Abstract
The imidazopyridine drug zolpidem (
Introduction
Most people experience insomnia at some time during their lives. Because of its high incidence, and also because its symptoms are usually mild and transient, the importance of insomnia is frequently underestimated. However, as a chronic disorder, which affects about 10% of the population, its treatment is often challenging and, moreover, it is associated with a substantial number of comorbid symptoms.1–3 Various conditions are associated with insomnia and can contribute to its development. They can be related to neurological or psychiatric disorders, which in turn may be aggravated by a deficiency of restorative sleep and daytime fatigue. Insomnia can also result from a primary dysfunction or an age-related decline in the circadian system.
Insomnia is characterized by one or more of the following: difficulty falling asleep [e.g. sleep onset latency (SOL) of more than 30 minutes], insufficient sleep [e.g. total sleep time (TST) of less that 5.5-6 hours], numerous nocturnal awakenings, early morning awakenings with inability to resume sleep, or nonrestorative sleep. Common daytime complaints include somnolence, fatigue, irritability, and difficulty concentrating and performing everyday tasks. Because insomnia is associated with reductions in attention span, affected individuals can often be impulsive and experience impaired judgment, and thus are at an increased risk for having injuries at home or work, or involvement in accidents while driving. Psychiatric and other medical illnesses, including cardiovascular diseases, weight gain and glucose intolerance, are other conditions which include insomnia in their overall symptom complex. 4
The International Classification of Sleep Disorders (ICSD-2) 5 considers severity criteria as a guide to be applied in conjunction with consideration of the patient's clinical status. Mild insomnia refers to complaints of an insufficient amount of sleep almost every night or of not feeling rested the following day. There is little or no impairment in social and/or occupational functioning. Moderate and severe insomnia refers to complaints of experiencing an insufficient amount of sleep every night or of not being rested after the impaired sleep episode, accompanied by moderate and severe impairment of social and/or occupational functioning, respectively. The challenge for clinical treatment is to select the therapy which is most appropriate for these differing degrees of severity.
The last several decades have seen an evolution in thinking about the classes of medications which are to be preferred for treating insomnia. The benzodiazepines (BZDs) were introduced in the 1970s, and rapidly increased in popularity because of their efficacy and better safety compared to the barbiturates, carbamates, chloral derivatives and methaqualone.
6
In recent years, however, prescriptions for BZDs have progressively declined, especially because of their associated side effect profile, including their tendency to promote dependence, the occurrence of rebound insomnia following the withdrawal of short- and intermediate-acting derivatives, and the loss of efficacy after several weeks of treatment. The clinical need for medications which did not have these side effects was an important factor leading to the development of structurally dissimilar non-BZD hypnotics. These included the sedating antihistamines, the melatonin receptor agonists ramelteon and tasimelteon, certain antidepressants, and the so-called z-drugs, i.e. the cyclopyrrolones zopiclone and eszopiclone, the pyrazolopyrimidine zaleplon, and the imidazopyridine derivative, zolpidem (
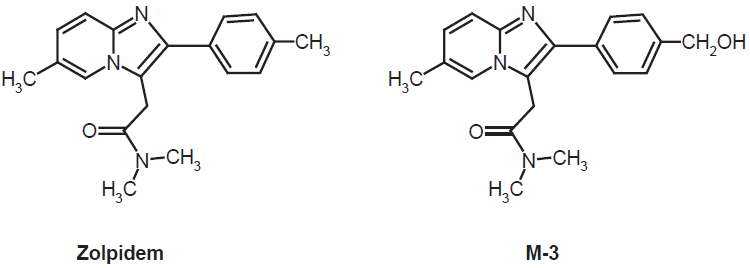
Chemical structures of zolpidem and its main, monohydroxylated metabolite, M-3, which accounts for 80% of the primary products. Other, moderately abundant metabolites are hydroxylated at the other aliphatic methyl groups. In secondary, urinary metabolites, the aromatebound methyl residues can be further oxidized to carboxylic acids.
Zolpidem has proved to be a suitable hypnotic, especially with regard to efficacy in sleep initiation. As will be discussed below, it is relatively well tolerable and almost devoid of the side effects typically associated with BDZs. The low incidence of side effects is, in part, a consequence of a relatively short half-life in the circulation. At usual doses of immediate-release (IR) zolpidem, the peak plasma concentrations are attained between about 45 min and 2 h after intake [Ambien® (zolpidem tartrate) 10 mg: 0.9 h], 7 a kinetics profile that corresponds well with the time course of psychomotor tests, in which the maximum efficacy was found around 1.5 h followed by a rapid decline. 8 The pharmacokinetics of such a short-acting drug, when given as an IR formulation, may be not ideal in terms of promoting sleep maintenance. To respond to the clinical need for an agent which could reduce the number of nocturnal awakenings, zolpidem extended release (ER; = modified release: MR; = controlled release: CR; Ambien CR™ 12.5 or 6.25 mg; Sanofi-Aventis) was developed, the characteristics of which will be in the focus of this article.
