Abstract
The dopamine agonist pramipexole (PRA) ((S)-2-Amino-4,5,6,7-tetrahydro-6-(propylamino) benzothiazole dihydrochloride; molecular formula C10H17N3S-2(HCl), is a D3 selective compound, approved in 1997 for the treatment of Parkinson disease and in 2006 for that of idiopathic restless legs syndrome (RLS). Because of its tolerability, safety and half-life, PRA is favored over levodopa and on the other ergot derivate dopamine agonists, and it is considered nowadays one of the first choices in the therapy of RLS. PRA is rapidly and completely absorbed after oral administration, its protein binding is around 15%, it is almost unaffected by hepatic metabolism and excreted by urine unchanged. PRA has a linear pharmacokinetics, with a half-life ranging between 8 and 14 hours. Double-blind, placebo-controlled studies demonstrated that PRA, even at low dosages and since the first nights of administration, is significantly effective on the typical sensitive symptoms of RLS, on the periodic leg movements during sleep, and in improving the quality of life of patients with RLS. A subjective improvement of sleep quality is usually also reported by the patients, but the polysomnographic assessment gave less solid results on objective sleep parameters. The most common PRA related side effects include headache, nausea and orthostatic hypotension. Data on the long-term therapy of PRA in RLS, and on the efficacy of PRA in symptomatic forms of RLS are warranted to better delineate the role of PRA in RLS treatment.
Introduction
Restless leg syndrome (RLS) is an often under diagnosed sleep-related sensorimotor disorder which affects up to 10% of the adult general population. 1 The essential main clinical features of the syndrome, that are also mandatory for the diagnosis, are the following: an urge to move the legs because of a disagreeable sensation, an appearance or a worsening of the symptoms during rest or inactivity, a partial or total recovery of the symptoms by movement, and an appearance or a worsening of the symptoms at evening or night time. 2 Depending on the severity and on the frequency of occurrence of the symptoms, RLS is often associated to insomnia and reduced quality of life. 3 Although not necessary for the diagnosis, the polysomnographic recording may help in the differential diagnosis of complex cases and, in more than 80% of patients, unmasks periodic leg movements (PLM) during sleep. 4 PLM are repetitive leg jerks characterized by a triple flexion movement of the lower limbs, usually associated to electroencephalographic arousals and to an increasing of the heart rate, which can contribute to sleep disruption. 5 A PLM index (number of PLM per hour) higher than 10 is usually considered pathological (Fig. 1).
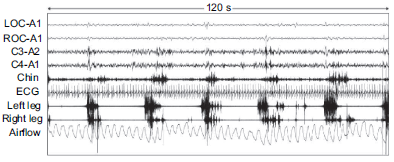
Polysomnographic sample of periodic leg movements during stage 2 of NREM sleep.
The idiopathic one is the most common form of RLS and can be inherited as an autosomal dominant trait. 6 Since now, eight loci (Chromosome 2q, 7 14q, 8 9p, 9 20p, 2q, 10 4q, 11 17p 11 and 19p 12 ) have been identified by using the linkage analysis, and in all but one 7 the model of inheritance was always dominant. Recently, two large genome-wide scan studies recognized three genomic regions encoding the intronic variants of the genes MEIS 1, BTBD9 and MAP2K5.13,14
The presence of uremia, pregnancy, hyposideremia, rheumatoid arthritis, and several neurological disorders involving either the central (CNS) or the peripheral nervous system, differentiate the secondary to the idiopathic form of RLS. 15 In primary RLS form the neurological examination, as well as the majority of instrumental diagnostic investigations, are normal. The age of onset or RLS may vary widely from childhood to more than 80 years, but usually the syndrome affects adult population. Patients with primary familiar form are more likely to have an early-onset RLS than those with secondary form. 16 The natural course of RLS is generally chronic with only sporadic and irregular periods of remission. 2
