Abstract
Restless Legs Syndrome (RLS) is a neurological disorder characterized by unpleasant sensations in the legs and an irresistible urge to move them to relieve discomfort. Evidence-based guidelines consider dopaminergic therapy to be the mainstay of treatment for RLS. Ropinirole is a modern, non-ergoline dopamine agonist which has been widely studied for the treatment of moderate-to-severe primary RLS. Data from placebo-controlled studies show that ropinirole significantly improves the symptoms of RLS. These improvements are supported by data from individual studies and pooled analyses. In addition, ropinirole significantly enhances the objective measures of RLS motor symptoms, such as periodic leg movements, and subjective measures of sleep. Ropinirole is generally well-tolerated, but a newly developed extended-release formulation may benefit patients who warrant an extended duration of therapy.
Introduction
Restless Legs Syndrome (RLS) is a neurological disorder affecting up to ten percent of the population.1–3 The RLS Epidemiology, Symptoms, and Treatment (REST) primary care study found the prevalence of RLS symptoms in a primary care population of over 20,000 respondents from the United States and Europe to be 11.1%, with 9.6% of patients reporting symptoms at least weekly and 3% reporting moderately severe symptoms causing appreciable distress. 4 Clinically relevant RLS with significant sleep disturbances is both common and underdiagnosed. 5 At least 5% of adults experience frequent RLS symptoms, while older adults and women are affected even more often. 6 Clinically relevant RLS occurring at least twice a week causes distress in up about 3% of the adult population and in about 0.5% of children aged 8 to 11, increasing to 1% in adolescents 12 to 17.4,7,8 The disorder is characterized by an urge to move the legs, usually accompanied or caused by uncomfortable leg and feet sensations including feelings of burning, tickling or crawling, pain cramping, numbness or weakness. For most people with RLS, symptoms begin or worsen during periods of rest or inactivity. Moving the legs temporarily relieves the symptoms. Symptoms typically worsen or occur only in the evening or at night, disturbing the sleep of the affected persons or their partners. 9 Confounding conditions have to be excluded. 10 RLS can also severely impair daily life, and may lead to depression (17%-27%) and/ or anxiety (8%-23%).4,5,11,12 RLS is associated with periodic leg movements (PLMs) in about 80% of patients and they are supportive clinical features of RLS.11,13,14 Sleep disturbance is caused by both the sensory symptoms of RLS and the occurrence of involuntary PLMs characterized by extension of the big toe in variable combinations with flexion of the ankle, knee, and, sometimes, hip which may occur while the patient is awake (PLMA), thus preventing sleep onset, or while they are asleep (PLMS), which may result in arousal to a lighter stage of sleep or from sleep to wake. The resulting impairment of either quantity or quality of night-time sleep may also lead to significant daytime fatigue and weariness with a negative effect on mood. 15
RLS is currently most common in people over the age of 60, but it can develop at any age and may be related to conditions such as anemia, pregnancy or diabetes. The condition may be also familial: primary RLS is a highly familial trait with heritability estimates of about 50%. 16 RLS is considered a polygenic disorder in which a number of genetic variants contribute to the phenotype. Linkage studies in families with RLS revealed several loci of genetic determination but have not yet led to the identification of disease-causing sequence variants.
Although the pathophysiology of RLS is still unclear, dopaminergic dysfunction appears to play a central role, with the etiology of RLS being linked to dopaminergic regulation. Central dopamine signaling exhibits a daily rhythm with a nadir in the evening 17 thereby supporting the pathophysiologic role of brain dopaminergic hypoactivity in RLS-PLMs, 18 and may explain the circadian recurrence of the related symptoms. In particular, dysfunction or atrophy of a supraspinally located dopaminergic region in the hypothalamus (hypothalamic A11 nucleus) with descending pathways that target the preganglionic sympathetic neurons, dorsal horn region, interneurons and somatic motor neurons, has been hypothesised as being intimately linked with RLS-PLMs.19,20
The pathology of the primary dopaminergic system has not been clearly demonstrated and is complicate by the fact that the dopaminergic neurons of the A11 system appeared normal in six RLS autopsy brains. 21 Thus, RLS remains a complex multivarious disorder that may implicate many brain areas, including the striatal regions, 22 thalamic structures, 23 and regions belonging to the medial pain system (thalamus, anterior cingulated cortex and insula) where abnormal dopamine and opioid activity have been found.24,25
However, a striking advance was the first documented efficacy of dopaminergic drugs, highlighting the involvement of the dopamine system in RLS.26,27 Dopaminergic drugs are today considered the treatment of choice for patients requiring pharmacologic interventions, and have been increasingly used to treat RLS and periodic limb movement disorder [PLMD, currently defined by a PLMS index–-number of PLMS (extension of the big toe in variable combinations with flexion of the ankle, knee, and, sometimes, hip) per hour of sleep–-of 5 or greater associated with otherwise unexplained sleep-wake complaint and requiring a polysomnographic confirmation and the exclusion of other causes of sleep disturbances]. 28
