Abstract
Lung cancer is the most common cause of cancer mortality worldwide and even with current advances in chemotherapy the outcome is disappointing. The advances in molecular biology have enabled us to identify various targets for anticancer treatments. The VEGF-VEGFR signalling pathway plays a significant role in tumor angiogenesis and has been a major target for the development of various anticancer drugs. Bevacizumab is a recombinant humanized monoclonal antibody, which binds to VEGF and has shown efficacy in various tumors. Bevacizumab has been licensed in first line treatment of non-small cell lung cancer (NSCLC) in combination with chemotherapy. We are reviewing the evidence of the benefit and toxicity of Bevacizumab in combination with chemotherapy and other targeted agents in advanced NSCLC.
Introduction
Lung cancer is the most common cause of cancer mortality worldwide for both men and women, causing approximately 1.2 million deaths per year. 1 In the United States in 2007, it was estimated that there would be 215,000 new cases of lung cancer and 162,000 deaths. 2 Around 1953, lung cancer became the most common cause of cancer deaths in men, and in 1985 it became the leading cause of cancer deaths in women.
Lung cancer deaths have begun to decline in men, reflecting a decrease in smoking. 3 The rise in the death rate in women appears to have reached a plateau, although almost half of all lung cancer deaths now occur in women. Approximately 95 percent of all lung cancers are classified as either small cell lung cancer (SCLC) or non-small cell lung cancer (NSCLC). Other cell types comprise about 5 percent of malignancies arising in the lung.
Patients with advanced NSCLC are managed palliatively with systemic chemotherapy and/or local palliative modalities. The availability of multiple active agents has led to the development of numerous two-drug chemotherapy regimens. The use of such doublets has become the standard of care for the initial palliative management of patients with advanced NSCLC.
Despite the large number of randomized trials4,5 and a meta-analysis of 17 trials, 6 no single regimen has emerged as the best choice for the treatment of advanced NSCLC. Eastern Cooperative Oncology Group (ECOG) study 4 randomly assigned 1155 patients to one of four doublets: Gemcitabine plus Cisplatin, Docetaxel plus Cisplatin, Paclitaxel plus Carboplatin, and Cisplatin plus Paclitaxel. Overall response rate was 19%, median survival was 7.9 months, and one-year survival was 33%. The survival rates were similar in all four groups. The meta-analysis 6 included a total of 4792 patients and found that the use of a platinum-based doublet regimen was associated with a slightly higher survival at one year (relative risk of survival 1.08, 95% CI 1.01-1.16). The recommendation from the American Society of Clinical Oncology (ASCO) guidelines is that that first-line therapy should consist of a platinum-based doublet regimen. 7
Inspite of the advances made in chemotherapy the outcome of NSCLC is still disappointing. Progressive advances in tumor and cell biology has enabled us to understand the mechanism of oncogenesis at a molecular level and identify various molecular targets for anti cancer treatment. Two of the most successful targets in clinical practise for NSCLC have been Epidermal growth factor receptor (EGFR) and Vascular endothelial growth factor receptor (VEGFR). Erlotinib (Tarceva, OSI Pharmaceuticals, Melville, NY) is an EGFR inhibitor and now has an established role in patients who have progressed on chemotherapy. 8 The VEGF-VEGFR signalling pathway plays a major role in tumor angiogenesis. Vascular endothelial growth factor (VEGF) combines with Type 1(Flt-1) and 2(KDR) VEGFR resulting in activation of various intracellular signalling pathways giving rise to endothelial cell proliferation and new vessel formation.
