Abstract
Background:
Clinical trials investigating the effects of beta-blockers (BBs) on cancer are underway. Evidence from preclinical research suggests that BBs could serve as anticancer agents and immune boosters. There is conflicting evidence regarding the effect of BB use on clinical outcomes in patients with breast cancer.
Objectives:
The study aimed to determine whether BB use is associated with progression-free survival (PFS) and overall survival (OS) in patients receiving anti-human epidermal growth factor receptor 2 (HER2) treatment for advanced breast cancer.
Design:
Retrospective hospital-based study.
Methods:
The participants enrolled were breast cancer patients with advanced HER2-positive status who initiated trastuzumab monotherapy or concomitant therapy with trastuzumab and any dose of BB. The patients were enrolled between January 2012 and May 2021 and divided into three groups based on whether they received a BB or not in the therapeutic regimen: BB−/trastuzumab+, BB+ (non-selective)/trastuzumab+, and BB+ (selective)/trastuzumab+. PFS and OS were the primary and secondary endpoints, respectively.
Results:
The estimated median PFS in the BB−/trastuzumab+, BB+ (non-selective)/trastuzumab+, and BB+ (selective)/trastuzumab+ groups was 51.93, 21.50, and 20.77 months, respectively. The corresponding OS was 56.70, 29.10, and 27.17 months. The intergroup differences in these durations were significant. Both PFS [adjusted hazard ratio (HR): 2.21, 95% confidence interval (CI): 1.56–3.12; p < 0.001]) and OS (adjusted HR: 2.46, 95% CI: 1.69–3.57; p < 0.001) were worse when BBs were used.
Conclusion:
Our study provides important evidence that BB use potentially has a negative effect on patients with HER2-positive advanced breast cancer. Nevertheless, despite the study’s results, cardiovascular disease (CVD) should be appropriately treated in patients with HER2-positive advanced breast cancer. Other types of drugs can be used to treat CVD, but BB use should be avoided. Large real-world database and prospective studies should be conducted to validate the results of this study.
Plain language summary
• Evidence from preclinical research suggests that beta-blockers (BBs) could serve as anticancer agents and immune boosters.
• Beta-blockers could therefore be a potential therapy for cancers.
• Trastuzumab is a drug that affects the overall survival (OS) and progression-free survival (PFS) of patients with HER2-positive breast cancer by binding to the extracellular domain of HER2.
• This study investigates the effect of BBs on trastuzumab therapy in patients with advanced breast cancer.
• This retrospective study was conducted between January 2012 and May 2021.
• Patients with HER2-positive advanced breast cancer who were treated using trastuzumab monotherapy or trastuzumab concomitantly with any dose of a BB were recruited and divided into three groups.
• One group received only the trastuzumab (BB−/trastuzumab+), another group received both BB+ (non-selective) and trastuzumab [BB+ (non-selective)/trastuzumab+], and the third group received both BB+ (selective) and trastuzumab [BB+ (selective)/trastuzumab+].
• The PFS and OS were determined and compared between the treatment groups.
• We enrolled 221 patients (mean age: 56.1 ± 11.1 years) in the study.
• The estimated median PFS and OS were significantly lower in the BB+ (non-selective)/trastuzumab+ and BB+ (selective)/trastuzumab+ groups than in the BB−/trastuzumab+ group.
• The use of BBs was associated with worse PFS and OS in patients with HER2-positive advanced breast cancer.
• Trastuzumab treatment was independently associated with poorer PFS and OS for patients who used BB prior to initiating trastuzumab therapy for advanced HER2-positive breast cancer.
• BB use potentially has a negative effect on patients with HER2-positive advanced breast cancer.
• Future studies with larger sample sizes are needed to validate our findings.
