Abstract
Vinorelbine is a semi-synthetic vinca alkaloid that has been shown active in many tumor types and is currently registered for the treatment of advanced breast cancer (ABC) and non-small cell lung cancer (NSCLC), and it is used in other cancer type such as prostate cancer, lymphomas, gynecologic malignancies. This agent has a generally favorable safety profile, and may be suitable for use in special populations such as the elderly and/or frail patient. However, with the taxanes established as standard second line treatment for ABC after failure of an anthracycline, Vinorelbine has been generally relegated for use as third line therapy, in competition with the oral compound Capecitabine. Combination regimens of Vinorelbine and Gemcitabine have been indeed widely studied, and have been demonstrated to be safe and effective. More recently, the exciting results observed with the combination of Vinorelbine and Trastuzumab in patients with Her-2 overexpressing tumors, as well as the development of a reliable formulation and revised schedule of oral Vinorelbine appear to have revived the interest in this compound in the management of this disease. Yet, data from properly designed randomized clinical trials are eagerly awaited to confirm the amount of available data which are mostly based on phase II studies.
Introduction
Despite the recent trend toward treatment of early stage breast cancer aggressively with anthracyclines and taxanes, nearly half of those women will have metastatic recurrence. Advanced breast cancer (ABC) generally responds to chemotherapy yet, given the usual prior exposure to these drugs, many women facing first-line therapy for recurrent disease will now have developed anthracycline- and taxane-refractory metastatic breast cancer, presenting a major therapeutic challenge. A number of established drugs have proved to be effective in this setting: Capecitabine; Gemcitabine; Vinorelbine; and new drugs are currently being developed, such as camptothecins, epothilones, mitosis inhibitor E7389, the ascidian-derived anticancer compound trabectedin and others.
ABC is however an incurable disease, with a median survival of 2-3 years, with response rates to monotherapy ranging from about 20%-75% in chemotherapy-naïve patients. 1 Moreover, the quantity and quality of the available evidence regarding the efficacy of currently used drugs in patients with locally advanced or metastatic breast cancer previously treated with both a taxane and an anthracycline are extremely limited. 2
Vinorelbine (5‘-noranhydrovinblastine) is a semisynthetic vinca alkaloid, from the Catharanthus alkaloid family. The clinical development of Vinorelbine started in the 1980s and, although a derivative of Vinblastine, Vinorelbine is structurally different, with an 8-membered ring as opposed to the 9-membered ring characteristic of naturally occurring vinca alkaloids.
These structural modifications give highly specific antimitotic properties to Vinorelbine, as well as enhanced lipophilicity.
Similarly to other vinca alkaloids, Vinorelbine induces cytotoxic effects by inhibiting polymerization of tubulin dimers into microtubules, disrupting mitotic spindle formation and preventing cell division. Thus, its activity is cell cycle specific. Vinorelbine is selective for mitotic microtubules; it has minimal to no activity (except in higher concentrations) on axonal or other microtubule classes, with a reduction in the frequency and severity of neuro-toxicity, typically observed with other microtubule agents. 3 Vinorelbine is extensively metabolized in the liver through cytochrome P450 iso-enzymes of the CYP3A subfamily. Biliary excretion is the predominant route of elimination, accounting for at least 50% of an intravenous dose. Extensive biliary excretion of metabolites also appears to occur.4,5 The elimination half-life after intravenous administration is 42 to 45 hours and 24 to 56 hours after oral administration.5,6 Pharmacokinetics in elderly patients (older than 65 years) is similar to those reported in younger people. 7
This report reviews the most important studies involving Vinorelbine in ABC with particular regard to the results of randomized trials in order to evaluate its role in this subset of patients.
Vinorelbine represents both an active and a well tolerated treatment for metastatic breast cancer in patients who display anthracycline and/or taxanesrefractory disease, or who are not suitable for such treatments. Some concerns, though, have been raised about a potential cross-resistance between taxanes and Vinorelbine, and contradictory evidence exists on the use of Vinorelbine in patients with disease refractory to taxanes. In a phase II trial, no responses were seen with Vinorelbine among 14 patients with ABC refractory to taxanes, 8 whereas other studies indicate possible responses in patients treated with Vinorelbine for ABC judged taxane-refractory.9–11 In the next paragraphs single agent activity and combinations regimens with Vinorelbine are discussed, as well as combinations with targeted therapy, namely Trastuzumab.
