Abstract
Most patients with hereditary or chronic acquired anemias are dependent on regular red cell transfusions. Untreated iron overload from transfusions is responsible for morbidity and mortality in patients with thalassemia major. However, clinical consequences of parenchymal iron overload have been reported not only in thalassemia major but also in patients with myelodysplastic syndrome. The current standard in iron chelation therapy is deferoxamine mesylate (Desferal®). Deferasirox is the first oral iron chelator approved in the Europe Union for use in patients with transfusional iron overload with different diseases. The aim of this review is to examine the properties and management of Deferasirox.
Introduction to Iron Overload
Many patients with hereditary or chronic acquired anemias are dependent on regular red cell transfusions. Examples of such hereditary anemias include β-thalassemia major, sickle cell disease, Diamond Blackfan and Fanconi's anemia;1,2 of acquired anemias myelodysplastic syndromes (MDS), aplastic anemia and primary myelofibrosis.
Iron loss by the human body is minimal; we are the only species that has difficulties in maintaining iron balance and, as a consequence, repeated blood transfusions lead to iron overload.1,3
Untreated iron overload from transfusion is responsible for morbidity and mortality in patients with thalassemia major.4,5 Clinical consequences have also been reported in acquired chronic anemias requiring regular transfusions. 6
Each blood unit contains 200-250 mg of iron. 7 If left untreated, iron accumulates in the liver, heart, anterior pituitary, pancreas and joints leading to organ dysfunction and early death. 1 Iron overload became detectable in the liver after approximately 20 Packed Red Blood Cell (PRBC) 8 transfusions leading to ferritin levels of about 1,000 mg/L.
In patients with β-thalassemia major, transfusional chronic iron overload complications, most frequently associated with mortality, are those related to myocardial dysfunction. Other common complications include hepatic fibrosis, 9 liver cirrhosis, diabetes mellitus, osteoporosis and impaired growth and development in children.2,3,10
During recent years methodologies to assess iron overload have progressed with new technologies. Today with direct (hepatic iron concentration on biopsy species) or indirect measurements (serum ferritin, transferrin saturation and magnetic imagine resonance-MRI) adequate and quantitative evaluation of body iron can be obtained. Particularly MRI T2* and MRI R2, 11 are validated as non-invasive methods to assess cardiac and hepatic iron, respectively. The Italian society of Hematology has recently extensively reviewed methodology to assess iron overload.12,13
The majority of clinical research regarding iron overload has been conducted in patients with β- thalassemia major and limited data is available for other patient populations. 10
However, clinical consequences of parenchymal iron overload have been reported not only in thalassemia major but also in patients with myelodysplastic syndrome. For the management of MDS patients blood transfusions represent the main component of supportive care. Ninety percent of all MDS patients with permanent anemia become dependent on blood transfusions to maintain their quality of life and to survive. Therefore they are likely to develop iron overload, proportionally to the duration and intensity of transfusion dependency. 13
Introduction to Deferasirox
The current standard in iron chelation therapy is deferoxamine mesylate (Desferal®). 12 Long-term data collected since the drug was introduced in the early 1970s has demonstrated its safety and high efficacy in reducing stored iron and related complications, thus resulting in decreased mortality in iron-overloaded thalassemia subjects. 14
Unfortunately, deferoxamine has a short plasma half-life and an insufficient oral bioavailability. These features require an intravenous or subcutaneous application of the drug over a period of 8 to 12 hours, usually five to seven times a week. Alternatively, deferoxamine can be administered as a subcutaneous bolus injection with a markedly reduced efficacy. 15 As a consequence of the cumbersome administration, the compliance of the deferoxamine therapy is often poor, resulting in limited efficacy. In MDS patients the association with severe thrombocytopenia and neutropenia further limits the applicability of deferoxamine.