Mechanism of Action and Preclinical Pharmacology
The imidazopyridine compound zolpidem was synthesized by Synthélabo Recherche in the early 1980s,9,10 and its therapeutic potential for the treatment of sleep disorders was recognized soon after.11,12
Zolpidem shares with the BDZs the property of binding to α subunits (the “BDZ receptor”) of the GABAA receptor/chloride channel complex, however, with a decisive difference in selectivity concerning the subtypes. BDZs are relatively non-selective in this regard and, therefore, cause antiexcitatory hyperpolarizations in many places in the CNS. These actions lead to more generalized neuronal inhibitions and numerous effects exceeding sedation and sleep promotion, such as anxiolytic, anticonvulsant and myorelaxant actions. The structurally unrelated compound zolpidem, however, displays a relative, but not absolute selectivity towards a specific BDZ binding site, the so-called BZ1 receptor (ω1 receptor), based on a particularly high affinity to the α1 subunit.13–15 Among the GABAA receptors, usually consisting of α, β, and γ subunits (alternately δ subunits), multiple isoforms such as α1-α6, β1-β3, and γ1-γ3, lead to a spectrum of differentially regulated or pharmacologically accessible variants. The high α1/α5 affinity ratio of zolpidem can be taken as a characteristic of this compound. 16 While both α1 and β2 knockout mice show decreases in sleep time, the α1 knockouts are considerably more resistant to zolpidem. 17 Similar conclusions were drawn from investigations on H101R mice expressing zolpidem-insensitive subunits. 16 Via α1 binding, zolpidem acts as an allosteric modulator of the GABAA receptor/chloride channel complex. On the other hand, the anxiolytic, anticonvulsant, myorelaxant, ataxic and also several withdrawal effects of GABAergic sedatives are largely associated with the α2- and α3-containing receptor complexes, which accounts for zolpidem's relative absence of anticonvulsant and myorelaxant effects.18,19 More importantly for a hypnotic drug, zolpidem's relative selectivity reduces the probability of withdrawal effects, with their potential for dependence and abuse. Additionally, the distribution of BZ1 receptors within the CNS leads to a more specific action spectrum. This receptor subtype is predominantly localized in lamina IV of the sensorimotor and in the ventral thalamic cortex, but also in olfactory bulb, pons, inferior colliculus, globus pallidus, pars reticulata of the substantia nigra and the cerebellar molecular layer.20,21 Taken together, the relative α1 specificity of zolpidem and the presence of binding sites in relevant structures could especially explain the preservation of deep sleep stages 3 and 4 by this drug, contrary to the suppressive deep sleep actions observed with BZDs. 22
Consistent with the findings of its binding properties, preclinical studies have shown zolpidem to exhibit predominantly sedative-hypnotic effects, although anticonvulsant and myorelaxant activities have also been reported, however, with a different dose dependence. In rats, zolpidem is more potent in suppressing locomotor activity (sedative effect) than pentylenetetrazol-induced convulsions (anti-convulsant activity) and rotarod performance (muscle relaxation). In contrast to zolpidem, the BZD hypnotics produce a sedative effect with doses which are similar to or greater than those producing anticonvulsant or myorelaxant effects.23,24
To what extent zolpidem interacts with the hypothalamic 25 and pontine26,27 sleep switches, which control sleep induction and sleep stages, respectively, is less clear, although GABAergic mechanisms are involved in both of them. In the case of the hypothalamic sleep switch, which is strongly influenced by the circadian pacemaker, the suprachiasmatic nucleus, α4δ-containing GABAA receptors seem to play a major role,25,28 findings that are in accordance with the sleep-promoting actions of gaboxadol and, perhaps, also with a particular role of extrasynaptic GABAA receptors.28,29 With regard to the pontine REM/non-REM (REM/NREM) switch, the situation is complicated inasmuch as GABAergic inhibitions are present at both sides of the switch. 26 Although NREM sleep is favored by zolpidem, the influences of zolpidem on the stage distribution within the sleep pattern, apart from deepening sleep, are moderate. It remains to be clarified whether this switch system is less affected by the drug, or in a more balanced way. A spectral analysis of sleep EEGs in curarized rats has revealed that its power density in non-REM (NREM) sleep is predominantly increased in the low frequency band (1.0-4.0 Hz). In freely moving animals, zolpidem has been found to augment the duration of NREM sleep and to reduce wakefulness. The effect endured during sub-chronic administration with no rebound occurring after abrupt withdrawal.30,31 On the other hand, variable results have been reported in regard to REM sleep. Depoortere et al 30 for instance, described an increase of REM sleep in rats recorded during the light period, whereas Renger et al 32 reported the opposite effect.
Metabolism and Pharmacokinetic Profile in Healthy Adults and Populations at Risk
Zolpidem is rapidly absorbed and extensively distributed to body tissues, including the brain. Bioavailability is reported to be in the range of 70%. 33 A large fraction is bound to plasma protein (92.5%), independently of concentrations between 40 and 790 ng/mL. Because of the extensive protein binding, its direct excretion is negligible and clearance is predominantly a metabolic one. Zolpidem is metabolized by several cytochrome P450 monooxygenases, but predominantly by CYP3A4, to inactive metabolites.34,35 The principal primary metabolite, M-3, which accounts for more than 80% of the products, is a compound monohydroxylated at the methyl group of the benzene moiety 35 (Fig. 1). Differently hydroxylated and some further oxidized metabolites are also produced, including compounds carrying carboxyl groups. After oral administration, the more polar zolpidem metabolites, which poorly bind to plasma proteins, are readily excreted.33,36–38 Zolpidem carboxylic acid is found as a major urinary product. 39
After a single therapeutic dose of 10 mg zolpidem IR to healthy adults, peak plasma concentrations (Tmax) are attained after 45-60 min, but high levels in the maximum range may be found up to 2 h, 40 and the mean terminal-phase elimination half-life (t1/2) amounts to about 2.4 hours.7,21 Zolpidem clearance is reported to be lower in women than in men. 41 This is in agreement with a positive correlation between testosterone levels and clearance as observed in males. 42
The metabolic clearance of zolpidem is reduced in elderly patients, aged 66 years and older, resulting in increases in maximum plasma concentration (Cmax), area under the concentration curve (AUC), and prolonged terminal-phase elimination (t1/2), the latter amounting to approximately 2.9 hours. 43 Thus, a reduction of the initial dose from 10 to 5 mg in the elderly is recommended. These changes may be partially related to hormonal declines, 42 but largely reflect an age-related decrease in hepatic metabolism. Accordingly, studies on populations at risk have shown that zolpidem clearance is impaired in patients with severe hepatic insufficiency, thus requiring dose reduction, but not in patients with compromised renal function. Therefore, dose adjustment is generally not required in patients with renal failure.38,43
Zolpidem tartrate ER, in its formulation as Ambien CR™ 12.5 mg, consists of a coated two-layer tablet. The primary release comprising about 60% of the content is as fast as that of zolpidem tartrate IR 10 mg and the rise in plasma concentration within the first 30 min does not differ from the IR values. Between 45 and 120 min, plasma levels are very similar in healthy male adults, but the ER formulation leads to a subsequently higher concentration over more than 8 h.7,40,44 The maximum is considerably broadened and values between 45 min and 3.5 h differ only by about 12%.7,21 The mean peak concentration (Cmax) was reported to be 134 ng/mL (range 69-197 ng/mL), at a median Tmax of 1.5 h7,21 (higher value reported by ref. 40: Tmax = 2.4 h), while the mean AUC was 740 ng × h/mL (range: 295-1359 ng × h/mL). 7 The mean elimination half-life, t1/2, was 2.8 h (range 1.62-4.05 h). The substantial interindividual variation in Cmax and AUC is largely explained by differences in resorption, but not so much by metabolic clearance. Correspondingly, studies on food effects (intake while fasting or 30 min after a meal) showed that half-life was not changed, whereas food intake reduced Cmax and AUC by 30 and 23%, respectively. 7 In view of these findings, administration of zolpidem ER (and also zolpidem IR) with or directly after a meal should not be recommended.
