg NS as compared to placebo in a double-blind, multicenter, crossover study across 151 attacks in 52 cluster headache sufferers. Headache relief at 30 minutes was reported in 63.3% of cluster headache sufferers who received the 10 mg dose and in 50% of those who received the 5 mg dose. 83 Thus, zolmitriptan NS is effective for treatment of cluster headache attacks and the 10 mg dose may be more effective than the 5 mg dose.
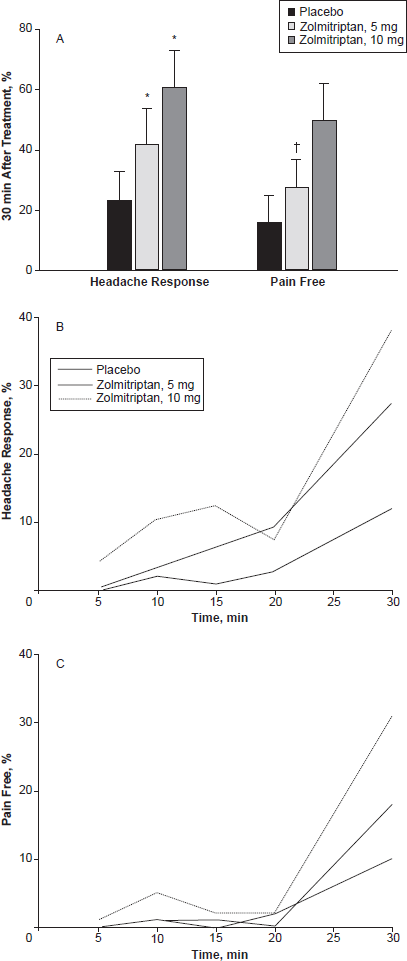
Efficacy results for Zolmitriptan in cluster headache. Headache response, a reduction of headache intensity from very severe, severe, or moderate to mild or no pain, and pain-free rates (no pain) at 30 minutes after treatment with zolmitriptan vs. placebo (A). *P < 0.002; P < 0.003. Data for headache response (B) and pain-free rates (C) are demonstrated as they cumulate with time from treatment at time zero to 30 minutes post dose. 82
Satisfaction and Preference of Treatment with Zolmitriptan
Patient satisfaction
The majority of patients who use zolmitriptan 5 mg nasal spray are satisfied with zolmitriptan as acute abortive therapy and wished to continue using it. Specifically, in a recent post marketing surveillance survey, Diener et al showed that 72.9% of approximately 1800 migraineurs who used zolmitriptan NS to treat migraine attacks of any severity and who were followed up within 4 months, rated it as better than their previous therapy. In addition approximately 58% of patients reported they were pain free at one hour, 62% reported they were able to resume usual daily activities and 88.8% wished to continue to use zolmitriptan NS. 80
Patient preference
Zolmitriptan has been evaluated in regards to patient preference of formulation, taste of medication and in regards to preference of zolmitriptan to other therapies. First, in regards to formulation preference, preference for one of the three formulations of zolmitriptan (tablet, ODT or NS) was evaluated in a prospective, randomized, open, crossover, longitudinal study. 101 A total of 48 patients were treated with the different zolmitriptan formulations. Patients who had never received zolmitriptan were treated with either the zolmitriptan 2.5 mg tablets or 2.5 mg Orally Disintegrating Tablets (ODT) for one month, before being crossed over to receive the alternative formulation for the second month followed by zolmitriptan 5 mg NS. Patients were then allowed to choose the formulation(s) of their choice for the next seven months. Individual preferences and changes in headache-related disability scores and adverse events were evaluated. Although at baseline, most patients preferred conventional tablets, after 4 months, 46.9% of patients preferred zolmitriptan ODT, 43.8% zolmitriptan nasal spray, and 6.3% the conventional tablet. Disability scores decreased significantly with treatment with zolmitriptan and it was well tolerated probably indicating that newer zolmitriptan formulations might be preferred over the conventional tablets. 101
In an international, open-label, 6-month clinical study, 595 patients treated 7171 migraine attacks with zolmitriptan ODT. 97 Zolmitriptan 2.5 mg (ODT) was used by patients to treat upto three migraine attacks per month. The main reasons why patients wanted to continue using zolmitriptan ODT included effectiveness [93.2% (of 380)], ease of use (80.8%) and rapid effect (78.4%). Over 74% of patients were able to resume usual activities within 2 h of treatment, and over two-thirds of patients (70.9%) preferred zolmitriptan ODT to their usual migraine therapies. Most patients were satisfied or very satisfied with zolmitriptan ODT and preferred it to their usual therapy (including other triptans). PCPs wished to continue prescribing zolmitriptan ODT for over two-thirds of patients. The withdrawal rate because of adverse events in this study was very low (3.4%) and similar to that seen in controlled studies of zolmitriptan ODT.73,98
Patient preference was also evaluated in an open post marketing surveillance study of 12,919 patients, treating 36,510 migraine attacks with zolmitriptan 2.5 mg Compared with all previous therapies like herbal remedies, homeopathic, magnesium and analgesics, 85% of patients preferred zolmitriptan on efficacy grounds, while 56% considered it to be better tolerated. 111
Second, in a study evaluating only taste preference, patients received either the ODT of either zolmitriptan or rizatriptan to assess the flavor and after-taste. Zolmitriptan (81%) was preferred overall as compared to rizatriptan (68%). In a second study, 60% patients preferred zolmitriptan 2.5 mg ODT as compared to 40% for sumatriptan 50 mg tablet users in the treatment of acute migraine. A total of 77% of patients who received zolmitriptan ODT, considered it to be an effective migraine treatment, as compared to 63% of patients who were treated with the conventional sumatriptan tablet.71,75
