Abstract
Insomnia continues to be a major public health concern and to have a pronounced and detrimental effect on health care costs, productivity, and quality of life. Efficacious pharmacological and nonpharmacological interventions have emerged over the past few decades, giving providers several options for addressing this disruptive condition; these are herein reviewed.
Benzodiazepines have long been the pharmacological treatment of choice for insomnia; however, novel hypnotics being developed by the pharmaceutical industry show promise in addressing insomnia with fewer side effects. From the nonpharmacological perspective, several different single-component interventions such as stimulus control have been shown to be effective, although a combination of these approaches (eg, cognitive-behavioral therapy for insomnia) also enjoy strong empirical support. Future research should continue to examine how sequencing of treatment components, treating different patient cohorts, and combinations of pharmacological and non-pharmacological treatments influence patient outcome.
Introduction
Insomnia, broadly defined as difficulty initiating and maintaining sleep, is a pervasive problem, impacting the lives and health of millions of people in most industrialized countries 1 with approximately 5%–10% of the population experiencing persistent or chronic insomnia. 2 Insomnia is associated with considerable societal costs and individual burden ranging from higher health care costs, impaired work performance, and poor quality of life to increased morbidity and mortality.3,4 The majority of insomnia complaints occur within the context of medical conditions and psychiatric disorders and typically do not remit with treatment of the co-occurring condition. 5 In fact, insomnia can contribute to illness severity 6 and temper responses to treatment 7 for these other conditions.
Given both the scope and the significance of insomnia as a public health problem, delivery of efficacious and effective forms of treatment is paramount. Unfortunately, less than half of patients with insomnia seek care for their condition and many turn to ineffective management strategies such as alcohol use or long-term use of other-the-counter medications. 2 Fortunately, a variety of both pharmacologic and nonpharmacologic approaches are available for the management of insomnia, which are reviewed herein.
Current Pharmacological Options for the Management of Insomnia
Pharmacotherapy represents 1 potential treatment approach for insomnia. Other approaches, including cognitive behavioral therapy, are reviewed elsewhere in this article. In general, pharmacotherapy is thought to be most efficacious when coupled with behavioral interventions. 8
Medications approved by the U.S. Food and Drug Administration (FDA) that have indications for the treatment of insomnia include several older- and several newer-generation medications. The older generation medications include 2 barbiturates: butabarbital and secobarbital, which have largely fallen out of favor; as well as several benzodiazepines: flurazepam hydrochloride, quazepam, triazolam, estazolam, and temazepam. The newer generation of medications, which are used much more frequently, includes 3 benzodiazepine receptor agonists (zolpidem, zaleplon, and eszopiclone).
Additionally, ramelteon, a selective melatonin receptor agonist, is approved for treatment of insomnia. Most recently, doxepin hydrochloride, a tricyclic antidepressant, received FDA indication for insomnia. Furthermore, 2 over the counter antihistamines, diphenhydramine and doxylamine, are also FDA approved. Off-label use of medications, without FDA indication for the treatment of insomnia, is nearly as frequent as prescription of medications with FDA indications for insomnia. 9 The agents most commonly utilized include antidepressants such as trazodone, amitriptyline, and mirtazapine, antipsychotics, such as quetiapine and risperidone, and other benzodiazepines.
In addition, there is widespread use of over-the-counter supplements and herbal preparations, such as melatonin, valerian root, kava kava, lavender, and chamomile. The characteristics, metabolism, pharmacokinetics and dosing of FDA-approved medications with an indication for insomnia are summarized in Table 1.
Characteristics of FDA approved medications for the treatment of insomnia.