Smaller tumors of 1-2 mm derive their blood supply from the surrounding vessels, but to grow more than 2 mm, the tumor needs to establish its own blood supply 9 by promoting angiogenesis thereby resulting in tumor migration and metastasis. Tumor blood vessels are irregularly shaped, dilated, tortuous and can have dead ends. These vascular networks are often leaky and hemorrhagic, partly due to the overproduction of vascular endothelial growth factor (VEGF). 9 Drugs (chemotherapy) that rely on these vascular networks to get into the tumor cells are supplied at suboptimal levels due to these abnormal vessels.10,11
The significant role played by VEGF- VEGFR pathway has made it a major target for the development of anti-cancer drugs. The first VEGF inhibitor antibody that has shown promising results in clinical trials and has been approved in the USA, Europe and elsewhere was Bevacizumab (Avastin; Genentech, Inc, South San Francisco, CA). 12
Bevacizumab is a recombinant humanized monoclonal IgG1 antibody, which binds to VEGF thereby preventing the activation of VEGFR (Flt-1 and KDR). It also normalizes tumor vasculature resulting in more effective delivery of chemotherapy. 10,11
The recommended dose of Bevacizumab in non-small cell lung cancer is 7.5 mg/kg or 15 mg/kg bodyweight, given every 3 weeks as an intravenous infusion. It can be used in combination with platinum based chemotherapy for up to 6 cycles followed by use as a single agent until disease progression. The estimated half-life of Bevacizumab is approximately 20 days (range 11-50 days) and the predicted time to reach steady state is 100 days. 13
Bevacizumab as First Line Treatment in Advanced or Metastatic NSCLC (Except Squamous cell)
A phase II trial evaluated the addition of Bevacizumab to Carboplatin and Paclitaxel. 14 99 patients with histologically confirmed stage IIIB/IV, or recurrent NSCLC were randomly assigned to Bevacizumab 7.5 mg/kg (n = 32) or 15 mg/kg (n = 35) plus Carboplatin (AUC = 6) and Paclitaxel (200 mg/m2) every 3 weeks or Carboplatin and Paclitaxel alone (n = 32).
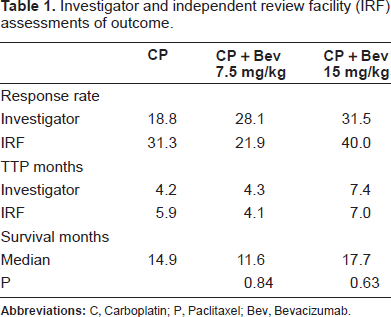
This study (Table 1) concluded that treatment with Carboplatin and Paclitaxel plus Bevacizumab (15 mg/kg) resulted in a higher RR (31.5% vs. 18.8%), longer median TTP (7.4 vs. 4.2 months, p = 0.023) and a modest increase in survival (17.7 vs. 14.9 months, p = 0.63). The results of this trial led to the development of a Phase III study by the Eastern Cooperative Oncology group (E4599 study). 15 This trial and most of the subsequent trials excluded squamous cell NSCLC in view of the increased risk of pulmonary hemorrhage in this trial (see below).
Investigator and independent review facility (IRF) assessments of outcome.
Two key studies resulted in the licensing of Bevacizumab as first line treatment in NSCLC. First was the E4599 study 15 which was a trial conducted mainly in the United States and second was the AVAiL study16–18 which confirmed the efficacy of Bevacizumab in NSCLC in a global population.
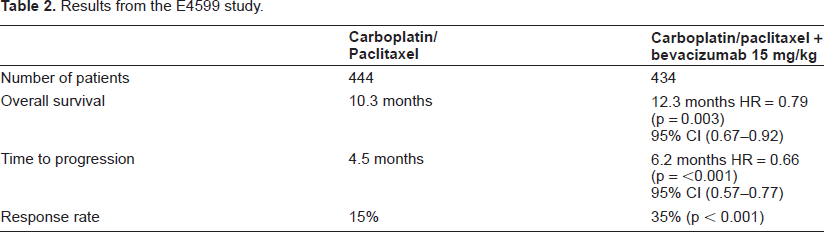
The E4599 study 14 was a randomized, phase III study of Bevacizumab in combination with Carbo platin and Paclitaxel in patients with recurrent or advanced NSCLC (stage IIIB or IV) with non-squamous histology. A total of 878 patients were recruited. Patients were randomized to 6 cycles of Carboplatin AUC 6 day 1 and Paclitaxel 200 mg/m2 day 1 with or without Bevacizumab 15 mg/kg body weight day 1 every 3 weeks. Bevacizumab was continued after chemotherapy untill disease progression. The primary end point was overall survival (OS) and secondary endpoints included time to progression (TTP), response rate (RR) and tolerability.
