Abstract
Deferiprone is an orally active iron-chelating agent used in the management of transfusion-related hemosiderosis. It has been in clinical use for over 20 years and has been shown to be effective in reducing cardiac iron load and improving cardiac function. As cardiac siderosis is the leading cause of death in patients with transfusion-dependent thalassemia, deferiprone helps to improve the overall prognosis of these patients. It is relatively well tolerated with gastrointestinal symptoms being the commonest side effects. Agranulocytosis (0.5%), neutropenia (9%), thrombocytopenia (up to 45%) and arthropathy (20%) are the most important side effects and may require discontinuation of therapy. Regular monitoring of blood counts is recommended for patients on deferiprone therapy.
Introduction
Thalassemia is a genetic disorder resulting from mutations of the α- or β-globin genes with reduced or ineffective globin chain synthesis. Mutations of the α-globin genes leads to excess γ and β chains, which produces unstable tetramers: γ4 (hemoglobin Bart’s) and β4 (hemoglobin H), respectively. Mutations of the β-globin genes result in absent (β0 thalassemia) or reduced (β+ thalassemia) β-globin production. This generates excess α chains, which aggregate and precipitate in the red cells, leading to chronic hemolysis and destruction of red cells and their precursors in the bone marrow or peripheral blood. In patients with thalassemia, chronic hemolysis and ineffective erythropoiesis lead to varying severity of anemia. Patients may have severe anemia and present with intrauterine death from hydrops fetalis or may present with mild asymptomatic anemia [Weatherall, 1997].
Patients with thalassemia major require regular blood transfusions from childhood to support normal growth and development and reduce extramedullary hematopoiesis. As there is no physiologic excretory pathway for iron, regular blood transfusions lead to iron accumulation in the body. In addition, patients with thalassemia major have decreased expression of hepcidin and increased expression of ferroportin which results in increased iron absorption through the gastrointestinal (GI) tract [Weizer-Stern et al. 2006; Gardenghi et al. 2007]. Transfusional iron overload and increased GI iron absorption have been postulated to account for the secondary hemosiderosis in these patients.
Secondary hemosiderosis in patients with transfusion-dependent thalassemia is associated with endocrinopathies, including diabetes mellitus (DM), hypothyroidism and hypogonadism [De Sanctis et al. 2004]; hepatic cirrhosis; and cardiac complications like cardiomyopathy and arrhythmias. Cardiac siderosis related complications are the leading cause of death in these patients [Zurlo et al. 1989; Ceci et al. 2006; Aessopos et al. 2008].
Deferoxamine has been the standard iron chelator in clinical practice since the 1970s and has helped to improve the prognosis of patients with thalassemia major. It is given as a continuous subcutaneous infusion over 8–10 h, 5–7 days per week. Owing to the taxing nature of administration, patients tend to be poorly compliant and, hence, a significant proportion of patients continue to develop iron-related complications despite the availability of deferoxamine [Modell et al. 2000].
Deferiprone was the first oral chelator to be introduced for clinical use. It can bind iron in conditions of iron overload and is able to excrete excess iron from the body. Since the first publication of the use of deferiprone in humans in 1987 [Kontoghiorghes et al. 1987], numerous clinical studies have published reports on the efficacy of deferiprone in accessing various iron tissue stores in the body as well as on its safety profile. The objective of this review is to summarize data on the clinical efficacy and safety of deferiprone in patients with transfusion-dependent thalassemia.
Pharmacology
Deferiprone is an orally active bidentate hydroxypyridinone that binds with iron to form a stable 1:3 iron–chelator complex. It has excellent oral bioavailability. It is rapidly absorbed after oral ingestion, with peak plasma levels at 45–60 min after ingestion. Ingestion with meals does not alter the absorption. It is metabolized in the body by hepatic glucuronidation to an inactive metabolite. The chelator complex and free drug metabolite are mainly excreted in the urine [Al-Refaie et al. 1995b].
Deferiprone was approved for clinical use in Europe in 1999. The US Food and Drug Administration (FDA) approved deferiprone recently, in 2011, for the treatment of transfusional iron overload in patients with thalassemia when their current chelation therapy is inadequate. The recommended dosage for deferiprone is 50–75 mg/kg/day in three divided doses, up to a maximum of 100 mg/kg/day. Dosage should be adjusted based on the patient’s response and therapeutic goals.