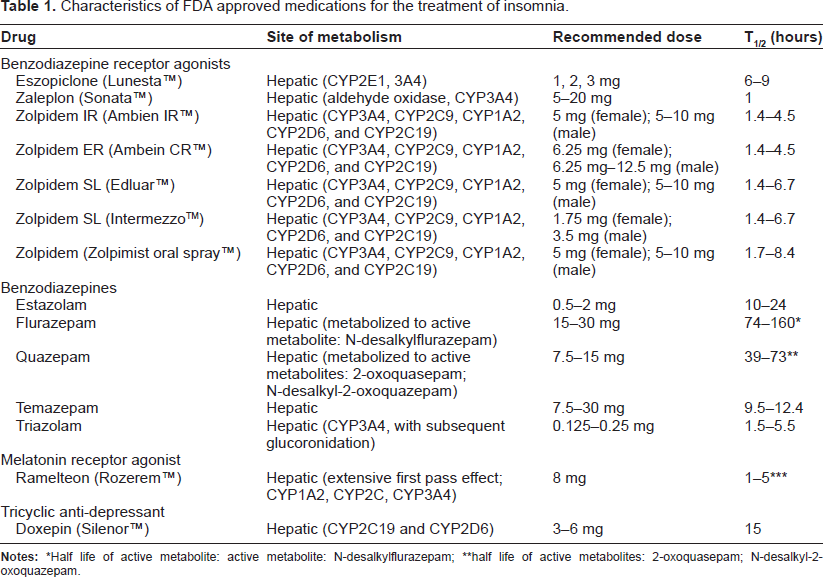
Half life of active metabolite: active metabolite: N-desalkylflurazepam;
half life of active metabolites: 2-oxoquasepam; N-desalkyl-2-oxoquazepam.
Benzodiazapine receptor agonists
In 1992, zolpidem was approved for use in the United States. Since that time, 2 other benzodiazepine receptor agonists (BRA) have been approved for clinical use. These have become the most widely-prescribed class of medication for the treatment of insomnia. 9 BRAs are structurally dissimilar from benzodiazepines. They target action at gamma aminobutyric acid (GABA) type A receptor complexes, with preferential affinity for the α-1 subunit more than others subtypes. This theoretically affords the medication greater sedative hypnotic properties, and less impact on anxiolysis and muscle relaxation relative to benzodiazepines. As such, these medications tend to have fewer cognitive and psychomotor side effects compared to benzodiazepines. 10 They have been demonstrated to reduce sleep latency (time to sleep onset), reduce the frequency of nocturnal awakenings, and increase sleep duration (except for zaleplon). 11 In addition, it has been described that zolpidem can impact perception of wake and sleep. In 1 trial, when insomniacs were awoken from sleep, they were more likely to report having been asleep after administration of zolpidem, compared to after placebo, whereas there was no such difference between placebo and the benzodiazepine, flurazapem. 12
Zaleplon has the shortest half-life of three agents at approximately 1 hour; as such, its greatest utility is in helping to promote sleep onset. 13 In general, patients should allow at least 4 hours of sleep time after taking zaleplon. It is not associated with persistent morning sleepiness. It is indicated for short-term treatment of insomnia. Headache is the most common adverse reaction. 14
Zolpidem has an intermediate half-life, between 1.4–6.7 hours. It is available in several formulations: as an immediate-release preparation (intended for sleep initiation insomnia) and as an extended-release preparation (intended for sleep initiation and sleep maintenance insomnia). It is also available as a lower-dose dissolvable tablet to be taken during the middle of the night for sleep maintenance insomnia. In addition, a metered dose oral spray is also available. The most common side effects in order of prevalence are headache, somnolence and dizziness. 14 In January 2013, the FDA released a new recommendation that the bedtime dose of zolpidem be lowered, as new data showed that patients may be at risk for next morning impairment for activities requiring mental alertness. The FDA recommended lowering the dose of zolpidem for women from 10 mg to 5 mg for immediate-release preparations and from 12.5 mg to 6.25 mg in controlled-release preparations. 15 Complex sleep-related behaviors (parasomnias) such as sleep-driving (ie, driving while not fully awake), preparing and eating food, making phone calls, or having sex while not fully awake, usually with no memory of the event, have been reported. 16 Similar behaviors could theoretically be observed with the other BRAs.
Eszopiclone is the active dextrorotatory stereoisomer of zopiclone (a BRA that is approved in Canada, Australia, and Europe). It is the longest acting of the BRAs, with a half-life of 6–9 hours. It is intended for use in patients with sleep-initiation and sleep-maintenance insomnia. Adverse effects are similar to the other BRAs. In addition, patients will intermittently experience an unpleasant or metallic taste from the medication. 14
An additional concern with the use of these agents is the possibility of rebound insomnia, as can be seen with benzodiazepines. 17 In addition, one must be alert to the risk of withdrawal and dependence. These are often the greatest concerns patients have when considering this type of therapy. Further, patients often have a reluctance to feel “dependent” on any type of sleep aide.
