Abstract
With approximately 240,890 new cases expected in 2011, prostate cancer remains the leading cause of non-melanoma cancer deaths in men. Immunotherapies using viral vector-based delivery systems targeting tumor-specific antigens are being studied. Viral vector-based delivery systems present tumor-targeted antigens (TAAs) to the immune system while breaking self-tolerance. Modified vaccinia ankara has been combined with the oncofetal antigen 5T4 to create TroVax for the treatment of castration-resistant prostate cancer (CRPC). The 5T4 antigen is highly expressed in a large number of carcinomas, including prostate cancer, but is rarely expressed in healthy tissue. TroVax has been demonstrated to be safe and highly immunogenic, both as monotherapy and in combination with other standard of care therapies in colorectal, renal cell, and prostate cancer. With minimal side effects and the ability to produce a strong immunogenic response, TroVax (MVA-5T4) is a viable addition to the treatment of prostate cancer.
Introduction
Prostate cancer is the leading cause of non-melanoma cancer deaths in men with an estimated 240,890 new cases for 2011. 1 The largest challenge lies with treating patients who develop castration-resistant prostate cancer (CRPC). Treatment for prostate cancer consists of prostatectomy, chemotherapy, and radiotherapy. More recently, immunotherapy has become of particular interest as primary therapy or combination therapy. The first and only Food and Drug Administration (FDA) vaccine for prostate cancer to date is sipuleucel-T (Provenge). In patients with asymptomatic or minimally symptomatic CRPC, sipuleucel-T has been shown to extend median survival to 25.8 months from 21.7 months. 2
Cancer vaccine development has explored using viral vectors to elicit an immune response against tumor-specific antigens.3,4 Research in viral vector-based therapy diverges into two thrusts: (1) identifying the optimal tumor associated antigens (TAAs) and (2) identifying the most suitable delivery system. Cancer cells generally show some degree of genetic instability, which can produce an array of aberrant proteins that are hypo- or hyperglycosylated and highly expressed or expressed at the wrong development stage. These proteins can be used as antigens to target tumor cells. Optimal TAAs are minimally, if at all, expressed in normal tissue and have homogenous high-level expression on the surface of a broad range of tumors. TAAs that are associated in some way with disease progression are of particular interest because of their ability to induce an immune response and also actively interfere with tumorigenesis.
Once the appropriate TAAs are identified, the focus becomes finding the most suitable viral vector-based therapy to present the TAAs to elicit immunity. Although a wide variety of viral vectors have been studied, the best vectors induce potent, long-lasting cellular and humoral response to the TAA across a wide patient population without compromising the patient's health and safety. Vaccinia virus, a member of the poxvirus family, has been developed as a recombinant expression vector.5,6 In animal studies, the vaccinia virus has been shown to produce both antibody and CTL responses to the exogenous proteins, which aids in creating an optimal environment for the induction of an efficacious immune response. The proteins expressed by carcinomas are usually presented to the immune system as self-proteins that consequently elicit no immune response. Recombinant vaccine viruses expressing murine homologues of TAA, which are classified as self-antigens, have been shown to induce TAA-specific immune response in animal models, thus illustrating that such constructs are able to overcome immune tolerance to self-antigens. 7 Viral delivery systems help break self-tolerance and trigger an immune response to the TAA. Such responses are able to prevent tumor establishment and, in some cases, are able to actively treat established tumors. Recombinant vaccinia viruses expressing the self-antigen CEA has been constructed and have been evaluated for toxicity and, to a lesser extent, efficacy in late stage colorectal cancer. 8 These viruses were found to be well tolerated and both antibody and cellular immune responses to the self- antigen CEA were reported. 9
The modified vaccinia ankara (MVA) virus has been extensively studied as a viral vector delivery system that has a well-documented safety profile and proven ability to generate a potent immune response. The antigen 5T4 is normally expressed in the placenta and rarely in fully developed healthy tissues; however, it is highly expressed in a range of human carcinomas, including prostate cancer.10–12 Research about the 5T4 antigen has linked it to altering cell adhesion, motility, and morphology. The oncofetal antigen 5T4 has been combined with the MVA delivery virus the formation of TroVax (Oxford BioMedica). Consistent with histology studies in other epithelial cancers, significant expression of 5T4 has been detected in the majority of primary prostate cancers (16/19, 84%) studied. Unlike other self-antigen TAAs (eg, CEA), 5T4 expression appears to be tumor specific with only low level expression reported in the gut. Tumor cells are considered poor immunogens. Although there is no single known mechanism to explain poor tumor immunogenicity in all experimental models studied, the molecular basis can be conceptually into 4 distinct groupings: (1) lack of expression of co-stimulatory molecules essential for effective immune induction, (2) production of immune-inhibitory substances, (3) poor antigen processing and presentation, and (4) variability in the expression of antigen by tumors. The first two groups are shared with normal cells in the body; however, the latter two have to do with the inherent genetic instability of cancer cells.