Efficacy of deferiprone
The goal of deferiprone therapy is to remove excess iron from the body and inactivate current iron deposits with the aim of achieving negative iron balance. Assessment of efficacy can be estimated in the short term by measuring the net iron balance, and in the long term by measuring serum ferritin concentrations and iron concentrations in the various organs, especially in the liver and the myocardium.
Net iron balance
Pharmacokinetic studies with deferiprone have shown an incremental dose–response relationship in achieving increased urinary excretion [Olivieri et al. 1990; Collins et al. 1994], although higher doses are associated with greater side effects [Al-Refaie et al. 1995a]. The studies also demonstrated that a dose of 75 mg/kg/day of deferiprone was comparable to 50 mg/kg/day of deferoxamine in achieving negative iron balance [Olivieri et al. 1990; Collins et al. 1994]. A Cochrane systematic review did not find a consistent effect on reduction of iron overload between deferiprone and deferoxamine, with one trial favoring deferiprone and three trials favoring deferoxamine in increasing urinary iron excretion [Roberts et al. 2007]. A recent meta-analysis on iron chelation therapy has shown greater urinary iron excretion in patients receiving combination therapy with deferiprone and deferoxamine compared with patients receiving deferiprone or deferoxamine alone (
Serum ferritin concentration
Serum ferritin concentration is a good surrogate marker to estimate the body iron load. Ferritin plays a critical role in cellular iron homeostasis and is a site for iron storage. Deferiprone has been shown to be effective at decreasing or stabilizing serum ferritin concentrations [Al-Refaie et al. 1995a; Hoffbrand et al. 1998; Galanello, 2007], with greater decline seen in patients with higher baseline ferritin concentrations (greater than 2500 μg/liter) prior to starting deferiprone [Cohen et al. 2003]. Single values of ferritin may be misleading, as ferritin is an acute phase reactant and may be elevated in inflammatory processes, but monitoring serum ferritin concentration over time has been shown to be useful to trend the response to chelation therapy in patients [Galanello, 2007].
Cardiac magnetic resonance imaging
Cardiac magnetic resonance imaging (MRI) T2* has been shown to be a good surrogate for tissue iron concentration and is inversely related to tissue iron load [Anderson et al. 2001]. This technique has high reproducibility in measuring cardiac iron load (coefficient of variation 5.0%) [Anderson et al. 2001]. It has been shown across multiple studies (Table 1) that deferiprone is significantly more effective than deferoxamine at reducing cardiac iron load, improving left ventricular ejection fraction and reducing prevalence of cardiac dysfunction [Anderson et al. 2002; Piga et al. 2003; Borgna-Pignatti et al. 2006; Pennell et al. 2006; Perifanis et al. 2007]. Deferiprone therapy, either as monotherapy or in combination with deferoxamine, has been shown to improve right ventricular ejection fraction which may further contribute to improved cardiac function [Smith et al. 2011; Alpendurada et al. 2012]. Use of deferiprone has been shown to yield a 40.7–46.4% and 25.5–29.6% relative risk reduction for heart failure in patients with reduced and normal baseline ejection fraction, respectively [Pennell et al. 2011]. There have been reports of dramatic improvement in myocardial function after addition of deferiprone to the chelation regimen in patients with end-stage heart failure [Wu et al. 2004; Porcu et al. 2007]. Combination therapy with deferiprone and deferoxamine showed a two-to threefold increase in cardiac MRI T2* scores and 9% increase in mean left ventricular ejection fraction, suggesting that combination therapy may be more effective in preventing or reversing cardiac function [Farmaki et al. 2011].
Reports on efficacy of deferiprone on cardiac function and cardiac iron load.

IQR, interquartile range; SD, standard deviation.
Iron-induced mitochondrial damage with loss of mitochondrial cellular energy production has been postulated to cause cardiac disease in these patients [Pennell et al. 2011]. Iron exposure has been postulated to generate reactive oxygen species that damage the mitochondrial membrane and its DNA, resulting in impaired synthesis of respiratory chain subunits. Iron may bind directly to respiratory chain enzymes to alter their catalytic activity by converting amino acid residues to carbonyl derivatives. Impaired mitochondrial functioning leads to reduced adenosine triphosphate production, which in turn, adversely affects myocardial functioning. As deferiprone has a lower molecular weight and is lipophilic, it is postulated that it is able to penetrate the myocardial cells and remove intracellular toxic iron stores, particularly within mitochondria, and hence, help reduce cardiac siderosis [Piga et al. 2010; Pennell et al. 2011].