Benzodiazepines
For much of the latter half of the 20th century benzodiazepines were the most commonly used pharmacologic treatment for insomnia. Benzodiazepines enhance the effect of GABA, an inhibitory neurotransmitter, at the GABAA receptor. They do this by increasing the opening frequency of the GABAA receptor. They have varied clinical properties, including sedative hypnotic effects, anxiolysis, anticonvulsant effects, and muscle relaxant properties. Most benzodiazepines are highly lipophilic, and as such enter the central nervous system rapidly. There are five benzodiazepines that have FDA indication for the treatment of insomnia: flurazepam hydrochloride, quazepam, triazolam, estazolam, and temazepam. In addition, a number of other, off-label, benzodiazepines are commonly used in clinical practice for the treatment of insomnia. Benzodiazepines vary widely in their rapidity of onset as well as duration of action. Multiple benzodiazepines have been shown to decrease sleep latency, reduce the number of nocturnal awakenings and increase sleep duration. 18 Adverse effects of benzodiazepines include morning sedation, respiratory depression, dizziness, cognitive impairment, withdrawal potential, and dependence. 19
Melatonin agonist
Ramelteon is a melatonin receptor agonist that selectively binds to the MT1 and MT2 receptors in the suprachiasmatic nucleus of the hypothalamus. It is thought that these are the same receptors that endogenous melatonin acts upon. In contrast to benzodiazepines and benzodiazepine receptor agonists, this is not a GABAergic agent. In a randomized, double-blind, placebo-controlled trial, ramelteon produced a modest reduction in patient reports of sleep latency. It had an inconsistent effect on wakefulness after sleep onset. 20 There are no published studies in humans comparing the efficacy of ramelteon to melatonin. Like melatonin, ramelteon can be utilized in patients with circadian rhythm disturbances. It has been shown to have phase-shifting effects on endogenous circadian rhythms. 16 It is generally a well-tolerated medication, with the most common side effect being dizziness and somnolence. Post-marketing reports have been published of complex behaviors (parasomnia), 21 increased prolactin levels, and decreased testosterone levels. It is one of only 2 agents with an FDA indication for insomnia that is not categorized as a controlled substance. 22
Antidepressants
Doxepin, a tricyclic antidepressant that was originally introduced in 1969 for treatment of depression, is also not a controlled substance. It had been observed for many years that doxepin seemed to have hypnotic effects in patients treated for depression. In 2010 it was approved for the treatment of sleep maintenance insomnia by the FDA. Doxepin acts as a norepinephrine and serotonin reuptake inhibitor. It also has very strong antagonistic effects on Histamine1 and Histamine2 receptors, strong antagonism on 5-HT2, α1-adrenergic, mACh and moderate antagonism to 5-HT1 receptors. Doses usually used for the treatment of depression range between 50–200 mg. At lower doses, it is postulated that doxepin has selective antagonism for histamine1 without activity at the other receptors. 2 randomized, double-blind, placebo-controlled trials demonstrated that low dose doxepin (3–6 mg) led to a significant improvement in sleep maintenance and early morning awakenings. They reported no rebound insomnia or withdrawal effects upon discontinuation of the medication.23,24
Off label use of medications
Various classes of medications that have sedative properties are utilized for the treatment of insomnia, without FDA-labeled indication. At times a medication may be used that can be helpful in treating a comorbid condition, such as the use of a sedating antidepressant. This process often takes advantage of medication side effects, such as central anticholinergic or histamine activity. Trazodone, a serotonin antagonist and reuptake inhibitor, is one of the most commonly used agents for the treatment of insomnia. There is limited data about its utility. One randomized, double-blind study compared trazodone 50 mg, zolpidem 10 mg, and placebo over a 2-week period. Both trazodone and zolpidem led to reduced sleep latency and increased sleep duration of non-depressed, primary insomniacs. Zolpidem was found to be somewhat more efficacious at the doses studied. 25 Other antidepressants that are commonly used include amitriptyline and mirtazapine. These medications are often limited by morning sedation and anti-cholinergic side effects.