Vinorelbine: Single Agent Activity
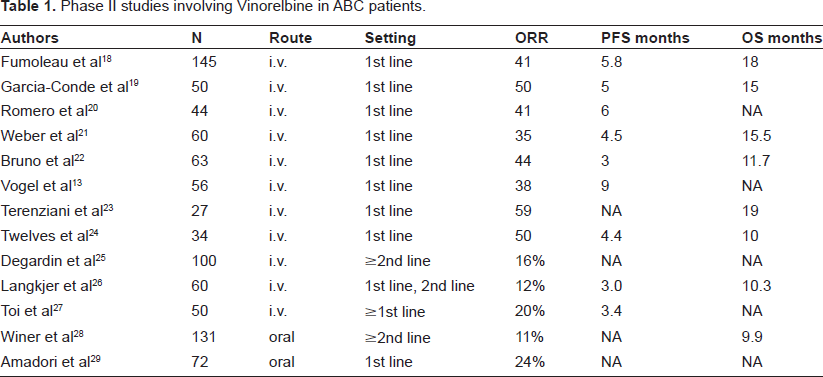
Many phase II studies have demonstrated the activity and tolerability of intravenous administration of Vinorelbine, with response rates ranging from 40%-60% in chemo-naïve disease and with good toler-ability, 12 Table 1. Vinorelbine rarely induces total alopecia or severe gastrointestinal events or symptomatic cardiac events. The major dose-limiting hematological toxicity associated with Vinorelbine is neutropenia. Common non-hematological side-effects include G1-G2 peripheral neuropathy, constipation and phlebitis.
Phase II studies involving Vinorelbine in ABC patients.
Because of its clinical activity and its mild toxicity profile and, in particular, the absence of alopecia could be particularly attractive for women.
Indeed, for all the above mentioned reasons, Vinorelbine as single agent represents a valid alternative in the treatment of metastatic breast cancer in elderly patients.
Sorio and colleagues demonstrated that Vinorelbine administered intravenously at the dose of 30 mg/m2 on days 1 and 8 every 3 weeks was well tolerated. Responses were observed in 6 out of 20 evaluable patients (ORR 30%) and no correlation between dosage, age and toxicity was observed. 7 In another study, Vinorelbine was administered weekly at the same dosage (30 mg/m2). The dose-limiting toxicity observed was hematologic, easily recovered with dose adjustment. Asthenia, gastroenteric toxicity and injection site reactions were the predominant non-hematologic adverse events. Neurotoxicity and alopecia were relatively infrequent and mild.13–15
Only two randomized phase III trials are available testing Vinorelbine single agent,16,17 Table 2. Vinorelbine given with schedule 30 mg/m2 (days 1 and 8) 16 has been shown to be as effective as single agent as in the combination with Gemcitabine as for overall survival, whereas the combination significantly improves progression-free survival (PFS), when toxicity was significantly higher in the combination arm.
Phase III randomized studies involving single agent vinorelbine in ABC patients.
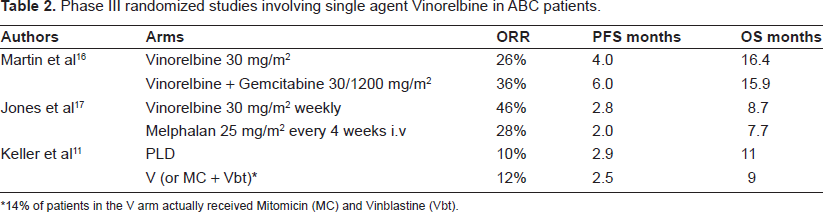
14% of patients in the V arm actually received Mitomicin (MC) and Vinblastine (Vbt).
The study by Jones and colleagues explored Vinorelbine with schedule 30 mg/m2 weekly, 17 showing a survival benefit in anthracycline-refractory ABC with use of Vinorelbine versus Melphalan, with comparable toxicity and quality of life.
