Abstract
Hepatocellular carcinoma (HCC) is the most common liver cancer and the third leading cause of cancer death. It has been a major worldwide health problem with more new cases being diagnosed each year. The current available therapies for patients with advanced HCC are extremely limited. Therefore, it is of great clinical interests to develop more effective therapies for systemic treatment of advanced HCC. Several promising target-based drugs have been tested in a number of clinical trials. One breakthrough of these efforts is the approved clinical use of sorafenib in patients with advanced HCC. Targeted therapies are becoming an attractive option for the treatment of advanced HCC. In this review, we summarize the most recent progress in clinical targeted treatments of advanced HCC.
Introduction
Hepatocellular carcinoma (HCC) is the most common liver cancer and the third leading cause of cancer death. It has been one of the major global health problems with increasing new cases coming up each year. The major risk factors for HCC development are hepatitis B and C viral infections, which lead to liver cirrhosis and account for 75% of HCC cases.1,2 Other etiologic factors include toxic chemical exposure, alcohol abuse, and intake of aflatoxin-contaminated food.3,4 HCC has long been a major disease in developing countries, with most cases occurring in Eastern Asia and sub-Saharan Africa; however, there is a rising incidence in Western countries and many emerging cases are associated with hepatitis C infection, obesity/type 2 diabetes, and nonalcoholic fatty liver disease (NAFLD) such as nonalcoholic steatohepatitis (NASH).5,6 The worldwide incidence of HCC is over 600,000 cases every year.7,8
Treatment of HCC is most effective in the early stages. If diagnosed early, major treatment options are surgery and interventional radiological therapy. Surgical options include surgical resection, cryo-surgery, radiofrequency ablation (RFA), and liver transplantation, while interventional radiological therapy includes hepatic artery chemoembolization, percutaneous ethanol injection, percutaneous radio frequency ablation, and cisplatin gel infusion. Surgery is generally believed the best treatment option for a cure. Surgical resection is recommended for patients who have relatively functional livers with unifocal tumors (<5 cm). The 5-year overall survival rate can be increased up to 5-fold in well-selected patients after surgical resection. 9 Liver transplantation is another option for treatment of early stage HCC. The most commonly used criteria for candidate selection is the Milan criteria proposed by Mazzaferro et al in 1996. 10 Patients with HCC who either have one tumor (≤5 cm) or have 2 to 3 tumors (≤3 cm) could be selected for liver transplantation. The 5-year overall survival rate for patients who have received liver transplantation is up to 70%. 11 However, the supply of donor livers is quite limited. Therefore, selection of candidates becomes more important to ensure the best outcome. On the other hand, some surgeons have suggested more inclusive parameters than the Milan criteria in an effort to benefit more patients.
Unfortunately, patients with HCC are usually found to be in the advanced stages at the time of diagnosis, which severely limits these surgical options. A widely used conventional treatment for patients with advanced HCC is chemotherapy. However, response rates to such treatments are poor or nonexistent given the fact that HCC is a highly aggressive tumor that often shows resistance to chemotherapy. Therefore, there is an urgent clinical need to develop more effective therapies for patients with advanced HCC.
Recent discoveries in the molecular mechanisms of HCC pathogenesis have created many opportunities for developing targeted therapies for the treatment of advanced HCC. Some key signaling pathways have been identified, such as epidermal growth factor receptor (EGFR) pathway, insulin-like growth factor receptor (IGFR) pathway, vascular endothelial growth factor (VEGF) pathway, and so on. Targeting these molecular pathways provides a potential to generate new therapeutic approaches for the treatment of advanced HCC. One successful example is the recently FDA approved clinical use of sorafenib, a multikinase inhibitor, for patients with advanced HCC.12,13 This approval unveils a new era of targeted therapy in HCC and encourages more targeted agents being developed and tested in the systemic treatment of advanced HCC.
In this review, we will focus on some key targets relevant to the signaling pathways implicated in HCC pathogenesis and evaluate the features of such targeted therapies in clinical trials.
HCC-Associated Molecular Pathways and Epigenetic Regulation
The occurrence of tumor development in liver requires genetic/epigenetic aberrations in normal hepatocytes, which could be induced by risk factors as mentioned above. The malignant transformation of HCC, which is called hepatogenesis, is a very complex and multistep process that may take years to develop. The most frequent and direct risk factor for HCC is liver cirrhosis, which is mostly developed by hepatitis infection and chronic inflammation. The pathogenesis of HCC eventually results in certain genetic/epigenetic alternations, such as point mutation, chromosomal amplification/deletion, genomic instability, gene methylation, and histone deacetylation, which ultimately lead to the dysregulation of many molecular signaling pathways. Generally, the activation of specific oncogenes such as
Angiogenesis-Related Pathways
HCC is characterized as a hypervascular solid tumor with a high tendency towards vascular invasion. 17 The angiogenesis in HCC is essential for the tumor growth by supplying oxygen and nutrients and is the end result of a complex network of orchestrated action among tumor cells, endothelial cells, and growth factors. The major mediator of the angiogenesis in HCC is vascular endothelial growth factor (VEGF).18,19 Circulating VEGF is a key proangiogenic factor which binds to its receptors (VEGFR1–3, mainly 2) on endothelial cells and induces the dimerization and autophosphorylation of the receptor, which activates the downstream signal pathway eventually leading to angiogenesis (Fig. 1). 20 Elevated expression level of VEGF has been found in most HCC, which is associated with poorer prognosis and decreased survival. 21 Apart from VEGF, other growth factors including the platelet-derived growth factor (PDGF), fibroblast growth factor (FGF), and angiopoietin have been shown to make a significant contribution to the angiogenesis in HCC. 22 These growth factors thus appear to be attractive antiangiogenesis candidates for targeted therapies.