Over-the-counter supplements often contain diphenhydramine or doxylamine, first generation histamine H1 antagonists. They are used for their mild sedating properties, mediated through central histamine blockade. One study demonstrated that people develop rapid tolerance to the sedative effects of diphenhydramine. Tolerance was observed in this daytime trial within 4 days of starting diphenhydramine 50 mg BID. 26 Further, use of these medications can be limited by next-morning sedation, grogginess, and anticholinergic side effects, including cognitive impairment, urinary retention, and xerostomia. The routine use of diphenhydramine to treat chronic insomnia is not recommended.
Sedating antipsychotic agents are also frequently used to treat insomnia. The most commonly prescribed agents are quetiapine, olanzapine, and risperidone. Thus far, the data has been largely inconclusive regarding the safety and efficacy of these agents for the treatment of insomnia.27,28 There are multiple potential side effects associated with these medications, the most notable being weight gain, metabolic syndrome, hyperprolactinemia, sudden death, and an increased risk of mortality when used to treat psychiatric symptoms in elderly dementia patients.
Novel hypnotics
There are several classes of novel hypnotics that are currently being evaluated. These include orexin receptor antagonists almorexant and suvorexant. Suvorexant (MK-4305) is a dual orexin receptor antagonist that is a novel treatment for insomnia. It has completed phase III clinical trials. It demonstrated decreased waking after sleep onset, increased sleep efficiency and total sleep time in healthy subjects without sleep disorders.29,30 In addition, there is interest in agents that are specific serotonin subtype 2A antagonists. This includes volinanserin, eplivanserin, and pimavanserin amongst several others.
Eplivanserin is an inverse agonist on the serotonin receptor subtype 5-HT2A. In contrast to other sedating drugs acting on 5-HT2A receptors (eg, mirtazapine, risperidone), eplivanserin has minimal affinity to dopamine, histamine and adrenergic receptors. 31 Theoretically, this may lead to fewer potential side effects.
Propofol, an anesthetic agent that is most commonly used for the induction of anesthesia, was reported in 1 study to be a potential therapeutic target for the treatment of insomnia. Propofol is proposed to work through potentiation of the GABAA receptor, enhancing GABAergic transmission, and also as a sodium channel blocker. A randomized, double-blind, placebo-controlled study tested the use of propofol induced anesthesia in patients with refractory chronic primary insomnia. 103 patients were randomized to receive either physiological saline (placebo) or propofol in a 2-hour continuous intravenous infusion for 5 consecutive nights. They reported improvement in insomnia characteristics immediately after the therapy, which persisted for 6 months. 32
Summary of pharmacological treatments of insomnia
There are a wide variety of agents that are utilized for the treatment of insomnia, including over-the-counter sleep aids, alcohol, herbal remedies, and prescription medications. Further, of prescription medications, off-label use of medications is nearly as frequent as the use of medications with an indication for the treatment of insomnia.
It is not uncommon that patients will have tried over-the-counter sleep aids, alcohol, or other substances to help promote sleep and eliminate insomnia. Whenever pharmacotherapy is being considered for the treatment of insomnia, one must always be alert to the presence of other sleep disorders that contribute to poor quality sleep, such as sleep disordered breathing or circadian rhythm disturbances. One must also be alert to other medical co-morbidities that may be contributing to insomnia, such as nocturnal dyspnea, nocturia, and chronic pain.
Additionally, drug choice may be influenced by the presence of particular medical co-morbidities, such as psychiatric disease, liver disease, and respiratory disease. Further, a careful review of current medications is useful to determine if a patient may be taking medications that can contribute to insomnia (corticosteroids, stimulants, and diuretics for example). In addition, the side effect profile of a medication must also be considered. Finally, and of much importance, is that thought should be given to the duration of treatment. Although long-term use of many of the medications reviewed is not uncommon and there are no FDA restrictions on the duration of treatment, the empirical support for long term use is limited and addresses use of 6–12 months duration. Thus, providers need to consider how long this treatment will be used, and ought to counsel patients accordingly before instituting any particular treatment.