Introduction
Breast cancer is the most common malignancy diagnosed globally, with more than 2 million cases reported each year. 1 Among women in the United States, breast cancer is the most common and second-leading cause of cancer-related death. 2 Approximately 20% of the affected patients show overexpression of the human epidermal growth factor receptor 2 (HER2). Breast cancer patients with HER2 amplification are at a higher risk for metastasis than other affected patients. 3 Trastuzumab affects the overall survival (OS) and progression-free survival (PFS) of patients with HER2-positive breast cancer by binding to the extracellular domain of HER2.4,5 Anti-HER2 therapies are generally well tolerated; however, they can also cause cardiotoxicity, which worsens when anthracyclines are added to the treatment regimen.6,7 Clinical trials investigating the effects of beta-blockers (BBs) on cancer are underway. Evidence from preclinical research suggests that BBs could serve as anticancer agents and immune boosters.8,9 BBs may inhibit catecholamines released by the sympathetic nervous system in response to stress, which regulate cell survival, proliferation, and motility. 10 β2 Receptor stimulation may lead to resistance to anti-HER2 therapies; therefore, BBs could result in re-sensitization to anti-HER2 treatments. 11 There is conflicting evidence regarding the effect of BB use on clinical outcomes in patients with breast cancer. Studies have suggested that the use of BBs in patients with breast cancer can reduce the risk of recurrence or mortality12–16; however, some other related studies have suggested the contrary.10,17–22 Moreover, some meta-analyses have been conducted to determine whether the use of BB affects the prognosis of women with breast cancer; nevertheless, the results of these meta-analyses were also inconsistent.23–30 Pooled analyses of the clinical trial data published in 2020 for patients initiating anti-HER2 therapy for advanced breast cancer and a sensitivity analysis of patients with preexisting cardiovascular disease (CVD) both showed that BB use was associated with a worse OS rate. 30 In 2022, a population-based cohort study and meta-analysis included 30,060 breast cancer patients assessing the association between BB use and survival. When stratified by molecular subtype, the use of BB was associated with prolonged breast cancer-specific survival only in triple-negative breast cancer patients. 22 Thus, in this study, we aimed to determine whether BB use is associated with PFS and OS in patients receiving anti-HER2 treatment for advanced breast cancer.
Material and methods
Study design and patient population
This retrospective cohort study was conducted from January 2012 to May 2021 at a regional teaching hospital in Taiwan. The inclusion criteria were as follows: (1) patients first diagnosed with breast cancer with International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) codes C50.0–C50.9; (2) patients with HER2-positive advanced breast cancer with who were treated using trastuzumab monotherapy or trastuzumab concomitantly with any dose of a BB (propranolol, carvedilol, atenolol, or bisoprolol); (3) patients with cytologically or histologically confirmed stage IV breast cancer; (4) patients with a left ventricular ejection fraction of ⩾50%; (5) patient’s physical functional status at the beginning of treatment was according to the Eastern Cooperative Oncology Group (ECOG) performance status (PS) rating ⩽2 (ECOG PS of 0: normal with no limitations; 1: not my normal self, but able to be up and about with fairly normal activities; 2: not feeling up to most things, but in bed or chair less than half the day; 3: able to do little activity and spend most of the day in bed or chair; 4: pretty much bed-ridden, rarely out of bed; 5: dead.). Patients are generally considered not suitable for chemotherapy if their ECOG PS rating is >2; and (6) patients aged >20 years. Patients whose electronic medical records were incomplete and patients with a history of cancer were excluded.
Non-selective and selective BB exposure
The patients were divided into three groups: BB−/trastuzumab+ (BB non-user), BB+ (non-selective)/trastuzumab+ (non-selective BB user), and BB+ (selective)/trastuzumab+ (selective BB user). The BB non-user group consisted of patients who were neither on non-selective BB nor selective BB therapy (control group). The non-selective BB user group comprised patients who received non-selective BB (propranolol or carvedilol) therapy within 90 days prior to trastuzumab treatment. Finally, the selective BB user group included patients who received selective BB (atenolol or bisoprolol) therapy within 90 days prior to trastuzumab treatment.
Statistical analysis
PFS was the primary endpoint and OS was the secondary endpoint of this study. PFS was defined as the interval between trastuzumab initiation to disease progression [progression assessed by the physicians using the Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 criteria and recorded in the progression note] or death, whereas OS was defined as the interval between trastuzumab initiation and death. The day of trastuzumab initiation was designated as the index date. Patients were followed-up until an endpoint was reached or until 31 May 2022.
The pre-treatment characteristics available included BB use status, age, ECOG PS, number of metastases, existence of brain metastasis, estrogen/progesterone receptor (ER/PR) status, Ki-67 expression, and progression note recoding of hypertension and CVD includes coronary artery disease, arrhythmia, stroke, and heart failure. These were analyzed by performing univariate and adjusted analyses.