Diagram of signaling pathways stimulated by ligand binding to the tyrosine kinase receptor.
Growth-Related Pathways
In addition to increased angiogenesis, dysregulation of signaling pathways associated with cell growth has also been shown to be critical in the development of HCC (Fig. 1). Molecular aberrations of growth pathways are directly involved in cell proliferation and the pathogenesis of HCC regarding tumor growth, invasion, and metastasis. Some key signaling pathways are the hepatocyte growth factor receptor pathway (HGFR), epidermal growth factor receptor (EGFR) pathway, insulin-like growth factor 1 receptor (IGF-1R) pathway, and PI3K/Akt/mTOR pathway. Other pathways such as the Wnt-β catenin pathway and the hedgehog pathway are implicated in HCC, but the therapies targeting these pathways are only in their early stages.
HGF/C-Met Pathway
HGF is secreted by stellate cells and is an important growth factor for liver regeneration and injury repair. 23 Its receptor is c-Met, a typical receptor tyrosine kinase (RTK). Numerous studies have shown that the HGF/c-Met signaling pathway is required for normal mammalian development and is involved in diverse cellular responses such as cell proliferation, survival, and differentiation depending on the particular cell type and the microenvironment. Under physiological conditions, the HGF/c-Met pathway does not contribute significantly to the liver homeostasis; however, in circumstances of liver removal or injury, the pathway is heavily activated as an important mediator in the liver regeneration processes. 24 Deregulation and activation of c-Met may result in unregulated cell growth and differentiation, thus contributing to malignant transformation. Like many other RTK pathways, HGF/c-Met pathway is activated by the binding of HGF to its receptor c-MET, which results in the receptor autophosphorylation and the activation of the downstream intracellular signaling pathways including the PI3K and mitogen-activated protein kinase (MAPK) pathways. c-Met overexpression or enhanced activation relative to normal tissues has been noted in many human cancers. In HCC, about 20% to 48% of samples from surgical specimens showed elevated levels of c-Met, which is associated with poor outcome in patients. 25 Although overexpression of c-Met has been found to correlate with the increased malignancy of human HCC, there is still lack of understanding regarding the big picture of the exact role of this pathway in the pathogenesis of HCC.
EGFR Pathway
EGFR, also called ErbB1 and Her1, is a member of an RTK family of 4 related receptors, which includes ErbB2/Her2/Neu, ErbB3/Her3, and ErbB4/Her4. 26 Every member of this family has some identified ligands except ErbB2/Her2/Neu. The most common ligand for EGFR is epidermal growth factor (EGF), but EGFR also binds to other ligands such as transforming growth factor α (TGF-α), amphiregulin, and β-cellulin. 27 Upon binding with its ligands, EGFR undergoes receptor homodimerization or heterodimerization with ErbB2 or ErbB3, resulting in receptor tyrosine kinase activation and autophosphorylation. The main downstream signaling pathways include the PI3K/Akt/mTOR pathway, the Ras/Raf/MEK/ERK1/2-MAPK pathways, and the signal transducer and activator of transcription pathways (STAT 3 and 5). Activation of these pathways stimulates cellular division, survival, and apoptosis, and any alteration in these pathways aids tumor growth and progression. 28
IGF-1R Pathway
IGF-1R basically has two ligands: IGF-1 and IGF-2. Binding of these ligands to a cysteine-rich domain in the extracellular α subunit of IGF-1R triggers self-dimerization and autophosphorylation of three tyrosines in the activation loop of the tyrosine kinase domain in the cytoplasmic portion of the β subunit, which results in amplification of tyrosine kinase activity and further autophosphorylation of additional tyrosine residues. 29 These phosphotyrosine containing motifs are binding sites for adaptor and effector molecules in receptor signaling pathways. Binding of the adaptor protein Shc and the docking protein IRS-1 to IGF-1R activates the MAPK and PI3/Akt/mTOR signaling pathways, respectively. Genetic studies have demonstrated that mice deficient in IGF-1R die during the development; mice with only one copy of IGF-1R gene exhibit almost half body mass reduction.30–32 IGF-1R pathway plays a crucial role in cell proliferation, differentiation, and apoptosis.
Enhanced expression of IGF-1R and IGF-2 has been correlated with disease implication in HCC.33–35 On the other hand, IGF binding proteins such as IGFBP-3, which regulate the circulating concentration of IGF and IGF in the tumor microenvironment, are downregulated. 36 Further, the expression of IGF-2R, a receptor with a relative short intracellular tail, which functions to clear IGF-2 from cell surface to attenuate the IGF-1R signaling, is also downregulated in some HCCs. 37
IGF-1R has long been a target for the treatment of various tumors due to its overexpression on cell surface. Recent evidence shows that IGF-1R undergoes nuclear translocation in cancer cells and autoregulates its gene expression.38–40 We have further found that gefitinib resistant HCC cells have an increased level of the IGF-1R nuclear translocation. 41 The biological role of this nuclear IGF-1R in cancer cells remains unclear; thus, nuclear IGF-1R will merit attention when designing future IGF-1R targeted therapies for HCC.