The first oral iron chelator Deferiprone (Ferriprox®) was introduced 20 years ago. The use of the drug remained limited to second-line therapy for patients with thalassemia major. The risk of neutropenia and agranulocytosis, with a frequency of about 0.5%-1% in thalassemia were the major limiting factors.16–18 Recently a specific chelator effect on myocardial iron has been reported from retrospective analysis with a relevant decrease of cardiac deaths in thalassemia. 19 Sporadic effects has been reported in MDS. Deferiprone is currently not licensed to MDS patients.
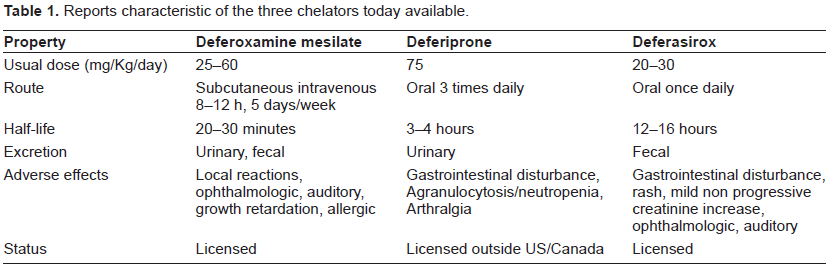
Deferasirox (Exjade®), an orally administered once daily iron chelator was first approved in the United States in 2005 and in the European Union in 2006. (Table 1 reports characteristics of the three chelators available today). The aim of this review is examine the proprieties and management relating to the use of Deferasirox.
Reports characteristic of the three chelators today available.
Pharmacodynamic Properties
Deferasirox is a tridentate iron chelator (Fig. 1), with a higher affinity for iron as Fe3+ than Fe2+ and minimal affinity for other divalent metal ions such as zinc or copper. 20
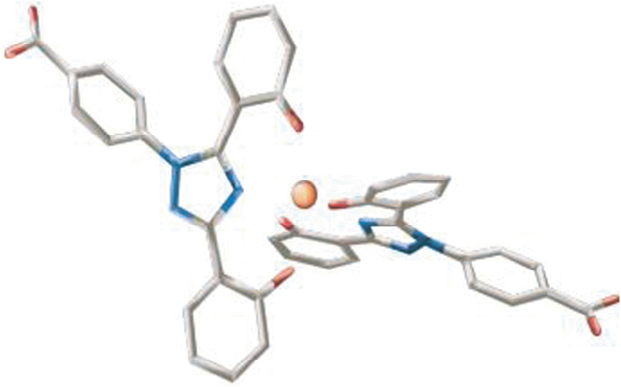
Deferasirox is an orally active tridentate iron chelator with 3 polar interaction sites in the binding pocket.
The active molecule is a highly lipofilic, 99% protein-bound, N-substituted bis-hydroxypheniltriazole.21,22 Two molecules are required to form a stable complex with each iron atom (Fe3+) because three polar interactions sites in the binding pocket results in Deferasirox binding with iron in a 2:1 ratio.
Orally administered Deferasirox, dispersed in water, orange or apple juice, has an absolute bioavailability of 70%. Presence of food increases the Deferasirox bioavailability to a variable extent. To ensure appropriate systemic exposure Deferasirox should be taken 30 minutes before food. Serum concentration is proportional to the dose administered. 23
Pharmacokinetic data recommend Deferasirox dosage of 20 mg/kg/day. Deferasirox reaches maximum plasma concentration (C max) at a median time of 1-2 hours after repeated administration of 10 or 20 mg/Kg/day. Median time to C max was independent of dose. 24
The main pathway of Deferasirox metabolism is hepatic predominantly by glucoronidation with biliary excretion. Deferasirox is mainly excreted in the feces (83%), predominantly as the unchanged drug.25,26 A small proportion (6%-8%) of unchanged Deferasirox and metabolites is excreted in urine25,27,28 with phenol glucuronide as main metabolite. 26
As the iron–Deferasirox complex has a mean elimination half-life of 17.2 hours after a single dose of Deferasirox 20 mg/kg plasma levels are maintained within the therapeutic range over a 24 hours, therefore providing 24 hours chelation coverage and protection from toxic labile iron with a once daily administration.