The results (Table 2) showed the median OS was 12.3 months in the group receiving chemotherapy and Bevacizumab, as opposed to 10.3 months in the chemotherapy only group (hazard ratio for death, 0.79; P=0.003). The median TTP in the two groups was 6.2 and 4.5 months, (hazard ratio for disease progression, 0.66; P<0.001), and RR was 35% and 15% respectively.
Results from the E4599 study.
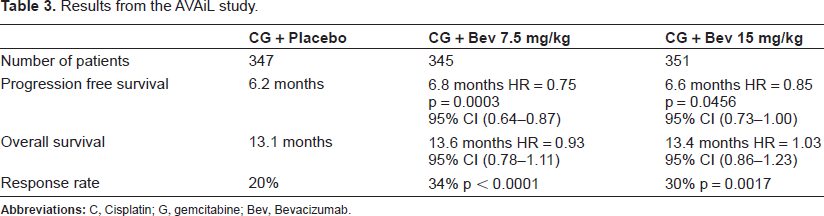
The AVAiL study16–18 was a randomized, double blind, phase III study of Bevacizumab in combination with Cisplatin and Gemcitabine, versus placebo in chemotherapy naïve patients with advanced (stage IIIb/IV) or recurrent non-squamous NSCLC. A total of 1043 patients were recruited. Cisplatin 80 mg/m2 day1 and Gemcitabine 1250 mg/m2 day 1 + 8 was given with either Bevacizumab 7.5 mg/kg or 15 mg/kg or placebo day 1 and was given 3 weekly. Bevacizumab or placebo was continued after chemotherapy until disease progression.
The primary end point was Progression free survival (PFS) and secondary end points were OS, RR, duration of response and safety.
The study (Table 3) showed that the addition of Bevacizumab to Cisplatin and Gemcitabine resulted in improvement of PFS but it did not translate into OS. This could be because of heterogeneity in post protocol second-line therapies. The OS in the chemotherapy + placebo arm was more than 13 months, which is the longest reported for NSCLC.
Results from the AVAiL study.
Various other Phase 2 trials have been reported and are summarized in Table 4. In all of these trials Bevacizumab (15 mg/kg) has been used in combination with first line chemotherapy in advanced or recurrent NSCLC.
Phase II trials of bevacizumab with chemotherapy in 1st line.
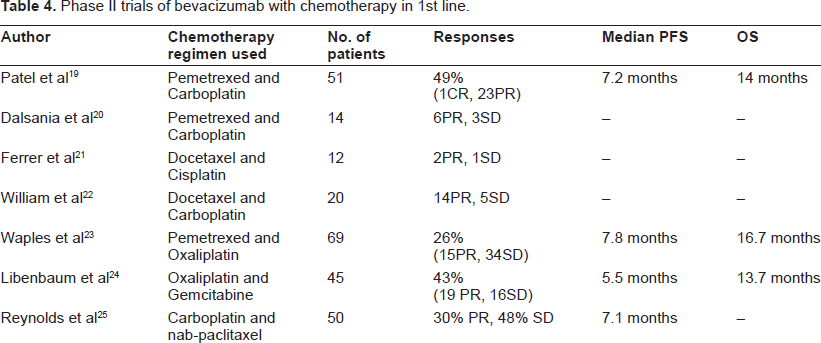
These studies further support the evidence from the large phase 3 trials regarding the use of bevacizumab in combination with various platinum based doublet combination in non-squamous NSCLC.
Bevacizumab as Second Line Treatment in Advanced or Metastatic NSCLC (Except Squamous Cell)
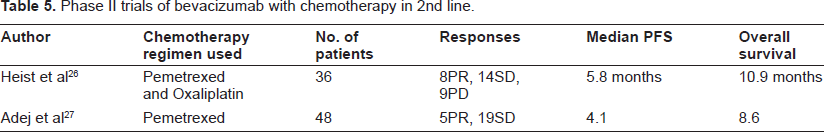
Bevacizumab has been combined with chemotherapy agents after failure of 1st line chemotherapy agents in Phase II trials and are summarized in Table 5.