Liver iron concentration
Liver biopsy is considered the gold standard to measure liver iron concentration (LIC) but is an invasive procedure and is associated with significant risks. Despite newer noninvasive modalities, including superconducting quantum interference device (SQUID), MRI T2* and more recently, MRI R2 [Christoforidis et al. 2009], data on efficacy on LIC is lacking. Using MRI T2* values as a surrogate for LIC, studies have shown significantly lower LIC in patients on deferoxamine compared with deferiprone [Anderson et al. 2002; Perifanis et al. 2007]. However, there was no difference in reduction of LIC when measured on liver biopsy between deferiprone and deferoxamine groups [Maggio et al. 2002; Pennell et al. 2006] (Table 2). Patients on combination therapy with deferiprone and deferoxamine had a significantly greater reduction in liver iron concentration compared with deferiprone alone (
Reports on efficacy of deferiprone on hepatic iron load.

IQR, interquartile range; MRI, magnetic resonance imaging; SD, standard deviation.
Endocrinopathies
There has been a gradual decline in the incidences of hypothyroidism, DM, hypoparathyroidism and hypogonadism with long-term deferiprone therapy [Gamberini et al. 2008]. Serum ferritin concentrations above 2000 μg/liter are associated with hypogonadism and levels above 3000 μg/liter are associated with hypoparathyroidism, hypothyroidism and DM [Gamberini et al. 2008]. As combination therapy with deferiprone and deferoxamine is more effective in reducing total body iron load, combination therapy has been shown to reverse abnormalities of glucose metabolism and improve gonadal function more effectively [Farmaki et al. 2011]. Patients treated with combination therapy had significantly reduced mean glucose levels on oral glucose tolerance testing. Gonadal function and fertility improved in men and women, with successful pregnancy achieved in some patients [Farmaki et al. 2011].
Long-term survival
As cardiac siderosis is the leading cause of death in patients with transfusion-dependent thalassemia, reduction in cardiac iron load by deferiprone, either as monotherapy or in combination with deferoxamine, has been shown to remarkably decrease prevalence of cardiac disease and cardiac death and improve overall survival [Borgna-Pignatti et al. 2006; Telfer et al. 2009].
Safety profile
Deferiprone has been in clinical use for over 20 years. Its safety has been extensively evaluated across multiple studies, both in the short term as well as long term (Table 3).
Safety profile of deferiprone.
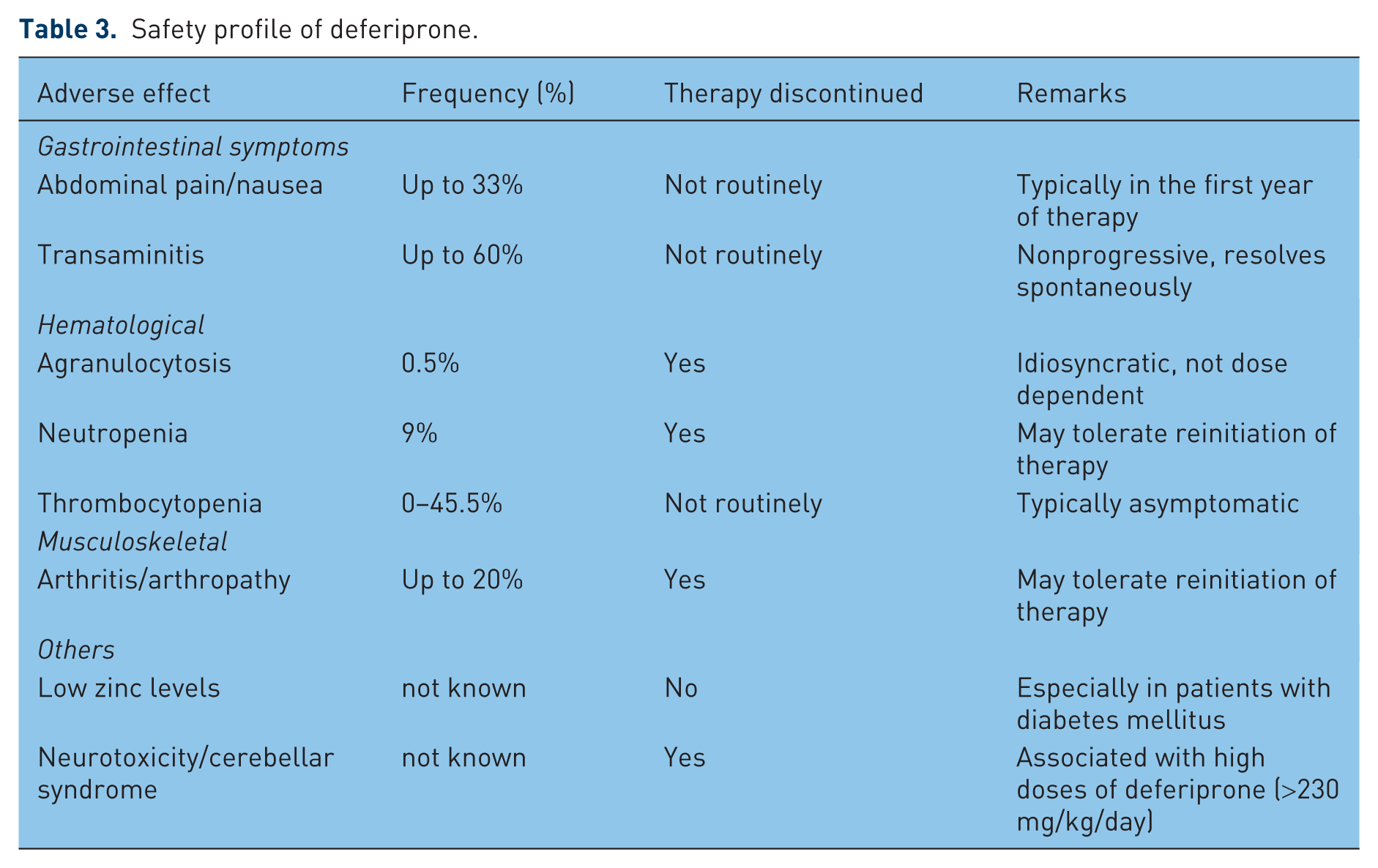
Gastrointestinal symptoms
GI-related symptoms including abdominal pain and nausea are the commonest adverse reactions related to deferiprone use. The incidence can be as high as 33% but is most commonly seen within the first year of therapy [Cohen et al. 2003]. The symptoms are mild to moderate and resolve without discontinuation of therapy in the majority of patients.