There are a growing number of evidence-based practice parameters for RLS.29–32 Among the dopaminergic agents, ropinirole, a selective dopamine agonist with a nonergoline structure, is currently licensed for the treatment of patients with moderate to severe RLS clinical symptomatology following several clinical trials.33–39 RLS clinical trial programs with ropinirole are the largest published to date, and show that ropinirole produces rapid and significant reductions in the sensory and motor symptoms of RLS associated with significant improvements in sleep and quality of life. Moreover, these benefits are maintained during long-term treatment (months/years) without significant adverse effects or drug interactions. Ropinirole is thus a valuable tool in the management of RLS.32,40
Mechanism of Action, Metabolism and Pharmacokinetic Profile of Ropinirole
Ropinirole is a selective non-ergoline dopamine D2-like receptor agonist with affinity for the D2, D3 and D4 receptor subtypes. It has negligible affinity for the dopamine D1 receptor or other common neurotransmitter receptors, and is about 20 times more active at the central D3 than at the D2 receptor.41–43 Ropinirole binds to both central and peripheral dopamine receptors. In the central nervous system, ropinirole binds to postsynaptic dopamine D2 receptors, acting as a replacement stimulus for the dopaminergic neurotransmission. The action of ropinirole at presynaptic central dopamine receptors can reduce the turnover and release of dopamine from presynaptic terminals producing a neuroprotective effect by decreasing the amount of toxic metabolites and therefore oxidative stress. 44 In the periphery, the action of ropinirole at dopamine D2 receptors increases the sympathetic activity tone. Activity at peripheral presynaptic dopamine receptors results primarily in hypotension and nausea, 45 effects that are reduced by slow titration of the dosage.
Ropinirole is rapidly and almost completely absorbed, as demonstrated by the similar urinary excretion of radioactivity after oral and intravenous administration of approximately 10% or less. 46 The systemic pharmacokinetics of ropinirole are variable. Maximal plasma concentration (Cmax) are reached after about 1.5 hours [time to reach maximal concentration (tmax),range 0.5 to 4 hours], and the elimination half-life appears to be approximately 3 hours. The average bioavailability of the oral tablet formulation is approximately 50%. 47
Like other lipophilic amines, ropinirole is rapidly and extensively distributed. The volume of distribution at the steady state is approximately 7.2 L/kg after oral administration. Ropinirole shows low plasma protein binding (10% to 40%) irrespective of its plasma concentration.48,49 The blood/plasma ratio of 1.2 suggests that ropinirole is taken up to some extent by blood cells.50,51
Most of the absorbed dose of ropinirole is cleared by metabolism in the liver, with only 10% of the administered dose being excreted as unchanged ropinirole.46,52 All the pharmacological activity achieved by oral intake can be attributed to the parent molecule, the metabolites of ropinirole being practically inactive.46,53,54 The main enzyme responsible for ropinirole metabolism is the cytochrome P450 (CYP) enzyme CYP1A2, with a low affinity component, CYP3A, making only a minor contribution. 53 Human CYP1A2 has considerable individual variations in its activity and is induced by smoking. 55 The excretion of ropinirole-derived products is predominantly via the urine, with a mean terminal elimination half-life of about 6 hours (range 2 to 10 hours) independent of the dose.46,52,56
The average dose-normalized Cmax and area under the concentration-time curve (AUC) are about twice as high at steady state after 3 times daily administration than following single dose administration. There is no marked difference in the clearance of ropinirole between single and repeat dose administration. When single doses of ropinirole in the range of 2 to 12 mg are administered, Cmax and AUC increase approximately in proportion to the administered dose. This linear pharmacokinetic behavior of ropinirole is also indicated by its constant oral clearance over a wide dose range. 56
Food intake affects the pharmacokinetics parameters of ropinirole. The mean Cmax was reduced by 25% following a high-fat breakfast and the median tmax was delayed by 2.6 hours compared with results obtained in the fasting state. There was also a small but significant reduction (13%) in the AUC for ropinirole (95% confidence interval 77% to 98%). Thus, food reduces the rate of absorption but only minimally affects the extent of ropinirole absorption, these effects not being expected to result in any clinical consequence. 57
Ropinirole is thus rapidly and almost completely absorbed and shows an approximately linear pharmacokinetic after single dose and steady-state administration. Population studies have shown that the pharmacokinetics of ropinirole are unaffected by sex, mild-to-moderate renal impairment or several common concomitant diseases or drug treatments. However, the CYP1A2 inhibitor ciprofloxacin produce increases in the plasma concentrations of ropinirole when these 2 drugs are coadministered, but no interaction is seen with theophylline which, like ropinirole, is also a substrate for CYP1A2. Clearance of the drug is slower in patients older than 65 years compared to younger subjects, and in women taking hormone replacement therapy compared with those who are not, but as the drug is titrated for effectiveness, this is of little clinical concern. There is no clear plasma concentration-effect relationship for ropinirole.58–60
Clinical Studies
Ropinirole has been widely studied for the treatment of RLS in a comprehensive clinical development program. Data from individual studies and pooled analyses document that ropinirole significantly improves RLS symptoms and significantly enhances objective measures of RLS motor symptoms, such as PLMs, and subjective measures of sleep.61–64 Randomized, placebo- controlled trials have been qualified for this review.