Phase II trials of bevacizumab with chemotherapy in 2nd line.
Bevacizumab with Erlotinib in Advanced or Metastatic NSCLC
Since multilevel cross-stimulation exists among the targets of the new biological anticancer agents, multi-targeted therapy may be a better therapeutic option. Combining VEGF and EGF receptor blockade may produce additive effects as they share a common downstream signaling pathway. Activation of EGFR signaling can up regulate the production of VEGF in human cancer cells and EGFR blockade can cause inhibition of the secretion of VEGF. 28
The combination of Erlotinib with Bevacizumab has been evaluated in an open label phase I/II study 29 in patients with recurrent non-squamous NSCLC. The phase I part of the study (n = 40) assessed the maximum tolerated dose of this drug combination and found that the 15 mg/kg dose of Bevacizumab every three weeks with 150 mg/day oral Erlotinib was an acceptable regimen. There were no dose limiting toxicities observed during the phase I part of the trial.
Patients (n = 34) who had relapsed after at least one platinum based chemotherapy regimen for metastatic disease were entered into the phase II part of the trial. Of the total of 40 patients, eight patients (20.0%; 95% CI, 7.6% to 32.4%) had partial responses and 26 (65.0%; 95% CI, 50.2% to 79.8%) had stable disease as their best response. The median OS for the 34 patients treated at the phase II dose was 12.6 months, and PFS was 6.2 months.
Groen et al 30 conducted a phase II study of Erlotinib (150 mg/day) and Bevacizumab (15 mg/kg) given every 21 days, in patients with previously untreated stage IIIb/IV NSCLC. Primary endpoint was no progression (NPR) at 6 weeks. Secondary endpoints were objective response rate (ORR), TTP and OS. 47 patients were included with a median follow up of 15.2 months. NPR after 6 weeks was 75% and ORR was 26%. Median TPP was 4 months and median OS 6.9 months.
Faoro et al. 31 looked at sequential treatment of Bevacizumab with Erlotinib and chemotherapy for first line treatment of clinical stage IIIB or IV NSCLC. Treatment consisted of 3 modules. Module A: up to 4 cycles of Bevacizumab (15 mg/kg) every 21 days and Erlotinib (150 mg qd). Module B: up to 4 cycles of Carboplatin (AUC = 6), Paclitaxel (200 mg/m2) and Bevacizumab (15 mg/kg) every 21 days. Patients who had either stable disease or any response in module A progressed to module C: Bevacizumab every 21 days and Erlotinib until progressive disease.
A total of 48 patients were enrolled in the study. The primary objective was to determine the rate of non-progressive disease after module A. In module A, 63% (n = 30) of patients had 4 cycles. Lack of progression was seen in 64% (n = 31), and 12% had a Partial Response. 37 patients were treated on module B, with 36 evaluable for response resulting in partial response in 21% (n = 8), stable disease in 45% (n = 17) and progressive disease in 29% (n = 11). 23 patients were treated on module C (median 7 cycles per patient). 19 patients were evaluated for response: 17% (n = 4) had partial response, 39% (n = 9) had stable disease and 26% (n = 6) had progressive disease. 10 patients remained on study. Median survival for all patients was 12.9 months (95% CI = 7.1 to 18.6 months). 1-year survival rate was 49%. Patients who had stable disease or partial response in module A had a median survival of 23.2 months compared to 6.6 months in progressors, (p < 0.001).