Finally, patient satisfaction was evaluated in the REALIZE phase II study. This study consisted of 851 migraine patients treating upto 3 attacks found that 75.7% reported being satisfied or very satisfied with zolmitriptan nasal spray. The majority of patients (59.8%) were willing to use zolmitriptan nasal spray in the future and 57.8% patients preferred zolmitriptan nasal spray over previous therapies. 99 Migraine patients also show preference for zolmitriptain in the Zolmitriptan Evaluation versus Sumatriptan Trial (ZEST), in which 218 migraine patients reported preferring the zolmitriptan 2.5 mg orally disintegrating tablet (ODT) to sumatriptan 50 mg conventional tablet. 76 Zolmitriptan ODT was also noted found to be significantly more effective, least disruptive, easier to be more convenient to take as compared to sumatriptan tablet. 76
Safety and Tolerability
Most adverse events seen with the triptans, including zolmitriptan, are generally mild and transient; hence this makes them very well-tolerated. Specifically adverse events reported with the use of zolmitriptan were transient, generally of mild or moderate intensity, did not require intervention and did not require withdrawal of treatment. In one analysis of zolmitriptan 2.5 mg and recipients of placebo had a similar proportion of severe adverse events (4% vs. 5%, respectively). This value was slightly higher in recipients of zolmitriptan 5 mg (7%). 94 In addition less than half of patients in clinical trials have reported adverse events. Patients commonly reported nausea, dizziness, paresthesias, somnolence and chest tightness for the oral formulation. Unusual taste, local nasopharyngeal symptoms and pharyngitis for the nasal spray formulation.58,43,63–65
Uncontrolled hypertension, coronary artery disease, arrhythmias associated with accessory pathways such as Wolff-Parkinson-White syndrome, any condition associated with risk of stroke and concomitant use of MAO-A inhibitors, ergotamine or other 5-HT agonists use are considered contraindications to the use of zolmitriptan. 39 And like other triptans and ergots, there is concern with zolmitriptan about vasoconstriction. Although rare, triptan use has been associated with serious cardiovascular events, including myocardial infarction, stroke, and arrhythmia. In particular, the potential for triptans to be associated with hypertension and chest pain have raised the most concern.27,95 However although blood pressure elevations do occur, they have shown to be minimal and of no clinical significance in patients without cardiovascular disease. Importantly, almost all serious adverse events with triptans have occurred in patients with who had other diseases which mimicked migraine or contraindications to triptans. 95 Indeed, the cardiovascular risk with triptans has been shown to be rare, with a serious adverse event occurring in only about one per one million attacks. Furthermore, a study by Hall et al evaluated 13,664 participants without cardiovascular risk factors who were prescribed a triptan and observed that there was no significant increased risk of cardiovascular morbidity or mortality with the use of triptans. 96 Furthermore it should be noted that up to 15% of patients report non-cardiac chest symptoms with triptan use. 13 In summary, with proper patient selection, triptans, such as zolmitriptan, are both effective and very safe migraine abortive therapies.
Conclusions
The pathophysiology of migraine is complex. Current data suggests that migraine is a chronic low serotonin state interictally, with sudden rises in 5-HT release during acute migraine attacks. The importance of serotonin in the pathophysiology of migraine is further supported by the efficacy of pharmacological agents that modulate serotonin such as the 5-HT1B/1D agonists like zolmitriptan. Zolmitriptan's pharmacokinetics, pharmacodynamics, tolerability and safety have been well studied. The different formulations (tablet, orally dissolving tablet and nasal spray) allow a wider range of choices for the patient's individual needs. In addition multiple double-blind placebo controlled studies have demonstrated zolmitriptan's efficacy in the treatment of migraine and with proper patient selection, zolmitriptan is not only effective but also very safe to use.
Disclosure
The authors report no conflicts of interest.