The pharmacodynamic properties of Deferasirox in patients with transfusional iron overload has been evaluated using three key parameters (1: iron balance 2: myocardial iron levels, 3: labile plasma iron).
Iron balance (iron excretion: iron intake)
The effect of oral deferasirox on iron balance in patients with β-thalassemia and transfusional chronic iron overload has been examined in phase II eIII trials. These results indicate that deferasirox dosages <20 mg/kg/day are unlikely to maintain net iron balance or achieve net iron excretion 29 in patients receiving >2 packed red blood cells units/month. At a deferasirox dose of 20 mg/kg per day, neutral iron balance was achieved in 46% of patients with highest transfusional iron intake and negative iron balance in 75% of patients with lowest transfusional iron intake; 30 mg/kg per day produced successful control of iron stores in 96% of patients with a low rate of transfusional iron intake. 30
This data has been obtained using serum ferritin and hepatic iron concentration as quantitative markers of iron overload (see efficacy studies discussed below).
Myocardial iron levels
Myocardial iron levels are estimated by calculating the T2* phase using MRI (T2* > 20 msec is considered normal, 8-20 msec indicates moderate iron overload, <8 indicates severe myocardial iron overload).31,32 In vitro 33 and in vivo 34 preclinical data indicate that Deferasirox has the capacity to enter and chelate iron from myocardial cells.
One study in a gerbil model of iron overload found that daily separated doses of Deferasirox reduced cardiac iron more effectively than once-daily dosing, although the difference between dosing regimens was not statistically significant. Variation in the pharmacokinetics of Deferasirox between humans and gerbils could explain the differences between the dosing regimens in human iron overload. 35 In a study of 29 patients who received Deferasirox in two pivotal studies as described earlier36,37 the T2* magnetic resonance imaging showed a significant reduction in cardiac iron burden after 12 months of Deferasirox treatment, which was maintained after 2 years. 38
Preliminary clinical data reported that myocardial iron levels were significantly reduced after Deferasirox treatment in patients with transfusional chronic iron overload. 38 T2* mean improvement of 18.0 ms to 23.1 ms. 39 Data from a prospective, single-arm, multicenter trial in patients with β-thalassemia also show that after 6 months of Deferasirox treatment patients experienced significant improvements in cardiac T2*, liver iron concentration (LIC) and labile plasma iron. 40 These results show that in 93% of the patients, Deferasirox achieved a negative cardiac and liver iron balance.
Labile plasma iron (LPI)
LPI and non transferrin bound iron (NTBI) are the effectors of iron related tissue damage by OH– formation, lipid degradation and intracellular damage. 41
Labile plasma iron can be transported intracellularly and lead to excessive storage of iron 42 so it can be used as an indicator of iron overload and its changes can be used to assess effectiveness of chelation therapy. In patients with β-thalassemia and transfusional chronic iron overload who were treated with Deferasirox 20-30 mg/kg/day LPI was significantly reduced. 43 Of relevance Deferasirox pharmacokinetic provides efficient 24-hours/day protections from LPI and NTBI. However so far LPI and NTBI are experimental measurements to be validated and diffused.