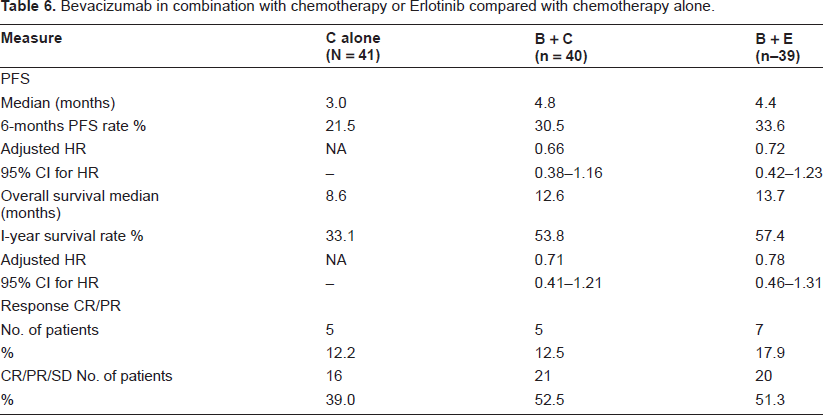
Herbst et al 32 looked at efficacy and safety of Bevacizumab in combination with chemotherapy or Erlotinib compared with chemotherapy alone for treatment of recurrent or refractory NSCLC in a phase II study as measured by PFS. Although not statistically significant, relative to chemotherapy alone, the risk of disease progression or death was 0.66 (95% CI -0.38 to 1.16) among patients treated with Bevacizumab + chemotherapy and 0.72 (95% CI -0.42 to 1.23) among patients treated with Bevacizumab + Erlotinib. One-year survival rate was 57.4% for Bevacizumab + Erlotinib and 53.8% for Bevacizumab + Chemotherapy compared with 33.1% for Chemotherapy alone (Table 6).
Bevacizumab in combination with chemotherapy or Erlotinib compared with chemotherapy alone.
Two phase III trials of Bevacizumab and Erlotinib combination have recently reported their results. The ATLAS Trial 33 evaluated the efficacy of Bevacizumab with or without Erlotinib, given after Bevacizumab plus chemotherapy and the BeTA Lung Trial 34 assessed the efficacy of Erlotinib with or without Bevacizumab in the second-line setting.
ATLAS 33 was a global, multicenter, randomized, Phase III, double blind, placebo-controlled study that had enrolled 1,160 patients with locally advanced, recurrent or metastatic NSCLC (including patients with treated brain metastasis, squamous histology and anticoagulated with LMWH). Patients were initially treated with four cycles of Bevacizumab in combination with the investigators’ choice of platinum-based chemotherapy regimens. If the patients did not progress or get significant toxicity, they were randomized (n = 768) to receive maintenance therapy with Bevacizumab plus Erlotinib or Bevacizumab plus placebo until disease progression. The study's primary endpoint was PFS. PFS assessment began from the start of the maintenance phase of the study after initial treatment with four cycles of Bevacizumab and chemotherapy. Secondary endpoints included OS, incidence of all adverse events, selected Grade 3 or more adverse events, and incidence of treatment discontinuation for reasons other than disease progression. It was announced in February 2009 that the study was stopped early on the recommendation of an independent data safety monitoring board after a pre-planned interim analysis showed that combining Erlotinib and Bevacizumab significantly improved PFS, compared to Bevacizumab plus placebo. The median PFS for Bevacizumab plus Erlotinib was 4.8 months compared to 3.7 months with Bevacizumab plus placebo (HR = 0.722 (95% CI: 0.592-0.881), p = 0.0012).
The BeTA Lung Trial 34 was a phase III, randomized trial comparing Erlotinib plus Bevacizumab with Erlotinib plus Placebo in the treatment of non squamous stage IIIB/IV NSCLC in the second line setting. 636 patients pre-treated with standard first-line chemotherapy were enrolled. Primary end point was OS and secondary endpoints were PFS and ORR. The results were presented at the Multidisciplinary Symposium in Thoracic Oncology in Chicago, 2008. Median OS was 9.3 months in the combination arm compared with 9.2 months in the single-agent arm [p = 0.75,HR: 0.97 (95% CI 0.80 to 1.18)]. Median PFS was 3.4 months in the combination arm compared with 1.7 months in the single agent arm [p = 0.0001,HR: 0.62 (95% CI 0.52 to 0.75)]. The ORR was 12.6% in the combination arm compared with 6.2% in the single agent arm (p = 0.006). The authors concluded that the addition of Bevacizumab to Erlotinib does not confer a survival benefit in the second-line treatment of advanced NSCLC. However, the significantly greater PFS and ORR values do provide evidence of clinical activity.