PI3K/Akt/mTOR Pathway
The PI3K/Akt/mTOR pathway can be activated by many RTK as described above and plays an essential role in the cell survival, growth, and proliferation. 42 Phosphatidylinositol 3 kinase (PI3K) has two subunits: one subunit p85 as the adaptor and another subunit p110 as the kinase. When associated with cytoplasmic tails of some relevant RTKs, PI3K phosphorates phosphatidylinositol 3,4,5-triphosphate (PIP3) to generate phosphatidylinositol 4,5-biphosphate (PIP2) and activates Akt (also known as protein kinase B, PKB) through phosphoinositide-dependent kinase (PDK). The PI3K pathway can be suppressed via dephosphorylation of PIP3 by phospharase and tensin homologue (PTEN). 43
The major mediator and regulator of this pathway is mammalian target of rapamycin (mTOR) (Fig. 1). Once activated by PI3K, mTOR promotes cell growth and proliferation by stimulating protein synthesis through phosphorylation of 4E-BPs and the S6 kinases. In addition to receiving signals from Akt, mTOR can be directly activated by the cell's environmental growth factors and nutrients. 44 When the cell needs additional resources, mTOR can increase nutrient uptake and promote angiogenesis. Further, mTOR can increase the activity of Akt and thus enhance the effect of this signaling pathway. 45
The signaling pathway that includes mTOR is highly active in many cancer cells, including HCC.46,47 Abnormal association of PI3K to RTK or loss of function of PTEN leads to uncontrolled activation of this pathway and causes carcinogenesis. Actually, activation of the PI3K/Akt/mTOR pathway is associated with poor prognosis in many cancers and is linked to resistance to many types of therapy.
Other Emerging Pathways
Apart from the above described signaling pathways that are under active clinical trials for targeted therapies in HCC, there are an increasing number of other signaling pathways that are found to be involved in the pathogenesis of HCC. These pathways include the Notch signaling pathway, 48 the hedgehog signaling pathway, 49 the transforming growth factor β (TGF-β) signaling pathway,50,51 and the Wnt-β-catenin pathway.52,53 Although these pathways play an important role in HCC, drugs targeting these pathways are in early stages, and further basic and clinical studies are needed before relevant clinical targeted therapies can be developed.
Epigenetic Regulation and microRNA
Despite deregulation of various signaling pathways through classic genetic mutations, which either activate oncogenes or inhibit tumor suppressor genes, cancer development can be triggered by epigenetic mechanisms that alter gene expression through DNA modification without changing DNA sequences. Generally, there are 3 types of epigenetic mechanisms: gene methylation/demethylation, histone acetylation/deacetylation, and noncoding RNAs such as microRNAs (Fig. 2).

Mechanisms of epigenetic regulation in cancer gene expression.
Gene methylation can occur in the promoter region, rich with CpG islands. In cancer cells, tumor suppressor genes are usually silenced due to the hypermethylation of their promoter regions. Methylated promoter region inhibits the binding of critical transcriptional factors to the gene thus block the transcription. On the other hand, some genes such as those involved in tumor invasion and metastasis that are supposed to be silent under normal physiological condition are activated in cancer cells, resulting in cancer progression.
DNA methylation has been found to have a close connection with chromatin configuration.54,55 While unmethylated DNA is usually associated with loose chromatin configuration that is active for gene transcription, methylated DNA is associated with tight chromatin configuration that is inactive for gene transcription. The state of chromatin configuration is regulated by histone acetyltransferases (HATs), which add acetyl groups to histone H3 and H4 tails, and histone deacetylases (HDACs), which remove acetyl groups from the tails. Methylated DNA can recruit HDACs to condense chromatin structure.
microRNAs (miRNAs) are a group of noncoding small molecules with 18 to 23 nucleotides in length. miRNAs are post-transcriptional gene regulators that play crucial roles in almost every normal cellular process. Alteration of miRNA levels can cause the activation of oncogenes or the inhibition of tumor suppressor genes, leading to cellular transformation and cancer development. In most cases of cancer, miRNAs are usually found to be downregulated. 56 Recent evidence demonstrated that miRNAs can be regulated by DNA methylation.57,58
Since aberration of epigenetic regulation is closely associated with tumor formation, key enzymes involved in these mechanisms such as HATs and HDACs have becoming attractive targets of therapeutic treatment for various cancers.59,60
Mechanism of Action, Safety, and Efficacy of Targeted Therapies in HCC
Agents that specifically disrupt targets in HCC associated molecular pathways are designed and tested to suppress the development of liver cancer. There are basically two types of these agents for advanced HCC systemic therapy: small molecule kinase inhibitors and monoclonal antibodies. Some targeted therapies based on these agents are summarized in Figures 2 and 3. Based on the HCC relevant signaling pathways as described above, agents that disrupt these pathways are reviewed and discussed in regards to the molecular action, efficacy, safety, and metabolism.