Concerning the low-dose formulation designed for the elderly (Ambien CR™ 6.25 mg), the following pharmacokinetic parameters were determined for healthy subjects of 65 years and older: 7 Cmax 70.6 ng/mL (range 35-161 ng/mL); Tmax 2.0 h; AUC 413 ng × h/mL; (range 124-1190 ng × h/mL) elimination t1/2 2.9 h (range 1.59-5.50 h). Not unexpectedly, the interindividual variation is larger in this group, and for some individuals the higher dosage may be more appropriate.
Although this has not been studied in detail for zolpidem tartrate ER (Ambien CR™), the experience with IR formulations would imply a caveat for individuals with hepatic insufficiencies as well, but not so much for those with renal impairment.
Clinical Studies
Basic findings with zolpidem IR during normal sleep and in induced, transient insomnia
Zolpidem's suitability as a hypnotic drug in humans has been investigated in several studies and results of the IR formulations have been repeatedly reviewed, though not always in any detail.45–55 With regard to the basic hypnotic properties and for reasons of comparison, the main findings obtained with zolpidem IR, based on questionnaires (subjective self evaluation) and on objective measures obtained by polysomnography (PSG), will be summarized first. The efficacy endpoints used in many studies have included sleep onset latency (SOL; or latency to persistent sleep: LPS), REM onset latency (REMOL), wake time after sleep onset (WASO), number of awakenings, total sleep time (TST), sleep efficiency (SE), quality of sleep, daytime ability to function, daytime alertness, and sense of physical well-being. Various populations have been included in studies assessing zolpidem's efficacy and safety: healthy volunteers, poor sleepers, non-elderly and elderly patients with chronic primary insomnia, and patients with secondary or comorbid insomnia.
In healthy young and middle-aged adults not affected by insomnia, earlier, usually placebo-controlled, trials with single-dose treatments in rather limited samples led to the following results. Nicholson and Pascoe, 56 double-blind study using 10, 20 and 30 mg zolpidem in 6 young and 6 middle-aged adults: Decreased stage 2 sleep and augmented stage 4 sleep in the young adults, reduction in the number of awakenings, of WASO and stage 1 sleep, and increased stage 3 sleep and REMOL in the middle-aged adults, who also reported subjective reduction of SOL and improvement of sleep quality. A double-blind study of 8 subjects using 10 to 40 mg zolpidem by Blois and Gaillard 57 found increases in stage 2 (40 mg) and stage 4 sleep (20 and 40 mg) and reductions in REM sleep (40 mg), whereas sleep induction and maintenance were not significantly altered, although subjects reported a subjective impression of decreased SOL and improved quality of sleep. Lund et al 58 using 10 or 20 mg in 10 subjects reported decreases in NREM sleep latency and stage 1 sleep, at 10 mg. Merlotti et al 59 studied the effects of 2.5 to 20 mg zolpidem in 12 volunteers and found decreases in objectively evaluated SOL and REM sleep percentage, reductions in the number of awakenings, while simultaneously total sleep time was increased. Brunner et al 60 investigated the effects of 10 mg zolpidem on subjective and polysomnographic (PSG) sleep measures in 8 healthy young men and did not detect significant changes in PSG data, except for a reduction in REM sleep, but neither in sleep induction nor in sleep maintenance.
Quantitative EEG spectral analyses in healthy normal sleepers 60 revealed that 10 mg zolpidem caused a significant depression of power density in the low-frequency range of NREM sleep episodes. By contrast, Patat et al 61 observed a significant increase of delta wave power and a reduction of alpha activity after oral (20 mg) or intravenous (10 mg) administration of zolpidem. On the other hand, Ferrillo et al 62 did not observe significant differences in slow-wave spectral power between placebo and zolpidem 10 mg. Feinberg et al 63 made use of the period-amplitude analysis method to determine the effect of zolpidem (10 mg) on delta, sigma, and beta bands of night sleep EEG. The amplitude of delta waves was significantly reduced, whereas its incidence was slightly but not significantly increased. Sigma and beta waves showed minor changes in amplitude or incidence that did not attain significance. As noted by Okuma and Monti, 64 discrepancies in the findings of these different studies could have reflected variations in experimental design, drug dose, and methodology of data analysis.
Scharf et al 65 used polysomnography and subjective reports to study 30 healthy elderly non-insomniac subjects and found a significant reduction of LPS and WASO and increased SE. Slow wave sleep (SWS; stages 3 and 4) was not modified. Doses of 10 and 20 mg also caused slight but significant decreases in REM sleep.
The value of studies involving asymptomatic volunteers has qualified significance inasmuch as their sleep variables are within normal limits. However, these investigations, which are based on relatively small samples, have a rather preliminary character and only a limited number of dependent measures. Nevertheless, they show that 5 to 10 mg zolpidem can induce objectively demonstrable hypnotic effects that only slightly disrupt sleep architecture in subjects with normal sleep.
The effects of zolpidem on transient insomnia have been studied using the so-called
Walsh et al 72 made use of a 3-hour phase-shift in sleep time as a model of transient insomnia. Slightly fewer than 50% of the subjects with normal sleep did show transient sleep disturbance. In the group whose sleep was significantly altered, zolpidem (5, 10, 15 or 20 mg) reduced LPS, WASO, stage 1 sleep and the percentage of REM sleep, whereas TST and stage 2 sleep were augmented relative to placebo. Subjective estimates of SOL and TST closely corresponded to objective measures. Erman et al 73 combined the first night effect and a 2-hour phase advance in a single night laboratory-based study protocol, to compare the effects of zolpidem 10 mg or placebo on sleep variables in 358 healthy young adults. Zolpidem significantly reduced the number of awakenings and WASO and additionally increased SE, whereas the percentage of time spent in different sleep stages was not modified. Subjective outcome measures, including SOL, number of awakenings, WASO, TST, and quality of sleep, were also significantly superior to placebo.
The efficacy of zolpidem for facilitating sleep initiation and sleep maintenance as well as for enhancing subjective daytime alertness and performance following a rapid travel across 5-9 time zones (jet lag) was evaluated in a double-blind study by Jamieson et al. 74 Zolpidem 10 mg or placebo were administered for 3 consecutive nights following an eastward bound transatlantic flight. Compared to placebo, zolpidem significantly increased TST (night 1), improved sleep quality (nights 1, 2 and 3), and reduced the number of nocturnal awakenings (nights 1 and 2), whereas SOL and WASO did not differ significantly.
