Abstract
Restless Legs Syndrome (RLS) is one of the most common neurological diseases characterized by an urge to move the legs, often associated with unpleasant sensations relieved by movement. It is engendered by rest, and is worse in the evening or at night. Patients affected by severe RLS should be treated pharmacologically. Dopamine-agonists represent the first-line treatment for RLS symptoms. Pramipexole is a non-ergot derived dopamine agonist with a high selectivity for D2 and D3 receptors. At doses comprised between 0.125 and 0.75 mg, pramipexole improves subjective symptoms and objective signs of primary RLS even after the first administration. In addition, pramipexole seems to be safe and well tolerated. However, physicians should be aware that augmentation and compulsive behaviours might occur in their RLS patients treated with pramipexole. Further studies are needed to confirm the efficacy of pramipexole in uremic RLS and in children affected by the sleep disorder.
Introduction
Restless Legs Syndrome (RLS) is included among the sleep-related movement disorders in the last version of the International Classification of Sleep Disorders. 1
RLS is characterized by an urge to move the legs often associated with unpleasant sensations, engendered by rest, is relieved by movement and is worse in the evening or at night. Diagnosis of RLS is clinical and based on the above reported clinical characteristics of the sleep disturbance.2,3 In order to increase the sensibility and specificity of RLS diagnosis, physicians should perform a careful patient history followed by a meticulous physical and neurological examination.
Many patients with RLS (almost 80%–90%) present periodic, involuntary and stereotyped jerks in the lower limbs known as Periodic Legs Movements (PLM). 4 PLM may occur both while awake (PLMW) or during sleep (PLMS). The presence of PLM represents a supportive feature for RLS diagnosis. Unfortunately, the presence of PLM has a low specificity; in fact, these movements may occur also in other sleep complaints (e.g. sleep-disordered breathing, narcolepsy, REM behavior disorder) and in elderly subjects. 1
RLS can be distinguished in primary and secondary forms. Although not mandatory, an important characteristic of primary RLS is a positive family history, 5 thus a genetic basis for this form has been hypothesized. 6 RLS has been associated with iron deficiency, uremia, pregnancy and use of neuroleptics. 7 In addition, RLS may occur as a side effect related to the administration of antidepressants. For example, mirtazapine, a noradrenergic and specific serotonergic antidepressant, is able to provoke or impair RLS symptoms in almost one-third of patients treated with this compound. A significantly lower risk is associated with reboxetine, a selective noradrenergic reuptake inhibitor. 8 Recent case-control studies reported novel associations that should be confirmed by further surveys.9–14
Prevalence of RLS in Caucasian and North American general population is 5%–10%. 15 The prevalence of RLS increases with age and is higher in women than in men.16–18 Almost 3% of the general population is affected by a severe form of the sleep disorder that might lead to relevant consequences, 19 such as psychological distress, 20 cognitive dysfunction, 21 impaired quality of life, 22 and cardiovascular diseases. 23
Patients affected by mild and intermittent RLS may undergo non-pharmacologic treatment. This approach consists of behavioral therapy, sleep hygiene and lifestyle interventions (avoiding caffeine and alcohol intake, heavy meals and drugs able to induce RLS). Otherwise, subjects reporting frequent and severe RLS symptoms should be treated pharmacologically. 24
Dopaminergic agents represent the first-line therapy for RLS. Since chronic use of levodopa is limited by the occurrence of augmentation (up to 60% of patients treated with this drug) 25 and rebound (up to 46% of patients treated with a single bedtime dose of regular-release levodopa), 26 this compound is recommended as on demand treatment for RLS patients reporting intermittent symptoms.