In a more recent trial performed in a population of 301 women with ABC who had failed a taxane as first or second line therapy, Vinorelbine (which was the chosen comparator in 85% of the cases; n = 129) was shown equivalent to Pegylated Liposomal Doxorubicin (PLD), a novel Doxorubicin formulation with comparable efficacy and better safety profile. 11 Only the subgroup of patients without previous exposure to anthracyclines (17% of the study population) had a somewhat better PFS with PLD.
Combination Regimens with Vinorelbine
One of the virtues of Vinorelbine is the fact that, in view of its mild toxicity profile, it can be easily combined with virtually any other cytotoxic. A large number of phase II trials have been performed which have generally shown encouraging activity with an acceptable safety profile (Table 3). Examples include Paclitaxel30–37 Docetaxel;38–46 Doxorubicin;47–51 Epirubicin52–54 Mitoxantrone;55–57 Pegylated Liposomal Doxorubicin;14,58–61 Gemcitabine;62–73 5-Fluorouracil;74–79 platinum compounds;80–83 and Capecitabine,84–88 which will be separately discussed. Several triplets have also been investigated but increased toxicity should be expected.89–92
Combination regimens with Vinorelbine.
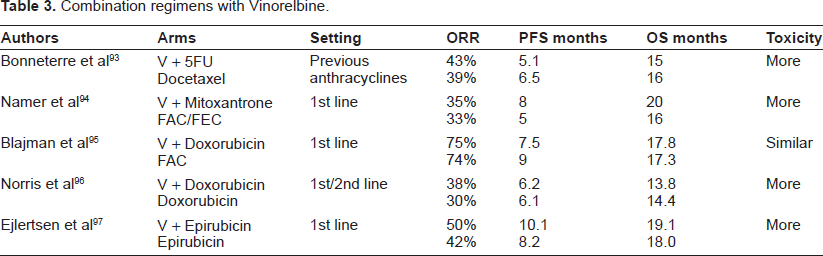
Some of these combinations were eventually investigated in phase III trials, and will be discussed in more detail in the following section.
The combination of Vinorelbine and 5-Fluorouracil, one of the most extensively studied in patients with ABC, was compared to Docetaxel in a small randomized phase III trial (sample size 176 patients). 93 Although there were no differences in terms of efficacy, the combination resulted in more toxicity, with also toxic deaths registered, as compared to single agent Docetaxel. Yet, it must be noticed that high doses of 5-Fluouracil (750 mg/m2/day for 5 days) have been administered in this trial. Randomized trials have evaluated the addition of Vinorelbine to an anthracycline such as Mitoxantrone, 94 Doxorubicin,95,96 and Epirubicin 97 against standard anthracycline arms. Only one of these trials suggested an advantage for the combination in terms of response rates and progression-free survival, at the price of higher toxicities for the combination arms. Generally, toxicities for the combination arms were statistically increased in the combination arms in trials comparing single agent versus combination with Vinorelbine.
These results are in line with those of other randomized clinical trials in ABC, showing that combination chemotherapy is usually not superior to optimal sequential treatment, and is likely to result in more toxicity, therefore the use of combination chemotherapy regimens in advanced settings should be limited to those patients requiring rapid shrinking of life-threatening tumors.
Vinorelbine and Capecitabine, Phase II Studies
Vinorelbine is currently occupying a delicate place in the management of ABC especially for third line therapy after failure of anthracyclines and taxanes, in open competition with the oral compound Capecitabine, an oral fluoropyrimidine carbamate which is enzymatically converted via a 3 enzymatic step to 5‘-deoxy-5-fluorocytidine (5‘-DFCR), 5‘-deoxy-5-fluorouridine (5‘-DFUR), and finally 5-FU. To date, no randomized phase III trial comparing directly Vinorelbine vs. Capecitabine has been performed. The limited data coming from a phase II study 98 and retrospective data sets99,100 show similar activity of the two agents or, in the case of retrospective data, higher activity for Capecitabine. In particular, safety profiles are different, with more G3-G4 hematological toxicity for Vinorelbine, and more grade 3 cutaneous toxicity for Capecitabine. In addition, Capecitabine and Vinorelbine have been compared in a pharmacoeconomic analysis performed in the United States. Mean monthly cost for patients with advanced breast cancer was higher for patients receiving Vinorelbine when compared to matched patients receiving Capecitabine and the main difference in cost was dictated by management of treatment related toxicity and chemotherapy i.v. administration. 101
Capecitabine is an attractive combination partner since it displays a high single agent activity and a non overlapping toxicity profile, with only minimal myelodepression and no alopecia. Moreover, there is a synergistic effect due to Vinorelbine mediated up-regulation of thymidine phosphorilase (TP), which has a crucial role in the 3-step conversion of Capecitabine to 5-FU at tumor site. 102
The combination of Capecitabine and Vinorelbine has been investigated in a variety of phase I studies and phase II studies, which are illustrated in Table 4.