Prostate Cancer
Prostate cancer is the most common non-cutaneous cancer in men. Approximately 1 in 6 men will be diagnosed with prostate cancer during their lifetime. 13 In 2011, an estimated 240,890 new cases of prostate cancer will be diagnosed, and an estimated 33,720 men will die from advanced prostate cancer. While early stage therapies such as prostatectomy and radiotherapy are successful in about 80% of patients,14,15 the remaining 20% of patients and patients with advanced stages of prostate cancer undergo hormonal therapies. Patients who fail hormonal therapy and reach CRPC disease state have a limited number of approved treatment options available.
Currently, there are 4 methods of treatment: (1) immunotherapies including sipuleucel-T, which was approved in 2010, (2) androgen deprivation through bilateral orchiectomy, Luteinizing hormone-releasing hormone (LHRH) agonists, or Gonadotropin releasing hormone (GnRH) agonists, (3) chemotherapy including docetaxel, which was approved in 2004 and cabazitaxel, approved in 2010, and (4) second-line hormone manipulation using first-generation androgen-receptor antagonist, steroid hydrolyase inhibitor such as abiraterone (approved in 2011) with prednisone. Only docetaxel, abiraterone, cabazitaxel, and sipuleucel-T have been approved by the FDA for the treatment of CRPC. These approved treatment options are palliative with a median overall survival of just over 2 years.
Docetaxel is a cell-cycle specific agent, which translates into being cytotoxic to all dividing cells, not just tumor cells. This leads to some of the common side effects of docetaxel, including neutropenia, anemia, neuropathy, alopecia, and nail damage. Current research targets therapeutic strategies that have a more favorable toxicity profile. Innovative therapies have demonstrated an increase in overall survival in comparison to docetaxel. Cabazitaxel was approved as a second-line therapy primarily based on the results of a phase III study that showed an increased overall median survival benefit of 2.4 months for men with docetaxel-pretreated metastatic CRPC receiving cabazitaxel (with prednisone) compared to mitoxantrone (with prednisone) (15.1 months vs. 12.7 months). 16 Phase III trials of abiraterone demonstrated extended median survival by 3.6 months (15.8 months vs. 11.2 months in the placebo). 17 Sipuleucel-T has a demonstrated 4-month improvement in overall survival (25.8 months vs. 21.7 months). 2 Research in vaccine treatments focuses on working with first-line therapies to extend the current overall survival rate while providing limited side effects.
While historically chemotherapy using docetaxel was not considered an effective treatment for CRPC with a median 2 month improvement in overall survival, 18 the recent ability to monitor prostate- specific antigens (PSAs) has been identified as a valid surrogate endpoint. Monitoring the rate and degree of decline in PSA following radical prostatectomy, radiation therapy, or androgen deprivation may help determine the clinical benefit of treatment. A >50% decline in serum PSA appears to define a subgroup of patients with hormone refractory metastatic prostate cancer with improved survival.19–21 However, this is only an effective method of measurement in patients in the early stages of CRPC who have low tumor burden. Because these patients have low tumor burden and a long life expectancy in comparison to patients in advanced stages of CRPC, they are ideal candidates for vaccine therapy. Prostate cancer in these patients is generally considered a slow- growing tumor, which may allow adequate time for a vaccine to activate the immune system by recognizing TAAs. A prostate cancer vaccine is a promising alternative therapy that aims to eradicate tumors with tumor-specific or tissue-specific antigens, thereby targeting specific cancer cells. However, many antigens overly expressed or unique to tumor cells have been identified.
