Abstract
Non-small cell lung cancer (NSCLC) is the most deadly cancer in the world. Vinorelbine is an active vinca-alkaloid with a broad spectrum of anti-tumour activity. In particular, it has high activity in NSCLC, where a synergy with cisplatin has been demonstrated. Recently, oral vinorelbine has shown a similar activity and efficacy as compared to the intravenous formulation, giving to the patient an easier way of administration and increasing his comfort. Although surgery remains the best chance of cure for patients with resectable NSCLC, it is now accepted that postoperative cisplatin-based adjuvant chemotherapy significantly improves survival in patients with NSCLC. The combination of cisplatin and vinorelbine is the most active among all the adopted schedules. Patients presenting with locally advanced disease at diagnosis benefit from concomitant chemotherapy with cisplatin-vinorelbine regimen in combination with thoracic radiotherapy. Platinum-based combinations have become the standard of care for treating NSCLC. Vinorelbine–cisplatin combination is one of the proposed doublets for treating advanced, metastatic, NSCLC. Because of its favorable toxicity profile, vinorelbine has become a major compound as a treatment for elderly patients with advanced NSCLC.
Lung cancer is the leading cause of cancer–-related mortality in the Western World and is rapidly emerging as a major cause of mortality in the developing countries with recent statistics showing that, globally, it accounts for more than 1,000,000 deaths each year. 1 As small-cell lung cancer represents approximately 15% of lung cancers, 2 non small cell lung cancer (NSCLC) represents roughly 85% of lung cancers overall. Most NSCLCs are directly attributable to cigarette smoking. The predominant type of NSCLC used to be squamous cell carcinoma (SCC). However, due to the changes in tobacco blends and the use of filters in cigarettes, the relative incidence of SCC has decreased and adenocarcinoma (ADC) has become the dominant cell type. Historically, surgery has provided the best chance of cure for patients with resectable NSCLC. Whenever surgery has not been an option for patients with resectable cancers, radiotherapy has been used of the primary tumor and regional lymphatics. Platinum-based combinations have become the standard of care for treating advanced, metastatic, NSCLC.
Vinorelbine is a semi-synthetic second generation vinca-alkaloid that differs from earlier vinca-alkaloids 3 in that it has a substitution on the catharantine rather than the vindoline moieity of the molecule. 4 Vinorelbine exerts its cytostatic effect by binding to tubulin, the basic protein subunit of cellular microtubules, resulting in disruption of the mitotic spindle apparatus during metaphase. Vinorelbine has been demonstrated to have strong inhibitory effects on the polymerisation of mitotic microtubules, equal to vincristine, but to have a lesser effect on axonal microtubules implying that neurotoxicity is less likely to be a problem with vinorelbine as compared with the other vinca-alkaloids. Vinorelbine blocks the progression of cells at the G2/M stage of the cell cycle and produces apoptosis in NSCLC cell lines. In preclinical studies vinorelbine has shown a high level of activity against experimental tumors than have other vinca alkaloids. Vinorelbine has been shown to have indicated activity against non-small cell lung cancer5–7 and metastatic breast cancer.8–11 Because of its favorable toxicity profile, vinorelbine has been investigated as a treatment for elderly patients with advanced NSCLC. 12 Recently, an oral form of this agent has been developed. Oral vinorelbine was found to have acceptable and reliable pharmacokinetic profiles at clinically relevant dosage levels. Oral vinorelbine was found to have approximately 40% bioavailability; thus, a dose of 80 mg/m2 orally is the equivalent of 30 mg/m2 intravenously (IV), and 60 mg/m2 orally is the equivalent of 25 mg/m2 intravenously. 13 Oral vinorelbine is generally well tolerated with a mild-to-moderate toxicity, easily manageable with standard treatment. 14 Studies also concluded a lack of food effect on the administration of oral vinorelbine. In addition, no drug-drug interactions were found with a variety of commonly used antineoplastic agents. 15 Oral vinorelbine can therefore be considered a good alternative to IV administration. 16 Furthermore, the efficacy and safety of the alternating vinorelbine schedule (IV on day 1, oral on days 8, 15 and 22) in combination with cisplatin in advanced NSCLC was shown to be similar to those of the standard regimen using exclusively IV vinorelbine, whereas ease of administration and patient comfort may favor this approach.17–19
Adjuvant Chemotherapy
Surgical resection remains the treatment of choice for early-stage non-small cell lung cancer, yet 5-year survival rates for pathological stages I and II disease are only 60 to 70% and 35 to 40% respectively. 20 Recurrences tend to occur at extrathoracic sites, suggesting the presence of micrometastatic disease at the time of surgery, leading to the hypothesis that adjuvant systemic therapy should improve outcomes in resected NSCLC. 21
Four randomized clinical trials explored the role of the vinorelbine–-cisplatin combination in the adjuvant setting after complete resection of a non small-cell lung cancer (Table 1).