In summary, zolpidem tended to improve disturbed sleep in several models of transient insomnia in normal sleepers. Disregarding the doses used, the most consistent findings included reductions in SOL, number of awakenings and WASO, and increases in TST, SE and quality of sleep. However, in several instances, the subjects spent significantly less time in REM sleep.
Zolpidem may, therefore, be useful in treating transient insomnia associated with acute stress, travel across time zones or sleeping in unfamiliar surroundings.
Zolpidem IR in sleep pathology–-poor sleepers and chronic insomniacs
According to Declerck, 75 complaints of poor quality sleep occur frequently in general medical practice, accounting for 4%-5% of all consultations. Poor sleepers generally do not consider themselves to be insomniacs, despite having difficulties in getting to sleep and staying asleep. They usually have no significant psychological symptoms and seldom take hypnotic medication. 75 Three studies have assessed the efficacy of zolpidem on disrupted sleep of poor sleepers.76–78 In all these studies, investigators made use of a sleep laboratory to obtain objective evidence of zolpidem's effects on sleep induction, continuity measures, and sleep architecture. Taken together, these findings indicate that zolpidem 10 mg can significantly reduce stage 2 SOL and WASO (two studies), and increase TST (three studies) in poor sleepers. In two of the investigations, however, the increase in TST only occurred on a limited number of nights. Concerning sleep architecture, stage 1 sleep was not significantly modified (two studies), whereas stage 2 sleep and SWS were increased (two studies). Zolpidem's effects on REM sleep duration, however, were not entirely consistent. Although this parameter was increased after 10 mg zolpidem in one study, a 20 mg dose of zolpidem was found to reduce REM sleep time and to lengthen REMOL in another one. Rebound insomnia was reported for the first withdrawal night (one report). Rebound effects upon discontinuation were also observed in another study. 79
Among women with subjective complaints of insomnia quantitative EEG analysis did not reveal significant differences between zolpidem and placebo concerning nocturnal delta activity, which progressively declined in amplitude and duration in either case. 80 Benoit et al 81 also found that zolpidem had almost no influence on nocturnal delta activity, even in young individuals with longstanding histories of poor sleep.
Considerable research attention has been given to the suitability of zolpidem IR for chronic insomnia, representing the most challenging sleep disorder. The effectiveness of zolpidem IR in this regard has been investigated in 13 studies involving a total of 1757 outpatients and 1807 inpatients.11,12,82–92 Six of the studies used PSG and either a single-blind or double-blind experimental design to evaluate the effect of zolpidem IR 10 mg on sleep quality in patients with chronic primary insomnia. The drug was administered for periods ranging from 7 to 35 nights. In most of the studies, the patients had not been taking hypnotic medication prior to the study for periods ranging from 4 to 30 nights. The outcome was, again, heterogeneous: (1) stage 2 SOL was reduced in three studies (range: 13.0-40.1 min); (2) WASO was decreased in two studies (1 missing value); (3) the number of nocturnal awakenings was not significantly modified (2 missing values); (4) values corresponding to TST were found to be increased in three studies (range: 32.0-62.5 min) (1 missing value); (5) SE was increased in three studies (range: 3.0%-8.3%) (1 missing value). NREM sleep architecture findings were: stage 2 sleep was increased in three studies; stage 3 and stage 4 sleep were not significantly modified (3 missing values), whereas total SWS (stage 3 + stage 4) was increased in two studies (2 missing values). In relation to REM sleep variables, REM latency was increased in one study whereas REM sleep, in min or as percentage of TST, was not significantly modified (1 missing value). More recent studies have mainly focussed on the comparison with other hypnotics93–95 or were designed for testing EEG methodologies, 96 but despite these differences in emphasis their findings were consistent with those of the earlier data.
Spectral EEG analyses in patients with moderate-to-severe chronic primary insomnia under treatment with daily doses of 10 mg zolpidem for 15 nights showed only moderate effects. 97 Power density of NREM sleep was significantly increased in the delta (0.25-1.0 Hz) band only during the first two-hour interval, both during short- and intermediate-term treatments. In contrast to BZD hypnotics, zolpidem did not suppress low frequency EEG activity in poor sleepers nor in patients with chronic primary insomnia.
In chronic primary insomnia, both the polysomnographic and EEG spectral studies demonstrated predominantly early nocturnal effects, findings that are consistent with the known pharmacokinetics of zolpidem. Results with zolpidem IR 10 mg indicate that the compound is capable of reducing stage 2 SOL and WASO, and, with certain qualifications, can augment TST and SE. With respect to sleep architecture, zolpidem tended to increase stage 2 sleep and SWS while REM sleep was not significantly modified. In summary, these results demonstrate limited suitability of zolpidem IR for treating chronic insomnia, especially in improving sleep induction. With regard to other objective measures, beneficial effects were largely restricted to statistical tendencies or not confirmed in all studies. However, the subjective rating by patients or caregivers was sometimes better than the outcome of the objective parameters (details not included). However, some of the studies are affected by methodological shortcomings such as inadequate sample sizes as well as confounding variables such as age, gender, diagnostic criteria and the simultaneous administration of other psychotropic drugs.
The predominant reduction of stage 2 SOL or WASO during the first part of the night, in conjunction with few or no reductions in the number of nocturnal awakenings, is most easily interpreted to be the result of zolpidem's less than ideal pharmacokinetics, i.e. its short elimination half-life and lack of active metabolites. This would also explain the inconsistencies observed in supporting sleep maintenance. However, the subjective feeling of being more refreshed on awakening, as reported by many patients, should not be underrated, even when not supported by objective measures. Therefore, the development of ER formulations of zolpidem appeared to have therapeutic potential, or was at least justified for improving its pharmacokinetic profile.
Long-term nightly versus non-nightly administration
Despite the proliferation of numerous sleeping aids, the development of a new sleep promoting agent is justified in view of the disruptiveness of primary chronic insomnia to overall health, as well as by the resistance of the condition to long-term treatment with classic hypnotics, which, additionally, are often linked to undesirable side effects. The long-term use of hypnotic drugs has been generally discouraged on the grounds of risk of rebound insomnia, withdrawal reactions and/or dependence.98–101 In fact, this recommendation is mainly relevant to the BZDs. The recommendation has nevertheless been extended to the recently introduced non-BZD hypnotics, which have only a low potential of causing rebound insomnia, dependence, or other adverse effects on health.102,103 In this respect, evidence from accumulated clinical practice and controlled studies tends to indicate that long-term pharmacological treatment of insomnia with zolpidem is efficacious and safe.104–106 More recent studies on zolpidem ER over 6 months support this conclusion, as will be discussed below.