Bevacizumab with Cetuximab in Advanced or Metastatic NSCLC
A phase II study SWOG 0536 35 (Southwest Oncology Group) was presented at the 2008 Chicago Multidisciplinary Symposium in Thoracic Oncology. It evaluated the addition of the anti-EGFR mAb Cetuximab to Carboplatin, Paclitaxel, and Bevacizumab for 6 cycles followed by Cetuximab and Bevacizumab in advanced (Stage IIIB or IV), non squamous NSCLC. The primary end point was to evaluate the frequency and severity of hemorrhage toxicity. The secondary end points were PFS, OS, RR, non-hemorrhage toxicities and molecular correlative studies to investigate predictors of efficacy. 110 patients were registered of which 104 were evaluable. Pulmonary hemorrhage rate (severe) was 2%. The disease control rate was 74%, PFS was 7 months and OS was 14 months. The authors concluded that the combination was safe and effective.
Safety Profile of Bevacizumab with Chemotherapy
The first set of data regarding the safety of antiVEGF treatment in humans was published in 2001. Results from this preliminary phase I trial, 36 which included 25 patients with various different malignancies showed infrequent grade 3-4 toxicity with the use of the drug. The side effects that occurred were anemia (1 patient), dyspnea (1 patient) and serious bleeding (2 patients). Common, but less severe toxicity included headache, fever and asthenia.
Since then several studies have been conducted examining the use of Bevacizumab both on its own and in conjunction with various chemotherapy regimes. From its use in the treatment of other malignancies (e.g. colorectal cancer; 37 breast cancer) 38 the more common adverse effects of Bevacizumab are thought to be hypertension, gastrointestinal perforation, and arterial thrombosis.
The Phase II trial of Bevacizumab in NSCLC by Johnson et al 14 examined the use of Paclitaxel and Carboplatin with Bevacizumab in 99 patients with advanced or recurrent non-small cell lung cancer, including squamous cell lung cancers. The addition of Bevacizumab to this chemotherapy regimen did appear to change the toxicity profile, with symptoms such as diarrhea, headache and fever occurring much more often, however, these were predominantly experienced at grade 1-2. Hypertension was to be increased with the use of Bevacizumab, with 2 patients having grade 3 and above hypertension in the high dose arm. The risk of leucopenia, though significant in the chemotherapy alone arm (31.3%) was higher with the addition of Bevacizumab (46.9% in the low dose arm; 55.9% in the high dose arm). There was a significant increase in the risk of bleeding (epistaxis, hemoptysis and other hemorrhage) and 6 patients had a major life-threatening bleed, with four of these being fatal. There was no particular association with being exposed to a higher dose of Bevacizumab, as four of the six were actually on the lower dose. Predisposing factors for major hemorrhage appeared to be central tumors (near major vessels), cavitation, and necrosis within the lesions. Also of particular interest was that 4 of the 6 patients had squamous cell histology. Further analysis suggested non-squamous histology to be of much lower risk of pulmonary hemorrhage (4 of 13 with squamous versus only 2 of 54 with adenocarcinomas).
Subsequently the safety of Bevacizumab has been examined in several phase II and the two major phase III trials, however, these trials excluded squamous histology.
In the ECOG E4599 15 study the rates of neutropenia, headache, hypertension, bleeding events, proteinuria, febrile neutropenia, thrombocytopenia and rash were statistically significantly higher in the Paclitaxel-Carboplatin-Bevacizumab arm, compared with the Paclitaxel-Carboplatin arm. Grade 3 or more hypertension was noted in 7% of those in the Paclitaxel-Carboplatin-Bevacizumab arm compared with 0.7% of those without Bevacizumab. Seventeen deaths were thought to be toxicity-related within this trial, with 15 of these being in the Paclitaxel-Carboplatin-Bevacizumab arm (5 associated with bleeding; 5 with complications associated with febrile neutropenia; 2 each to gastrointestinal bleeds and cerebrovascular events; and 1 due to probable Pulmonary embolus).