In general, when considering one's choice of agents it is imperative to first characterize the type of insomnia (sleep onset and/or sleep maintenance), as well as associated medical comorbidities. Zolpidem and zaleplon may be first-line agents for sleep-onset insomnia, as well as other benzodiazepines or ramelteon. In patients with sleep-maintenance insomnia, extended release preparations of zolpidem, eszopiclone, doxepin, and other benzodiazepines can be considered first-line agents.
Current Nonpharmacological Options for the Management of Insomnia
Sleep hygiene education
Sleep hygiene education is often used by physicians as an initial approach to addressing insomnia and other sleep difficulties and can be generally described as the promotion of behaviors that are intended to increase the quantity and quality of sleep that an individual obtains each night. Through psychoeducation and general advice, patients are encouraged to limit behaviors thought to interfere with sleep such as the ingestion of nicotine or alcohol and to establish an environment that is conducive to sleep (eg, regulating room temperature).33,34
Sleep hygiene education traditionally consisted of a list of 10 recommendations 33 : (1) avoid oversleeping, (2) establish a regular wake time, (3) exercise daily (4) reduce potentially disruptive noise, (5) regulate the room temperature, (6) have a light snack before bed to regulate hunger, (7) avoid chronic use of hypnotics, (8) avoid caffeine in the evening, (9) avoid alcohol prior to sleep, and (10) do not fight inability to sleep, rather, turn on light and do something else. Subsequent conceptualizations of sleep hygiene have expanded the list of recommendations to include things such as eliminating the bedroom clock 35 or adding hot baths 36 or relaxing activities 37 to the pre-sleep routine, further blurring the lines between sleep hygiene education and other interventions.
While it is generally accepted that poor sleep hygiene does adversely affect sleep, sleep hygiene education has typically been found to be ineffective as a stand-alone therapy for insomnia.38,39 A partial explanation for this may be revealed in the finding that although insomnia patients are often just as knowledgeable regarding sleep hygiene rules, they are less likely to follow these rules when compared to healthy sleepers. 40 Further, while it may worsen or complicate a patient's sleep difficulties, the presence of poor sleep hygiene alone is unlikely sufficient to be the primary cause of insomnia. 41
Despite these findings, and although it is often used as the control condition of studies examining the efficacy of other sleep interventions, sleep hygiene education is often successfully combined with other approaches as part of a larger treatment package.eg,42
Stimulus control
Stimulus control has been identified as one of the more effective treatments available for addressing insomnia.8,38 The intervention is predicated on learning theory and seeks to re-establish positive cognitions and break counterproductive associations regarding the bed, bedtime routine, and sleep. Through repeated episodes of insomnia, sleep-related stimuli are thought to become paired with the negative cognitions generated by being unable to fall asleep. The mind and body gradually become trained to associate the bed, sleep, and sleep-related stimuli with frustration, anger, and wakefulness. 43
Stimulus control, in turn, is designed to reduce the amount of time that sleep-related stimuli and negative cognitions are paired for, serving to break counterproductive associations between bed and sleep. This is typically accomplished through adherence to several rules concerning sleep behavior. The patient is advised to: (1) keep a fixed wake time every day, regardless of the night's sleep, (2) avoid napping, (3) use the bed/bedroom only (or almost exclusively) for sleep and intimacy, (4) lie down to go to sleep only when sleepy, and (5) leave the bed if unable to sleep after approximately 15–20 minutes of wakefulness. These rules all serve to curtail the amount of total wake time the patient spends in bed.