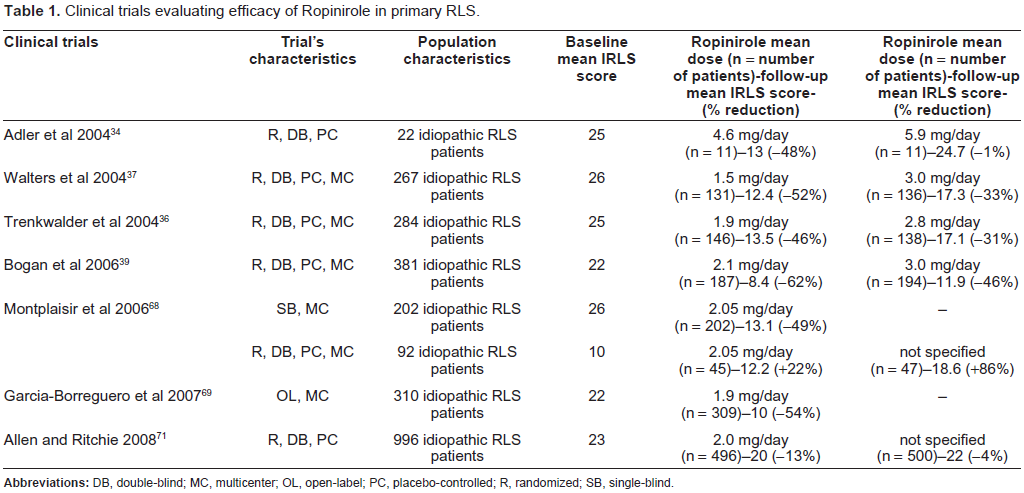
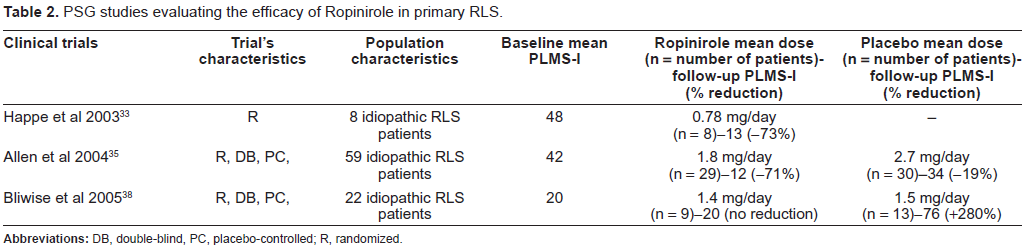
The tolerability and efficacy of a randomized treatment with either gabapentin (a structural analogue of γ-aminobutyric acid) or ropinirole in patients with idiopathic RLS (diagnosed according to the international RLS study group–-IRLSSG criteria) 65 were evaluated in a 4-week open clinical trial. 33 Patients were treated with either 300 mg gabapentin (8 patients; 5 women; mean age: 56 ± 9.2 years, range: 47-74) or 0.5 mg ropinirole (8 patients; 6 women; mean age: 63.4 ± 7.6 years, range: 49-72) as the initial dose. The dose was up-titrated until relief of symptoms was achieved [gabapentin mean dosage: 800 ± 397 mg (range: 300-1,200); ropinirole mean dosage: 0.78 ± 0.47 mg (range: 0.12-1.50)]. The IRLSSG questionnaire 66 scores improved significantly (p ≤ 0.018) in both groups, whereas the scores of the Epworth Sleepiness Scale (ESS) 67 remained unchanged and within normal limits. Polysomnography showed a reduction of PLMS (p < 0.03) and of PLMS index (PLMS-I, the number of PLMS per hour of sleep; p < 0.02) in both groups. Sleep efficiency was superior in the gabapentin group. Side-effects were only mild and mostly transient (numbness, dizziness, sleepiness and headache not leading to discontinuation of treatment in gabapentin group; transient nausea and sleepiness not leading to discontinuation of treatment in ropinirole group). After 6-10 months of follow-up, RLS symptoms were still improved in most patients.