Randomized clinical trials comparing adjuvant chemotherapy versus observation for patients with resected NSCLC.
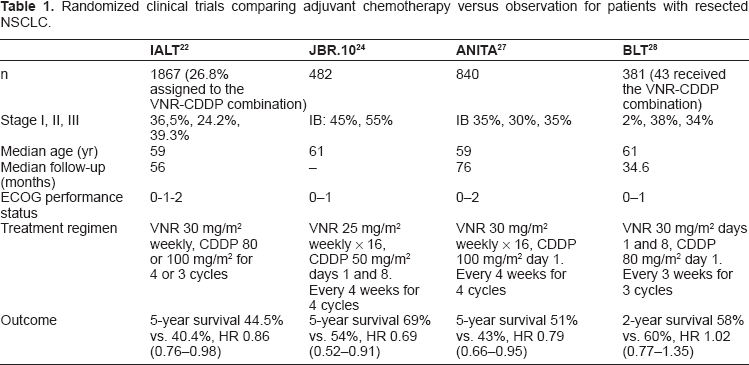
The International Adjuvant Lung Cancer Trial (IALT) was the first major trial to show a significant benefit to adjuvant chemotherapy in resected NSCLC. 22 A total of 1867 patients underwent randomization after complete resection of NSCLC. They were allocated either to three or four cycles of cisplatin-based chemotherapy or to observation; the main end point was overall survival. Vinorelbine was the drug allocated with cisplatin in 26.8% of patients. Patients assigned to chemotherapy had a significantly higher survival rate than those assigned to observation (44.5% vs. 40.4% at five years [469 deaths vs. 504]; hazard ratio for death-HR-0.86; 95 percent confidence interval–-95% CI: 0.76 to 0.98; P < 0.03) as well as a significantly higher disease-free survival rate (39.4% vs. 34.3% at 5 years [518 events vs. 577]; HR, 0.83; 95% CI, 0.74 to 0.94; P < 0.003). However, the results were not confirmed with a median follow-up of 7.5 years. 23
The JBR.10 trial randomly assigned patients with completely resected stage IB or stage II non-small-cell lung cancer to vinorelbine plus cisplatin (n = 242) or to observation (n = 240). 24 The primary end point, overall survival, was significantly prolonged in the chemotherapy group as compared with the observation group (94 vs. 73 months; HR for death, 0.69; P = 0.04), as was relapse-free survival (not reached vs. 46.7 months; HR for recurrence, 0.60; P < 0.001). Five-year survival rates were 69 percent and 54 percent, respectively (P = 0.03). The benefit in survival for adjuvant chemotherapy with cisplatin and vinorelbine was recently confirmed with a prolonged median follow-up of 9.3 years 25 and was proved to be a highly cost effective treatment. 26
The ANITA (Adjuvant Navelbine International Trialist Association) trial aimed to compare the effect of adjuvant vinorelbine plus cisplatin versus observation on survival in patients with completely resected stage IB-IIIA NSCLC. 27 The primary end point was also overall survival. 840 patients were randomly assigned to observation (n = 433) or to 30 mg/m2 vinorelbine plus 100 mg/m2 cisplatin (n = 407). After a median follow-up of 76 months (range 43–116), median survival was 65.7 months (95% CI: 47.9–88.5) in the chemotherapy group and 43.7 (95% CI: 35.7–52.3) months in the observation group. Adjusted risk for death was significantly reduced in patients assigned chemotherapy compared with controls (HR, 0.80 [95% CI: 0.66–0.96]; P = 0.017). Overall survival at 5 years with chemotherapy improved by 8·6%, which was maintained at 7 years (8.4%).