Sateia and Pigeon 107 have proposed that long-term treatment with hypnotic medications could be used in patients with persistent insomnia, given the qualification that it is not secondary to mental disorders, neurological diseases, medical conditions, nor the effect of a substance of abuse or medication. In addition, advocacy of this approach is restricted to patients for whom all non-pharmacological therapies have proven to be ineffective. Circumstances in which the long-term administration of hypnotic drugs must be discontinued include the development of tolerance and dose escalation, the occurrence of severe adverse events, and the diagnosis of newly developed disabilities.
Non-nightly hypnotic administration has been proposed as an alternative option to nightly drug intake. To date, information on the efficacy and safety of non-nightly intake of hypnotic medication is available only for zolpidem. Administration of 10 mg zolpidem tartrate IR for five nights followed by two nights of placebo per week for two weeks induced an improvement of sleep that was comparable to nightly zolpidem treatment in patients with chronic insomnia. Rebound insomnia did not occur on the nights during which zolpidem was substituted by a placebo. 108 Nightly and non-nightly zolpidem treatment were compared using the same design except that the two placebo nights per week were randomly assigned. Again, improvements in sleep quality were equivalent in the two groups of patients.109,110 More recently, zolpidem IR 10 mg or placebo were administered for 12 weeks to patients with a diagnosis of chronic primary insomnia. The patients were instructed to take no fewer than three and no more that five pills per week. Sleep was evaluated daily with sleep diaries. Patients receiving zolpidem exhibited an improvement of sleep induction and maintenance that persisted over time. There was no evidence of either rebound insomnia or dose escalation. 106 Although a thorough comparison between the single-dose studies and those with long-term treatment would be required to convincingly judge the progress, the currently available data suggest that long-term non-nightly administration of zolpidem (10 mg) for patients with chronic primary insomnia is an effective and more conservative alternative to its continued nightly use or infrequent repetitions of zolpidem for recurrent symptoms of insomnia.
Zolpidem extended release
While early research had demonstrated the zolpidem IR's potential for sleep induction, as well as its effectiveness during the initial hours following sleep onset, 102 the assumption that an ER formulation might improve sleep maintenance throughout the night had to be thoroughly tested. In particular, the superiority of zolpidem ER over zolpidem IR should have been of interest, but most clinical studies of the ER formulation were limited to comparisons with placebo. The relative efficacy of zolpidem ER vs. IR vs. placebo has been particularly studied in healthy subjects. In 54 subjects who had been awakened 3, 4 or 5 h after adminstration of the tablet and exposed to a noise interfering with resumption of sleep, PSG measurements taken among the 4- or 5-h post-dose groups showed greater improvements of LPS associated with zolpidem ER compared to IR. 111 Findings were consistent with sedation and cognitive impairment data during awakenings. Another double-blind ER/IR comparison was conducted by Greenblatt et al 40 in 70 healthy young subjects. Apart from the pharmacokinetic differences, EEG data on the β amplitude demonstrated a significantly stronger effect of ER compared to IR, for both 0-8 h and 3-6 h after sleep onset.
Several placebo-controlled studies have been conducted in patients with primary chronic insomnia. In 212 middle-aged subjects (completed by 192 patients) treated nightly for 3 weeks, 112 PSG (on nights 1/2 and 15/16) and subjective measures (several nights) revealed often highly significant improvements in sleep quality, in particular with respect to LPS, WASO during first 6 h of sleep, PSG sleep efficiency, as well as self-reported SOL, WASO, number of awakenings and TST. In another study comprising 1025 insomnia patients, based on self-ratings,113,114 subjective SOL, WASO, number of awakenings and sleep quality were improved at a high significance level, as possible with the large samples. The efficacy of zolpidem ER was confirmed in a 6-months randomized, double-blind Phase IIIb trial on 1018 outpatients diagnosed with primary chronic insomnia,115,116 on the basis of subjective measures (Patients Global Impression and Clinical Global Impression-Improvement). Patients taking the drug 3-7 times per week showed consistent improvements in SOL, TST, number of awakenings, WASO and sleep quality, and also reported sustained improvements concerning morning sleepiness and ability to concentrate. In view of the usual difficulties in long-term management of chronic insomnia, these findings are significant, especially because of the efficacy over a relatively long period of time. In elderly patients with primary chronic insomnia, the low-dose formulation of zolpidem ER 6.25 mg proved to be similarly effective, in 3-week studies using both PSG and subjective measures,117–119 in accordance with earlier data of the Ambien-CR Prescribing Information. 120
In addition to subjective sleep parameters, a study in 752 otherwise healthy employees with chronic insomnia 121 investigated the effect of zolpidem ER 12.5 mg on work efficiency, using a standardized Work Limitations Questionnaire (WLQ). Throughout 24 weeks, highly significant improvements were reported for the WLQ parameters of “Time Management” and “Work Output.”
The efficacy of zolpidem ER was also demonstrated in patients with comorbid insomnia and major depressive disorder (MDD)122,123 or generalized anxiety disorder (GAD), 124 in either case with concurrent escitalopram therapy. In a multicenter, double-blind, randomized and placebo-controlled Phase I and II study, 122 adult MDD patients (21-64 years) were investigated (Phase I: 8 weeks, completed by 119 subjects treated with zolpidem ER/escitalopram and 125 with placebo/escitalopram; Phase II: additional 16 weeks, completed by 67 and 60 patients, respectively). Significant improvements were observed in most relevant subjective sleep measures (TST, WASO, nocturnal awakenings; sleep quality, SOL in Phase I, but not Phase II), and also in parameters of next-day functioning (morning energy, sleep impact on daily activities; and, in Phase I, morning concentration). No rebound insomnia occurred after discontinuation. Depressive symptoms remained unchanged. Another randomized Phase I and II trial in 379 MDD patients, using the Sleep Impact Scale (SIS), showed likewise improvements of sleep impact. 123
In GAD patients (21-64 years), a multicenter, double-blind, placebo-controlled study of 8 weeks duration (completed by 116 subjects treated with zolpidem ER/escitalopram and 126 with placebo/escitalopram), also demonstrated significant improvements in TST, WASO, SOL, sleep efficiency, and also next-day morning energy, morning concentration, and sleep impact on daily activities. 124 Again, no rebound insomnia was observed, and no substantial change in the psychiatric symptoms.