An international consensus conference yielded the Max Plank Institute criteria for diagnosing RLS augmentation. 27 Augmentation is characterized by an earlier onset of symptoms during the day, a shorter latency to onset of symptoms when at rest, an expansion of symptoms to the upper limbs and the trunk, an overall increase in the intensity of symptoms and a shorter-lived benefit of the medications. Besides, a validated instrument able to quantify the severity of augmentation (the Augmentation Severity Rating Scale) has been developed. 28 The exact pathophysiological mechanism causing augmentation is unknown, 29 but it seems that low ferritin levels have a main role in increasing its occurrence. 30
Since regular release levodopa shows a short half-life of 1 to 3 hours, patients treated with a single bedtime dose of this compound may report recurrence of RLS symptoms and PLMS during the second half of the night. This clinical condition, known as rebound, can be treated using a combination of regular release levodopa and sustained-release levodopa. 26 In contrast to rebound, augmentation is most prominent in the afternoon or evening, although in very severe cases it may be present at all hours.
Differently from levodopa, dopamine-agonists represent a useful treatment for daily RLS. 24 Since pergolide and cabergoline, two ergot-derived dopamineagonists, may cause valvular heart disease, non-ergot derived dopamine-agonists are usually used in clinical practice. 24 EMEA and the FDA approved ropinirole and pramipexole for the treatment of primary RLS.
Although direct comparative studies between ropinirole and pramipexole are lacking, a recent meta-analysis showed a greater decrease in the mean International RLS Study Group Rating Scale (IRLS) score and a higher Clinical Global Impression-Improvement (CGI-I) response rate for pramipexole compared to ropinirole. In addition, pramipexole had a significantly lower incidence of nausea, vomiting, and dizziness than ropinirole. 31
Pramipexole
Pramipexole has a selective action on the dopamine D2 and D3 receptors. When administered orally, pramipexole is readily absorbed from the gastrointestinal tract. Oral bioavailability is reported to be about 90%. Plasma protein-binding is <20%. Metabolism of pramipexole is minimal; in fact >90% of a dose of the dopamine-agonist is excreted unchanged in urine via renal tubular secretion. Pramipexole shows a half-life of 8–12 hours. Pramipexole half-life is prolonged to 36 hours in patients with a creatinine clearance of 30–50 ml/min.
In 1999 Montplaisir et al performed the first placebo-controlled study on the efficacy and safety of pramipexole in the treatment of idiopathic RLS. Ten patients were studied before and after two 1-month treatments (placebo and pramipexole) administered in a double-blind crossover fashion. The authors observed a good efficacy of pramipexole, both on leg discomfort (nine of the 10 patients reported a complete disappearance of RLS symptoms) and on PLMS (index reduced to normal values). 32
The efficacy of pramipexole on primary RLS was further confirmed by six large randomized, double blind, placebo-controlled studies (see Table 1).33–38 In a polysomnographic trial (the PRELUDE study), 109 patients were treated with either different fixed doses of pramipexole (0.125 mg, 0.25 mg, 0.5 mg and 0.75 mg) or placebo. All-active doses of pramipexole were able to significantly improve subjective symptoms (IRLS score) and objective signs (PLM index) of RLS. After a 3 weeks’ treatment (pramipexole 0.25, 0.5 and 0.75 mg), the percentage of patients assessed as either ‘much improved’ or ‘very much improved’, according to the CGI-I scale, was significantly higher than that of the placebo group. Besides, treatment with pramipexole at 0.125, 0.5 and 0.75 mg was able to show a significant reduction of sleep latency. Based on these results, the authors suggested to start RLS treatment using the lowest dose of pramipexole (0.125 mg/daily). 33 Recently, a dose-raging polysomnographic study on 107 patients affected by primary RLS confirmed the efficacy of pramipexole at the dose of 0.125 mg in several sleep-related facets of the sleep disorder. 37 The positive effects of pramipexole on RLS symptoms at a dosage between 0.125 and 0.75 mg/daily was confirmed by the other placebo-controlled trials (see Table 1).34–36,38