On the other hand, the surgical setting of the Big Lung Trial (BLT) randomised 97% of 381 patients to adjuvant chemotherapy or no chemotherapy. 28 Chemotherapy was three 3-weekly cycles of cisplatin/vindesine, mitomycin/ifosfamide/cisplatin, mitomycin/vinblastine/cisplatin or vinorelbine/cisplatin. This trial failed to observe a survival benefit with adjuvant chemotherapy. However, the vinorelbine–-cisplatin doublet was given to only 43 (22%) patients.
It is therefore admitted that postoperative cisplatin-based adjuvant chemotherapy significantly improves survival in patients with NSCLC. 21 The combination of cisplatin and vinorelbine seems to be the most active among all the adopted schedules. 29 The Lung Adjuvant Cisplatin Evaluation (LACE), collecting individual patient data from the five largest trials (4,584 patients) of cisplatin-based chemotherapy in completely resected patients, showed that postoperative cisplatin-based chemotherapy significantly improves survival in patients with NSCLC. 30 It has recently been shown that patients randomized to cisplatin-vinorelbine or observation were the largest subgroup (1888 patients) and the most homogeneous in terms of drug doses and eligibility. Survival improvement at 5 years was 8.9% with cisplatin-vinorelbine versus observation (HR 0.80, 95% CI: 0.70–0.91, P < 0.001). Stage was a significant predictor for survival (test for trend, P = 0.02; benefit at 5 years: 14.7% [stage III], 11.6% [stage II], and 1.8% [stage I]). Similar benefits were seen for disease-free survival. The overall result was statistically superior to that achieved by patients randomized to other chemotherapy or observation. Therefore, adjuvant cisplatin-vinorelbine provides a superior survival benefit and can be recommended in completely resected stages II and III non-small cell lung cancer. 31
Concomitant Chemoradiotherapy for Locally Advanced Inoperable NSCLC
At least one third of patients present with locally advanced (stage III) disease at diagnosis. Combined radiotherapy (RT) and cisplatin-based chemotherapy 32 has been shown to be superior to radiotherapy alone with a significant benefit demonstrated on survival. 33 Moreover, concomitant chemoradiotherapy (CT-RT), as compared to sequential CT-RT, improves survival of patients with locally advanced NSCLC, mainly due to the decrease of loco-regional progression, at the cost of increased acute oesophageal toxicity. 34 Preclinical studies have shown that both cisplatin and vinorelbine act as radiation enhancers. Cisplatin enhances the cytotoxic effects of radiation against tumor cells both in vitro and in vivo. 35 In addition, studies have shown that vinorelbine enhances the antitumor effects of radiation in vitro in a cell-cycle-dependent manner, with maximal effects when the cells are in the G2/M phase.36–38 A possible mechanism of vinorelbine-induced radiosensitization may at least in part be associated with impairment of DNA repair following radiation-induced DNA damage. 39
The cisplatin–-vinorelbine regimen was tested in a phase I trial to identify the maximum-tolerated dose (MTD) and dose-limiting toxicities (DLTs) of this regimen with concomitant thoracic radiation (TRT) in patients with advanced chest malignancies. Thirty-seven patients with advanced chest malignancies that required RT were enrolled onto this phase I study of standard chest radiation (30 daily 2-Gy fractions for a total of 60 Gy) and concurrent chemotherapy with cisplatin starting at 100 mg/m2 every 3 weeks and vinorelbine starting at 20 mg/m2/wk. Esophagitis was the DLT, with neutropenia occurring at higher dose levels. The recommended phase II dose was cisplatin 80 mg/m2 every 3 weeks and vinorelbine 15 mg/m2 given twice over 3 weeks on a day 1/day 8 schedule with concomitant chest RT. 40
In another phase I study, 18 patients received cisplatin (80 mg/m2) on day 1 and vinorelbine (20 mg/m2 in level 1, and 25 mg/m2 in level 2) on days 1 and 8 every 4 weeks for 4 cycles. TRT consisted of a single dose of 2 Gy once daily for 3 weeks followed by a rest of 4 days, and then the same TRT for 3 weeks to a total dose of 60 Gy. The recommended dose is the level 1 dose. 41
The cisplatin–-vinorelbine regimen in combination with thoracic radiotherapy was also tested in the context of phase II trials (Table 2).
Clinical phase II trials evaluating the combination of cisplatin and vinorelbine plus radiotherapy in locally advanced NSCLC.