Chi-squared test was used to compare categorical variables and one-way analysis of variance (ANOVA) was used to compare continuous variables among the three groups. A post hoc Bonferroni test was conducted for pairwise comparison of the groups. A Cox proportional hazard regression model was used to calculate hazard ratios (HRs) and 95% confidence intervals (CIs). Multivariable cox proportional hazard regression model was adjusted for age, ECOG PS, number of metastases, and brain metastases. A proportional hazard assumption was evaluated by the Kolmogorov-type Supremum Test, which was not violated. All statistical analyses were conducted using SPSS v28 (IBM Corp., Armonk, NY, USA) and SAS v9.3 (SAS Institute Inc., Cary, NC, USA) statistical software. P Values <0.05 were considered statistically significant for all analyses. In this study, the Cox regression model was devised with a 0.61 overall probability of death based on a sample of 221 observations, which resulted in more than 99% power to detect a regression coefficient of 2.66 at a 0.05 significance level.
Results
Between January 2012 and May 2021, 4259 patients were diagnosed with breast cancer and categorized as C50.0–C50.9 per the ICD-10-CM system. Of these, 320 were HER2 positive and used trastuzumab treatment. Overall, 221 women met the study’s eligibility criteria and were enrolled in the study. The mean patient age was 56.1 ± 11.1 years (Figure 1). The BB non-user, non-selective BB user, selective BB user groups comprised 127 (57.47%), 51 (23.08%), and 43 (19.45%) patients, respectively. The BBs used were propranolol (n = 46), carvedilol (n = 5), and bisoprolol (n = 43). Brain metastases were observed in 10.41% (n = 23) of the patients, and 97.74% (n = 216) had an ECOG PS score of 0–1 at the beginning of trastuzumab treatment. Furthermore, 41.63% (n = 92), 25.79% (n = 57), and 32.58% (n = 72) of the patients had ER and PR positive, ER or PR negative, and ER and PR negative statuses, respectively. Preexisting comorbidities at the start of trastuzumab therapy were CVD (3.17%, n = 7) and hypertension (23.53%, n = 52). Age, ER, and PR status were the only baseline characteristics exhibiting significant differences among the three groups (Table 1).
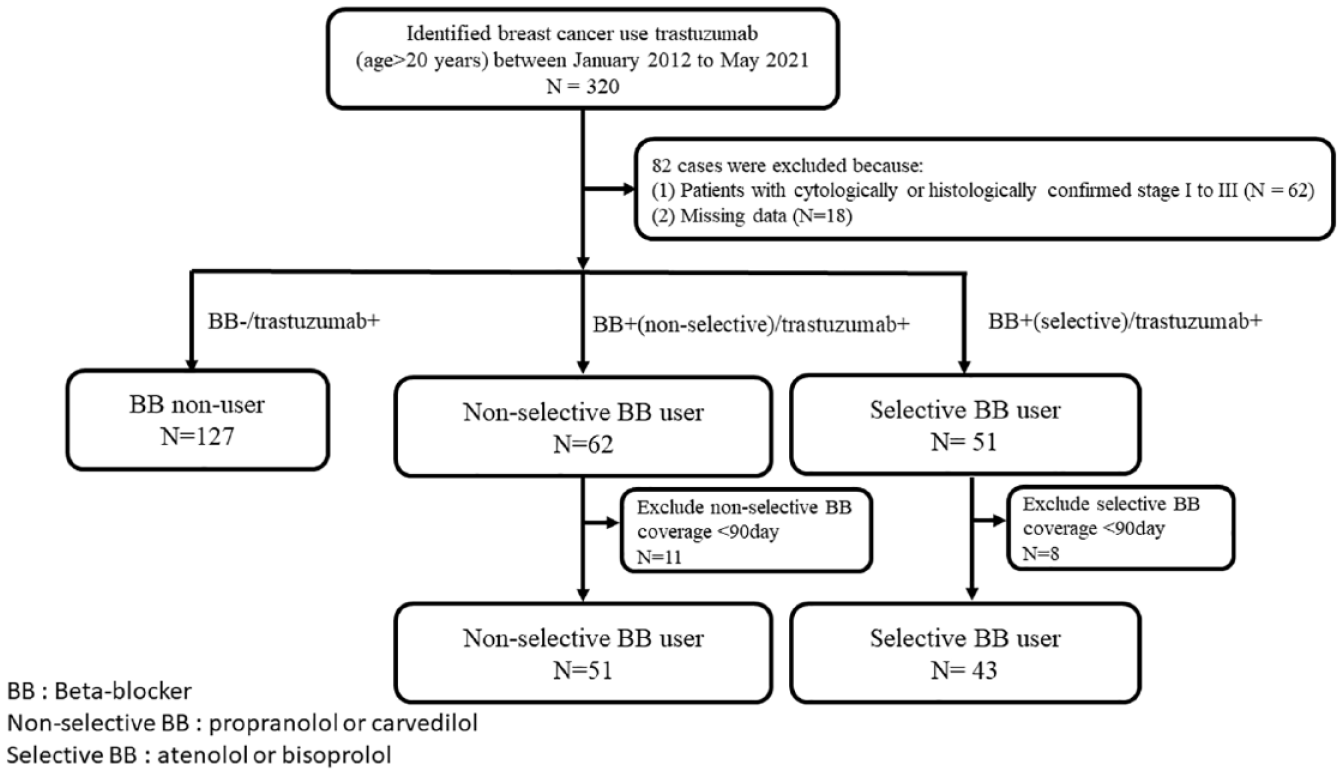
Study flow diagram.