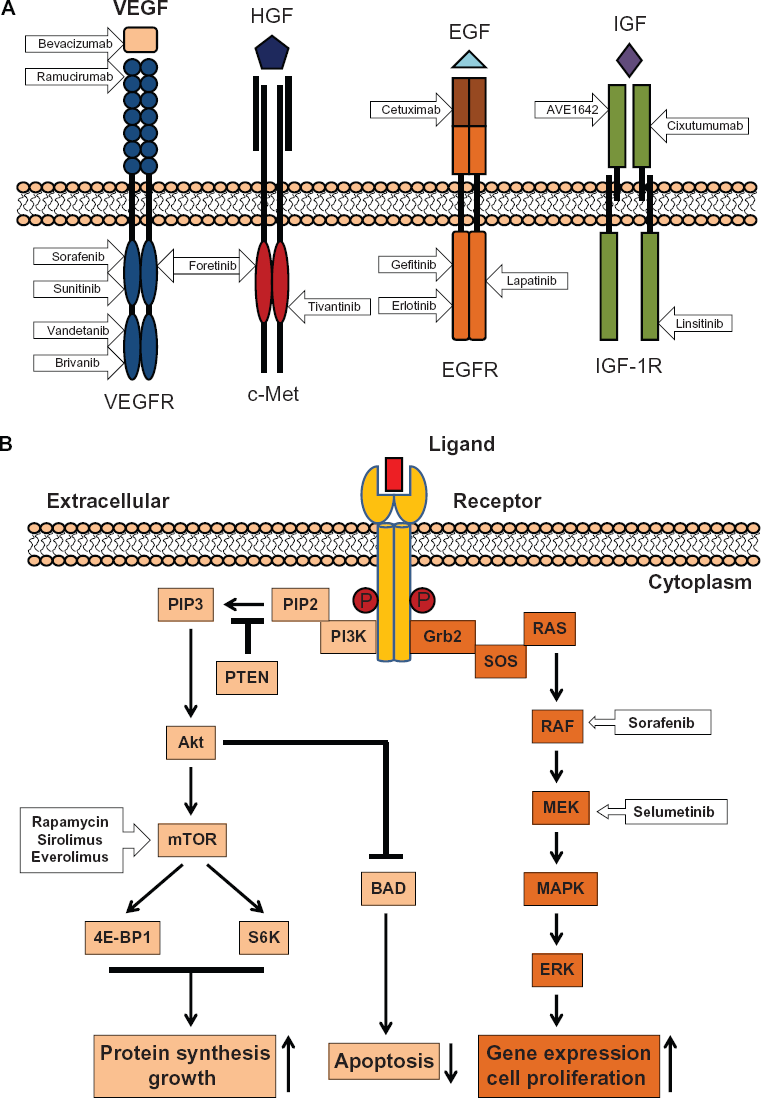
Molecular action of targeted agents in tyrosine kinase receptors and the corresponding signaling pathways. (
Anti-VEGFR Pathway
Sorafenib
Sorafenib (Nexavar/Bay43-9006, Bayer Pharmaceuticals Corporation, Leverkusen, Germany) is a multitargeted small molecule that inhibits the activity of VEGFR, PDGFR, FGF as well as RAF to block tumor proliferation and angiogenesis (Fig. 3A and B). Sorafenib has demonstrated safety and efficacy against a wide variety of cancers in numerous preclinical and clinical studies. 61
The pharmacokinetic characteristics of sorafenib regarding absorption, distribution, metabolism, and excretion have been evaluated. The peak plasma level of sorafenib occurs around 3 hours after dosing; however, there is no linear relationship between dosing and exposure. The bioavailability of sorafenib is reduced when taken with food.62–64 Since sorafenib is highly protein bound, it is estimated that there is a large volume of distribution. Metabolism of sorafenib is about 50%, and this drug is mainly eliminated through the liver. 63
Sorafenib established itself as an effective drug for the treatment of advanced HCC in recent clinical trials.64–66 In two recent, large, randomized, double-blinded phase III trials, sorafenib improved the overall survival (OS).67,68 In one trial, 602 Caucasian patients with advanced HCC were recruited and evaluated between sorafenib (299 patients) and placebo (303 patients). The median OS in the sorafenib treated group was 10.7 months compared with 7.9 months in the placebo group. Clearly, sorafenib increased OS out of placebo by total 44%. The hazard ratio for sorafenib/placebo was 0.69 based on 321 deaths. Sorafenib decreased the relative risk of death by 31%. 67 In another similar trial, 226 Asian-Pacific patients with advanced HCC (mostly developed from hepatitis B infection) were recruited and evaluated between sorafenib (150 patients) and placebo (76 patients). The median OS in the sorafenib treated group was 6.5 months compared with 4.2 months in the placebo group. 68 Although tolerance to sorafenib was found generally good in these two studies, there were treatment-related adverse events. The most frequently occurring events were diarrhea, fatigue, weight loss, and hand-foot skin reaction.
So far, sorafenib is the only drug shown to improve the overall survival in patients with advanced HCC and was approved for clinical use by the US Food and Drug Administration (FDA) in 2007. 12 Other countries such as European countries and China have also approved sorafenib for advanced HCC treatment. Sorafenib has thus become the standard of care for patients with advanced HCC.
More clinical trials are being tested to evaluate the use of sorafenib in combination with other targeted agents and conventional treatments such as chemotherapy. A finished randomized phase II clinical trial reported an overall survival rate of 13.7 months in patients treated with the combination of sorafenib and doxorubicin compared with 6.5 months in patients treated with doxorubicin alone. 69 Some other agents being tested in combination with sorafenib are AVE1642 (NCT00791544), erlotinib (NCT00901901), linsitinib (NCT01334710), cixutumab (NCT01008566 and NCT00906373), and temsirolimus (NCT01008917 and NCT01335074).