Characterization of TroVax
TAAs can broadly be divided into three major categories: (i) non-self-viral antigens (eg, E6/E7 from human papilloma virus), (ii) altered self-antigens (eg, MUC-1), and (iii) non-mutated self-antigens (eg, carcinoembryonic antigen). MVA-5T4 is a non-mutated self-antigen. As a 72-kDa surface glycoprotein, it is expressed in high levels in the placenta, specifically on trophoblasts, and in non-pregnant adult tissue, 5T4 is highly expressed in more than 80% of carcinomas of the breast, renal, colorectal, prostate, and ovaries.22,23
The exact function of 5T4 in tumor development has not yet been elucidated. In vitro studies of humans and mice have found that the over expression of 5T4 alters cell adhesion, motility, and morphology. The 5T4 antigen is most often associated with tissue containing undifferentiated progenitor cells or differentiating cells engaged in migration. 12 5T4 may play a common role in epitheliomesenchymal transition (EMT), a critical process that controls morphogenesis in multi cellular organisms and reactivates in the progression of carcinoma. Immunohistochemical analysis indicates that 5T4 expression is an indicator of poor prognosis in colorectal cancer. Additionally, when tumor cells are transfected with the cDNA encoding for 5T4, they display increased motility suggesting that expression of this molecule may induce metastatic properties in a tumor. 5T4 expression is not evident in normal prostate tissues but is present at low levels in benign prostatic hyperplasia (BPH). Expression in BPH contrasts with that identified in prostatic carcinomas. In BPH, expression is generally diffuse at low levels and localized to basal cells, whereas in primary carcinomas, expression is generally high and does not show any apparent polarization. These observations support the hypotheses that 5T4 expression increases as part of malignant progression and clearly indicate that prostate cancer is an appropriate indication for 5T4 targeted therapies.
Viral vector-based immunotherapy presents the self-antigen to the immune system in conjunction with a foreign viral vector, which helps overcome the poor immunogenicity associated with TAAs. The most studied and established viral vector is MVA, which was originally developed as a safe vaccine for smallpox. 24 It was derived from the vaccinia virus Ankara strain of more than 500 passages in primary chick embryo fibroblasts, after which it was found to be replication defective in all mammalian cell lines tested25,26 except Baby Hamster Kidney cells (BHK-21). 27 No complications were reported when MVA was administered to over 120,000 recipients, many of which were at risk for vaccine complications.28,29
Clinical Efficacy
Preclinical murine models have demonstrated that TroVax is safe and highly effective in the prophylactic and active treatment settings. Clinical trials have focused on castration-resistant prostate cancer that has proven unresponsive to conventional systemic cytotoxic chemotherapy as well as metastasized renal cell cancers and colorectal cancers. The first study of TroVax in humans was a dose escalating study in colorectal cancer patients, which demonstrated that TroVax was safe and well tolerated. 30 To date, limited research has been conducted using TroVax to treat CRPC.
An open-label, phase II, single-center study assessed TroVax alone and in combination with granulocyte macrophage-colony stimulating factor (GM-CSF) in 27 patients with castration-resistant prostate cancer. 31 Patients received 5 × 10 8 plaque-forming units/mL TroVax intramuscularly alone or in combination with 250 μg/m2 for 14 days in every 28-day cycle for a total of 12 cycles. Of the 27 enrolled patients, 25 were evaluable for treatment (12 on the TroVax arm and 13 on the TroVax + GM-CSF arm). Two patients were not evaluable due to clinical response secondary to clinical deterioration that lead to the inability to receive the minimum required therapy of 4 TroVax ± 2 GM-CSF cycles. Of the 25 evaluable patients, one was withdrawn due to a hepatitis C infection. Patients were evaluated for adverse events and clinical benefit, including time-to-progression (TTP). TTP was defined as the number of days from the day the subject started TroVax to the date of the subject's disease progression, and it was calculated using the Kaplan-Meier methodology. Disease progression was defined as at least one of the following: PSA progression (25% increase over baselines or nadir on at least 2 consecutive measurements) and an increase in the absolute PSA level by ≥5 ng/mL on at least 2 measurements at least 2 weeks apart, new disease by bone scan, or progressive disease on CT scan. There were no grade 3 or 4 toxicities, and treatment-related adverse events include myalgia, bone discomfort, low-grade temperature elevation, and injection site irritation.