Characteristics of the study population.

Categorical variable: chi-square test; continuous variable: one-way ANOVA. Different superscript alphabets within the column indicate statistically significant differences between groups at p < 0.05 by post hoc Bonferroni test.
ANOVA, analysis of variance; BB, beta-blockers; CVD, cardiovascular diseases; ECOG, Eastern Cooperative Oncology Group; ER, estrogen receptor; HT, hypertension; PR, progesterone receptor; PS, performance status; Q1, first quartile; Q3, third quartile; SD, standard deviation.
Primary endpoints
In all, 151 (68.33%) of the 221 patients exhibited disease progression or died at the end of the study. The estimated median PFS of the entire cohort was 39.3 months [95% CI: 32.84–45.76; first quartile (Q1)–third quartile (Q3): 18.1–65.0]. Moreover, the estimated median PFS for the BB non-user, non-selective BB user, and selective BB user groups was 51.93 (95% CI: 48.47–55.40; Q1–Q3: 31.2–71.9), 21.50 (95% CI: 14.94–28.06; Q1–Q3: 13.5–42.6), and 20.77 (95% CI: 17.21–24.33; Q1–Q3: 11.9–28.7) months, respectively. Kaplan–Meier analysis showed a significant difference (log rank test: p < 0.001) in PFS rates among the three groups (Figure 2). Subgroup analysis revealed significant differences between the BB non-user and non-selective BB user groups (log rank test: p < 0.001) and between the BB non-user and selective BB user groups (log rank test: p < 0.001). The PFS of the non-selective BB user and selective BB user group did not differ significantly (log rank test: p = 0.619). The univariate analysis performed using the hazard model showed significant associations between PFS and BB use (yes versus no), ECOG PS score (1 versus 0 and 2 versus 0), number of metastases (2 versus 1 and 3 versus 1), and brain metastases (yes versus no), with HR values equaling 2.30 (95% CI: 1.67–3.17; p < 0.001), 2.37 (95% CI: 1.71–3.29; p < 0.001), 3.62 (95% CI: 1.32–9.97; p = 0.013), 2.31 (95% CI: 1.56–3.43; p < 0.001), 2.43 (95% CI: 1.55–3.81; p < 0.001), and 2.70 (95% CI: 1.64–4.43; p < 0.001), respectively. After adjusting for age, ECOG PS score, number of metastases, and brain metastases, BB use (yes versus no) was found to be associated with significant worsening of PFS (adjusted HR: 2.21, 95% CI: 1.56–3.12; p < 0.001) (Table 2). In addition, further analysis comparing selective and non-selective BB users with non-users suggested statistically significant positive associations for both BB types (adjusted HR: 2.72, 95% CI: 1.80–4.12 and 1.88, 95% CI: 1.26–2.82, respectively; Supplemental Table S1). Although higher magnitude associations were observed for selective BBs, no statistically significant difference was detected between selective versus non-selective BB users (adjusted HR: 1.34, 95% CI: 0.86–2.10; p = 0.195) (Supplemental Table S1).