Safety and Adverse Effects
With regard to the relatively similar phamacokinetic time courses of zolpidem IR and ER, adverse effects should be expected to be mainly the same, except for the possibility of differences seen on the next day. In fact, the safety profiles of the two formulations were very similar.125,126 The most commonly observed adverse events associated with the use of zolpidem IR had been headache, drowsiness, dizziness, nausea, diarrhea, and myalgia, but these complaints occurred only with moderate frequency. Headaches and dizziness were sometimes even less than in placebo groups. 114 The nature of adverse events reported in adult patients receiving zolpidem IR 10 mg was similar to that described in studies that included elderly patients being given zolpidem IR 5 mg.53,127 The most commonly observed adverse events observed in adults and elderly patients treated with zolpidem ER (at daily doses of 12.5 and 6.25 mg, respectively) were next-day somnolence, headaches, and dizziness.7,99,120
As in most medications, strict contraindications for zolpidem ER exist only for hypersensitivity to the drug itself or to other ingredients of the tablets. 21 Rare cases of presumably anaphylactic responses with angioedema, dyspnea, nausea, eventually throat closing have been observed, 21 but these should be regarded as exceptional and are found with numerous medications. It should be noted that the experience with zolpidem and concomitant systemic illnesses is still rather limited. Nevertheless, several reasons for precaution have to be considered and require surveillance of the patient.7,21 These include hepatic impairment (including reduced hepatic metabolism in the elderly), concomitant use of CYP3A4 inhibitors, respiratory diseases, as with sedative/hypnotics in general, (especially sleep apnea and chronic obstructive pulmonary disease), pregnancy category C, concomitant use of CNS depressants, worsening of psychiatric disorders (in particular, depression), and, individually possible but nevertheless rare, withdrawal effects. Drug interactions are known or can be expected for substances interfering with CYP3A4, such as the inhibitors ciprofloxacin, clarithromycin, erythromycin, doxycycline, fluoxetine, fluvoxamine, isoniazid, or verapamil, which lead to reduced metabolic clearance, whereas CYP3A4 inducers such as carbamazepine, phenobarbital, phenytoin or rifampicin (rifampin) can substantially decrease plasma levels and efficacy of zolpidem.7,21,128 Rifampicin given 17 h before zolpidem caused reductions of AUC by up to 73%. The relevance of interactions mentioned can be individually variable, especially when the main effects of the drugs are CNS targeted. This is the case with, e.g. fluoxetine, which only moderately enhances blood levels of zolpidem,129,130 but, in its capacity as a serotonin uptake inhibitor, can lead, when coadministered with zolpidem, to hallucinations in individual cases.131,132 This combination may also favor dependence, 133 which is otherwise rare with zolpidem. Concomitant use of zolpidem and ethanol has shown additive effects, 128 findings which are in accordance with the actions of these compounds at the GABAA receptor level in animal studies. 134
No indications exist for mutagenic, carcinogenic or reproductive effects of zolpidem, and no teratogenic potential was demonstrable during prenatal development, but some effects of reduced ossification were detected at high doses in rats, which gave rise to the recommended precaution in pregnancy category C. 21 In rats, the no-effect dose of embryo/fetal toxicity was 4 mg zolpidem base/(kg × d).
Zolpidem contrasts with BDZs regarding aftereffects, behavioral and cognitive impairments. At recommended doses, zolpidem IR usually causes only minimal rebound insomnia.49,98 On the other hand, there is objective evidence of impaired sleep in the elderly on the first post-treatment night at levels above the 5 mg dose, which is recommended for this clinical group.135,136 Rebound insomnia has been described during the first night after abrupt discontinuation of zolpidem ER 12.5 mg in adult patients with chronic primary insomnia. 112
Impairment of short-term memory, which is a particularly undesirable effect of BZD hypnotics, has been also investigated in the case of zolpidem. Controlled studies in adults utilizing objective measures of memory yielded no consistent evidence of next-day memory impairment following the administration of zolpidem IR 10 mg. 53 At recommeded doses of 12.5 or 6.25 mg for middle-aged or elderly patients, respectively, zolpidem ER did not decrease vigilance, memory, cognitive or psychomotor functions 8 h post-dose. 21
Short-term and long-term use of zolpidem 10 mg/day has not been shown to produce tolerance.87,137,138 In addition, the potential for tolerance with zolpidem 10 mg/day used as needed is very low.106,139
There is no clear evidence that abrupt discontinuation of zolpidem generally promotes withdrawal symptoms, although rebound insomnia has been observed in some cases, as mentioned above. Nevertheless, discontinuation has occasionally been linked to symptoms of fatigue, nausea, flushing, lightheadedness, emesis, stomach cramps, panic attacks, nervousness, and abdominal discomfort, but, these symptoms by far have not been generally experienced. In view of zolpidem's lack of or rarely demonstrated tolerance and withdrawal effects, its dependence and abuse potential are low. Nevertheless, cases of abuse and dependence have been described.110,140,141 In patients with a history of drug abuse, the effect of zolpidem 40 mg was similar to that of diazepam 20 mg. Notwithstanding the above, zolpidem abuse and dependence are rare, and have been seen mainly in drug abusers with or without a concomitant psychiatric disorder. 110
Several case reports have described behavioral changes caused by zolpidem, including the occurrence of bizarre behaviors, agitation and sleep-related complex behaviors, such sleep eating, sleepwalking with object manipulation, sleep conversations, sleep driving, sleep sex and sleep shopping, frequently with amnesia for the event.142–151 There is some indication for a higher probability of behavioral abnormalities with zolpidem ER than IR. 148
Behavioral changes as described are not a peculiarity of zolpidem. In fact, they are not uncommon with GABAergic drugs in general, and are relatively frequently observed with BZDs as well, although the spectrum of effects may vary among agents. 149 One may be inclined to regard these phenomena as rare, and most of them represent, in fact, single cases without general importance for the usefulness of zolpidem. However, in a recent retrospective evaluation of 255 Taiwanese patients treated with zolpidem, 151 complaints were reported by 5.1% concerning somnambulance and other unusal sleep-related behavior.
With these exceptions, which may exclude a certain subpopulation from treatment with zolpidem, the general and overwhelming impression remains that zolpidem, whether IR or ER, is a remarkably safe hypnotic, having few adverse effects in conjunction with considerable efficacy relative to other options in sleep medicine, especially in comparison to BDZs.