The efficacy of ropinirole for the treatment of RLS was polysomnographically evaluated in 65 patients with RLS plus PLMS by means of a double-blinded, placebo-controlled, parallel-group, 12-week study. 35 Inclusion criteria were primary RLS (diagnosed according to the IRLSSG criteria), a PLMS-I >5, an IRLS score ≥15, and a minimum of 15 nights of RLS symptoms in the months prior to study entry. Of the 65 patients, 59 completed the post-baseline polysomnography (PSG) assessments and were included in the analysis of the study primary end points. There were 29 patients (17 women; mean age: 55.4 ± 10.3 years, range: 37-76) in the ropinirole group, and 30 (17 women; mean age: 53.3 ± 12.5 years, range: 30-79) in the placebo group. PLMS-I decreased more with ropinirole (48.5 to 11.8) than with placebo (35.7 to 34.2) (adjusted treatment difference: -27.2; 95% confidence interval: -39.1, -15.4; p < 0.0001). PLMS-I associated with arousal decreased from 7.0 to 2.5 with ropinirole but increased from 4.2 to 6.0 with placebo (adjusted treatment difference: -4.3; 95% confidence interval: -7.6, -1.1: p = 0.0096). PLMA-I decreased from 56.5 to 23.6 with ropinirole but increased from 46.6 to 56.1 with placebo (adjusted treatment difference: -39.5; 95% confidence interval: -56.9, -22.1; p < 0.0001). Ropinirole treatment significantly improved patients’ ability to initiate sleep (p < 0.05) and the amount of NREM sleep stage 2 compared with placebo (p < 0.001). Sleep adequacy (measured on the subjective Medical Outcomes Study sleep scale) was significantly improved with ropinirole treatment (adjusted treatment difference: 12.1; 95% confidence interval: 1.1, 23.1; p = 0.0316). In contrast, the placebo group showed a greater increase in NREM sleep stage 3-4 (p < 0.01). No serious adverse events occurred in either group. In conclusion, this study showed that ropinirole was effective in the treatment of both the sleep and waking symptoms of RLS.
Twenty-two patients (16 women; mean age: 60 ± 13 years, range: 40-83) meeting the IRLSSG criteria for primary RLS 65 and with an IRLSSG scale score >10 (IRLSSG, 2003) were enrolled in a double-blind, placebo-controlled, crossover study of ropinirole (0.5 to 6.0 mg/day) for RLS. 34 Patients had to be off RLS medication for at least 2 weeks before the baseline visit. Subjects were randomized to receive 4 weeks of placebo then ropinirole, or ropinirole then placebo. The study was completed by18 patients. The mean dose taken during ropinirole treatment was 4.6 mg/day (range: 1-6 mg), with 14 patients taking the full 6 mg/day. The mean dose of placebo taken was 5.9 mg/day with all but one patient taking 6 mg/day. Efficacy was assessed using the IRLS and ESS scales as well as an RLS diary that was completed twice a week. Mean RLS scores were lower at the end of the ropinirole treatment (13 ± 12) compared with the end of the placebo treatment (24.7 ± 7.2) (p < 0.001). The incidence of complete response (i.e. RLS score = 0) was 36% (8/22) during ropinirole treatment and 0% (0/22) during placebo treatment. The ESS scores did not change. Diary sheets for 19 patients revealed the mean rate of RLS to be 23% during placebo treatment and falling from 50% to 12% following treatment with ropinirole. Two patients prematurely discontinued the study drug during ropinirole treatment (1 due to lack of efficacy, 1 because of dizziness, nausea, and vomiting) and 1 patient during placebo treatment (because of syncope). Thus, the degree of improvement of RLS symptoms with ropinirole was approximately 50% (using the RLS rating scale and diary data).
A double-blind, randomized, 12-week, multinational (46 centers from Australia, Europe, and North America) study compared the efficacy and safety of ropinirole and placebo in RLS. 37 All participants had a diagnosis of primary RLS using IRLSSG criteria, a score at baseline of at least 15 on IRLS and at least 15 nights of RLS symptoms during the previous month. Patients were required to discontinue any current RLS medications, or those affecting sleep, for 5 half-lives or 7 nights, whichever was longer, before baseline. In total, 267 outpatients with moderate-to-severe RLS were randomly assigned to ropinirole (0.25-4.0 mg/day) or placebo, 1 to 3 hours before bedtime [ropinirole group: 131 patients (76 females); mean age: 54.9 ± 10.8 years, range: 29-77; placebo group : 136 patients (83 females); mean age: 56 ± 11.2 years, range: 29-79]. The primary end point was the change in IRLS score at week 12. Key secondary endpoints were the percentage of patients showing significant improvement on the Clinical Global Impression-Improvement (CGI-I) scale at week 12 and changes in the IRLS and CGI-I scale scores at week 1. Other measures included the Medical Outcomes Study sleep scale and the RLS Quality of Life questionnaire. Improvements were significantly greater for ropinirole than placebo for change in IRLS score at week 12 [-11.2 ± 0.76 vs. -8.7 ± 0.75,respectively; adjusted treatment difference: -2.5 (95% confidence interval: -4.6,-0.4); p = 0.0197]. All key secondary endpoints, sleep and QoL parameters improved significantly. Adverse events were typical for dopamine agonists; disease augmentation (see later), although not directly assessed, was not reported during treatment. In conclusion, ropinirole improved symptoms, associated sleep disturbance, and QoL of RLS patients and was generally well-tolerated.