Phase II studies in first and subsequent lines of Vinorelbine and Capecitabine for ABC.
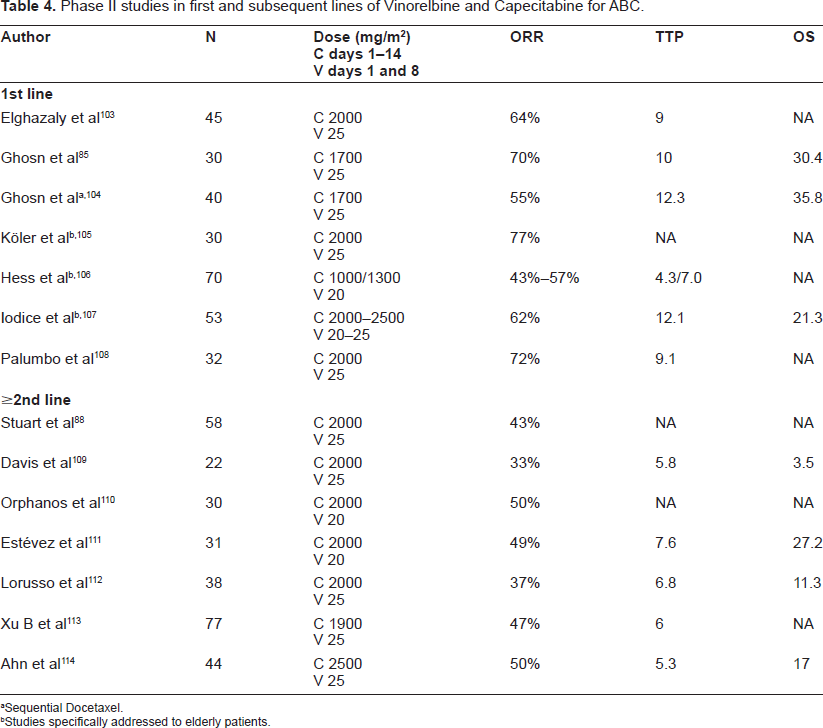
Sequential Docetaxel.
Studies specifically addressed to elderly patients.
Recently, an oral formulation of Vinorelbine has become available. Some studies also evaluated an all oral combination of Vinorelbine and Capecitabine, which could represent an attractive option in particular subsets of patients, such as elderly patients.