KM plot for PFS by BB use among HER2-positive advanced breast cancer patients treated with trastuzumab (log rank test: p ⩽ 0.001).
Cox proportional hazards model for evaluating the effect of the clinical variables on PFS.

Adjusted for age, ECOG PS, number of metastases, and brain metastases.
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; ER, estrogen receptor; HR, hazard ratio; HT, hypertension; PFS, progression-free survival; PR, progesterone receptor; PS, performance status.
BB use with higher HR was associated with poorer PFS regardless of patients’ ER/PR receptor status (positive or negative) (Supplemental Table S3).
Secondary endpoints
Eventually, 134 (60.63%) of the 221 patients died. The estimated median OS of the entire study cohort was 45.63 (95% CI: 37.93–53.33; Q1–Q3: 26.4–71.9) months. Moreover, the estimated median OS of the BB non-user, non-selective BB user, and selective BB user groups was 56.70 (95% CI: 52.03–61.37; Q1–Q3: 41.3–95.9), 29.10 (95% CI: 21.37–36.83; Q1–Q3: 19.0–45.5), and 27.17 (95% CI: 20.57–33.77; Q1–Q3: 16.1–54.0) months, respectively. Kaplan–Meier analysis of the cumulative OS rates showed a significant intergroup difference (log rank test: p < 0.001) (Figure 3). Subgroup analysis revealed significant differences between the BB non-user and non-selective BB user groups (log rank test: p < 0.001) and between the BB non-user and selective BB user groups (log rank test: p < 0.001). OS was not significantly different between the non-selective BB user and selective BB user groups (log rank test: p = 0.766). The results of the univariate analysis using the hazard model were significant for BB use (yes versus no), ECOG PS score (1 versus 0 and 2 versus 0), number of metastases (2 versus 1 and 3 versus 1), and brain metastases (yes versus no), with HR values for OS equaling 2.66 (95% CI: 1.88–3.76; p < 0.001), 2.93 (95% CI: 2.07–4.16; p < 0.001), 3.96 (95% CI: 1.43–10.99; p = 0.008), 2.16 (95% CI: 1.43–3.27; p < 0.001), 2.39 (95% CI: 1.49–3.83; p < 0.001), and 2.67 (95% CI: 1.58–4.49; p < 0.001), respectively. After adjusting for covariables, the OS was found to significantly worsen with BB use (adjusted HR: 2.46, 95% CI: 1.69–3.57; p < 0.001) (Table 3). In addition, further analysis comparing selective and non-selective BB users with non-users suggested statistically significant positive associations for both BB types (adjusted HR: 2.97, 95% CI: 1.91–4.62 and 2.10, 95% CI: 1.36–3.25, respectively; Supplemental Table S2). Although higher magnitude associations were observed for selective BBs, no statistically significant difference was detected between selective versus non-selective BB users (adjusted HR: 1.27, 95% CI: 0.80–2.02; p = 0.316; Supplemental Table S2).
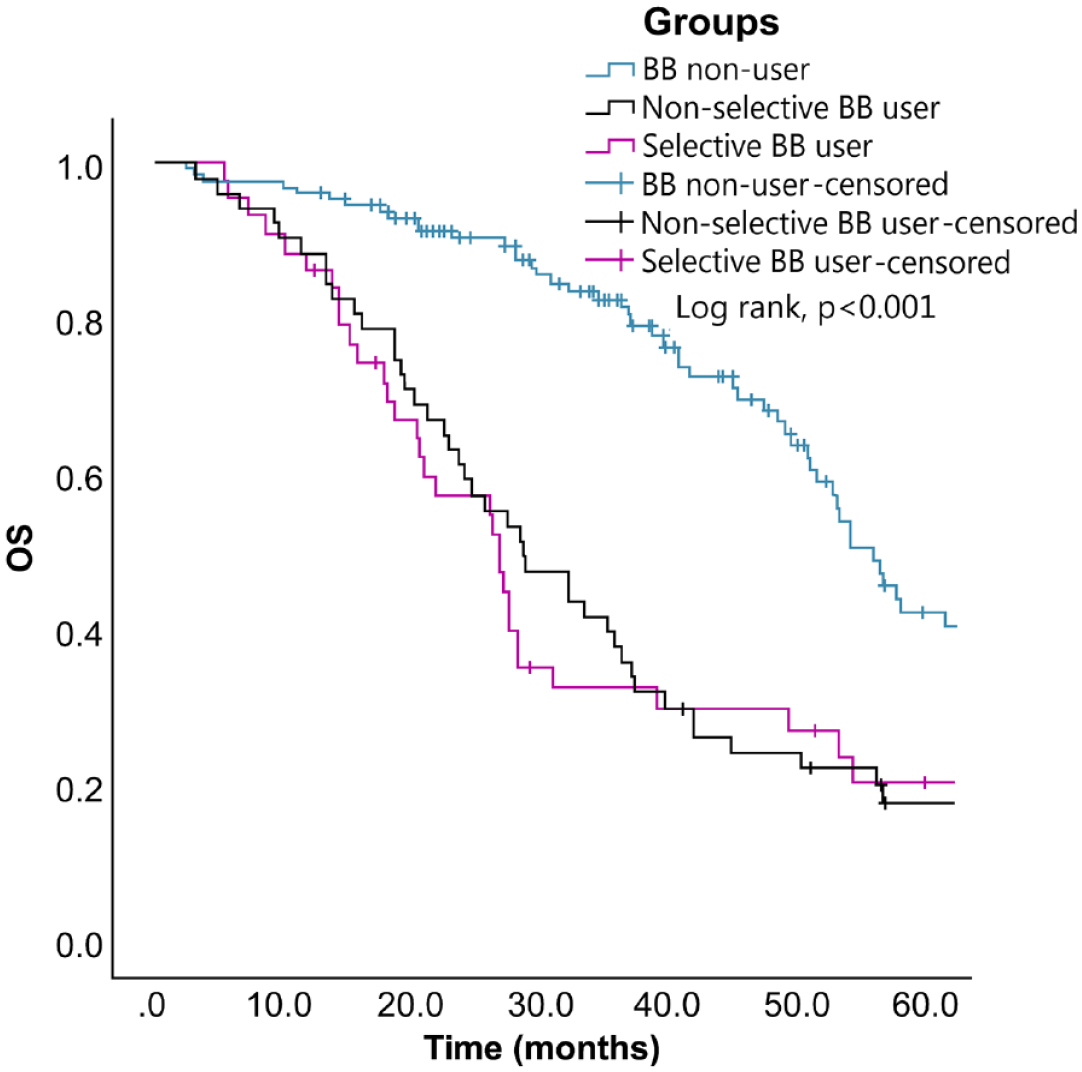
KM plot for OS by BB use among HER2-positive advanced breast cancer patients treated with trastuzumab (log rank test: p ⩽ 0.001).
Cox proportional hazards model for evaluating the effect of clinical variables on OS.

Adjusted for age, ECOG PS, number of metastases, and brain metastases.
CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; ER, estrogen receptor; HR, hazard ratio; HT, hypertension; OS, overall survival; PR, progesterone receptor; PS, performance status.