Double-blind trials on pramipexole performed on large populations

Long-term efficacy of pramipexole was investigated by Montplaisir et al in 195 consecutive RLS patients treated with the dopamine-agonist for more than 1 year. Of the forty-four patients that discontinued pramipexole, only 12 because of a lack of efficacy. The remaining 152 patients took a mean pramipexole dose of 0.59 ± 0.31 mg (range 0.125–2.25 mg). Patients who continued pramipexole for more than 1 year reported a mean decrease in RLS symptom severity of 80.0% ± 20.8% at follow-up compared with baseline. In addition, difficulties in falling asleep and nocturnal awakenings due to RLS were significantly reduced after pramipexole treatment. 39 A recent 26-week, open-label trial (extension of the Prelude study) demonstrated that pramipexole administered at dosages of 0.125–0.75 mg/daily was able to improve RLS symptoms (IRLS and CGI-I), quality of life (SF-36) and sleep (sleep diary). 40
Two studies investigated the acute effect of pramipexole on RLS symptoms. In an open-label trial our group demonstrated that pramipexole 0.125 mg improved sensory symptoms (IRLS) and motor signs (PLM indexes) of idiopathic RLS already after the first administration. The sleep macro- and micro-structure improved as well. 41 These results were confirmed by Manconi et al in a single-blind study that evaluated 32 consecutive primary RLS patients treated with a single-dose of pramipexole 0.25 mg (18 patients) or placebo (14 patients). 42 The acute effect of pramipexole on RLS might be useful not only for improving treatment compliance but also as ex-juvantibus test for differential diagnosis between RLS and RLS-mimics.
Only one study evaluated the effects of pramipexole on patients affected by uremic RLS. This small, uncontrolled trial showed that pramipexole at dosages of 0.125–0.75 mg/daily was able to improve IRLS score and PLM index. The daily dose of pramipexole should not exceed 0.75 mg/daily in uremic patients. 43
Pramipexole is not approved by EMEA and FDA for the treatment of RLS in children. However, Guilleminault et al reported that this drug was able to control RLS symptoms in two children affected by confusional arousals. 44 Although there is limited experience on the use of dopaminergic agents in children, levodopa and dopamine agonists result in long-term improvements in children with RLS and PLMD. 45 This is particularly true for children affected by Attention-Deficit Hyperactivity Disorder (ADHD) and RLS in which dopaminergics may also resolve ADHD symptoms. 46
Experimental studies show that pramipexole can affect embryo-fetal development at maternally toxic doses in rats. Since clinical experience regarding the safety of pramipexole and other dopaminergic agents during pregnancy in humans is limited, these drugs are included in the Pregnancy Category C by the FDA. The pregnant woman affected by RLS should be reassured that the sleep disorder generally resolves quickly postpartum. Iron and folic acid supplements might be useful to improve RLS symptoms during pregnancy.
Pramipexole has a favorable safety profile. Nausea, vomiting, headache, nasopharyngitis, fatigue, insomnia and somnolence were the most common Adverse Events (AE) reported in the six large placebo-controlled trials (see Table 2). AE were transient and mild or moderate in severity; in fact, drug discontinuation due to AE was uncommon (see Table 2).33–38
Prevalence of Adverse Events (AE) due to pramipexole and placebo in the double-blind trials performed on large populations.