Sunitinib
Sunitinib (Sutent/SU11248, Pfizer Inc., New York, USA) is another oral multikinase inhibitor like sorafenib that mainly targets VEGFRs and PDGFRs (Fig. 3A). This drug is well studied and has been approved for the treatment of renal cell carcinoma and gastrointestinal stromal tumors that are refractory to imatinib. 70
The absorption of sunitinib peaks within 6 to 12 hours after dosing. Taking the drug with food or not has a marginal effect on the exposure. 71 Like sorafenib, sunitinib is highly protein bound and has a large volume of distribution. Sunitinib is metabolized by CYP3A4 and is excreted mainly via feces. 72
While early clinical trials in patients with advanced HCC showed that this drug has encouraging results on efficacy and tolerance,73,74 the most recent phase III SUN 1170 clinical trial showed that use of sunitinib increased serious drug-related adverse events. This trial was subsequently discontinued. Thus, sunitinib is no longer tested for the treatment of advanced HCC. 75
Vandetanib
Vandetanib (Zactima/ZD6474, AstraZeneca, Wilmington, USA) is an oral tyrosine kinase inhibitor of VEGFR (Fig. 3A). This compound also inhibits the activities of EGFR and the rearranged during transfection (RET) tyrosine kinase receptor. Vandetanib has been approved by FDA for the treatment of medullary thyroid cancer. In the treatment of advanced HCC, vandetanib was shown to exert inhibitory effects on cell proliferation and migration in preclinical models. 76 A phase II, placebo-controlled clinical trial (69 patients) showed limited efficacy and safety in patients with advanced HCC. However, it seems further evaluation with a larger population of patients is needed to confirm the benefit of vandetanib on HCC. The most common adverse effect profile of vandetanib is diarrhea and rash. 77
Brivanib
Brivanib (BMS-582664, Bristol-Meyers Squibb, New York, USA) is an oral selective inhibitor of VEGFR and EGFR (Fig. 3A). Brivanib demonstrated significant antitumor activity in HCC xenograft models by decreasing tumor microvessel density and increasing apoptosis of tumor cells. 78 Brivanib has been evaluated as a first line and a second line agent in advanced HCC treatments. As a first line therapy in a phase II clinical trial, brivanib achieved a median 10 months OS. 79 In a second line therapy after prior treatment with sorafenib, the median OS was 9.8 months. Major drug-related adverse events were fatigue, nausea, loss of appetite, diarrhea, and hypertension. 80 More clinical trials are currently underway to further evaluate the antitumor activity in patients with advanced HCC.
Bevacizumab
Bevacizumab (Avastin, Genetech, San Francisco, USA and Roche, Basel, Switzerland) is a recombinant humanized monoclonal antibody neutralizing the biological activity of VEGF (Fig. 3A). Bevacizumab has been approved for the treatments of colorectal, non-small-cell lung, and breast cancers. It is the first antibody against VEGF to be tested in HCC and has been evaluated as both single agent and in combination with other drugs.
A phase II trial including a total of 46 patients with locally advanced HCC showed promising results: 13% of patients showed partial responses and 65% of patients obtained disease stabilization for up to 6 months. 81 However, since some patients with severe diseases were excluded from this trial, the clinical benefit obtained from bevacizumab treatment seems only suitable to well-selected patients. Benefit for a broad range of patients shown in a large scale study is needed to further endorse the activity of bevacizumab in advanced HCC. The major drug-related adverse event was severe hemorrhagic complications (11%); other drug-related adverse events were arterial thrombosis (6%) and hypertension (15%).
The combination use of bevacizumab with other chemotherapy agents such as gemcitabine, capecitabine, and erlotinib has been evaluated in some phase II trials and showed moderate clinical benefit with reasonable safety.82–84
Ramucirumab
Ramucirumab (IMC-1121B, ImClone Systems Inc., New York, USA) is a fully human immunoglobulin G1 monoclonal antibody specifically developed to bind to the extracellular VEGF-binding domain of VEGFR-2 and block the activation of its receptor tyrosine kinase and the downstream signaling pathways (Fig. 3A).
Ramucirumab was tested in various solid tumors including HCC in a phase I trial and showed objective antitumor activity with reasonable safety. The most frequently reported serious events included hypertension (13.5%), abdominal pain (10.8%), and anorexia, vomiting, headache, and proteinuria (each in 5.4% of patients). 85
Preliminary data from a phase II study of 42 patients with advanced HCC showed that, as a single agent in a first-line monotherapy, ramucirumab achieved a disease control rate of 50% and a median progression-free survival (PFS) of 4.3 months. 86 Based on this positive study, a phase III clinical trial in HCC comparing ramucirumab with placebo for the second-line treatment after sorafenib is underway (Table 1, NCT01140347).