Even though the pathogenesis of the RLS is still unknown, there are several pieces of evidence that address to a possible dysfunction of the descending dopaminergic neurons from the hypothalamic A11 region to the intermediolateral and dorsal spinal gray matter. 17 RLS worsens with antidopaminergic treatment, 18 can be induced by hyposideremia (iron is the coenzyme of the tyrosine-hydroxylase, the limiting enzyme in the dopamine synthesis), 19 may be associated with Parkinson diseases, 20 and shows a circadian trend of the symptoms and PLM distribution which is inversely related to the levels of blood and cerebro-spinal fluid dopamine. 21 As well as in reward and cognitive processes, dopamine is also strongly implicated in regulation of locomotor activity and movement in general. 22 A possible connection between the dopaminergic and the nociceptive system may be illustrated by the regulation of the sensitive input in the dorsal horns of spinal cord, operated by descending dopaminergic neurons. 23 In mice, the A11 area consists of two groups of approximately 200 dopaminergic neurons symmetrically located in the posterior hypothalamus, which receive afferents from the endogenous circadian pace-maker (suprachiasmatic nucleus) and projects to diencephalus, brainstem and spinal cord (intermedio-lateralis and dorsal horns gray matter). Immunohistochemstry demonstrated a positivity of the A11 neurons for the tyrosine hydroxylase (TH). 24 The A11 axons extend over the whole spinal cord to the D2 subfamily receptors, which have been observed to have the highest densities in the superficial layers of the dorsal horn at cervical and lumbar levels. The connection between A11 and intermedio-lateralis spinal column it is supposed to be implicated in the hypothalamic control of vegetative functions, while the one to the dorsal gray matter may explicate regulation of the sensory input from the posterior root. Dopamine production, correlation to circadian endogenous pace-maker, and regulation of the spinal sensory input, represent the three main aspects which suggest that a dysfunction of this pathway could induce a RLS like symptomatology. Low doses of dopamine and pergolide reduce the monosynaptic spinal stretch reflex amplitude in WT but not in D3 knock-out (KO) mice. 25 These results stand for a limitation of the spinal reflex by dopamine and for an excitation of the spinal reflex by D3-antagonists acting in both cases on D3 receptors. However, should be noted that Earley et al found no histopathological abnormalities examining the A11 dopaminergic neurons in autopsy brains of 6 patients with RLS. 26
Mainly, the dopaminergic hypothesis in RLS is supported by the fact that both RLS and PLM respond dramatically to dopamine-agonists medication, even at very low dosages and since the first night of administration.27,28 Akpinar was the first to observe an effectiveness of the L-Dopa plus benserazide in RLS. 29 Nowadays L-Dopa is not frequently used because of its short half life and the consequent high incidence of rebound of symptoms within the night. 30 The possible typical side effects (mainly represented by the risk of cardiopulmonary fibrosis) due to the ergot derivates actually limit the use of bromocriptine, pergolide and cabergoline, which often need a pre-medication with the peripheral dopamine-antagonist domperidone. 31 For their effectiveness, half-life and safety, in the last two decades, the nonergot derivates dopamine agonists, such as pramipexole (PRA), ropinirole and rotigotine have become the first line treatment in RLS. 32 Alternative non dopamine-agonists treatments are represented by opioids, antiepileptics, benzodiazepines and clonidine. 33
Mechanism of Action, Metabolism and Pharmacokinetic Profile
PRA (S)-2-Amino-4,5,6,7-tetrahydro-6-(propylamino) benzothiazole dihydrochloride; molecular formula C10H17N3S-2(HCl), molecular weight 284.25 (Fig. 2), is a nonergot derivate dopamine agonist compound from Boehringer Ingelheim research, first approved in 1997 for the treatment of the idiopathic Parkinson Disease. Afterward, PRA was approved in November 2006 in U.S. by the Food and Drug Administration, and in April 2006 throughout the European Union for the symptomatic treatment of moderate to severe idiopathic RLS. PRA is commercially available under the trade names of Mirapexin®, Sifrol®, Pexola®, Mirapex® with three different tablets doses: 0.088 mg of PRA base (equivalent to 0.125 mg PRA salt), 0.18 mg of PRA base (equivalent to 0.25 mg of PRA salt), 0.7 mg of PRA base (equivalent to 1.0 mg of PRA salt).
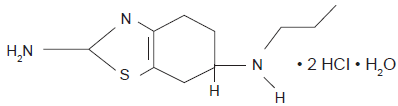
Chemical structure of pramipexole.