A 12 week, prospective, double blind, randomised comparison study involving 284 patients from 10 European countries assessed the efficacy, safety and tolerability of ropinirole in the treatment of RLS patients. 36 All participants had a score of ≥15 on the IRLS scale. Patients were randomised to receive either ropinirole 0.25-4.0 mg once daily or placebo. There were 146 patients (88 women; mean age: 54 ± 11.1years, range: 30-78) in the ropinirole group, and 138 (91 women; mean age: 56.2 ± 11.2 years, range: 28-77) in the placebo group. The primary efficacy end point was the mean change from baseline to week 12 in the total IRLS score. Global improvements, evaluated with the Clinical Global Impression (CGI) scale, and improvements in sleep, health-related quality of life (QoL; using generic and disease specific measures), work and other activities were also assessed. 112/146 patients (76.7%) taking ropinirole and 109/138 (79.0%) taking placebo completed the study. Improvement in the IRLS score at week 12 with ropinirole (mean dose: 1.90 ± 1.13 mg/day) was greater than with placebo (-11.04 ± 0.719 versus -8.03 ± 0.738 points; adjusted treatment difference: -3.01; 95% confidence interval: -5.03 to -0.99; p = 0.0036). More patients in the ropinirole group (53.4%) showed improvement on the CGI scale at week 12 than in the placebo group (40.9%) [adjusted odds ratio = 1.7 (1.02 to 2.69); p = 0.0416)]. Significant differences on both the IRLS and CGI scales favoring ropinirole were apparent by week 1. Ropinirole was also associated with significantly greater improvements in sleep and QoL endpoints. The most common adverse events were nausea and headache. In conclusion, ropinirole was well-tolerated and improved RLS compared with placebo, with benefits apparent by week 1.
To test the clinical efficacy of ropinirole in the treatment of RLS, after undergoing successful open-label titration and dose adjustment with ropinirole for RLS symptoms over a period of 4 weeks, 22 RLS patients (13 women; mean age: 50.8 years, range: 46.4-55.2) were randomized to receive either placebo (13 patients) or ropinirole (9 patients; 0.25-1.5 mg at bedtime, maximum daily dosage 6 mg) for 2 additional weeks. 38 Outcome measures included assessment of PLMS recorded with nocturnal PSG and RLS symptoms as assessed with the IRLSSG rating scale. Secondary outcomes included sleep macroarchitecture. Ropinirole, at a mean dose of 1.4 mg significantly decreased PLMS and RLS symptoms as measured by the IRLS. Sleep macroarchitecture did not change. The mean PLMS-I during NREM sleep in the placebo group was 19.2 (range: 4.6-33.9) at baseline and 76.4 (range: 37.3-115.5) at week 6, compared with 19.7 (range: 0-45.6) in the ropinirole group at baseline, and 19.8 (range: 0-44.4) at week 6. No patients dropped out of the study due to adverse events. Side-effects included nausea, headache, and daytime somnolence and were dose-related. The majority of patients (20/22) elected to continue treatment with ropinirole upon study completion.
A multicenter, 12 week, double-blind, placebo-controlled, flexible-dose study enrolled 381 patients randomized to ropinirole [187 patients (109 women); mean age: 52.2 ± 12.8 years, range: 18-79] or placebo [194 patients (123 women); mean age: 52.4 ± 13.1 years, range: 19-78], 0.25-4.0 mg as needed and tolerated dose, once daily, 1 to 3 hours before bedtime. 39 The primary end point was the mean change from baseline to week 12 in the IRLS total score. Key secondary efficacy measures included the Clinical Global Impression-Improvement scale. One-hundred-sixty-four (87.7%) of the 187 patients randomized to ropinirole and 167 (86.1%) of 194 randomized to placebo completed the study. Significant treatment differences favouring ropinirole were observed for change in IRLS total score at week 12 (adjusted mean treatment difference: -3.7; 95% confidence interval: -5.4 to -2.0; P < 0.001) and for all of the 3 key secondary end points: mean change from baseline in IRLS total score at week 1, and proportion of patients who were much/very much improved on the CGI improvement scale at weeks 1 and 12. Ropinirole was associated with significantly greater improvements in subjective measures of sleep disturbance, quantity and adequacy, quality of life, and anxiety. Although treatment differences favouring ropinirole in daytime somnolence were observed, they were not statistically significant (P = 0.10). Ropinirole was generally well-tolerated (7 patients in the ropinirole group and 9 patients in the placebo group dropped out of the study due to adverse events) with an adverse event profile consistent with other dopamine agonists.
To investigate the long-term efficacy of ropinirole in patients with RLS and to assess the potential for relapse after the discontinuation of active treatment, 202 patients (51 women; mean age: 52.9 ± 13.5 years, range: 18-79) with primary RLS, who had experienced symptoms on 15 nights during the previous month and had an IRLS score ≥ 15, received single-blind ropinirole for 24 weeks. Patients underwent a pre-entry washout period of five half-lives or seven nights. The second part of the study randomly assigned 92/202 patients of the initial population to doubleblind treatment with either ropinirole (45 patients) or placebo (47 patients) for a further 12 weeks. 68 At week 20 of the single-blind treatment phase, after which no more changes in dose were allowed, the mean and median doses of ropinirole were 2.05 and 2.00 mg/day, respectively. At week 24, 10 patients (18%) were receiving the maximum dose of ropinirole, 4.0 mg/day. The primary efficacy variable was the proportion of patients relapsing during double-blind treatment. Additional efficacy measures included time to relapse, withdrawals due to lack of efficacy, improvement on the CGI improvement scale, change in IRLS score during double-blind treatment, and changes in sleep and quality of life (QoL) parameters. Significantly fewer patients relapsed on ropinirole than on placebo (32.6% vs. 57.8%; p = 0.0156). Time to relapse was longer with ropinirole and more patients withdrew due to lack of efficacy with placebo. Patients showed improvements in IRLS and CGI-I scores, sleep and QoL parameters with single-blind ropinirole, which were better maintained when ropinirole was continued during the doubleblind phase, but reduced with placebo. Thirty-seven patients in the single-blind phase and 1 patient in the double-blind phase dropped out of the study due to adverse events.