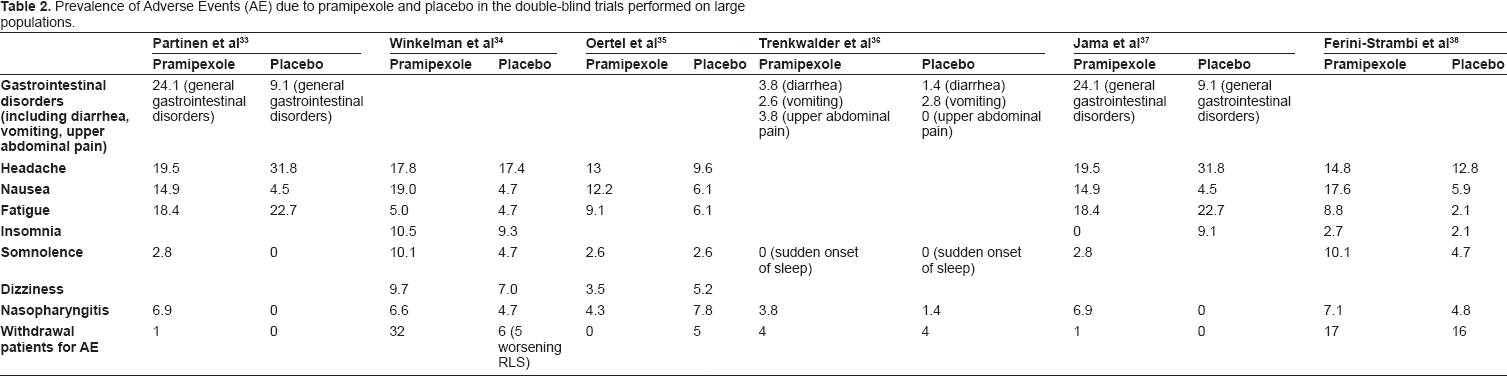
Although cases of augmentation were not reported, aggravation of RLS during pramipexole treatment was observed in 3.7% and 4.6% of patients in two placebo-controlled trials.33,37 Three studies focused their interest on the prevalence of augmentation during pramipexole treatment.39,47,48 Prevalence of this AE ranged between 22.4% and 32%.39,47 This rate is significantly lower than that reported during levodopa treatment (see above).
Differently from levodopa (see above), rebound appears to be infrequent during treatment with pramipexole, indeed the studies of Montplaisir et al and Winkelman and Johnston reported 13.8% and no patients, respectively, affected by a worsening of RLS symptoms during the second half of the night.39,47
It is known that dopaminergic agents may cause Sudden Onset of Sleep (SOS) in subjects affected by Parkinson's Disease (PD). 49 Differently, dopaminergics, in particular pramipexole, do not seem to be associated with SOS in RLS patients.33–38 A recent polysomnographic study confirms this suggestion. 50
Dopamine-agonists may cause compulsive behaviors (gambling, hypersexuality, excessive shopping, compulsive eating and internet/computer addiction) in PD patients. 51 Several case-reports observed similar clinical conditions also in RLS patients.52–54
A recent epidemiological study confirmed that compulsive behaviors might occur in RLS patients, even if with a lower prevalence than in subjects with PD. An increased impulsivity correlated with younger age and larger doses of dopamine-agonists could explain, at least in part, the greater side effects in parkinsonian patients. Besides pramipexole tended to have higher impulsivity associations than ropinirole. 55 An excessive stimulation of D3 dopamine receptor in the nucleus accumbens might cause compulsive behaviors during treatment with dopamine-agonists.
Conclusions
Dopamine-agonists represent the first-line treatment for patients with RLS. In particular, pramipexole demonstrated to be effective in treating subjects affected by moderate-to-severe primary RLS. Treatment should be started using the lowest dosage (0.125 mg) taken 2–3 hours before bedtime. Pramipexole should be able to improve RLS symptomatology already after the first administration. If pramipexole 0.125 mg/daily is not effective in controlling symptoms and signs of the sleep disorder, the dosage of the dopamine agonist may be increased. Most patients respond to a pramipexole dose ranging between 0.125 and 0.75 mg/daily. Pramipexole presents a favorable safety profile characterized by transient and mild or moderate AE. Physicians should pay attention to the occurrence of augmentation and compulsive behaviors during pramipexole treatment in their RLS patients. There is a strong relationship between the dosage of dopaminergic agents and the likelihood of these AE, so patients affected by severe forms of the sleep disorder might be prone to them. Therefore, in order to avoid augmentation and compulsive behaviors, the lowest possible doses should be used and, in any case, without passing the maximum regulatory dose (maximum approved dose of pramipexole for RLS treatment is 0.5 mg in the United States and 0.75 mg in Europe). Higher dosages can be administered in patients affected by severe or very severe RLS, but physicians must be aware that this is “off label” and under their personal responsibility. Further studies are needed to confirm the preliminary data reporting pramipexole as effective in treating uremic RLS and children affected by the sleep-related movement disorder.
Disclosures
This study was conducted independently from any commercial entity and it did not receive any funding. The authors have no other financial disclosures to make and they report no conflict of interest.