Efficacy Relative to other Sedative/Hypnotics
To date, comparative studies with other sedative/hypnotics are limited to zolpidem IR, so that the advances by the longer-acting formulation cannot be definitely judged on this basis. 7
The hypnotic effect of zolpidem IR with long-acting (flurazepam), intermediate-acting (flunitrazepam, temazepam), and short-acting (triazolam) BZD hypnotics have been compared in a number of trials. Zolpidem 10 mg or 20 mg was more effective than flurazepam 30 mg for sleep outcomes in patients with chronic insomnia. However, zolpidem 20 mg was associated with more adverse events. 152 In one study comparing zolpidem 20 mg to flunitrazepam 2 mg and placebo in healthy young subjects, both drugs improved the subjective assessment of the ease of getting to sleep. In contrast to zolpidem, the BZD derivative impaired performance on tests of memory and psychomotor coordination and increased the subjective rating of sleepiness. 153 In one study of zolpidem 10 mg versus flunitrazepam 2 mg and placebo, both zolpidem and flunitrazepam similarly reduced stage 2 SOL in subjects with mild insomnia. In addition, zolpidem decreased WASO and REM sleep percentage and increased slow wave sleep during the first 2 hours of the recording. Flunitrazepam, however, reduced stage 1 sleep during the first 2 hours, increased stage 2 sleep during the second 2-hour period and decreased REM sleep for the three 2-hour recording periods. The maximum delta activity was also significantly decreased. 80 In another study of chronic primary insomnia, zolpidem 10 mg was reported to be inefficient, whereas flunitrazepam 1 mg improved sleep maintenance, as judged by the increase of total sleep time and sleep efficiency. Flunitrazepam augmented stage 2 sleep and decreased slow wave sleep and REM sleep, but also significantly impaired attention and memory relative to zolpidem and placebo, while zolpidem did not differ in this regard from placebo. 154 At a higher dose of 20 mg, zolpidem IR was similarly effective as flunitrazepam 2 mg, in patients with moderate to severe comorbid insomnia precipitated by neuropsychiatric disorders. Both hypnotics produced remarkable improvements in SOL and TST, number of awakenings and WASO, without any significant difference between the active medications for the variables studied. In this case, neither zolpidem nor flunitrazepam induced alterations in anterograde memory nor in morning psychomotor performance. 155
In non-elderly patients with chronic insomnia, zolpidem 10 mg and temazepam 20 mg significantly reduced SOL and number of awakenings, increased TST to a similar extent and had comparable effects regarding rebound insomnia. 156 In elderly subjects with chronic insomnia, zolpidem 5 mg and temazepam 15 mg had a similar outcome concerning efficacy and safety during a 28 days administration period. There was no evidence of loss of effect vs. placebo over time for any of the two treatment groups. 157
Several studies compared zolpidem with triazolam. Kanno et al 158 reported that zolpidem 10 mg increased SWS, reduced total wake time and stage 1 sleep, whereas triazolam 0.25 mg increased stage 2 sleep in subjects with normal sleep. After withdrawal, triazolam, but not zolpidem, increased wake time and REM sleep and caused worsening of the mood in the morning. Parrino et al 159 tested the efficacy of zolpidem 10 mg and triazolam 0.25 mg on both conventional and cyclic alternating pattern (CAP) parameters under basal and perturbed environmental conditions (acoustic perturbation produced by a continuous 55 dB white noise). In the placebo condition, acoustic perturbation produced a situational insomnia. Zolpidem induced a significant decrease of WASO during the nights with experimental noise disturbance. In addition, CAP time and rate were significantly lower during zolpidem and triazolam administration. In two other trials of zolpidem 10 mg compared to triazolam 0.25 or 0.5 mg, both hypnotic drugs showed significant efficacy compared to placebo in poor sleepers and in patients with chronic primary insomnia. SOL, TST, sleep efficiency, WASO and number of awakenings were improved after zolpidem 10 mg and triazolam 0.25 mg in poor sleepers. Slow wave sleep was increased after zolpidem but reduced after triazolam. 160 Zolpidem 10 mg and triazolam 0.5 mg also increased TST in patients with chronic primary insomnia. This increase observed in the zolpidem group was associated with a greater number of sleep cycles. In addition, zolpidem decreased total wake time, whereas triazolam augmented REM latency. 161 When the active treatment was discontinued, pronounced symptoms of rebound insomnia were observed in the triazolam group, but not with zolpidem. In patients with comorbid insomnia, zolpidem 20 mg and triazolam 0.5 mg were similar in efficacy and occurrence of adverse events during 3 months of administration. 162 Similar results were also obtained with zolpidem 5 mg and triazolam 0.125 mg in elderly patients with chronic insomnia. 163 In contrast, Ochs et al 164 found zolpidem 5 mg to be more effective than triazolam 0.125 mg in elderly subjects with long-term insomnia.
In conclusion, most comparisons that included poor sleepers and patients with primary and secondary insomnia found zolpidem to be similar to BZDs in efficacy, but superior to BZD's in terms of adverse events.
Several studies have compared zolpidem with other z-drugs. One crossover trial of three nights of treatment in healthy young subjects found similar rises in SWS by zolpidem 10 mg and zopiclone 7.5 mg and no differences concerning mood and physical status in the morning, while slow wave sleep was significantly increased; however, zopiclone augmented stage 2 sleep and reduced TST and stage 1 sleep. 165 In the above-mentioned study by Parrino et al, 159 zopiclone 7.5 mg was also included. Zolpidem and zopiclone similarly reduced sleep fragmentation, as judged by a significant decrease of WASO. Moreover, zolpidem diminished sleep instability, as shown by significantly lower values of CAP time and rate. Tsutsui et al 166 performed a two-week, doubleblind trial of zolpidem 10 mg versus zopiclone 7.5 mg in 479 patients with chronic primary insomnia. Again, sleep outcomes were significantly improved relative to placebo by the two hypnotics to an almost similar extent. However, more patients medicated with zopiclone had rebound insomnia and experienced drug-related adverse events. Erman 167 compared the hypnotic efficacy of eszopiclone, the S-isomer of racemic zopiclone, to zolpidem and placebo in patients with chronic primary insomnia. Eszopiclone 2.5 and 3 mg and zolpidem 10 mg were more effective than placebo for objective SOL and TST. Eszopiclone, in addition, significantly reduced WASO. Subjective measures of next-day effects including morning sleepiness, daytime alertness, and daytime ability to function did not differ among the hypnotic drugs. In conclusion, the IR formulations gave similar results on most parameters, but the cyclopyrrolones were more effective in lengthening TST, which might be related to differences in mean elimination half-life between drugs.