A multicentre, 52-week, open-label continuation study conducted at 57 centres in 11 countries 69 involving 310 patients (women 186; mean age: 56.5 ± 11 years, range: 25-80) evaluated the longterm efficacy and safety of ropinirole in patients with idiopathic RLS. Eligible patients came from four previously published parent studies (i.e. pooled data) and were invited to participate.36,37,68,70 At parent study entry, all patients had a score of ≥15 on the IRLS. In this continuation study, all participants received ropinirole, 0.25-4.0 mg once daily, for 52 weeks. The primary study objective was to evaluate the safety of ropinirole. Efficacy was assessed by change in IRLS score, and by CGI scale and improvements in measures of sleep, work productivity and quality of life. Overall, 251 (81.0%) patients completed the study. The mean ropinirole dose at study end was 1.90 mg/day. A total of 282 patients (91.3%) reported adverse events that were mild or moderate in intensity for the majority. The most common adverse event was nausea. Adverse events led to discontinuation in 8.7% of patients. At week 52, IRLS scores improved by an average of 12.0 points from baseline, and 82.8% of patients were “much improved” or “very much improved” on the CGI-improvement scale. Ropinirole treatment was also associated with improvements in measures of sleep and quality of life.
Ropinirole provides relief of symptoms regardless of age at RLS symptom onset. Pooled data from four 12-week, randomized, double-blind, placebo-controlled studies of ropinirole in patients with moderate-to-severe primary RLS were analyzed post-hoc [ropinirole group: 496 patients (293 women),mean age: 53.6 ± 11.8 years, range: 18-79,age-at-onset: 35 ± 17; placebo group: 500 patients (315 women),mean age: 54.4 ± 12 years, range: 19-79, age-at-onset: 35.7 ± 17]. 71 The relation between age-at-onset and response to treatment, based on change from the baseline IRLSSG rating scale total score and the proportion of responders (rated much/very much improved) on the CGI-I scale, was explored. The range of age-at-onset of RLS symptoms was 2-75 years. No relationship was observed between the age-at-onset of RLS symptoms and baseline IRLS total score (correlation r: -0.06), or between dose administered at week 12 (last-observation-carried-forward, LOCF) and age-at-onset (r: -0.04). The age-at-onset by treatment interaction was non-significant (p = 0.952 for the IRLS and p = 0.716 for the CGI-I scale) indicating that there was no significant relation between age-at-onset and the magnitude of ropinirole treatment effect.
From the above reported data, ropinirole seems to be the most extensively studied drug for RLS in class I studies. For primary RLS, ropinirole significantly reduced the IRLS scale score and improved quality of life (Table 1). Ropinirole significantly improved polysomnographic indices of sleep disruption, i.e. PLMS-I, PLMS-A and sleep latency (Table 2). Thus, ropinirole has a level A rating of efficacy in primary RLS as demonstrated by the several evidence-based reviews of therapy in RLS, and by the published guidelines following an evidence-based analysis of the drug trials for RLS treatment.30,32,64
Clinical trials evaluating efficacy of Ropinirole in primary RLS.
PSG studies evaluating the efficacy of Ropinirole in primary RLS.
Data are much more scant on ropinirole treatment of secondary RLS, but some literature reports suggest that ropinirole is equally effective. An open, randomized, levodopa-controlled crossover study investigated the efficacy of ropinirole versus levodopa slow release (SR) in eleven uremic RLS patients on chronic hemodialysis (4 women; mean age: 56.2 ± 8.7 years; mean RLS duration: 3 ± 1.4 years; chronic hemodialysis mean duration: 6.4 ± 3.7 years). 72 The patients received either levodopa SR or ropinirole for 6 weeks, followed by a washout week, then the alternate treatment for 6 weeks. Patients rated the severity of RLS by means of IRLS scale, the CGI scale and sleep diaries. Under treatment with levodopa SR, 1 patient presented severe vomiting, leading to study discontinuation. The 10 patients who completed the study reported a 33.5% improvement (from 16.7 ± 3.2 to 11.1 ± 4; P < 0.001) of IRLS scale scores during levodopa SR treatment and a 73.5% improvement (from 16.6 ± 2.8 to 4.4 ± 3.8; P < 0.001) during ropinirole treatment. By the end of the study the mean levodopa SR dosage was 190 mg/d and the mean ropinirole dosage was 1.45 mg/d. Ropinirole was superior to levodopa SR in reducing IRLS scores (P < 0.001) and in increasing sleep time (P < 0.001). The patient CGI scale showed a significant difference favoring ropinirole (P < 0.01). There was no significant carryover or period effect for any outcome measure. Four patients reported a complete reversion of RLS symptoms during ropinirole treatment at doses ranging from 0.25-2 mg/d. Thus, all outcomes showed that ropinirole was more effective than levodopa SR in relieving symptoms in patients with uremic RLS on chronic hemodialysis.