BB use with higher HR was associated with poorer OS regardless of patients’ ER/PR receptor status (positive or negative) (Supplemental Table S3).
Discussion
Previous studies have suggested that BBs improve PFS and OS in breast cancer patients, but the benefits are still unclear. In 2015, a retrospective study showed that the combination of a BB plus trastuzumab improved PFS and OS significantly in patients having metastatic breast cancer with HER2 overexpression, suggesting the possibility of combining these two treatments for breast cancer with HER2 overexpression. 11 In 2011, a population-based study showed that breast cancer progression and mortality could be reduced by targeting the β2-adrenergic signaling pathway. 13 In 2011, the Life After Cancer Epidemiology study also showed that breast cancer recurrence, specific mortality, and overall mortality were associated with the use of BBs and/or angiotensin-converting enzyme inhibitors. Among patients who used BBs, recurrences and cause-specific deaths were lower. 17 In 2015, a meta-analysis suggested that BB use significantly reduced the risk of breast cancer mortality. 23
However, some studies have contradicted previous findings. In 2011, a retrospective cohort study conducted on a population-based basis found an HR value for OS equaling 1.09 (95% CI: 0.80–1.49) associated with the use of BB plus trastuzumab did not affect the survival of patients with breast cancer. 18 A nested case–control study conducted in 2013 in the UK Clinical Practice Research Datalink cohort showed little association between breast cancer-specific mortality and BB use. 19 According to another systematic review and meta-analysis published in 2016, BB use before the diagnosis of breast cancer did not improve cancer-specific or all-cause mortality. 25 In 2018, another meta-analysis involving 319,006 patients showed no evidence of an association between BB use and OS, all-cause mortality, and PFS. 26 According to a meta-analysis conducted in 2020, BB use was not significantly associated with breast cancer recurrence, breast cancer-related mortality, and death from any cause. 29 Pooled analyses of the clinical trial data published in 2020 for patients initiating anti-HER2 therapy for advanced breast cancer and a sensitivity analysis of patients with preexisting CVD both showed that BB use was associated with a worse OS rate. 30 In 2021, a systematic review and meta-analysis study showed there was no significant difference on recurrence-free survival observed according to non-selective or selective BB use in patients with early-stage breast cancer. 31
We found that BB use was associated with poorer PFS and OS in our current study. Comparison of selective and non-selective BB users with non-users suggested statistically significant positive associations for both BB types. Although higher magnitude associations were observed for selective BBs, no statistically significant difference was detected between selective versus non-selective BB users. Regardless of whether the ER/PR receptor status of the patients is positive or negative, BB use is associated with poorer PFS and OS.