Stimulus control therapy has been repeatedly found to be efficacious in the treatment of insomnia among both medicated and non-medicated patients. 44 In a 1994 meta-analysis of 59 treatment outcome studies consisting of 2,104 patients, Morin and colleagues reported that stimulus control was the most effective stand-alone therapy for the treatment of insomnia. 38 Participants receiving stimulus control therapy (lasting 6–8 weeks) saw a significant increase in total sleep time and mean reductions in sleep latency, time awake after sleep onset, and number of night awakenings. A subsequent 1995 meta-analysis by Murtagh and Greenwood confirmed significant improvements on sleep latency, nighttime awakenings, total sleep time, and self-rated sleep quality. 45 Currently, stimulus control therapy is supported as a standard in the treatment of insomnia by the American Academy of Sleep Medicine (AASM). 8
Relaxation
Relaxation training is another approach often used in the treatment of insomnia, both as a standalone therapy and as part of larger treatment packages such as cognitive behavioral therapy for insomnia (CBT-I). Earlier sleep onset may be the long-term goal of both provider and patient; however, reductions in basal levels of arousal are the primary aim of relaxation training. 47 This is achieved through teaching patients skills that can help them reduce the amount of autonomic and cognitive arousal they experience. 38
The form that relaxation training takes can be varied depending on patient preference and the efficacy that both provider and patient feel in practicing the skill. The most commonly observed components include: diaphragmatic breathing exercises, guided imagery, meditation, progressive muscle relaxation, and biofeedback. While learning relaxation skills is not intrusive or overly intensive, it takes consistent practice in order to achieve a sense of mastery and consistently reduce arousal.
In meta-analyses examining effects of relaxation training, it has been reported that the approach is effective in positively addressing several insomnia/ sleep related variables including sleep onset latency, time awake after sleep onset, sleep quality ratings, and number of nighttime awakenings.37,46,47 Findings on the impact that relaxation training has on total sleep time (TST) have been less clear. In their 1995 meta-analysis, Murtagh and Greenwood reported a small effect size in favor of increased TST, 45 while a subsequent meta-analysis has reported that the relationship between relaxation training and TST does not reach significance. 48
Cognitive therapy
In addition to being a primary component of CBT-I, cognitive therapy for insomnia 49 has been used as a stand-alone therapy. A cognitive approach to insomnia assumes a relationship between negative thoughts/ cognitions and sleep. Patients are typically preoccupied with perceived consequences of being unable to achieve sleep onset or return to sleep after an awakening (eg, “I will not be able to fall asleep and will get in an accident at work because I am overly fatigued”). Faulty cognitions often take the form of unrealistic expectations regarding sleep, misconceptions of the etiology of sleep problems, amplifying or catastrophizing the consequences associated with poor sleep, and performance anxiety related to previous attempts to control one's sleep. 49
Central to cognitive therapy for insomnia is the process of identifying and challenging the negative cognitions that interfere with sleep. The therapist works with the patient to explore the utility and validity of the faulty cognitions and replace them, which is in turn hypothesized to lead to a reduction in the anxiety associated with insomnia. Common cognitive strategies used to address insomnia include reappraisal, hypothesis testing, reattribution training, and attention shifting. 49 While cognitive therapy for insomnia has often been examined as a part of larger treatment packages, there is little empirical research specifically examining the unique contribution of cognitive therapy to the reduction of sleep disturbance. 50
One specific form of cognitive therapy for insomnia that has been the subject of randomized, controlled trials is paradoxical intention. This approach is based on the premise that anticipatory or performance anxiety is coupled with the patient's attempt to sleep. Patients are instructed to intentionally engage in the opposite and feared behavior, staying awake, which in turn may alleviate their performance-based anxiety. Based on positive, albeit limited data, the AASM currently recognizes paradoxical intention as an effective and recommended therapy (guideline) for the treatment of sleep onset insomnia. 8
Murtagh and Greenwood 45 reported that paradoxical intention reduced sleep onset latency and nocturnal awakenings, however, the effect on total sleep time was not significant. Subsequent to the Murtagh and Greenwood meta-analysis, a 2003 randomized controlled trial examining the effect of the paradoxical intention technique among 34 individuals suffering from sleep onset insomnia was conducted. It was reported that those in the paradoxical intention condition saw significant reductions in sleep onset latency as well as self-rated measures of sleep performance anxiety. 51
Sleep restriction therapy (SRT)
Sleep restriction therapy 52 is frequently used in the treatment of insomnia and is often one of the primary components of multimodal nonpharmacological treatment packages for insomnia. The therapy attempts to consolidate fragmented sleep by prolonging wakefulness (ie, limiting the amount of time that the patient can spend in bed attempting to sleep). This serves to increase the patient's sleep drive and leads to consolidated blocks of sleep with a decrease in sleep latency and fewer awakenings of shorter duration.