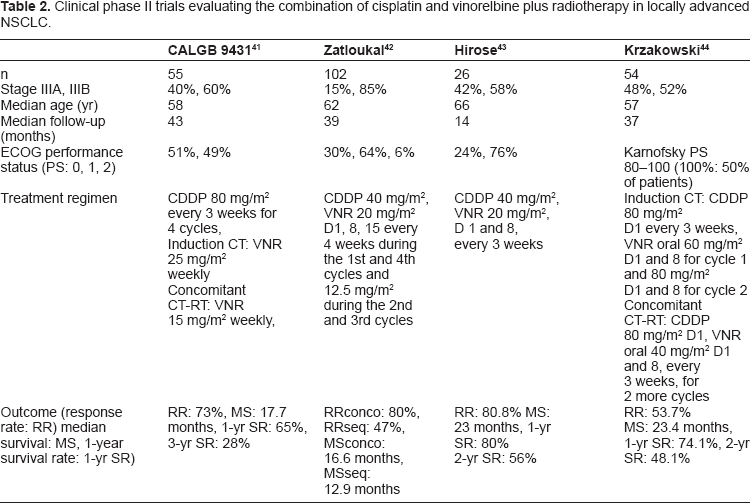
Cisplatin-based combinations with gemcitabine, paclitaxel or vinorelbine have been tested in a randomized CALGB phase II trial. 42 55 patients were randomized in the vinorelbine arm; cisplatin was administered for four cycles at 80 mg/m2 IV on days 1 and 22 (induction) and 43 and 64 (concomitant); vinorelbine was administered at 25 mg/m2 IV on days 1, 8, 15, 22, and 29 (induction chemotherapy) and at 15 mg/m2 IV on days 43, 50, 64, and 71 (concomitant chemoradiotherapy). Chest irradiation began on day 43 at 2 Gy/d (total dose, 66 Gy). After a median follow-up of 43 months, median survival was 17.7 (95% CI, 12.4–24.7) months. One-year survival rate was 65% (95% CI, 53%–78%), and 3-year survival rate was 23% (95% CI, 12%–34%). Grade 3 or 4 granulocytopenia was observed in 55% of patients during induction chemotherapy; other toxicities were mild to moderate. Tolerance was better with vinorelbine as compared to gemcitabine or paclitaxel during concomitant CT-RT: grade 3 or 4 granulocytopenia was seen in 27% of patients treated with vinorelbine. Only 13% and 12% of patients had grade 3 or 4 esophagitis. Grade 3 or 4 dyspnea as a possible measure of radiation pneumonitis was seen in 20% of patients. Radiation therapy interruptions were needed in seven of 55 patients.
Zatloukal et al compared the safety and efficacy of concurrent and sequential CT-RT, with CT consisting of a cisplatin and vinorelbine regimen, in 102 patients with locally advanced NSCLC. 43 The CT schedule consisted of up to four cycles of cisplatin 80 mg/m2 on day 1, and vinorelbine 25 mg/m2 at the first and fourth cycles (12.5 mg/m2 during the 2nd/3rd cycles) on days 1, 8, 15 of a 28-day cycle. Radiotherapy was prescribed at a dose of 60 Gy/30 fractions, given as five fractions per week for 6 weeks. In the concurrent arm (arm A), RT was started on day 4 of cycle 2; whilst in the sequential arm (arm B), RT started within 2 weeks after completion of CT. Overall survival was significantly longer in arm A (median survival 16.6 months) versus arm B (median survival 12.9 months) (HR = 0.61, 95% CI 0.39–0.93), and time to progression (TTP) was also significantly longer in arm A (11.9 months) versus arm B (8.5 months). The overall response rate was significantly higher in arm A, 80% compared with 47% in arm B (P = 0.001). WHO grade 3 or 4 toxicity was more frequent in arm A than in arm B, with a significantly greater incidence of leucopenia (53% versus 19%, P = 0.009) and nausea/vomiting (39% versus 15%, P = 0.044).