Molecular targeted agents used for clinical trials in HCC.*
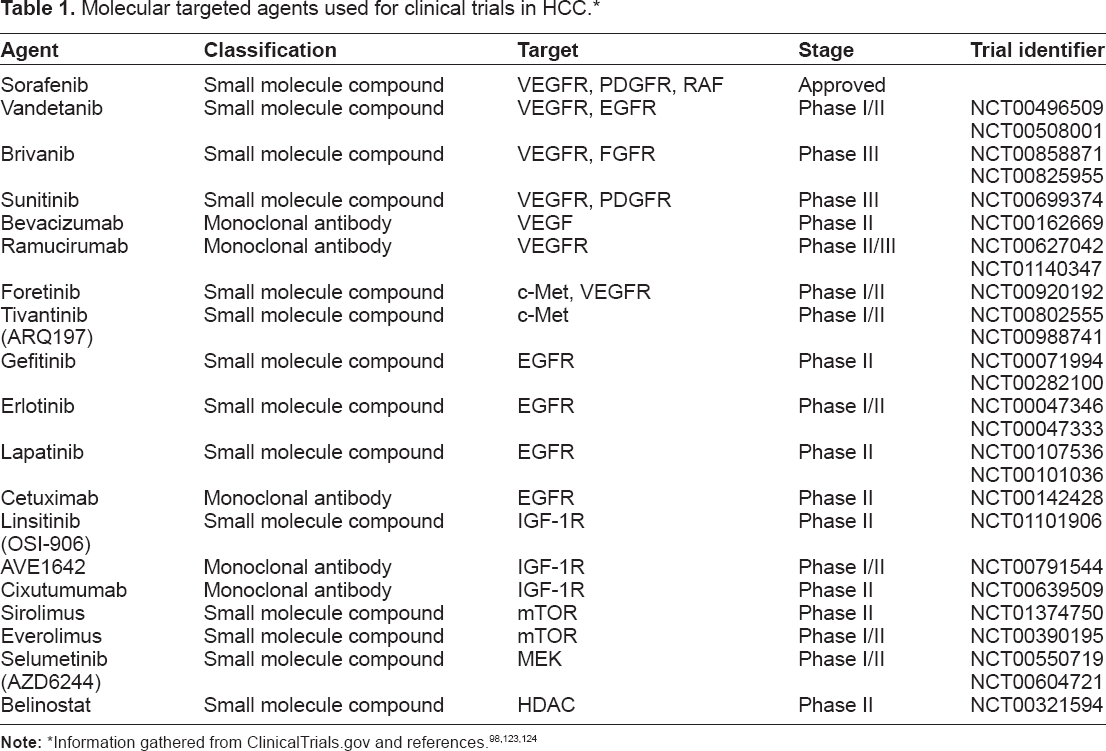
Information gathered from ClinicalTrials.gov and references.98,123,124
Anti-HGF/c-Met Pathway
Foretinib
Foretinib (XL880/GSK1363089, GlaxoSmithKline, Brentford, United Kingdom) is an oral selective small molecule inhibitor of c-Met and VEGFR-2 (Fig. 3A). Since overexpression of c-Met in HCC correlates well with the progress of this disease, 87 it is generally anticipated that targeting c-Met using foretinib could produce clinical benefit in patients with advanced HCC. Evidence supporting this notion is the recent observations that cells lacking c-Met expression do not respond well to c-Met inhibitor and that foretinib exhibits inhibitory effects on angiogenesis, cell proliferation, and tumor metastasis in preclinical HCC models.88,89
A phase I/II study in patients with advanced HCC is being tested for safety and tolerance of foretinib (NCT00920192).
Tivantinib
Tivantinib (ARQ197, ArQule, Inc., Woburn, USA) is the first in class orally available selective inhibitor of c-Met (Fig. 3A). This is also a novel, non-ATP-competitive c-Met inhibitor, the most advanced agent in a new class of trans-3,4-bisubstituted pyrrolidine-2 5-diones. 90
Results from a phase Ib clinical trial of 21 patients with advanced HCC showed antitumor activity with progression-free rates at 2 and 4 months of 59.7% and 39.8%, respectively. The study also found a manageable toxicity of tivantinib with anemia (43%), asthenia (43%), neutropenia (38%), leukopenia (33%), diarrhea (29%), anorexia (29%), and fatigue (24%). 91
On the basis of results from the phase Ib study, a global, randomized, double-blind, placebo-controlled, phase II clinical trial in patients with unresectable HCC is currently enrolling patients (NCT00988741).
Anti-EGFR pathway
Gefitinib
Gefitinib (Iressa/ZD1839, AstraZeneca) is the first small molecular inhibitor of EGFR tyrosine kinase (Fig. 3A). Gefitinib inhibits EGFR tyrosine kinase activity by binding to the ATP-binding site of the receptor and blocking the Ras signaling pathway.
Gefitinib is developed to treat cancers in which EGFR is overexpressed, such as lung cancer and breast cancer. Although demonstrated the ability to prevent HCC development in a murine model, 92 when applied to HCC in a phase II clinical trial with 31 patients enrolled, gefitinib induced a median OS of 6.5 months. Since the results did not meet the criterion for the second stage of the study, the authors concluded that this drug alone did not exhibit significant antitumor activity in patients with advanced HCC. 93
Our recent study showed that gefitinib resistance in HCC cells was associated with IGF-1R nuclear translocation and accumulation. 41 It seems that nuclear IGF-1R may play an important role in gefitinib resistance in HCC. Inhibiting IGF-1R expression using a specific miRNA may help improve the efficacy of gefitinib.