Initially, patients are asked to restrict the time that they spend in bed to the mean TST that is reported via sleep diaries completed during a one to two week baseline period preceding the beginning of SRT. A consistent rise time is set and the patient's bedtime is delayed in order to approximate the mean total sleep time. At subsequent appointments the patient's bedtime is adjusted based on sleep efficiency (TST divided by time in bed x 100%) scores since the last session. As the patient demonstrates sustained improvement (ie, mean sleep efficiency greater than 85% for seven days) time can be added to the sleep window in 15 or 20 minute intervals. Conversely, if sleep efficiencies fall below 80%, the therapist may opt to decrease the sleep window by 15 or 20 minutes.
In order to take full advantage of SRT it is recommended that the patient's bedtime be delayed, as opposed to advancing the rise time. Delaying the bedtime serves the dual purpose of helping to facilitate sleep onset in the patient with insomnia by increasing the sleep drive, and preventing the patient from coping with their insomnia by extending their opportunity for sleep. This approach may be contraindicated in some populations including those with histories of seizure disorder, mania, or fall risks.
Although only considered a guideline intervention when delivered on its own,8,46 the use of SRT is well supported by clinical research.36,38,52,53 Indeed, Morin and colleagues reported in their seminal 1994 meta-analysis that sleep restriction was one of the most effective standalone interventions for the treatment of insomnia and that SRT resulted in significant reductions in sleep onset latency and minutes of wake after sleep onset (WASO).
Cognitive behavioral therapy for insomnia (CBT-I)
While the aforementioned treatments have their utility as standalone approaches to the treatment of insomnia, they have perhaps received the most empirical support, and seen their most widespread implementation, as a part of the larger treatment package known as CBT-I. 54 The CBT-I approach combines psychoeducation with cognitive therapy, and behavioral treatment approaches such as stimulus control, sleep restriction therapy, and sleep hygiene. Using classical and operant conditioning principals as a theoretical framework, CBT-I targets thoughts, behaviors, and environmental variables that interfere with sleep onset and maintenance. 54
CBT-I techniques are used to reduce both physiological and cognitive arousal, while attempting to regulate the body's circadian and homeostatic influences on sleep via sleep scheduling. 54 Cognitive behavioral therapy for insomnia has been identified in the practice standards of the AASM as an effective treatment (standard) for chronic insomnia. 46
Meta-analyses of the existing research on CBT-I and insomnia have revealed that the treatment approach is effective in reducing sleep onset latency, WASO, and increasing sleep quality48,55 and established that behavioral interventions are an effective alternative to medication. 56 Further, it has been reported that the treatment gains achieved through CBT-I are less transient than the relief gained through medication and that they tend to persist for some time after the active treatment has ended. 57
In a recent dismantling study, 58 179 individuals with primary insomnia were randomized to 1 of 4 groups: stimulus control therapy (SCT), sleep restriction therapy, combined SCT and SRT, or waitlist control. All conditions saw improvements in sleep onset latency, WASO, TST, and sleep efficiency when compared to controls. However, the overall largest effects and greatest remission rates were observed in the combined SCT and SRT condition.
Brief interventions
Sleep science has not been immune to the trend in behavioral health of striving to create ever briefer and more compact treatments. One intervention that has recently emerged is intensive sleep retraining (ISR). 59 This brief, 25-hour treatment involves acute sleep deprivation and repeated attempts at sleep onset on the half hour. The patient is presented with multiple sleep onset opportunities to counter his or her conditioned insomnia. The authors reported that in the ISR-only condition, significant improvements in TST, sleep efficiency, and sleep onset latency were observed. However, even greater improvements were observed when ISR was combined with stimulus control therapy. Treatment gains are often observed rapidly (within 1 week) and were maintained at a 6-month follow-up.