A Japanese team assessed the efficacy and toxicity of concurrent chemoradiotherapy with divided schedule of cisplatin and vinorelbine in patients with locally advanced NSCLC. 44 Twenty-six patients were enrolled. Both cisplatin (40 mg/m2) and vinorelbine (20 mg/m2) were given on days 1 and 8 every 3 weeks. Beginning on day 2 of chemotherapy, thoracic radiotherapy was given (2 Gy per fraction) for a total dose of 60 Gy. Patients achieved an overall response rate of 80.8% (95% CI, 60.6%–93.4%). The median survival time was 23 months. Overall survival rates at 1 and 2 years were 80 and 56%, respectively. Hematologic toxicities included grade 3–4 neutropenia in 84.6% of patients, grade 3–4 thrombocytopenia in 3.8%, and grade 3–4 anemia in 61.5%. Two patients (7.7%) had grade 3 radiation esophagitis that resolved completely without dilation. Grade 3–4 radiation pneumonitis occurred in two patients (7.7%) and was treated with corticosteroids. Both patients had a good partial resolution of symptoms and radiographic abnormalities. There were no treatment-related deaths.
Oral vinorelbine in combination with cisplatin and radiotherapy was also tested in locally advanced unresectable NSCLC. 45 54 patients were treated with oral vinorelbine 60 mg/m2 day 1 and 8 cycle 1 and 80 mg/m2 day 1 and 8 cycle 2 (if no grade 3–4 toxicity) and cisplatin 80 mg/m2 day 1 every 3 weeks for 2 cycles as induction. Patients without progression received oral vinorelbine 40 mg/m2 day 1 and 8 and cisplatin 80 mg/m2 day 1 every 3 weeks for 2 more cycles with radiotherapy 66 Gy in 6.5 weeks. Toxicities during induction were as follows: neutropenia grade 3–4 (28%), febrile neutropenia (7%), nausea grade 3 (11%), vomiting grade 3–4 (9%), anorexia grade 3 (4%), diarrhea grade 4 (2%), constipation grade 3 (2%). Forty-seven out of 54 (87%) patients received concomitant chemoradiotherapy. Median radiotherapy delivered dose was 66 Gy. Tolerance was good: neutropenia grade 3 (9%); dysphagia grade 3 (4%); radiation dermatitis grade 3 (2%). Late pulmonary fibrosis was reported in one patient (1.8%). One month after completion of CT-RT, the overall response rate intent-to-treat in the 54 patients was 54% (95% CI 40%–67%). With a median follow-up of 37 months the median progression-free survival and overall survival were: 12.5 (95% CI 9.6–16.4) and 23.4 (95% CI 17.6–29.8) months, respectively.
Concomitant chemo-radiotherapy for locally advanced NSCLC has become a standard of care. A combination of cisplatin and vinorelbine with optimal RT doses may be used safely with high rates of treatment completion, good efficacy and a favorable safety profile.
Metastatic Disease
Vinorelbine has been extensively tested in combination in metastatic NSCLC, most commonly with cisplatin. The combination of cisplatin and vinorelbine has proven to be one of the new standard regimens for metastatic NSCLC, with objective responses ranging from 25 to 44% and median survival times of 8.0–9.5 months.46–48 It is beyond the scope of this paper to review all the clinical studies which have been performed. We will concentrate on the most significant of these studies.
In a phase II trial, vinorelbine achieved a 29% overall objective response rate with a median survival time of 33 weeks. 49
In a large randomized study, cisplatin and vinorelbine showed a clear survival advantage over cisplatin and vindesine. 46 612 patients were included in this study: 206 on vinorelbine-cisplatin (VNR-CDDP) arm, 200 on vindesine-cisplatin (VDS-CDDP) arm, and 206 on vinorelbine (VNR) as monotherapy. Vinorelbine was administered at a dose of 30 mg/m2 weekly, cisplatin at 120 mg/m2 on days 1 and 29 and then every 6 weeks, and vindesine at 3 mg/m2 weekly for 6 weeks and then every other week. Treatment was continued until progression or toxicity. An objective response rate was observed in 30% of patients in the VNR-CDDP arm versus 19% in the VDS-CDDP arm (P = 0.02) and 14% in the VNR arm (P < 0.001). The median duration of survival was 40 weeks in the VNR-CDDP arm, compared with 32 weeks in the VDS-CDDP arm and 31 weeks in the VNR arm. Comparison of survival among the three groups demonstrated an advantage for VNR-CDDP compared with VDS-CDDP (P = 0.04) and VNR (P = 0.01). Neutropenia was significantly higher in the VNR-CDDP group (P < 0.001), and neurotoxicity was more frequent with VDS-CDDP (P < 0.004).