Controlled studies are lacking and there is still insufficient evidence to make recommendations on the use of ropinirole in PLMD.30,32,64
Safety
The examined trials found that ropinirole treatment was generally well-tolerated, and its adverse event profile was typical of that of other dopaminergic agents. Mild and transient adverse events included nausea, headache, fatigue and dizziness and were those common to all dopaminergic agents (Table 3). Adverse events were not generally treatment-limiting and their overall frequency declined over time. Nausea was the most common adverse event but it was rarely severe and, in general, not treatment limiting. In addition to a manageable adverse event profile, long-term treatment with ropinirole raised no safety concerns as to changes in laboratory parameters or vital signs.
The most common adverse events (AE) during the Ropinirole trials in primary RLS.

Withdrawal form the studies refers only to lack of efficacy and/or AE: protocol deviation, lost to follow-up and other causes described in the original studies are not herewith reported.
The studies did not provide a definitive estimate of augmentation with ropinirole.
Particular safety aspects in the therapy of RLS are the so-called augmentation and rebound effects. Augmentation, the main complication of long-term dopaminergic treatment in RLS, was first described in 1996 73 in an uncontrolled study on 30 RLS patients treated with dopaminergic drugs (mainly L-DOPA). It was characterized by an earlier onset of symptoms in the afternoon (100% of patients), a shorter latency to onset of symptoms when at rest (56% of patients), an expansion of symptoms to the upper limbs and the trunk (11% of patients), an overall increase in the intensity of symptoms (96% of patients), and a shorter effect of the medication. Augmentation has been confirmed by subsequent reports,74,75 symptoms and signs of levodopa-related augmentation appearing related to plasma levodopa levels and closely mirroring the rapid rise and fall of plasma levodopa concentration, i.e. the levodopa plasma pharmacokinetic profile. 76 Another problem is the so-called rebound effect in which the symptoms of RLS reappear after the effect of the drug has worn off. This is similar to the ‘wearing off’ in Parkinson disease and manifests as early morning or late night symptoms. Rebound is related to the half-life of the drug, and occurs at the end of a dosing period when symptoms worsen. 77
Worsening of RLS with ropinirole possibly because of augmentation was observed in 4 out of 59 (7%) patients 35 and in 7 out of 309 (2.3%). 69 However, none of the previous ropinirole trials was designed to assess augmentation and currently we do not know the rate of augmentation in RLS patients taking ropinirole (Table 3).30,32,40
Pathological gambling (defined by the Diagnostic and Statistical Manual of Mental Disorders–-4th ed. as persistent and recurrent maladaptive gambling behavior characterized by at least 5 of the following actions: preoccupation with gambling; use of increasing amounts of money; inability to control, cut back, or stop gambling; irritability if not gambling; committing illegal acts to finance the behavior; lies to family or other persons to conceal the behavior; gambling to escape other problems; jeopardizing personal and professional relationships; or relying on others to relieve desperate financial situations caused by the behavior) 78 and other compulsive behaviors such as hypersexuality and punding (an intense fascination with complex, stereotypical, purposeless and repetitive behaviors like grooming, cleaning, assembly and disassembly of machines, and sorting and hoarding of objects) have also been described in patients on dopaminergic treatment for Parkinson's disease.79,80 Likewise, some patients with idiopathic RLS reported increased gambling and sexual desire after they began treatment with dopaminergic medications, ropinirole included.81,82 Moreover, punding and a variety of impulse control disorders have been reported anecdotally in RLS patients on ropinirole for RLS. 83 However, pathological gambling and punding have not yet been systematically assessed in controlled trials. Aberrant dopaminergic stimulation of the mesolimbic pathway from the ventral tegmental area to the nucleus accumbens may dysregulate normal reward pathways leading to these compulsive or addictive behaviors. Thus, patients with RLS should be cautioned about potential abnormal behaviors coinciding with the use of dopaminergic medications. However, further prospective studies and even functional imaging are needed to establish the relationship between dopaminergic medications and additive or compulsive behaviors associated with the RLS treatment.