Several limitations of this retrospective cohort study must be considered. The first and principal limitation was that the clinical data and medical records were obtained from the hospital information system. Thus, physician-related personal factors may be responsible for the incomplete records. Second, the sample size was small, especially for the selective BB user group, which did not permit an accurate assessment of the treatment response. Despite the adjustment analysis on age, brain metastases, ECOG PS, ER/PR status, CVD, and hypertension, this study may still have some confounding factors, such as important variables like body mass index and left ventricular ejection fraction not being included in the model. Consequently, the imbalance in these prognostic variables between the groups could potentially affect the results and cause bias. In addition, since breast cancer is a hormone-dependent cancer, changes in hormonal status during menopause may affect tumor characteristics. On the other hand, we have not explored the effects of other concomitant drugs with known potential anticancer effects, including angiotensin-converting enzyme inhibitors, metformin, and non-steroidal anti-inflammatory. We also did not examine the interaction between BB and other anti-hypertensive drugs due to the small sample size. Therefore, the results of this study should be confirmed by further prospective studies with larger populations.
Conclusion
Among patients with HER2-positive advanced breast cancer, BB used prior to initiating trastuzumab therapy compared with non-use was associated with poorer PFS and OS. Our study provides significant evidence that BB use may have a potentially negative impact on patients with HER2-positive advanced breast cancer, clinicians should be vigilant to ensure that patients also diagnosed with CVD are treated appropriately. Other types of drugs can be used to treat CVD, but BB should be avoided. Large real-world database and prospective studies are essential for validating the results of this study.
Supplemental Material
sj-docx-1-taw-10.1177_20420986231181338 – Supplemental material for Survival outcomes of beta-blocker usage in HER2-positive advanced breast cancer patients: a retrospective cohort study
Supplemental material, sj-docx-1-taw-10.1177_20420986231181338 for Survival outcomes of beta-blocker usage in HER2-positive advanced breast cancer patients: a retrospective cohort study by Hui-Hsia Hsieh, Tien-Yuan Wu, Chi-Hua Chen, Yu-Hung Kuo and Mann-Jen Hour in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986231181338 – Supplemental material for Survival outcomes of beta-blocker usage in HER2-positive advanced breast cancer patients: a retrospective cohort study
Supplemental material, sj-docx-2-taw-10.1177_20420986231181338 for Survival outcomes of beta-blocker usage in HER2-positive advanced breast cancer patients: a retrospective cohort study by Hui-Hsia Hsieh, Tien-Yuan Wu, Chi-Hua Chen, Yu-Hung Kuo and Mann-Jen Hour in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-3-taw-10.1177_20420986231181338 – Supplemental material for Survival outcomes of beta-blocker usage in HER2-positive advanced breast cancer patients: a retrospective cohort study
Supplemental material, sj-docx-3-taw-10.1177_20420986231181338 for Survival outcomes of beta-blocker usage in HER2-positive advanced breast cancer patients: a retrospective cohort study by Hui-Hsia Hsieh, Tien-Yuan Wu, Chi-Hua Chen, Yu-Hung Kuo and Mann-Jen Hour in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