Oral Vinorelbine
Among the options for treatment of anthracycline- and taxanes-pretreated advanced breast cancer, Capecitabine has been generally preferred by patients because of the convenience of the oral route, but recently a novel oral formulation of Vinorelbine (ONVB) which has apparently overcome the problem of erratic absorption has become available. In patients with NSCLC, oral and intravenous Vinorelbine have been shown to have comparable activity. 115
In pharmacokinetic studies, equivalence in bioavailability has been demonstrated between the oral and intravenous formulation. 116 Oral Vinorelbine is rapidly absorbed, with a Tmax of 0.75-1.4 hours.116,117 Bioavailability of soft-gelatine capsules, the third and most recent oral formulation of Vinorelbine, is 33%-43%,116,117 with 38% inter-individual variability. 116 Low bioavailability may be attributed to both incomplete absorption and to a first-pass effect (intestinal and hepatic). The problems with hematologic toxicity observed in earlier trials, 118 have been managed by administering the first course of ONVB at the dose of 60 mg/m2 days 1 and 8, and only if no toxicity is observed escalating to a dose of 80 mg/m2 days 1 and 8. This helped reducing the severe toxicity which was typically observed at first cycle, with the occurrence of even toxic deaths in the earlier trials. 118 This schedule has demonstrated moderate efficacy but is characterized by a high rate of neutropenia, the risk of which increases after dose escalation to 80 mg/m2. 119 Consequently close hematological monitoring is essential. 118 Furthermore, differently from intravenous formulation, oral Vinorelbine has been shown to be associated with frequent nausea, vomiting and diarrhea,118,119 and prophylactic antiemetics are recommended. 118 Nonetheless, oral formulation may be attractive for patients not willing to be treated with i.v. drugs and for elderly patients, a subset of patients in which it has been demonstrated fair safety of this formulation, though activity raises concerns. 120
Although experience with oral Vinorelbine is less extensive than with the intravenous formulation, the pivotal phase II trial that has investigated the new schedule in patients with ABC resulted in response rates of 31% and median duration of response of 4 months in previously untreated patients. 119 Furthermore, it has been shown that ONVB brings added value in terms of patients’ convenience allowing improved quality of life and reduced hospital stays while keeping optimal compliance to treatment schedule in a French experience. 121
All oral combinations of Vinorelbine and Capecitabine have been investigated in phase I and II studies, with overall response rates ranging from 26%-61%, median progression-free survival 6-10.5 months and median overall survival 10-48 months.87,122
In particular, when used in first line settings, response rates are in the range of 44%-51% with median progression free survival of 8.4 months.86,123 Among the major benefits for these combination regimens is the possibility of continuing therapy for prolonged periods in patients with controlled disease, whereas this would not be possible for anthracycline and/or taxanes containing regimens due to cumulative toxicity.
Recently, new metronomic schedules have been proposed, which can be appealing for the low toxicities and manageable side effects that render the schedule easily applicable to vulnerable populations such as elderly patients oral Vinorelbine 80 mg/m2 fractionated in days 1, 3, and 5, three week on-one week off, every 4 weeks. ORR 41%, with median time to disease progression was 7.1 months and median overall survival was 12.7 months. 124
Vinorelbine and HER2 Positive Disease
Trastuzumab, a 95% humanized monoclonal antibody targeted against the extracellular domain of the HER-2 receptor, is the standard of care for patients with HER2 over-expressing tumors (about 20% of breast carcinomas) in association with chemotherapy. In the pivotal trials of single agent Trastuzumab, response rates of 15% and 30% have been reported in previously treated 125 and untreated 126 patients. Trastuzumab is frequently combined with a taxane, however in patients who are not suitable for therapy with a taxane (e.g. prior exposure to taxanes, pre-existing peripheral neuropathy, patient's choice depending on potential side effects), Vinorelbine has been demonstrated an effective option.
In the preclinical setting, synergism between Trastuzumab and Vinorelbine has been demonstrated. 127 Vinorelbine and Trastuzumab have no overlapping toxic effects, which contrast with the use of Trastuzumab in combination with anthracyclines. 128
A phase I dose-finding study evaluated escalating doses of Vinorelbine in 12 patients. 129
Vinorelbine was given at doses of 20, 25 and 30 mg/m2 in successive cohorts, on a day 1 and 8 schedule every 21 days, in combination with Trastuzumab. Toxic effects were primarily hematological; one case of febrile neutropenia and one episode of acute cholecystitis were noted among six assessable patients receiving Vinorelbine 30 mg/m2 and this was therefore determined as the maximum tolerated dose.
Several phase II trials (Table 5) and one phase III trial 130 have now reported data for Trastuzumab in combination with Vinorelbine in the HER2-positive MBC setting. The trials have included >450 assessable patients, the majority (94%) having HER2-positive status, defined as an immunohistochemistry (IHC) score of 3+ or by FISH. In particular, in the phase III trial, 130 the Vinorelbine-Trastuzumab combination showed similar efficacy to the weekly taxanes-Trastuzumab combination, yet the trial was prematurely closed due to slow accrual.
Trials with combination of Trastuzumab and vinorelbine.