Though neutropenia is not usually associated with Bevacizumab, 5 patients deaths related to neutropenia were noted in the Paclitaxel-Carboplatin-Bevacizumab arm compared with 1 in the Paclitaxel-Carboplatin arm.
The AVAiL study,16–18 suggests a slightly higher rate of adverse effects in the high dose Bevacizumab arm, compared with the placebo and low dose Bevacizumab arm. The rate of grade 3 hypertension was noted to be significantly higher with 9% in the high dose Bevacizumab arm, and 6% in the low dose Bevacizumab, compared with 2% in the placebo arm.
The risk of pulmonary hemorrhage (all grades) was increased in the high dose and low dose Bevacizumab arms (7.0% and 9.7% respectively) compared with the placebo arm (4.9%). However, neither the risk of grade 3 + hemorrhage nor fatal pulmonary hemorrhage followed this trend.
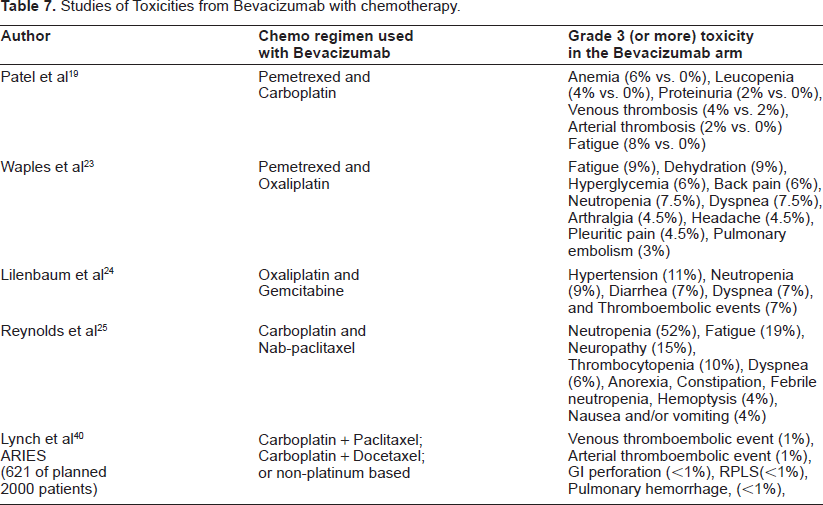
Bevacizumab has shown to be safe when combined with various chemotherapy combinations in NSCLC. MO19390 (SAiL) Study Group 39 looked at the safety profile of Bevacizumab in an open-label single-arm trial evaluating the safety of first-line Bevacizumab combined with various standard first line treatment of NSCLC. A total of 2008 patients were recruited. The primary end point was safety. Grade 3+ side effect reported were hypertension (0.7%), proteinuria (0.1%), gastrointestinal perforation (2.5%), wound healing perforation (0.2%), embolism (12.1%), congestive heart failure (0.4%), any bleeding (3.8%), CNS bleeding (0.4%) and hemoptysis (1%). The authors concluded that the side effects of Bevacizumab were mild to moderate, manageable and was consistent irrespective of the chemotherapy regimen used. Various other Phase II studies showing a multitude of toxicities (Grade 3 or more) of Bevacizumab along with different chemotherapy combinations are summarized in Table 7.
Studies of Toxicities from Bevacizumab with chemotherapy.
Most of the trials excluded patients with brain metastasis due to increased risk of bleed. Akerley et al 41
Safety Profile of Bevacizumab with Erlotinib
Groen et al 30 demonstrated Grade 3/4 events of rash (10%) when combining Bevacizumab and Erlotinib.
Faro et al looked at the use of Bevacizumab plus Erlotinib in their 3-stage phase II trial. 31 In two out of the three phases Erlotinib was combined with Bevacizumab, the first of these (module A) included grade 3 or 4 toxicity of rash (12%), neuropathy (6%), diarrhea (4%) and bowel perforation (4%). The second phase which involved (module C) had a 10% rate of neutropenia, 10% diarrhea, 10% thrombosis and 10% neuropathy rate (all grade 3-4).