Transient fluctuations in transaminases have been reported in up to 60% of patients [Al-Refaie et al. 1995a]. However, they resolve spontaneously and do not progress over time. Concerns related to an increased rate of hepatic fibrosis associated with deferiprone therapy was reported in an initial study [Olivieri et al. 1998]. Subsequent reviews have shown no evidence of progression of hepatic fibrosis [Wanless et al. 2002].
Agranulocytosis and neutropenia
Agranulocytosis, defined as absolute neutrophil count of less than 0.5 × 109/liter, is the most serious adverse effect associated with deferiprone therapy and occurs in around 0.5% of patients [Cohen et al. 2003; Hoffbrand et al. 2003]. It typically occurs in the first year of therapy, although it has been reported after up to 2 years of therapy. Neutropenia, defined as absolute neutrophil count between 0.5 and 1.5 × 109/liter, has been reported in up to 9% of patients and occurs more commonly in nonsplenectomized patients [Ceci et al. 2002; Cohen et al. 2003]. Agranulocytosis and neutropenia usually resolve spontaneously with discontinuation of the treatment but occasional patients have been given granulocyte colony-stimulating factor for faster recovery. Fatal outcomes related to agranulocytosis have been reported in the literature [Henter and Karlén, 2007]. Owing to a high risk of recurrence, reintroduction of deferiprone after an initial episode of agranulocytosis is not recommended, although patients with neutropenia tend to tolerate reintroduction better [Jamuar et al. 2011]. In the majority of patients, the mechanism for agranulocytosis and neutropenia remains unclear. It appears to be idiosyncratic and is not dose dependent [Pontikoglou and Papadaki, 2010]. A possible mechanism could be related to interaction of deferiprone with copper, which could affect copper-containing enzymes and copper-dependent biological processes that result in bone marrow toxicity [Pashalidis and Kontoghiorghes, 2000]. Cytomorphological and functional bone marrow examination on a patient with agranulocytosis while on deferiprone therapy showed block of myeloid lineage at the stage of promyelocytes and normal functional capacity of hematopoietic progenitor cells
Thrombocytopenia
Thrombocytopenia, defined as a platelet count of less than 150 × 109/liter, associated with deferiprone has not been described as an adverse effect in some studies [Cohen et al. 2003; Galanello, 2007], while in other studies, the incidence of thrombocytopenia has been reported from as low as 2.7% to as high as 45.5% [Naithani et al. 2005; Panigrahi et al. 2010; Jamuar et al. 2011]. None of the patients have been reported to have bleeding-related complications or required platelet transfusion. The mechanism for thrombocytopenia is not known.