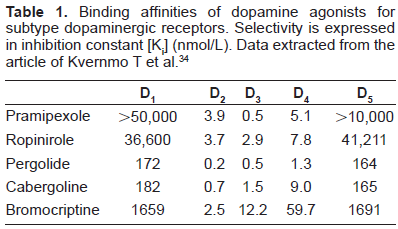
PRA acts as agonist on dopamine receptors, which consist on 5 different subtypes, classified in the 2 subfamilies of D1-like receptors (D1 and D5) and of D2-like receptors (D2, D3, D4). 34 PRA is a preferential D2 agonist with a high selectivity for D3 subtype receptors. Contrary to the ergot-derived dopamine agonists, PRA has a very low affinity for 5-HT2A and 5-HT2B receptors. 35 The action on receptors 5-HT2A and 5-HT2B probably accounts for the restrictive cardiac valvulopathies observed in patients treated chronically with ergot-derived. Possible targets of PRA in RLS are represent by the D3 receptors located in the dorsal spinal gray matter, with a supposed inhibitory modulation on the sensitive peripheral inputs. 23 Although less sustained by the literature, possible action of PRA on D2-D3 receptors in basal ganglia, might be important in the beneficial effects of PRA in RLS. 17
PRA is rapidly and completely absorbed after oral administration. Oral bioavailability is over 90% and the peak of plasma concentration is reached within 1-2 hours on an empty stomach or 3-4 hours with food although the extent of absorption remains unaffected. 36 Protein binding of PRA is 15%, and the volume of distribution of the drug is 7 liters/kg (~400 lt). PRA is almost unaffected by hepatic metabolism and 90% of the drug is excreted by urine unchanged and only 2% can found in the faeces. No potent cytochrome P450 inhibition has found for PRA. 37 PRA has a linear pharmacokinetics over its entire therapeutic range, and a low inter-subjects plasma concentration variability. The total clearance of PRA is about 500 ml/min, and the renal clearance is about 400 ml/min. 38 The half-life of PRA ranges between 8 and 14 hours, depending on the age and on the health state of the patients. Since the elimination of PRA depend on renal function, it should be used carefully and the dose should be adjusted in patients with renal failure. Subjects with a creatinine clearance higher than 20 ml/min do not require a reduction of the daily dose; however, should be underlined that PRA has not been systematically tested in hemodialytic patients or in subjects with severe renal failure. It is not required a dose adjustment in patients affected by hepatic diseases.
Clinical Studies
Most of the available literature data concern the clinical effects of PRA in Parkinson Disease.
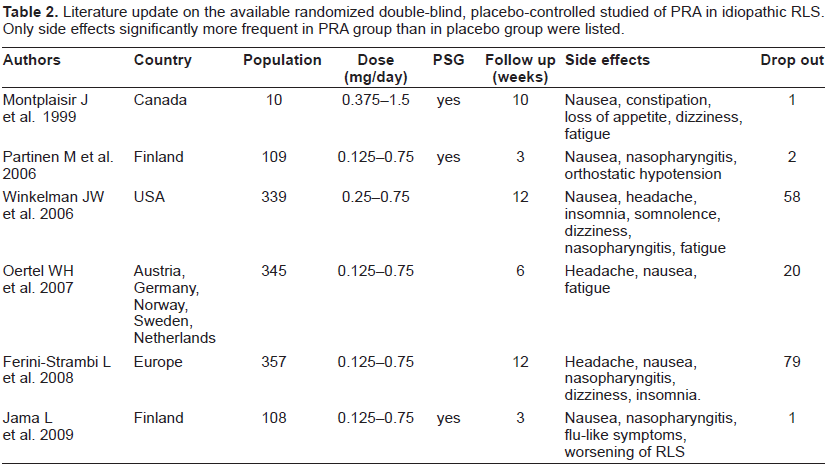
From 1998 to May 2009 (Pub Med database) 15 studies on PRA in RLS were published.27,39–51 Six of the above mentioned studies were conducted by a double-blind, placebo-controlled method39–44 (Table 2) and only two of these six studies included an instrumental polysomnographic evaluation.39,44
Binding affinities of dopamine agonists for subtype dopaminergic receptors. Selectivity is expressed in inhibition constant [Ki] (nmol/L). Data extracted from the article of Kvernmo T et al. 34
Literature update on the available randomized double-blind, placebo-controlled studied of PRA in idiopathic RLS. Only side effects significantly more frequent in PRA group than in placebo group were listed.