Following a first study showing that the combination of cisplatin and vinorelbine is a superior treatment when compared with single-agent cisplatin in the treatment of advanced NSCLC, 50 the vinorelbine-cisplatin doublet was then compared to paclitaxel plus carboplatin (TXL-CBDCA) in a landmark study by the Southwest Oncology Group. 47 Two hundred two patients received VNR-CDDP (vinorelbine 25 mg/m2/wk and cisplatin 100 mg/m2/d, day 1 every 28 days) and 206 patients received TXL-CBDCA (paclitaxel 225 mg/m2 with carboplatin area under the curve–-AUC–-of 6, day 1 every 21 days). The main end point of this trial was overall survival. The objective response rate was 28% in the VNR-CDDP arm and 25% in the TXL-CBDCA arm. Median survival was 8 months in both arms, with 1-year survival rates of 36% and 38%, respectively. Grade 3 and 4 leukopenia (P = 0.002) and neutropenia (P = 0.008) occurred more frequently on the VNR-CDDP arm. Grade 3 nausea and vomiting were higher on the VNR-CDDP arm (P = 0.001, P = 0.007), and grade 3 peripheral neuropathy was higher on the TXL-CBDCA arm (P < 0.001). More patients on the VNR-CDDP arm discontinued therapy because of toxicity (P = 0.001). No difference in QOL was observed. Overall costs on the TXL-CBDCA arm were higher than on the VNR-CDDP arm because of drug costs. 51
Scagliotti et al randomized 612 chemotherapy-naive patients to receive gemcitabine 1,250 mg/m2 days 1 and 8 plus cisplatin 75 mg/m2 day 2 every 21 days (Gem-CDDP arm), or paclitaxel 225 mg/m2 then carboplatin (AUC of 6 mg/mL·min), both on day 1 every 21 days (TXL-CBDCA arm), or vinorelbine 25 mg/m2/wk for 12 weeks then every other week plus cisplatin 100 mg/m2 day 1 every 28 days (VNR-CDDP arm). 48 The primary end point was in this trial the overall response rate which proved to be not significantly different among the three arms: Gem-CDDP 30%, TXL-CBDCA 32%, VNR-CDDP arm 30%. There were no differences in overall survival, time to disease progression, or time to treatment failure. Median survival for the Gem-CDDP, TXL-CBDCA, and VNR-CDDP groups was 9.8, 9.9, and 9.5 months, respectively. Neutropenia was significantly higher on the VNR-CDDP arm (Gem-CDDP 17% or TXL-CBDCA 35% versus VNR-CDDP 43% of cycles, P < 0.001), as was thrombocytopenia on the Gem-CDDP arm (Gem-CDDP 16% versus VNR-CDDP 0.1% of cycles, P < 0.001). Alopecia and peripheral neurotoxicity were most common on the TXL-CBDCA arm, as was nausea/vomiting on the VNR-CDDP arm (P < 0.05). The authors concluded that “these findings suggest that chemotherapy in NSCLC has reached a therapeutic plateau”.
Frail patients may be offered a non-platin doublet such as the vinorelbine–-gemcitabine 52 or vinorelbine–-paclitaxel 53 combination.
Recently, the availability of agents targeted against the EGF-R, 54 as well as the the anti-VEGF agent bevacizumab, 55 have provided some clinical benefit. The combination of cetuximab plus cisplatin/vinorelbine demonstrated an acceptable safety profile and the potential to improve activity over cisplatin/vinorelbine alone in a phase III clinical trial.56,57 However, this combination was not judged to be competitive enough to receive a formal approval by the European Medicines Agency. Clinical trials investigating the activity of targeted therapies in combination with chemotherapy are therefore eagerly awaited.
On the other hand, vinorelbine has been specially tested in the elderly population (Table 3). Quality of life (QoL) was explored in a first Italian trial which compared vinorelbine with supportive care alone. 58 Eligible patients were 70 years of age or older, had stage IV or IIIB NSCLC that was ineligible for radiotherapy, and had a performance status of 0-2. 191 of the 350 targeted patients were randomly assigned and data from 161 patients have been analyzed. Vinorelbine-treated patients scored better than control patients on QoL functioning scales, and they reported fewer lung cancer-related symptoms. There was a statistically significant survival advantage for patients receiving vinorelbine; median survival increased from 21 to 28 weeks in the vinorelbine-treated group. The relative hazard of death for vinorelbine-treated patients was 0.65 (95% CI, 0.45–0.93).