Efficacy Studies
The efficacy of oral Deferasirox in patients with transfusional chronic iron overload was evaluated in three randomized, open-label, multicenter trials comparing oral Deferasirox with subcutaneous Deferoxamine36,44,45 and two non comparative multicenter trials.29,46 The major of these trials enrolled pediatric and young adults patients with β-thalassemia major.29,36,44 The primary efficacy endpoint was the success of deferasirox in reducing or maintaining liver iron concentration which has been demonstrated to be proportional to body iron store,36,47 and serum ferritin level.29,44,45 Secondary endpoints included safety and tolerability. Cappellini et al in their phase III study evaluated the efficacy of Deferasirox in regularly transfused patients with β-thalassemia. They confirmed that in β-thalassemia patients, receiving 2 to 4 PRBCs per month, oral Deferasirox at a once-daily dose of 20 mg/Kg maintained or decreased liver iron concentration, to neutral iron balance and serum ferritin levels. Changes in LIC were dependent on the Deferasirox dose and amount of transfusional iron intake. In patients with baseline LIC > 7 mg iron/g dry weight (dw), Deferasirox initiated at a dose of 20 or 30 mg/kg/day produced statistically significant decreases in LIC. No reduction was seen in those with a baseline LIC < 7 mg Fe/g dw who received Deferasirox 5-10 mg/Kg/day. Doses 5-10 mg/Kg were too low to balance iron overload through transfusion in those patients. Similar observation have been found in other prospective non comparative trials evaluating the efficacy of Deferasirox in regularly transfused patients with myelodysplastic syndromes, Diamond Blackfan anemia, β-thalassemia major and other rare anemias. 37 A dose of Deferasirox 10 mg/Kg was able to maintain neutral iron balance in patients receiving not more than 2 PRBCs transfusions per month. 36
Extension and on Going Trials
In the subsequent ongoing 4-year extension trials, dose adjustments were permitted to enable patients to achieve their treatment goal. To date, patients have been receiving treatment for a median period of 3.4 years (range 0-4.5). Mean dose in the 5-10 mg/kg/day group increased in the extension trials reaching around 20-25 mg/kg/day after approximately 2 years of treatment. 48 Mean dose in the 20 mg/kg/day group has remained at around 20-25 mg/kg/day during the entire treatment period, while those in the 30 mg/day group decreased to around 25 mg/kg after approximately 18 months. Median serum ferritin levels in the lower dosage group (5-10 mg/Kg/day) steadily increased during the first 18 months of Deferasirox treatment. However, subsequent dose increases during the extension trials generally resulted in serum ferritin level decreasing to below baseline at around 32 months and for the remainder of the study. Deferasirox treatment in this subgroup was generally well tolerated (including doses >30 mg/Kg/day) with a low discontinuation rate. In the 20-mg/kg/day group, median serum ferritin levels were maintained throughout the treatment period and following, serum ferritin levels decreased overall from baseline to month 42. Overall, this data has shown that patients can achieve a therapeutic goal of maintenance or reduction in serum ferritin levels with appropriate dose adjustments.
A large 1-year, multicenter, open-label study aiming to enroll 1541 patients with various transfusion-related anemias is currently ongoing to evaluate the efficacy and safety of Deferasirox. 49 The study is evaluating the efficacy and safety of Deferasirox when initial dose is based on transfusion history with subsequent dose titration based on efficacy and safety markers. Baseline data from a subgroup of MDS patients enrolled in this study indicated significant iron burden above thresholds associated with increased morbidity and mortality. For the previously chelated patients with Deferoxamine, this indicates that their treatment regimen was not providing adequate management of their iron burden, while for the chelation-naïve patients this data clearly indicated a need for chelation therapy.
A GIMEMA (Gruppo Italiano Malattie Ematologiche dell'adulto) open label multicenter study for valuation safety, tolerability and efficacy of deferasirox in patients with myelodysplastic syndrome and transfusionally chronic iron overload, is ongoing in Italy. (www.clinicaltrials.gov NCT 00469560).
Safety and Tolerability
Evaluation of the safety and tolerability of Deferasirox has been a key objective of all pivotal clinical trials. Adverse events and serious adverse events have been carefully monitored throughout the programme and continue to be assessed in the extension phases. The clinical programme has shown that Deferasirox has a well-defined safety profile that is clinically manageable with regular monitoring both in adult and pediatric patients.36,45
Of the more than 1000 patients enrolled in core clinical studies, only 74 have discontinued treatment due to adverse events.29,44,45 The most frequent adverse events reported over a median 3.5 years of treatment included transient, mild-to-moderate gastrointestinal disturbances (nausea, vomiting, abdominal pain, constipation and diarrhea) and skin rash. Sporadic severe gastrointestinal disturbances were described (gastrointestinal bleeding and ulcers).