Lin et al performed the first open-label trial with a mean dose of 0.3 mg of PRA in 16 consecutive patients with RLS, who had previously experienced failure with other dopaminergic therapies. 47 The authors reported that, after 2-3 months of therapy, nocturnal leg restlessness, involuntary leg movements, and insomnia significantly decreased in 12, 10, and 11 patients, respectively. The first double-blind, placebo-controlled study was carried out by Montplaisir et al in 1999 on a small population of 10 patients with idiopathic RLS. 39 The authors observed a good efficacy of PRA after 1 month of treatment not only on leg discomfort (nine of the 10 patients reported a complete disappearance of RLS symptoms, while one reported a major improvement), but also on PLM (pramipexole produced a 98% decrease in both the number and the index of PLM compared with placebo). The largest series of patients included in single study was enrolled by Ferini-Strambi et al who evaluated 357 patients with RLS by a multi-centre randomized, double-blind, placebo-controlled trial, founding a significant superior improvement, in both sensitive symptoms and sleep complains, of PRA compared to placebo. 43 Winkelman et al conducted a 12-week, double-blind, randomized, placebo-controlled trial of fixed doses of pramipexole (0.25, 0.50, and 0.75 mg/day) in 344 patients with RLS. 41 PRA ameliorated significantly RLS symptoms compared to placebo with a positive correlation with the daily dose used, and significantly improved their quality of life measured by a specific questionnaire. Similar results have been obtained by the PRELUDE study, which included 109 patients treated with 0.125 to 0.75 mg/day of PRA per 3 weeks. 40 The authors, beside the significant improvement in RLS symptoms, also demonstrated a significant reduction of PLM compared to placebo. The same group of researchers repeated a 3 weeks, polysomnographic, double-blind, placebo-controlled study in 107 patients with RLS, obtaining comparable good results on symptoms and PLM. 44
Therefore, the literature agrees in define PRA as a well tolerated drug with a good efficacy in idiopathic RLS form in the control of both sensitive symptoms and PLM, at least at short follow up intervals.
The dose used in the available studies ranges between 0.125 mg and 1 mg per day, usually given as a single evening dose 2-3 hours before bedtime.
Low doses of PRA are effective in RLS even from the first night of administration on both sensitive and motor components.27,48 Manconi et al performed a single-blind placebo-controlled study in 32 consecutive idiopathic RLS de-novo patients, with a PLM index greater than 10. 27 Patients underwent two consecutive full-night polysomnographies, and received 0.25 mg of PRA or placebo at 9:00 p.m., before the second night of recording. Compared to placebo, PRA improved acutely RLS symptoms (VAS: from 7.4 ± 1.68 to 1.3 ± 1.62, p < 0.00001) and suppressed the PLM index (from 45.8 ± 33.56 to 9.4 ± 11.40, p < 0.0002). A mild improved was also observed on sleep quality, with an increasing in the percentage of stage 2 non-rapid eye movement (NREM). Moreover, the comparison with the placebo, demonstrated that PLM, as expected, are not influenced by the placebo effect. Similar results, concerning the acute efficacy of PRA on RLS, have been observed by Merlino et al. 48 The immediate efficacy of PRA might be important, not only for the obvious benefit to patients in obtaining rapid results, but also because it suggest two further possible interesting future drug applications: 1) the on-demand therapy, which may be helpful in patients with intermittent or mild symptoms and for patients (treated and untreated) who need to be rest for a long time, such as in air, train or car travel, and social events during the evening; 2) the use of a standard single dose of PRA as a diagnostic ex-juvantibus test in the differential diagnosis of complex cases. 27 However a validation study for a possible diagnostic pharmacological test in RLS is available at the moment only for Levodopa and not for PRA. 52
Almost no data are today available concerning the long effects of PRA in RLS. Montplaisir et al performed a telephone follow-up study on a cohort of 195 RLS patients. 51 After one year, 43 patients had discontinued pramipexole (20 because of side effects, 6 because of a lack of efficacy, 6 for both and 11 for reasons other than side effects), while the remaining 152 ones still reported a good efficacy on symptoms, confirming the effectiveness and the safety of PRA in the long-term treatment of RLS. In particular, patients who continued PRA for at least 1 year, reported a mean decrease in RLS symptom severity of 80.0% ± 20.8% (n = 152) and 95% of the patients (94.7%) reported a decrease in severity of 50% or more at follow-up compared with baseline.