Comparisons with zaleplon (5, 10, or 20 mg) were conducted by Elie et al 135 and Fry et al 136 in adults under age 65 with chronic insomnia. SOL was significantly lower with zaleplon 10 and 20 mg and zolpidem 10 mg compared to placebo at most timepoints during 4 weeks of treatment. While zaleplon 5 and 10 mg did not differ from placebo in number of awakenings, sleep duration, and sleep quality, zaleplon 20 mg and zolpidem 10 mg significantly improved sleep duration and sleep quality, but some rebound insomnia and withdrawal symptoms were observed after discontinuation of zolpidem treatment. In elderly patients, 65 years of age and older, with chronic primary insomnia, zaleplon 5 and 10 mg was compared to zolpidem 5 mg. Zaleplon 10 mg and zolpidem 5 mg significantly reduced SOL during 14 nights of the doubleblind treatment, whereas zaleplon 5 mg was effective during week 2 only. Subjective TST and sleep quality were improved with zolpidem 5 mg and zaleplon 10 mg, the latter during week 1 only. Again, rebound effects were observed after discontinuation of zolpidem. 79 In summary, zolpidem was moderately superior to zaleplon, at recommended starting doses, in both adult and elderly patients with chronic insomnia.
Place in Therapy
Compared to other sedative/hypnotics of the BZD group, zolpidem IR and ER clearly have advantages concerning next-day hangover effects, and considerably milder or less frequent rebound insomnia, development of tolerance, potential for dependence and abuse. These advantages are widely shared with other z-drugs, with some variations as discussed. Since the differences observed are, at least, partially due to those in bioavailability, the longer-acting ER formulation of zolpidem may have some disadvantages relative to IR. This may concern next-day effects–-which are, however, moderate–-and behavioral changes–-representing uncommon, but not negligible phenomena, concerning presumably only a certain subgroup of patients. These, assumed or real, disadvantages are certainly balanced out for patients with discontinuous sleep by the improvements in supporting sleep maintenance.
With regard to recommendations in the treatment of insomnia, one should have to distinguish between the different causes and clinical phenomenology of the various forms of this disorder. First, one should discriminate insomnia because of circadian disturbances and those of other etiology. If sleep difficulties are induced by circadian phase shifts (jet lag or shift work), by weak coupling of the endogenous oscillators to the environmental cycles or by abnormal endogenous period lengths, the use of melatoninergic drugs such as ramelteon, or melatonin extended release, which act as chronobiotics, may be more recommendable.168,169 If sleep disturbances are primarily associated with psychiatric disorders, in particular, depression, the usefulness of hypnotics needs to be carefully monitored, and interference with antidepressants has to be taken into consideration. Zolpidem may be used, and has already been successfully tested in MDD122,123 and GAD, 124 but due caution is still recommended for long-term treatment.7,21 In these cases, a drug like agomelatine may be an alternate medication of choice, inasmuch as it combines sleep-inducing, melatoninergic properties with actions of an antidepressant.169–171
In the other forms of insomnia, zolpidem is an effective option with a reduced risk for dependence and rebound insomnia. It is nearly free of hangover effects and does not require dose escalation during prolonged treatment. However, one should distinguish between IR and ER formulations. If sleep disturbances mainly concern sleep initiation, zolpidem IR 10 mg (or 5 mg in the elderly) should be fully sufficient, since the rise in plasma concentration is almost the same in the two formulations (cf. previous section). In addition to the possibility that undesired behavioral changes may be more frequent in the ER formulation, cost considerations could be also relevant. Zolpidem IR is meanwhile not only available as Ambien® 10 or 5 mg, but also as generic (Ivedal®, Nytamel®, Stilnoct®, Stilnox®, Zoldem®, Zolnod®, Zolpihexal®), and these preparations are considerably less expensive than the newly developed, patented Ambien CR™.
However, the situation is entirely different if the therapeutic objective is to improve sleep maintenance. In this case, the ER formulation is clearly indicated. Although more comparative studies on IR vs. ER would be desirable, the trials in which the two formulations have been investigated separately (see previous section) collectively indicate that sleep maintenance is more efficiently treated by zolpidem ER, including primary chronic insomnia. With regard to their side- and aftereffects, BZDs should be prescribed only if zolpidem ER fails.
Conclusions
The evaluation and management of insomnia is often challenging. Insomnia is a multidimensional disorder, and consequently any approach to its management should consider a combination of both pharmacological and nonpharmacological measures. In practice, pharmacological approaches tend to be used with greater frequency than psychotherapy and other treatment methods. In patients with chronic insomnia and a coexisting psychiatric, neurologic or medical condition, the underlying disorder needs to be treated appropriately.
In this regard, the imidazopyridine zolpidem has various advantages. Its higher selectivity towards α1 subunits of the GABAA receptor can be identified as cause of more specific sleep-related actions and reduction of some undesired adverse effects.
Oral zolpidem is a fast-acting drug, which is rapidly absorbed and extensively distributed to body tissues including the brain. Zolpidem is almost quantitatively metabolically cleared in the liver by cytochrome P450 isoenzymes to inactive metabolites. Therefore, renal failure does not substantially modify zolpidem's pharmacokinetic profile, but hepatic impairment is a matter of concern.
While zolpidem IR is mainly effective in treating sleep initiation difficulties, zolpidem ER is additionally suitable in supporting sleep maintenance and, therefore, an option in primary chronic insomnia. Various populations have been included in the studies that assessed the efficacy and safety of zolpidem: subjects with transient insomnia, poor sleepers, non-elderly and elderly patients with chronic primary insomnia, and patients with secondary or comorbid insomnia. Zolpidem, unlike BZD hypnotics, only moderately modifies SWS (stages 3 and 4) and REM sleep. Adverse effects are certainly less abundant and less severe than with BZDs. Abuse and dependence are rare and have been described mainly in drug abusers with or without a concomitant psychiatric disorder. A possible matter of concern has more recently arisen in terms of some behavioral changes reported for a small subpopulation. However, zolpidem ER–-and, where only the goal of sleep initiation is required, zolpidem IR–-can be recommended as both efficacious and reasonably safe medications.
Competing Interest Statement
The authors have declared association with the following companies:
S.R. Pandi-Perumal is a stockholder and the President and Chief Executive Officer of Somnogen Inc., a New York Corporation. He has declared no competing interests that might be perceived to influence the content of this article.
Dr. Salomon Z. Langer is a stockholder and the President and Chief Executive Officer of Euthymia Ltd. He has declared no competing interests that might be perceived to influence the content of this article.
All authors further declare that they have no proprietary, financial, professional, nor any other personal competing interest of any nature or kind in any product or services and/or company that could be construed to be a potential conflict of interest that might have influenced the views expressed in this manuscript.