Brief forms of CBT-I have been introduced, including a ‘primary-care friendly cognitive behavioral insomnia therapy’, which can be accomplished in 2 sessions. 60 Brief Behavioral Treatment for Insomnia (BBTI) also strives to accomplish the most therapeutic gain in the shortest amount of time. 61 BBTI involves an initial 45-minute session focused on education, stimulus control, and sleep restriction followed by a booster session 2 weeks later. Authors of both approaches report notable improvements in WASO, sleep quality, sleep efficiency, and sleep onset latency.60,61
Bright light or phototherapy
One alternative intervention that is frequently mentioned as a possible treatment for insomnia is bright light therapy, also known as phototherapy. This therapy involves exposure to light that is approximately 10,000 lux in intensity for varying periods of time. The treatment is believed to help patients adjust disrupted circadian rhythm cycles by advancing one's sleep propensity rhythm or “wake maintenance zone” to a more socially acceptable or desirable time for sleep. 62 For example, when the patient favors later bed and wake times, early morning exposure to bright light is indicated. However, if the patient tends to favor earlier bed and wake times, evening exposure to bright light is recommended.
It has been previously demonstrated that phase shifts can be achieved by exposure to 7000–12,000 lux for 2 to 3 days. 63 Bright light therapy has been reported to be effective in treating individuals with early morning awakening insomnia, 64 as well as older individuals with sleep maintenance insomnia. 65 Some have reported side-effects associated with bright light therapy, including insomnia, visual blurring, headaches, eye strain, and hypomania, particularly in individuals experiencing depression. 66
Summary of nonpharmacological treatments of insomnia
Strong evidence supports the efficacy of nonpharmacologic treatments for the management of insomnia. As early as 2005, an NIH consensus meeting concluded that such approaches should be the front-line treatment for insomnia. 19 Although individual non-pharmacological treatments have their own evidence bases, multimodal cognitive behavioral therapy for insomnia is increasingly viewed as the standard of care. There are very few (eg, short term exacerbation of daytime sleepiness in the initial weeks of sleep restriction) if any side effects associated with CBT-I. The primary concern is related to having good patient adherence, which can be addressed with standard therapeutic techniques. 54 A long-standing shortcoming in delivering this intervention has been the relative shortage of clinicians trained in the approach. 5 This shortage is being increasingly addressed through dissemination efforts 67 as well as the availability of internet-based approaches with good demonstrated efficacy. 68 As reviewed, newer approaches, 59 refinements stemming from dismantling studies, 58 and brief forms of these interventions continue to be developed.60,61
Conclusions
A wide variety of both pharmacologic and nonpharmacologic approaches are available for the management of insomnia. Moreover, these interventions have strong empirical support. Novel hypnotics in the development pipeline show some promise in maximizing efficacy while limiting side effects, although the latter remains an important goal to attain. Likewise, the field of sleep medicine with regard to nonpharmacological approaches has matured to a point where efficacy data is being complimented by effectiveness data for larger and more diverse populations. Dissemination of nonpharmacological approaches remains a barrier to shrinking the public health burden of insomnia, but recent efforts in this area also shows good promise. Currently, non-pharmacologic treatment, and especially CBT-I, should still be considered as the initial treatment of choice for chronic insomnia.8,19,46 Finally, one area that continues to require more thoughtful consideration is in identifying specific patient cohorts who may have enhanced outcomes by utilizing a combination of pharmacologic and nonpharmacologic approaches. Similarly, some efforts to identify the optimal sequencing or combination of treatments has been undertaken, 57 but more work is needed. In sum, clinicians have a wealth of intervention tools with which to manage insomnia; as in other areas of medicine, the near future of insomnia treatment will lie in increasingly personalized approaches.
Author Contributions
All authors jointly developed the structure and arguments for the paper; all authors contributed to the writing of the manuscript, made critical revisions, and reviewed and approved the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author Pigeon was on the Purdue Pharma, LLP Speakers bureau for Intermezzo (through July 2013) and received royalties from Ivy Press, Inc. for Sleep Manual: Training your Mind and Body to Achieve the Perfect Night's Sleep. Authors Bishop and Marcus have no competing interests.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
Footnotes
Acknowledgements
This material is, in part, the result of work supported with resources and the use of facilities at the Center of Excellence for Suicide Prevention, VA Medical Center, Canandaigua, NY and the Center for Integrated Healthcare, VA Medical Center, Syracuse, NY. The contents do not represent the views of the Department of Veterans Affairs or the United States Government.