Promising results have been also reported also by using PRA in the symptomatic form of RLS secondary to renal failure. 50
Safety
PRA is usually well tolerated by the RLS patients, and the majority of the adverse events are mild or transitory. The most frequent side effects met by the patients are represented by headache, fatigue, nausea and dizziness, that have been described usually with an incidence lower than 20%-30% of newly treated patients, during the first weeks of therapy.42–44 The above mentioned adverse events decreased in prevalence under 5% after few months of treatment. With a lower incidence, insomnia, nasopharyngitis, orthostatic hypotension, dry mouth, confusion and difficulty concentrating, have been also reported significantly more often by patients treated with PRA, compared to subjects who were receiving placebo. Contrary to what may happen in the treatment of Parkinson disease, at the common doses used for RLS patients, hallucinations are usually not reported. Side effects, lack of efficacy, fear of taking an antiparkinsonian drug for an extended period of time, fear of potential drugs interaction, and economical reasons, were cited, by Montplaisir et al among the causes of the long-term discontinuation of PRA. 51
A special mention should be reserved to daytime vigilance. While sleepiness an insomnia are possible side effects clearly observed in most of the past trial with PRA, during the last decade the attention of physicians was captured by the report of car accidents due to irresistible sleep attacks occurring in patients affected by Parkinson disease and treated by PRA or ropinirole. In the first series of 8 patients with motor vehicle mishaps, described by Frucht et al, 7 parkinsonian patients were taking PRA, and one ropinirole. 53 The authors reported that all attacks occurred after patients began taking pramipexole or ropinirole and stopped after the drugs were discontinued. These sleep attacks were put in relationship with a possible downregulation of dopaminergic input to the reticular activating system, maybe by acting on presynaptic receptors, which are stimulated by lower doses of these agents. 54 Several further cases have been described afterward, even with the levodopa and the other ergot or nonergoline dopamine-agonists derivates. 55 In the majority of the cases males were predominant, the sleep attacks occurred at both high and low doses of the drugs, with different durations of treatment (0-20 years), and with or without advice signs. 56 Since patients with Parkinson disease are often affected by overlapping sleep disorders, such as insomnia, REM behavior disorder, restless legs syndrome or sleep apnea, at the beginning the attribution of sleep attacks to dopamine agonists was thoroughly discussed. Concerning the low doses of PRA commonly used in RLS, there is no clear evidence of sleep attacks directly linked to the treatment. Winkelman et al in their controlled study on PRA in RLS, observed 4 cases of sleep attacks equally distributed between PRA (2 cases) and placebo (2 cases) group, 41 while Oertel et al found no sleep attacks among their patients. 42 Moreover, Möller et al performing a questionnaire survey among 156 RLS patients, found that subjects on dopaminergic therapy featured a lower risk of sleep attacks than untreated patients. 57 On the other hand, Micalef et al showed that a single oral dose of pramipexole (0.5 mg), and not of Levodopa or bromocriptine, induces sleepiness as assessed by MSLT, in healthy young subjects, and that this adverse event may occur without prior warning. 58
In the same way, previously reported in Parkinson disease,59,60 gambling or other abnormal compulsive behaviors, have been described to be associated with dopamine agonists treatment. In a series of 70 RLS patients, Driver-Dunckley et al observed an increased in gambling behavior in 7%, and in sexual desire in 5% of the interviewed subjects. 61 Two cases of punding, which consists of a complex impulse control disorder (pathological gambling, hypersexuality, excessive spending), with ropinirole and pergolide, have been recently described by Evans et al in two patients affected by RLS. 62 This latter possible side effect needs further prospective studies to confirm, at least in RLS, the link between dopamine agonists and compulsive behaviors. 63
Augmentation
Augmentation is a possible complication of long-term dopaminergic treatment of RLS, which consists on a worsening of RLS despite therapy, compare to the severity of symptoms before the onset of treatment. 64 Frequently reported features of augmentation are represented by: an anticipation of RLS symptoms respect to the usual time of onset, an anatomical extension of the symptoms to body parts previously not affected, a shortening of the latency of the symptoms appearance at rest, a worsening of symptoms severity with an increase of the drug dose. 65 Augmentation in RLS is typically induced by dopamine agonists and levodopa and is negatively correlated with the halflife of the molecule, with the highest prevalence seen for levodopa and the lowest for cabergoline. 66 The prevalence of augmentation in RLS patients chronically treated with PRA or ropinirole, ranges between 8 to 25%. 67 However, two reports of augmentation after long-term treatment with the non-dpoamine-agonist opioid tramadol, have also been published.68,69
Augmentation might be due to a hyper-dopaminergic stimulation of the D2 receptors subfamily at the spinal cord level or D1 at basal ganglia, and seems to be facilitated by low ferritin levels.70,71
Specific diagnostic criteria and a severity scale have been elaborated to evaluated the augmentation in RLS. 65
Efficacy
The end points of the previously cited clinical studies on PRA in RLS, focused on four possible symptomatic areas of interest: the typical sensitive RLS symptoms (disagreeable limb sensations and urge to move them), the sleep quality, the PLM, and finally the quality of life.