Most adverse events experienced by patients during treatment with Deferasirox resolved spontaneously; adverse events requiring dosage adjustments or interruptions included rash, gastrointestinal disorders, infections and elevated serum creatinine and transaminase levels. Mild, non-progressive increases in serum creatinine was observed in approximately one-third of patients in the pivotal clinical trials of Deferasirox. Creatinine levels returned spontaneously to baseline in more than two-thirds of patients who experienced these mild increases. There were no cases of moderate-to-severe renal insufficiency or renal failure, and no patients permanently discontinued therapy due to creatinine rises. Data from up to 3.5 years of treatment in 1034 patients 50 confirmed that these increases are non progressive. The majority of this data emerged from studies regarding β-thalassemic patients. Information about safety and tolerability of Deferasirox in patients affected by other anemias is scarce and further study in this area is needed. In fact this cohort of patients differs from the thalassemics for age and comorbidities. In particular in the Porter et al study, only 29 of 47 myelodysplastic patients completed the trial. The ongoing prospective open label GIMEMA study, will better evaluate the profile of safety and tolerability in this population.
Table 2 reports Deferasirox most common (>4%) adverse events registered during 3.5 years of treatment.
Most common (>4%) drug-related adverse events during 3.5 years of Deferasirox treatment.
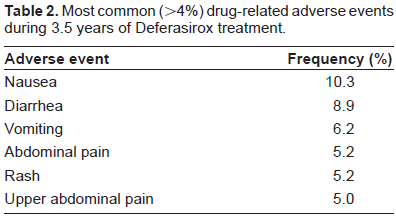
Table 3 reports Deferasirox-associated gastrointestinal adverse events and proposed management approaches.
Deferasirox-associated gastroenteric adverse events and proposed management approaches.

Post Marketing Surveillance
Post marketing experience in a large number of patients has provided additional data that further support the safety profile of Deferasirox in adults and children with a range of underlying anemias. Cases of acute renal failure have been reported following the postmarketing use of Deferasirox. 51
Other factors to explain renal failure were apparent in most of these cases, such as preexisting renal conditions, advanced age, comorbidity conditions, or concomitant medication that may depress renal function. 52 Neutropenia and thrombocytopenia have also been reported. However, most of these patients had preexisting hematologic disorders that are frequently associated with bone marrow failure and the relationship of these episodes to treatment with Deferasirox is uncertain and did not lead to label modification. 52
Product information for Deferasirox includes monitoring serum creatinine levels in patients who have preexisting renal conditions, are elderly, have comorbid conditions that may affect renal function, or are receiving medicinal products that depress renal function. Blood counts and liver function should also be monitored regularly. Most patients receiving Deferasirox had normal levels of serum alanine transaminase, although increased levels were reported and sporadic acute hepatic failure, sometimes fatal, occur especially in patient with a previous cirrhosis.36,44
Deferasirox Indication, Dosage, Monitoring
The Italian Society of Hematology guidelines for the management of iron overload in thalassemia major and related disorders recommended Deferasirox (in patients without severe iron overload) as the alternative therapy to Deferoxamina on the basis of its better safety profile compared with Deferiprone, which should be considered in the case of resistance or intolerance to Deferasirox. 12 Preliminary results from ongoing trials will probably lead to a larger and earlier indication. Recommendation for deferasirox dosage and patients monitoring in the United States or the European Union indicate a Deferasirox initial dosage of 20 mg/Kg/day. Deferasirox dose adjustment should depend on patient response, serum ferritin trends, goal of therapy and serum creatinin levels (Deferasirox doses adjustment is recommended to be performed in steps of 5-10 mg/kg/day every 3-6 month).25,27 The drug is contraindicated in patients with creatinin clearance <60 ml/min and it isn't recommended for patients with severe hepatic impairment as it has not been investigated in those patient populations.