The preliminary data from ATLAS Trial 42 having recruited 598 patients reported no unexpected safety events. Significant side effects of Bevacizumab with chemotherapy were hypertension, gastrointestinal perforation, arterio-thrombolic events, congestive heart failure and hemorrhage. The BeTA Lung Trial 34 reported Grade 3/4 rash and arteriothrombolic events to be higher in the Bevacizumab plus Erlotinib arm.
Conclusion
The U.S. Food and Drug Administration in October 2006 and the European Medicines Agency in August 2007 approved the use of Bevacizumab in combination with chemotherapy for the first-line treatment of patients with non-resectable, locally advanced, recurrent or metastatic, non-squamous, NSCLC.
Based on the data from the two large phase III trials (E4599 and AVAiL) and multiple phase II trials, we conclude that Bevacizumab can be safely combined with chemotherapy. As it also confers survival benefit, we suggest the standard treatment for first line, non-squamous, locally advanced or metastatic NSCLC should now be platinum based doublet chemotherapy with Bevacizumab.
Bevacizumab can be safely combined with other targeted therapy such as Erlotinib and Cetuximab. These combination needs to be further evaluated and its role in the routine treatment of NSCLC needs to be identified. Cetuximab is not currently routinely used in clinical practice in NSCLC, hence, any combination with it should be in the remit of a clinical trial only.
As Bevacizumab is expensive, it has a huge economic impact on the health care delivery systems and hence not yet been made standard first line treatment in many parts of the world.
There are many questions that still remain unanswered regarding the use of Bevacizumab in NSCLC. One of the most important questions is the optimal dose and duration of Bevacizumab treatment and the role of maintenance Bevacizumab. Bevacizumab 15 mg/kg was shown to have a better time to progression and response rate than 7.5 mg/kg in the Phase 2 trial 14 but similar benefit was not seen in the Phase 3 AVAiL study.16–18 Further studies will be required to decide on the optimum dose of Bevacizumab in NSCLC. In both the major phase III trials (E4599 and AVAil) Bevacizumab was given until disease progression. In view of the toxicities and the economic impact of Bevacizumab, we need to look at intermittent or shorter duration of Bevacizumab and see if the outcomes are similar.
We now know that Bevacizumab can safely be combined with other targeted therapy such as Erlotinib. The role of multi-targeted therapy along with platinum based chemotherapy now needs to be further investigated in the 1st line setting.
Most trials to date using Bevacizumab have excluded squamous cell cancers due to the risk of pulmonary hemorrhage. It may be interesting to look at this group to see if they benefit with the addition of Bevacizumab. The BRIDGE Trial 43 is looking at the safety of giving Bevacizumab with Carboplatin and Paclitaxel in patients with advanced, previously untreated NSCLC with squamous cell histology. Among the 27 patients who have received Bevacizumab, there have been 3 reports of pulmonary hemorrhage in 2 patients. Further updates from this trial would help guide us in the use of Bevacizumab in Squamous cell NSCLC.
The use of Bevacizumab is currently being evaluated in early NSCLC, both in the adjuvant and neoadjuvant settings. Bevacizumab and Chemotherapy for Operable NSCLC (BEACON) trial, recruiting at Memorial Sloan Kettering Cancer Center is looking at giving Bevacizumab and chemotherapy in stage IB–IIIA patients with resectable NSCLC. A U.S. Intergroup trial, ECOG 1505, is ongoing and is looking at adding Bevacizumab to platinum based chemotherapy in patients with resected stage IB, stage II, and stage III NSCLC. The results of these trials and various other ongoing and planned trials will hopefully further define the role of Bevacizumab in early NSCLC.
To date there has been no convincing data on predictive markers of response to Bevacizumab in NSCLC. Current evidence states that we should use Bevacizumab in all patients with NSCLC except those with squamous cell histology. There is a need to individualize treatment by having better patient selection criteria to improve response rates and reduce toxicity.
Disclosure
The authors report no conflicts of interest.