Randomised phase III clinical trials testing vinorelbine in the elderly population with advanced NSCLC.
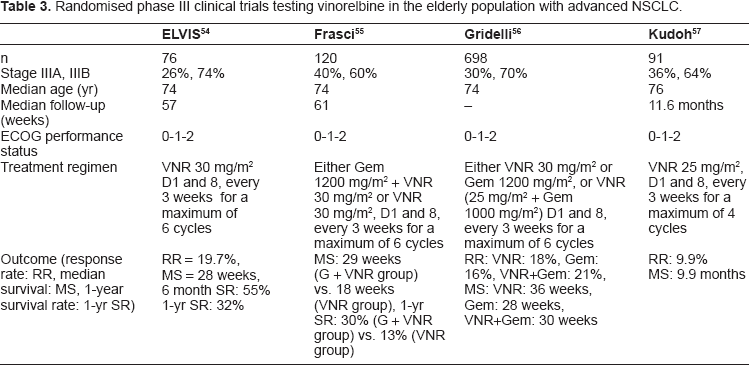
A second Italian study aimed to evaluate whether the addition of gemcitabine to vinorelbine improves survival, which was the primary end point, and QoL over vinorelbine alone among elderly patients with advanced NSCLC. 59 Patients with NSCLC aged > = 70 years with advanced disease were randomly allocated to receive vinorelbine 30 mg/m2 on days 1 and 8 every 3 weeks or gemcitabine 1,200 mg/m2 + vinorelbine 30 mg/m2 on days 1 and 8 every 3 weeks. In the doublet group, median survival time was 29 weeks and projected 1-year survival was 30%; these values were 18 weeks and 13% in the vinorelbine group. Combination therapy was also associated with a clear delay in symptom and QoL deterioration.
In a third study, Gridelli et al compared the effectiveness and toxicity of the combination of vinorelbine plus gemcitabine with those of each drug given alone in an open-label, randomized phase III trial in elderly patients with advanced NSCLC. 60 The primary end point was survival. Patients aged 70 years and older were randomly assigned to receive intravenous vinorelbine (30 mg/m2), gemcitabine (1200 mg/m2), or vinorelbine (25 mg/m2) plus gemcitabine (1000 mg/m2). All treatments were delivered on days 1 and 8 every 3 weeks for a maximum of six cycles. Of 698 patients available for intention-to-treat analysis, 233 were assigned to receive vinorelbine, 233 to gemcitabine, and 232 to vinorelbine plus gemcitabine. Compared with each single drug, the combination treatment did not improve survival. The hazard ratio of death for patients receiving the combination treatment was 1.17 (95% CI, 0.95–1.44) that of patients receiving vinorelbine and 1.06 (95% CI, 0.86–1.29) that of patients receiving gemcitabine. Although QoL was similar across the three treatment arms, the combination treatment was more toxic than the two drugs given singly.
A Japanese study randomised chemotherapy-naïve patients age 70 years or older between docetaxel or vinorelbine. 61 91 patients were allocated to the vinorelbine arm (25 mg/m2, days 1 and 8, every 21 days for four cycles). They obtained a median survival of 9.9 months.
Finally, a recent phase II trial confirmed that single-agent oral vinorelbine is extremely safe in elderly patients with advanced NSCLC and ECOG PS of two or more and may represent a valid option in this very special population. 62
Vinorelbine–-cisplatin combination is therefore one of the proposed and most active doublets for treating advanced, metastatic, NSCLC. 63 Vinorelbine alone is a valid option to propose to elderly or frail patients.
Conclusion
Despite recent advances in NSCLC treatment, clinical outcome of these patients still remains a challenge. A cisplatin-based doublet is recommended. The cisplatin-vinorelbine combination is among the most widely used association. It is definitely recommended as the most useful combination in the situation of post-operative adjuvant chemotherapy. A combination of cisplatin and vinorelbine with concomitant radiotherapy is a valid option for patients presenting with locally advanced, inoperable stage III NSCLC. For patients with metastatic NSCLC, vinorelbine-cisplatin doublet is one of the most active combination. The new oral formulation of vinorelbine is especially indicated for elderly patients or patients with performance status of 2 who may benefit from a single cytotoxic drug. Clinical trials that investigate the activity of targeted therapies in combination with conventional chemotherapy drugs are eagerly awaited.
Disclosure
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