Retroperitoneal and pleuropulmonary fibrosis and fibrotic cardiac valvulopathy with consequent restrictive valvular heart disease are known adverse reactions to ergot-derived dopamine-agonist treatment causing concern about the safety of these drugs.84–86 Though the most consistent data are derived from studies on Parkinson's disease, heart-valve abnormalities have also been reported in RLS patients taking ergot dopamine agonists. 87 The risk with non-ergot dopamine agonists, such as pramipexole or ropinirole, has been estimated to be low and similar to that of age-matched healthy controls. 85
Patient Preference
Ropinirole (marketed specifically for the RLS indication in some European countries) is available in preparations ranging from 0.25 mg 5 mg tablets. However, there are different regulations/recommendations worldwide, including formal licensing of ropinirole for treatment of RLS in Europe, USA, Australia, with each country carrying individual warnings for treatment. The US Food and Drug Administration has approved the first generic versions of “ropinirole hydrochloride” tablets for the treatment of moderate to severe RLS. Among the dopamine agonists, ropinirole is generally preferred because of a more favourable adverse-effect profile. 88 Moreover, the benefits of ropinirole in RLS treatment have been demonstrated through reduction in RLS symptoms,35–37,39,89 improvement in overall health status, 37 and improvement in quality of life. 68 Guidelines on RLS management assigned ropinirole a level A rating efficacy for relieving paraesthesia and motor restlessness.30,32,64
However, comparative studies investigating the efficacy of 2 drugs in RLS are rare and further investigations are needed. One (above reported) study in primary RLS compared gabapentin and ropinirole, finding them similarly effective and well-tolerated in the treatment of RLS. 33 Another study on uremic RLS patients undergoing hemodyalisis found ropinirole more effective than levodopa SR. 72 More randomized, double-blind crossover comparative studies with investigation of subjective ratings, polysomnography and follow-up are needed in RLS.
Place in Therapy
The mean effective daily dose of ropinirole reported in clinical trials was about 2 mg as a single dose at night; the maximum licensed dosage is 4 mg, although some studies used 6 mg. The superior efficacy of ropinirole versus placebo in the treatment of RLS was confirmed in a recent meta-analysis. 63 However, due to the possible interaction with other drugs and as many RLS patients who need treatment are over 65 years, dosage adjustment of ropinirole should be considered if hormone replacement therapy or ciprofloxacin treatment is started or discontinued during ropinirole therapy. In addition, smoker RLS patients should be advised that their smoking can significantly alter the efficacy of ropinirole.
Conversely, specific treatments cannot be indicated for PLMD 28 as the clinical significance of this sleep-related motor phenomenon is still debated, and many have failed to demonstrate an association between PLMD and symptoms of sleep and non-sleep disturbances.90–93 There are currently no established guidelines for treatment of the PLMD, and those that exist remain indecisive.30,32 Recommendations for PLMD therapy do include ropinirole, 94 but the evidence for this originates mostly from studies done on PLMS associated with RLS.4,31 Few trials have investigated the effects of dopaminergic drugs on the PLMD.93–95 Therefore, both the indications for treatment and the eventual drug choice for PLMD remain unclear. Moreover, clinical experience indicates that therapy of the PLMS and the associated sleep disorder may diverge: for instance, hypersomniacs with PLMS responded positively to psychostimulants but not to treatment of the PLMS with dopaminergic agents (personal communication). Interestingly, dopaminergic therapy for PLMS may even trigger RLS symptoms in the same patient, 96 this phenomenon probably representing the complication of the long-term dopaminergic treatment now called augmentation. 75 In patients presenting with symptoms of sleep disturbance such as excessive daytime sleepiness, insomnia or frequent awakening, the clinician is thus faced with deciding whether or not to treat those with a high PLMS index. The PLMS night-to-night variability further complicates the therapeutic decision. 97 In the final analysis, the decision whether or not to treat the PLMD must rest with the individual patient and his/her attending physician.
Conclusions
The results presented here provide evidence that ropinirole treatment significantly improves RLS symptoms. These benefits develop rapidly after starting treatment and are maintained during long-term therapy. As ropinirole is generally well-tolerated in patients with idiopathic and secondary RLS32,64,72 the drug is a valuable tool in the management of RLS.
Ropinirole is, however, currently approved for administration in a single dose and licensed only for treating moderate to severe RLS. Patients who needed treatment in the late afternoon or evening were excluded from many trials by definition. It is, thus, necessary to explore intermittent therapy given when needed in mild RLS forms or slow-release formulations for the treatment of RLS patients with late afternoon and evening symptoms. These two therapeutic approaches should also be addressed to reduce the long-term complications of dopaminergic treatment, namely augmentation. It is also clear that a substantial research effort is still needed to evaluate the pathophysiology, clinical presentation, treatment modalities and overall long-term outcome of RLS, in particular for patients with secondary forms of RLS (including RLS in pregnancy) and for children with RLS for whom no controlled clinical trials are yet available.98,99
Generally speaking, the only harmful effect of IRLS in clinical practice is that it interferes with sleep, but sometimes the sudden uncontrolled leg movements can be painful and cause pulled calf muscles, leaving the patient exhausted in the morning. Causes include pregnancy, lack of iron, vitamin B12 or folic acid, diabetes, Parkinson's disease, low thyroid function, several drugs including lithium, beta blockers, antidepressants, antihistamines, drinking coffee and alcohol and smoking, all conditions that may decrease the efficacy of ropinirole. Patients with RLS should avoid coffee, alcohol and smoking, while any vitamin B12 or iron deficiency should be corrected.
Disclosures
The authors report no conflicts of interest.