Table 4 reported recommendation for patients monitoring.
Recommendations for patients monitoring.

Myelodysplastic Syndrome and Iron Overload
Patients with MDS are usually adult–-elderly patient and have a range of age related comorbidities. They are a completely different clinical scenario in contrast to patients with thalassemia. 13 Only recently MDS-iron specific data is beginning to appear.
Low or intermediate I IPSS risk MDS patients with transfusion dependency can survive enough to develop iron related tissue damages. The new WHO classification for MDS evaluated disease prognosis with respect to risk factor in addition to age and gender and reported that transfusion dependency was associated with a short overall survival and leukemia free survival. 53
In a Malcovati study, patients were grouped according to transfusional burden i.e. <20, 20-40 or >40 RBC units received during the clinical course. The effect of transfusion burden was calculated as the number of transfusions per month, and a significant effect was found both on overall survival and leukemia free survival. Cardiac failure (51%), infection (31%), hemorrhage (8%) and hepatic cirrhosis (8%) were responsible for the majority of deaths. 54 Among low risk MDS patients who died as a result of a non-leukemic cause, cardiac failure was significantly more frequent in transfused patients.
The prognostic value of iron overload was also evaluated using serum ferritin as a marker of iron overload. A ferritin level of 1000 ng/ml was chosen as a threshold that distinguished between mild and clinically relevant iron burden. A ferritin level of 1000 ng/ml was reached after a median number of 21 RBC units. The development of secondary iron overload significantly affected overall survival (P < 0.001). After adjusting for transfusion burden there was a 30% increase in the hazard rate for every 500 ng/ml of increase in serum ferritin above the threshold.54,55 Conflicting results about the iron overload negative effect on survival are coming from retrospective studies. The majority of the studies suggest probable negative effects in low-risk, Intermediate-I MDS patients. This is usually attributed to longer survival and subsequent increased transfusion burden in these categories of patients.56,57
Cardiac iron accumulation has been demonstrated to develop in unchelated MDS patients, who received a very large (approximately 80 PBRCs units)8,58 amount of transfusional iron intake, but this has not been shown to be constantly associated with an impairment of cardiac function. Cardiac iron overload can cause dilated cardiomyopathy, manifesting as systolic or diastolic dysfunction. One study in 46 patients with MDS showed that >40% of patients have signs and symptoms of heart failure, which in some patients was accompanied by cardiac arrhythmias. 53
In MDS, the relationship between cardiac iron loading and cardiac function is more complex than thalassemias. This is dependent on the coexistence of several risk factors for cardiac disease. Hemoglobin level fluctuation, preloading and after loading transfusion related modification, phenomenon aging related, like hypertension–diabetes–arterioscleroses, have all independent effects on cardiac function and may all contribute to cardiac-related death. Large studies and multivariate analysis is required to clarify these effects, but it is reasonable to predict that cardiac iron could be an important co-factor for cardiac morbidity and/or mortality.
Iron overload and an elevated serum ferritin have been reported to be an important adverse prognostic factors for patients with congenital 59 or acquired anemia undergoing allogeneic stem cell transplantation. 60
Pretransplantation elevated serum ferritin level has been associated with lower overall and disease-free survival in patients with malignancies. The lower survival rate was attributable to a significant increase in transplant-related mortality.
Indication for Chelation Therapy in Myelodysplastic Syndrome
The most frequent cytopenia observed in myelodysplastic syndromes is anemia. The majority of MDS patients present with anemia at the time of diagnosis. About 60% of all MDS patients will experience severe anemia during the course of the disease.
The need of transfusion therapy is based upon clinical evaluation of anemia-related symptoms and existence of comorbidities. Preserving patient's quality of life (QoL) is the main goal of transfusion therapy. Managing the long-term efficacy of transfusion therapy of a patient in relationship to his QoL is essential as is improvement in organ function and survival.