Erlotinib
Erlotinib (Tarceva/OSI774, Genetech Inc. and Roche) is an oral specific inhibitor of EGFR tyrosine kinase (Fig. 3A). Erlotinib shares the common chemical backbone structure with gefitinib. The peak plasma levels occur 4 hours after dosing. It is not recommended to take erlotinib with food. Like gefitinib, erlotinib is highly protein bound and has an extensive distribution in tissues. It is predominantly metabolized by CYP3A4 and CYP3A5, and 91% of the dose of 100 mg was recovered over 11 days.94,95
As a more potent small molecule compound than gefitinib, erlotinib showed antitumor activity in a variety of solid tumors including advanced lung cancer and pancreatic cancer. Two phase II clinical studies using erlotinib as the first line therapy were conducted in patients with advanced HCC. The first trial (38 patients enrolled) reported a median OS of 13 months and the PFS at 6 months in 32% of patients. 96 The second trial (40 patients enrolled) reported a median OS of 10.75 months and the PFS at 4 months in 43% patients. 97 More clinical trials are ongoing to evaluate the combination of erlotinib with sorafenib and other agents. 98
Some of the major side effects found in a combined trial with bevacizumab were fatigue (20%), hypertension (15%), and gastrointestinal bleeding (12.5%). 99
Lapatinib
Lapatinib (Tyverb/GW572016, GlaxoSmithKline) is a dual tyrosine kinase inhibitor that blocks EGFR and HER2/NEU signaling pathways (Fig. 3A). Lapatinib is an orally active small molecule originally designed for the treatment of breast cancer with elevated expression of EGFR and HER2/NEU.
The pharmacokinetics of lapatinib were reported in some studies. The peak plasma concentration could be reached within 3 to 4 hours after dosing, and food does not significantly reduce its half life. 100 Lapatinib is highly protein bound, and the volume of distribution is large. CYP3A4 is the main metabolizing enzyme of lapatinib. Excretion of lapatinib is primarily through the liver. 101
Although HCC cells usually do not express a high level of EGFR and HER2/NEU, lapatinib was evaluated in two separate phase II clinical trials. In one study, 26 patients with advanced HCC were included. The median OS and PFS were 12.6 months and 1.9 months, respectively. Ten patients (40% of total enrolled) showed stable disease, and 6 patients (23% of total enrolled) had stable disease lasting more than 120 days. The most common drug-related adverse events observed in this trial were diarrhea (73%), nausea (54%), and rash (42%). 102 In another study, a total of 40 patients with advanced HCC were tested. The median OS and PFS were 6.2 months and 2.3 months, respectively. Although tolerance to this drug was high, only 5% of patients showed objective response. Results from this trial indicated that therapy with lapatinib in HCC did not meet the target efficacy rate. 103
Cetuximab
Cetuximab (Erbitux/IMC-C225, Bristol-Meyers Squibb, and Merck Serono, Geneva, Switzerland) is a chimeric monoclonal antibody neutralizing the extracellular domain of EGFR from the binding of EGF (Fig. 3A). This antibody drug is given by intravenous infusion and has been used in treatment of metastatic colorectal cancer and head and neck cancer.103,104
Evaluation of cetuximab as a single agent treatment in advanced HCC was tested in two phase II clinical trials. The median OS and PFS observed in the first trial with 30 patients enrolled were 9.6 months and 1.4 months, respectively. Although stable disease was found in 17% patients, no clinical responses were observed in this study. 105 In a similar designed second study, 27 patients within the total of 32 enrolled were evaluated for efficacy. Again, there were no clinical responses. The median PFS was 1.8 months, and the stable disease was observed in 44.4% patients for 8 weeks. 106 In combination with gemcitabine and oxaliplatin (GEMOX), cetuximab was evaluated in another phase II clinical trial. The clinical response rate was 23%. However, since no data are available for cetuximab as a single agent and GEMOX regimen showed antitumor activity before, the exact contribution of cetuximab in this trial remains unknown. 107
Anti-IGF-1R Pathway
Linsitinib
Linsitinib (OSI-906, OSI Phamaceuticals, Farmingdale, USA) is a novel dual tyrosine kinase inhibitor of IGF-1R and insulin receptor (IR) (Fig. 3A). Linsitinib is a small molecular compound that specifically binds to the intracellular domain of IGF-1R and IR and blocks the autophosphorylation of the receptors and the downstream signaling pathways. 108
A phase II clinical trial was initiated to evaluate its efficacy and safety in patients with advanced HCC as a second line of therapy for HCC refractory to a first line sorafenib therapy (NCT01101906). Unfortunately, the company recently announced the termination of this trial.
AVE1642
AVE1642 (Sanofi-Aventis, Paris, France) is a humanized monoclonal antibody that specifically blocks IGF-1R signaling by binding to the extracellular domain (Fig. 3A).
A phase I clinical trial reported that AVE1642 can be safely combined with active doses of sorafenib in patients with advanced HCC. This combination did not interfere the pharmacokinetics of either AVE1642 or sorafenib at the concentrations tested and achieved a mean PFS of 13.3 weeks in most patients. 109
Cixutumumab
Cixutumumab (IMC-A12, ImClone System Inc.) is also a humanized monoclonal antibody that specifically neutralizes the extracellular IGF-1 binding domain of IGF-1R and inactivates the subsequent signaling pathways (Fig. 3A).
Preclinical data showed that cixutumumab blocks proliferation, induces apoptosis, and delays tumor growth both in vitro and in vivo. 110 A recent phase II clinical trial in 22 patients with advanced HCC showed that a median OS was 8 months, and 7 patients (29% of total) had stable disease for at least 4 months. However, the study concluded that cixutumumab had no antitumor activity as a single agent in HCC, although an initial phase I study reported partial activity.111,112
Recent research progress may explain the lack of activities of antibody drugs targeting IGF-1R in clinical trials. IGF-1R has been found to migrate and accumulate in cancer cell nuclei, and drug-resistant cancer cells have increased levels of nuclear IGF-1R.38–41 It seems that direct inhibition of the expression of IGF-1R in cancer cells might improve the antitumor activity and cancer drug sensitivity. Nuclear IGF-1R should be considered in the future design of IGF-1R-based targeted therapies.