Disease progression was monitored by quantifying the levels of the surrogate marker PSA every 4 weeks and by performing CT scans and bone scans every 8 weeks. All 24 patients evaluable for response mounted a significant 5T4-specific immune response. Although it was too early to comment on differences in survival in the two arms, the median TTP was longer in patients who mounted a positive 5T4 specific enzyme-linked immunosorbent spot (ELISPOT) response. Patients in the TroVax alone arm had a significantly greater median TTP (4.05 months, range 2.30–10.30) compared to TroVax + GM-CSF (2.1 months, range 1.80–6.73). Six patients showed a reduction in the rate of PSA change in the TroVax alone arm (n = 4) and the TroVax + GM-CSF arm (n = 2). All patients experienced disease progression, and at 14.80 months follow up, 16 patients remained alive. There were no objective clinical (soft tissue or bone scan) responses; however, 3 patients had a stable bone scan, 2 patients had stable lymph nodes on CT scan of the abdomen and pelvis, and 4 patients had stable disease on bone scan and stable lymph nodes on CT scan of the abdomen and pelvis.
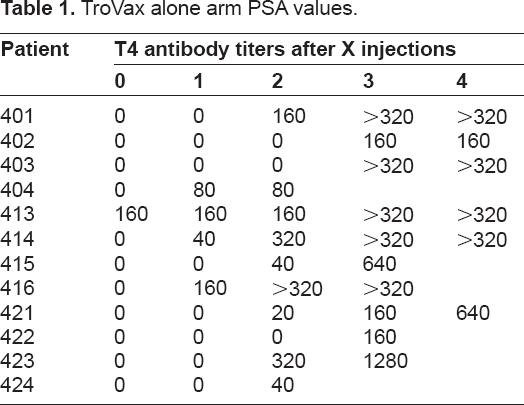
There were variable trends in the PSA responses in the small, heterogonous and heavily pre-treated population. Eight patients in the GM-CSF arm had a transient dip in the PSA in the first cycle only, but PSA levels have a tendency to return to original velocity (Tables 1 and 2). This trend was not observed in the TroVax only arm. Five patients from both arms showed maintenance in PSA during treatment. Twenty-two patients showed no retardation of PSA velocity during treatment. The majority of patients showed a significant decrease in PSA velocity when they moved off study onto taxanes, perhaps suggesting some sensitization to chemotherapy.
TroVax alone arm PSA values.
TroVax + GM-CSF arm PSA values.

The study demonstrated that TroVax was safe and well tolerated in patients with CRPC given alone or in combination with GM-CSF. Furthermore, treatment resulted in a 5T4-specific immune response in all patients. The magnitude of the cellular responses was encouraging, with 7 patients developing 5T4-specific T-cell frequencies in excess of 1 in 10,000 peripheral blood mononuclear cells (PBMCs) and 10 patients showing 5T4 antibody titers in excess of 1,000, which may be important for delaying disease progression. Of the 21 patients with pre-existing MVA responses, 10 showed a >2-fold increase in response after vaccination. The mean increase in MVA-specific PBMCs from baseline to the peak postvaccination frequency was 33.0-fold (range, 0.7 – ≥278.2) in the TroVax only arm and 30.3-fold (range, 0.8 – ≥331.1) in the TroVax + GM-CSF arm. Of the 9 patients who mounted 5T4-specific response, 4 received >2 prior therapies, 3 received ≤2 prior therapies, and 2 had no prior therapies. In this study, GM-CSF did not convincingly enhance 5T4-specific immune responses. Although 5T4-specific antibody responses were induced sooner and were of greater magnitude in patients who received TroVax + GM-CSF, this difference was only marked after the first TroVax vaccination, and, conversely, the frequency of 5T4-specific cellular responses was elevated in patients who received TroVax alone.