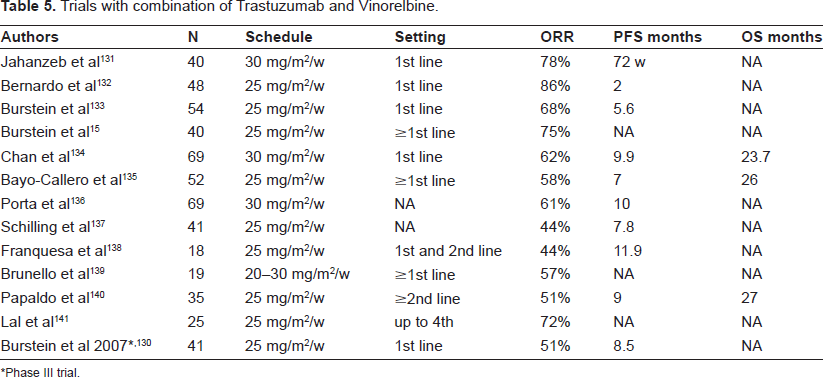
Phase III trial.
On the contrary of trials of combination taxanes-Trastuzumab, the studies of combination Vinorelbine-Trastuzumab demonstrate a more favorable therapeutic index, with rates of cardiomiopathy not higher than those reported with Trastuzumab single agent. Although it would be inappropriate to consider the combination Trastuzumab-Vinorelbine as standard treatment for patients with Her-2 overexpressing ABC in the absence of controlled trials, this schedule may be considered an acceptable option for patients willing to avoid the side effects associated with taxanes (such as alopecia), or for selected patients relapsing after previous exposure to taxanes, a situation likely to be more common with the increasing use of taxanes in the (neo-)adjuvant setting.
Schedules with three-weekly Trastuzumab have been studied as well, 142 with manageable toxicity and activity which falls in the range of expected response rates for weekly schedules, though at the lower limits.
The oral formulation of Vinorelbine has also been investigated in association with Trastuzumab for HER2 overexpressing tumors.
An observational study 143 demonstrated that the combination is an effective and safe treatment option for HER2 positive ABC, with response rates and time to progression depending mainly on line of treatment; a schedule which consists of intravenous Vinorelbine on day 1 and oral Vinorelbine on day 8 and Trastuzumab has also been studied 144 and results showed high antitumor efficacy, with overall response rate 62%, and excellent tolerability in 1st-line treatment of HER2 overexpressing ABC.
Conclusions
The increasing evidence that anthracyclines and taxanes improve the survival of patients with breast cancer in the adjuvant setting has led to a restricted use of these drugs in women with metastatic disease. 145 Combination regimens of Vinorelbine with anthracyclines or taxanes have been demonstrated to have same efficacy of the single agent (anthracycline or taxanes), in some cases improving the overall response rate, but usually at the price of increased toxicity that does not justify the use of such combinations. On the contrary, association with newer drugs such as Gemcitabine or Capecitabine have shown increased efficacy over the single agent, with manageable toxicity profile. Although Vinorelbine is clearly an active and well tolerated agent in BC, its use is currently limited by the lack of properly designed controlled trials. Generally, Vinorelbine as a single agent is not recommended in patients who are anthracycline naïve though it may be considered a reasonable option for patients willing to prioritize quality of life over maximum chance of tumor response. Single agent Vinorelbine or combinations of Vinorelbine and Capecitabine can be considered an alternative to the use of anthracyclines and taxanes in special populations of patients, such as the elderly and/or frail patients or in those willing to avoid alopecia and/or to prioritize other aspects of quality of life. The new oral formulation of Vinorelbine is particularly indicated for patients with disease characterized by low aggressiveness with or without visceral metastasis, or slowly progressing disease. In addition, recently Vinflunine, a novel fluorinated Vinca alkaloid, has become available and is currently under study for treatment of breast cancer progressing after anthracycline- and taxane-based chemotherapy.
The availability of Vinorelbine and other novel, well tolerated and effective treatments provides greater potential to tailor treatment to meet individual patient needs and, therefore, also provide the potential to improve patient outcomes.
Disclosures
The authors report no conflicts of interest.