Arthropathy/arthritis
Frequency of joint symptoms varies among studies and has been reported in up to 20% of patients receiving deferiprone [Al-Refaie et al. 1995a; Cohen et al. 2003; Jamuar et al. 2011]. Joint symptoms may present with pain alone or with swelling and redness, suggestive of arthritis. The symptoms can present even after many years of therapy. The symptoms may be severe enough to warrant discontinuation of therapy and may recur with reinitiation of deferiprone [Jamuar et al. 2011]. The underlying mechanism remains unknown, although it is postulated that low synovial fluid concentrations of deferiprone relative to iron may lead to formation of unstable and potentially damaging 1:1 and 1:2 iron–chelator complexes [Berkovitch et al. 1994]. Evaluation for a possible autoimmune cause with rheumatoid factor, antinuclear and anti-double-stranded DNA antibodies is usually negative [Jamuar et al. 2011].
Low zinc levels
Lower levels of zinc have been reported in patients on deferiprone therapy compared with controls, especially in patients with DM [Galanello, 2007]. However, the levels remain within the normal reference range and may not be clinically significant. Clinically significant zinc deficiency was seen more commonly in patients on higher doses of deferiprone [Al-Refaie et al. 1995a]. The lower zinc levels may be related to chelation of divalent zinc cations by deferiprone, resulting in increased 24 h urinary zinc excretion in patients on deferiprone [Bartakke et al. 2005].
Neurotoxicity/cerebellar syndrome
Two patients developed neurotoxicity and cerebellar syndrome (nystagmus, dystonia, axial hypotonia, ataxia and impaired motor coordination) when treated with high doses of deferiprone (>230 mg/kg/day). The symptoms resolved after deferiprone was discontinued. Deferiprone crosses the blood–brain barrier and the onset of these symptoms could be related to a direct toxic effect or excessive chelation of other divalent cations (like zinc and copper) that are essential cofactors for enzyme activity [Beau-Salinas et al. 2009].
Pregnancy and breast feeding
Animal studies suggest that deferiprone is embryotoxic and teratogenic. Women of childbearing age on deferiprone therapy should be counseled to avoid getting pregnant or advised to switch to an alternative iron chelator if they are planning to get pregnant. Nursing mothers should avoid deferiprone as well [Galanello, 2007].
Use in pediatric age group
In children aged 1–10 years old, liquid formulation of deferiprone given at the recommended dose of 50–100 mg/kg/day was shown to have a safety profile similar to that reported in older children or adults. The most frequent adverse reactions were transient increase in transaminases (12%) and GI disturbance (11%). Neutropenia, agranulocytosis and arthralgia were seen in 6%, 2% and 4% of patients, respectively. These adverse events resolved without any complications. Deferiprone was discontinued in the two patients with agranulocytosis and in one patient with arthralgia. Deferiprone was effective in reducing the mean serum ferritin levels from 2532±1463 μg/liter at baseline to 2176±1144 μg/liter after 6 months of therapy (
Conclusion
Deferiprone is an effective oral iron chelator and is able to reduce total body iron, especially intracellular iron stores from the myocardium. As cardiac siderosis is the leading cause of death in patients with transfusional iron overload, deferiprone is an effective therapy that improves the overall prognosis for these patients. Deferiprone is well tolerated by patients. Agranulocytosis, neutropenia, arthropathy and thrombocytopenia are the most important side effects. Careful monitoring of blood counts remains a critical component of therapy with deferiprone.
Footnotes
Funding
The authors did not receive any funding from any source for this study.
Conflict of interest statement
The authors have attended workshops and conferences sponsored by Apo-pharma, the manufacturers of deferiprone. However, they do not have any ongoing or long-term financial support or relationships that may pose a conflict of interest.