The subjective sensitive symptomatology is usually assessed in the clinical practice by the International RLS Rating Scale (IRLSRS), 72 which consist of a ten-question, validated and reliable self-administered questionnaire to measure the disease severity by a range of values from 0 to 40. Because of the IRLSRS results refers to the 4 weeks period of symptoms before the administration, often the effects of PRA for shorter intervals have been evaluated by using visual analogical scale (VAS) or the Clinical Global Impressions-Improvement (CGI-I) assessment. All of these instruments demonstrated that PRA is significantly more effective than placebo on RLS sensitive symptoms.40–44 However, a recent meta-analysis showed a notable placebo effect in the subjective evaluation of efficacy for PRA and for the other RLS medications, especially by using the IRLSRS. 73 The placebo effect is smaller for subjective and objective sleep parameters, and almost absent when PLMS were considered as the endpoint.
When the effects of PRA on sleep quality are tested by validated subjective questionnaire instruments, the percentage of patients complaining of sleep disruption decreases significantly after treatment, especially concerning the difficulty in falling asleep. When sleep parameters were assessed by the polysomnography, the improvement is less evident. In the acute fashion, PRA induced only a significant increment of the percentage of stage 2 NREM sleep. 27 Longer term polysomnographic results found no significant differences between PRA and placebo for the main variables, such as sleep efficiency, total sleep time, sleep latency, slow wave sleep percentage and number of awakenings.39,44 A tendency to a REM sleep suppression by PRA has been reported. 39
All the published studies reported a strong efficacy of PRA in reducing PLM either in acute or in chronic treatment.27,44
The quality of life in RLS was evaluated not only by the generic MOS 36-item Short-Form health survey, 74 but also by the specific validated RLS quality of life questionnaire (RLSQoL). 75 It consists in a 18 items self-administered questionnaire which takes in consideration the following areas: severity of RLS symptoms, evening activities, impact on morning activities regarding job or non-job appointments, concentrating in afternoon-evening, sexual activities. In both of the questionnaires (SF-36 and RLSQoL), a significant improvement of quality of life have been demonstrated for RLS patients after medication with PRA. 40
Place in Therapy
Since their notable efficacy and tolerability, dopamine-agonists are nowadays considered as the first choice for the treatment of RLS. At the present time, levodopa is not frequently used because of its short half-life and the consequent high incidence of symptoms rebound within the night and/or daytime augmentation.
Typical severe side effects, such as cardiopulmonary fibrosis, associated with ergot derivate molecules limit also the use of bromocriptine, pergolide and cabergoline, which, furthermore, often require a concomitant medication with the peripheral dopamine-antagonist domperidone to control nausea and vomiting. Due to their tolerability and half-life (approximately 6-8 hours), low evening doses of the D3-agonists PRA and ropinirole have become the first line treatment in RLS. However, follow-up studies demonstrated an augmentation effect also following the use of D3 non-ergoline agonists in about one-third of patients, appearing after at least 6 months of therapy. 76
Polysomnographic studies proved that ropinirole and PRA are noticeably effective in reducing both sensitive symptoms and PLM.77,78 For both of the mentioned drugs have been demonstrated also an acute efficacy. 27
A direct comparison between ropinirole and PRA is still not available. Quilici et al performed a meta-analysis of the literature to compare the efficacy and tolerability of PRA to ropinirole in RLS. 79 The analysis confirmed the significant efficacy for both drugs compared to placebo. Nausea, vomiting, dizziness, and somnolence were significantly higher for ropinirole than PRA, which also showed a superior reduction in the mean IRLS score, and a higher CGI-I response rate. This indirect comparison seems to favor PRA over ropinirole, but direct comparisons are warrant to achieve stronger evidences.
Conclusions
The nonergot derivate dopamine-agonist PRA is a well tolerated and effective treatment for the idiopathic form of RLS, with a demonstrated efficacy in improving the typical sensitive symptoms, the motor components (acatisia and PLM) and the quality of life of patients. PRA exerts its result also at low dosage and since the first night of treatment, and belong to the first line choice drugs in RLS therapy. Nausea and orthostatic hypotension, together with the augmentation phenomena, represent the main possible troubles met by the physicians in the management of the therapy. The long-term compliance and efficacy of PRA, a direct comparison of PRA to ropinirole, as well as the effect of PRA on sleep quality and on the secondary forms of RLS, represent the main topics to investigate by specific future researches.
Disclosures
The authors report no conflicts of interest.