Conclusion
TroVax has consistently proven to be safe and well tolerated as monotherapy and in combination with first-line standard-of-care therapies, which include chemotherapeutic agents, cytokine therapy, and surgical resection, for the treatment of renal cell, colorectal, and prostate cancer.
A phase III trial utilizing TroVax in renal cell cancer patients led to the development of an immune response surrogate. 32 This immune response surrogate helped identify an algorithm that predicts patients who are most likely to mount a strong 5T4 antibody response based on three pre-treatment blood tests: hemoglobin, hematocrit, and 5T4 antibody levels. Patients who had higher immune response levels had an increase in survival in comparison to patients who received a placebo. This was retrospectively confirmed in nine histories of phase I and II trials of renal, colorectal, and prostate cancer patients. This study indicates that the immune response surrogate could be used in future clinical trials to determine the patients who are best suited to receive TroVax.
A current phase II CRPC trial is utilizing the immune response surrogate. This trial will evaluate the role of combination therapy with TroVax plus docetaxel or docetaxel alone. Pre-clinical data suggested that some synergy might be observed by combining a taxand and microtubular-associated protein inhibitor estramustine phosphate (EMP). In subsequent randomized-based phase II trials, combinations with EMPdid result in significantly higher proportions of patients achieving PSA declines but at the cost of added toxicity. 33 The administration of cytotoxic chemotherapy can result in bone marrow suppression and has been perceived to have a negative effect on immune function. However, data suggests that taxane-based chemotherapy may actually exert beneficial immunomodulatory effects through a variety of mechanisms, including cytokine production and T-cell infiltration of tumor cells.34,35 Recently, concurrent docetaxel plus vaccine therapy has been safely administered without compromising the ability of the patient to mount a T-cell specific response. 36 It was observed that patients who received a vaccine following docetaxel therapy had an increase in progression-free survival, and patients who crossed over to docetaxel post vaccine demonstrated declining serum PSA levels while maintaining or increasing the T-cell response. A review of several cancer vaccine studies indicated that vaccines should be given followed by chemotherapy to enhance immune response. 37
One concentration of future research should focus on patient selection characteristics and suppressing or inhibiting the negative effects that cancer has on the immune system (e.g., the over expression of T-regulatory cells). Cytokines such as interferon-α and interleukins, which inhibit and kill CD8 + T suppressor cells, should be investigated in combination with TroVax because of their ability to suppress the immune system and facilitate delivery of TAAs. Currently, evidence suggests that patients with such characteristics as minimal tumor burden are more likely to respond to treatment as opposed to patients with advanced disease. TroVax has been demonstrated to have some clinical benefit, and trials evaluating other therapies combined with TroVax should be conducted to evaluate any synergistic effects.
Other concentrations of research include tumor suppression via T-cells and overcoming cytotoxic T lymphocyte antigen-4 (CTL-4). The antigen CTL-4 is a key negative regulator of adaptive immune responses and has a central role in maintaining peripheral tolerance and shaping emergent T-cell responses. Taxane-based therapy in combination with TroVax may also help suppress the immune system and present the TAAs. Taxane-based therapies promote apoptosis and disrupt cell division and may be a key to suppressing tumors. A current phase II trial is evaluating TroVax and docetaxel versus docetaxel alone in patients with prostate cancer to determine if there are any synergistic effects that would affect progression-free survival. 38 Patients will receive up to 10 cycles of docetaxel or 10 cycles of docetaxel plus TroVax. The study will compare progression-free survival between the two arms.
TroVax is a promising addition to the prostate cancer treatment arsenal as monotherapy or combination therapy because of its minimal side effects and demonstrated ability to elicit an immune response. Further research will help elucidate the most favorable patient characteristics for receiving the vaccine and help determine effective combination treatment strategies.
Disclosures
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