According to evidence-based guidelines, red blood cell transfusions will be the only therapeutic option offered to 40% of MDS patients diagnosed with anemia. 61
Uncontrolled studies, reviewed by Alessandrino et al, 62 have been published on the efficacy of iron chelation therapy on transfusion iron overload in patients with myelodysplastic syndrome. One unconfirmed study showed that subcutaneous bolus injection of Deferoxamine is an acceptable alternative to slow, pump-driven infusion. 63 Based on the limited available evidence, the Expert Panel of the Italian Society of Hematology 62 agreed that iron chelation should be considered as a therapy for myelodysplastic syndrome (The Italian guidelines have been published in pre Deferasirox era; a revision is currently in preparation). The Expert Panel of the British Society of Hematology 64 acknowledged that their recommendations for iron chelation treatment in myelodysplastic syndromes were based on limited data. The British experts concluded that iron chelation should be considered once a patient has received 5 gr iron (approximately 25 units of red cells) but only in patients for whom long-term transfusion therapy is likely, such as those with pure sideroblastic anemia or the 5q-syndrome. According the British guidelines, Deferoxamine 20 to 40 mg/kg should be administered by 12-hour subcutaneous infusion 5 to 7 days per week.
The U.S. Food and Drug Administration have approved Deferasirox for “treatment of chronic iron overload caused by blood transfusions in patients 2 years of age and older.” The therapeutic indications approved by the European Medicine Agency (EMEA) are more detailed, and with respect to myelodysplastic syndromes the drug is “indicated for the treatment of chronic iron overload due to blood transfusions when Deferoxamine therapy is contraindicated or inadequate.” Thus, Deferasirox is now available for treatment of transfusion iron overload in patients with myelodysplastic syndrome because Deferoxamine is inadequate in most of them. Data on the long-term safety and effectiveness are needed.
Recently retrospective data suggest that patients with iron overload related to allogeneic stem cell transplantation could benefit from iron chelation therapy. 60
Monitoring Therapy
For monitoring iron overload and response to therapy, serial measurements (monthly) of serum ferritin levels together with liver enzymes and inflammation parameters are recommend. Ferritin is a simple objective test and a relatively reliable and reproducible marker of iron overload unless the patient is suffering from an active hepatic disease or severe chronic inflammation. In such cases, serial imaging studies (MRI) may be required. 12
Place in Therapy
Currently Deferasirox is approved in over 85 countries worldwide, including the USA and Europe, and is under review in many more for the treatment of transfusional iron overload in adult and paediatric patients. Deferasirox is the first oral iron chelator approved in the EU for use in patients with transfusional iron overload from different disease processes. 50
Finally the problem of cost should be mentioned. A recent Italian study based on cost/benefit estimation from a societal perspective, quantified tariffs, expenses, generated an estimated mean direct cost of medical therapy in thalassemia (transfusion + deferoxamine chelation) of approximately 15,000 euros/year/patient. Iron chelation therapy with Deferasirox costs about twice, so it is expensive and clearly not commonly accessible worldwide.
Conclusions
Effective and tolerable chelation therapy with oral Deferasirox is an important development in the treatment of transfusional iron overload. It is significant to understand its capacity to provide constant chelation exposure and the potential to improve compliance. Results from up to 3.5 years of treatment in approximately 1000 patients have shown that deferasirox 20 mg/kg/day reduces overall iron burden in patients with transfusion-dependent anaemias.
In most patients, a starting dose of 20 mg/kg/day of deferasirox is suitable; however physicians should continue to monitor transfusional iron intake, serum ferritin, and safety markers on an ongoing basis to certify that any necessary dose adjustments are made in an opportune approach to meet patients’ needs. Deferasirox is generally well tolerated, with a manageable safety profile. 50
Disclosure
The authors report no conflicts of interest.