Anti-mTOR pathway
Sirolimus and Everolimus
Sirolimus and everolimus (RAD001, Novartis, Basel, Switzerland) are analogues of rapamycin and selective inhibitors of mTOR kinase (Fig. 3B). Since mTOR plays a pivotal role in signal transduction of many receptor tyrosine kinase induced signaling pathways, inhibiting mTOR should have the antitumor activity for a broad range of cancer stimuli.
Experimental studies using this rapamycin and its analogue everolimus (RAD001) in HCC cells showed significant effects in reducing tumor cell growth and improving cell survival.113,114 One recent phase I/II study in 28 patients with advanced HCC showed everolimus had clinical activity with a median OS of 8.4 months. 115 Promising results on the efficacy in patients with advanced HCC were also reported from two pilot clinical trials using sirolimus. The first study, with 21 patients enrolled, showed a median OS of 6.5 months and stable disease for at least 3 months in 24% of patients. 112 The second study, with 14 patients enrolled, observed a complete clinical response in 7% of patients and partial response in 33% of patients. In addition, sirolimus demonstrated a safe profile because no major drug-related adverse events were observed in these studies. 116
Several ongoing trials are currently recruiting patients with advanced HCC to determine the combinational effects of these drugs with sorafenib and conventional chemotherapeutic agents.
It is worth noting that cancer drug sensitivity can be improved in patients with certain gene mutations. For example, a recent study, using the whole genome sequencing technology, identified loss of function mutations in tuberous sclerosis complex 1 (
Anti-MEK/ERK Pathway
Selumetinib
Selumetinib (AZD6244/ARRY-142886, AstraZeneca) is an orally active new inhibitor that binds to MEK to block the RAF/MEK/ERK pathway, which appears to be the most significant signaling pathway in HCC development (Fig. 3B).
Selumetinib showed significant antitumor activity both in vitro with HCC cells and in vivo with xenografts. 118 However, when tested in a phase II study in 19 patients with advanced HCC, selumetinib failed to exhibit convincing antitumor activity. 119 Results from this clinical trial suggest that inhibition of RAF/MEK/ERK pathway using selumetinib as a single agent in the treatment of HCC is not effective.
Combinational use of selumetinib with other agents is under active evaluation in separate clinical trials.
Anti-epigenetic Regulation
Belinostat
Belinostat (PXD101, Spectrum Pharmaceuticals, Irvine, USA) is a small compound inhibitor of HDAC (Fig. 2). In a preclinical study with 3 HCC cell lines, belinostat showed inhibitory effects on cell growth on a dose-based manner and provided the basis for further clinical trials. 120
In a multicenter phase I/II clinical trial, belinostat was tested in patients (18 in phase I and 42 in phase II) with unresectable HCC and chronic liver disease. The pharmacokinetics of belinostat demonstrated a linear relationship from 600 to 1400 mg/m2 with no significant accumulation. When used with a 1400 mg/m2 dose in the phase II study, the stable disease rate was 45.2%. Among patients whose tumors expressed high levels of RAD23B, 58% achieved disease stabilization compared with 14% of those with low RAD23B expression in their tumors. Further, the median OS and PFS obtained from this trial were 6.6 and 2.64 months, respectively. 121
This clinical trial suggested that belinostat can safely stabilize unresectable HCC. Furthermore, the expression level of RAD23B may be eventually used as a biomarker for belinostat clinical response and as a helpful guide for a personalized medicine approach for HCC treatment. 122
Conclusion
Targeted therapies that specifically block tumor-associated signaling pathways using either small molecular compounds or humanized monoclonal antibodies to inhibit angiogenesis and tumor growth represent a new trend of promising novel treatments of HCC. The success of sorafenib in the treatment of patients with advanced HCC has established sorafenib as a new standard of care for patients with advanced HCC and encourages a number of novel agents being tested in clinical trials. Results of pharmacokinetics, safety, and efficacy obtained from those finished and ongoing trials will definitely provide valuable information for the future improvement of the targeted therapies in HCC.
While use as a single agent for the first-line therapy may not display full benefits to patients, sorafenib is being tested with either another targeted agent or conventional treatments such as chemotherapy and surgery as the second-line therapy. More clinical trials are thus needed for those emerging novel agents both as single agents and in combination with other therapies.
The design of targeted therapies is primary based on our understanding at the molecular level of the relationship between signaling pathways and the disease. Given the fact that HCC is highly heterogeneous and no dominant signaling pathway has been found so far, more investigations are warrant in the future to elucidate the molecular pathogenesis of this disease and the underlying mechanisms.
Author Contributions
Conceived and designed the experiments: ZW. Analyzed the data: ZW. Wrote the first draft of the manuscript: ZW. Contributed to the writing of the manuscript: ZW, CD, YL. Agree with manuscript results and conclusions: ZW, CD, YL. Jointly developed the structure and arguments for the paper: ZW, CD, YL. Made critical revisions and approved final version: ZW. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication the authors have provided signed confirmation of their compliance with ethical and legal obligations including but not limited to compliance with ICMJE authorship and competing interests guidelines, that the article is neither under consideration for publication nor published elsewhere, of their compliance with legal and ethical guidelines concerning human and animal research participants (if applicable), and that permission has been obtained for reproduction of any copyrighted material. This article was subject to blind, independent, expert peer review. The reviewers reported no competing interests. Provenance: the authors were invited to submit this paper.
