Abstract
Objective
We assessed the racial (Black–White) differences in glycemic control, prevalence of abnormal lipid profiles and factors influencing temporal trends in children with type 1 diabetes (T1DM).
Methods
This retrospective study was done in children with T1DM. The outcome measure was based on glycemic control and all lipid determinations which were stratified according to the published guidelines.
Results
The study included 181 children; 76.2% Whites and 23.8% Blacks. The mean glycated hemoglobin (A1C) was higher in Blacks than in Whites (p < 0.0001). Blacks had elevated total cholesterol (TC) (p = 0.0013), lower TC/HDL ratio (p < 0.0001) and higher concentration of HDL (<0.0001) when compared to Whites. The longitudinal analyses over a 5 year period showed changes in A1C significantly associated with changes in the lipid profiles. The lipid profiles in Blacks were more altered by the trend in A1C with changes in the TC (p = 0.0079), non-HDL (p < 0.0001) and HDL (p < 0.0001).
Conclusions
Black children with T1DM have poorer glycemic control. However they retained excellent levels of HDL when compared to Whites.
Introduction
Both diabetes and dyslipidemia are independent risk factors for cardiovascular disease. Moreover, in long standing type 1 diabetes (T1DM), there is an increased likelihood for atherosclerosis.1,2 Amongst the reports on prevalence of abnormal lipid profiles in children with T1DM,3–12 there is only few on temporal trends. 12 Limited information is available on Black-White difference on lipid abnormalities in children with T1DM. 13 It is well known that racial differences exist in glycemic control and development of diabetic complications in adults with type 2 DM.14,15
This retrospective study aimed to assess the racial differences in glycemic control and lipid profiles in children with T1DM. We also assessed the prevalence of abnormal lipid profiles in children with T1DM and characterized the factors influencing the trends in dyslipidemia in these subjects. Identification of such risk factors could lead to preventive measures and intervention strategies in this high risk population. To our knowledge this is the first study to address the temporal trends in glycemic controls and lipid profiles in children with T1DM with regard to race.
Methods
Children with T1DM, who are being followed by the department of Pediatric Endocrinology, Metabolism and Diabetes at the Children's Hospital of Birmingham, Alabama, were included in the study. Patient records over a 5 year period of time, 2001–2006 were abstracted. Patients were identified using the diagnosis code of 250.1 for T1DM. Only insulin dependent patients with positive autoimmune markers against islet cell or GAD-65 antigens and those with more than one lipid measurement were included in the study. Children with biochemical criteria for hypo or hyperthyroidism, those on thyroid medications, children who had hemoglo-binopathies, celiac disease and children with type 2 diabetes were excluded. All children with T1DM received similar diabetes education according to the endocrine division policy and patients were given similar instructions to contact the pediatric endocrinologist frequently for ongoing insulin adjustments to maintain euglycemia. In addition, the insulin dose was also adjusted during the clinic visits based on the blood sugar logs. All patients were initially on multiple daily dose injection (MDI).
The information on the race was collected based on the demographic data provided by the patient. We collected longitudinal data on BMI and A1C from the charts.
Due to the retrospective nature of the data collection we could not reliably ascertain the family history of lipid abnormalities and, hence, this issue is not included in this study. Once the insulin therapy was initiated lipid measurements were not taken under fasting conditions and hence, we did not incorporate triglyceride and LDL levels into our analysis. Non-HDL is calculated from the difference between the levels of total and HDL cholesterol. Non-HDL is a comprehensive measure of all potential atherogenic lipoproteins (VLDL, intermediate-density lipoprotein [IDL], LDL, and lipoprotein[a]). It can be calculated in the non-fasting state or in the setting of hypertriglyceridemia. The primary advantages of calculating non-HDL is due to the fact that it requires measurement of only TC and HDL, both of which can be measured reasonably accurately in a non-fasting sample. 16 Non-HDL cholesterol > 160mg/dL is designated as “high” according to ATP III guidelines. 17
Measurements of total cholesterol (TC) and HDL were done by standard enzymatic methods. Hypercholesterolemia was stratified according to the American Heart Association optimal lipid levels for children and adolescents 12 as: TC level < 170 mg/dL acceptable, 170–199 mg/dL borderline, >200 mg/dL high. Optimal HDL level for children with diabetes was defined as >35mg/dL. 18 A1C values measured by DCA2000 assay included in the analysis were those determined at the time of the lipid profile measurements.
Statistical analysis
Standard procedures in SAS® (Statistical Analysis Software) version 9.1 were used for descriptive and comparative statistical analysis. We have confirmed the normality of continuous variables. Linear correlation between A1C, BMI and lipid variables (TC, HDL) was examined using Pearson correlation coefficient. These correlations were further examined with linear regression model (ANOVA) with adjustments for age, gender and race factors. Longitudinal analysis with mixed linear model was used to evaluate the changes of A1C and lipid abnormality over time. The association between non-HDL, TC categories and race and sex were tested with Mantel-Haenszel statistics. A nominal p value of <0.05 was considered statistically significant. The data are expressed as mean ± S.D.
Results
After excluding those patients with the aforementioned exclusion criteria, the study included 181 children diagnosed with diabetes from age 2.6 to 16 years. Among these children, 94 (51.9%) are males and 87 (48.1%) are females. There were 76.2% Whites and 23.8% Blacks. The mean age of diagnosis of diabetes was not different between Black and White children (7.8 ± 2.9 years). The mean BMI did not differ statistically among races at the time of diagnosis and at follow up.
There were 181 patients with lipid profiles at two different time points, 115 with lipid profiles at three time points, 40 with lipid profiles at four time points, 7 children with five time points, and 3 children with 6 lipid profiles since diagnosis of diabetes. We examined all longitudinal data points for lipid values. The lipid measurements were further characterized based on the duration of diagnosis of diabetes.
Females had higher TC (p = 0.05), and non-HDL (p = 0.05) when compared to males. Based on all the observations, Black children had A1C of 10.6% ± 2.2% and White children had 8.7% ± 1.6% (p < 0.0001). In Blacks serum TC was 175.5 ± 35.0 mg/dl, whereas Whites had 164.9 ± 30.3 mg/dl (p = 0.0013). Similarly Black children had serum HDL of 66.6 ± 15.9 mg/dl, whereas White children had 56.3 ± 13.0 mg/dl (p < 0.0001).
In univariate analysis, there was a statistically significant association between A1C and TC (p < 0.001, Pearson correlation coefficient = 0.28). The multivariable regression analysis, after controlled for age, sex, BMI and race still showed a strong positive association with TC (p < 0.0001, β coefficient = 408.89), non-HDL (p < 0.0001, β coefficient = 486.62) and negative association with HDL (p = 0.02, β coefficient = –77.74) between the lipid variables and A1C. There was also a strong negative association between BMI and HDL (p < 0.0001, β coefficient = 0.69) and a strong positive association between BMI and non HDL (p = 0.004, β coefficient = 1.01), when adjusted for age, sex and race.
Table 1 classifies the lipid variables in terms of duration of diabetes. Figures 1 and 2 depict the mean changes in A1C, TC and HDL by race and duration. Mixed-model longitudinal data analysis, using A1C as the independent variable showed that the changes in A1C from baseline are strongly associated with the changes of TC (p < 0.0001, β = 0.000195), non-HDL (p < 0.0001, β = 0.000283), TC/HDL (p < 0.0001, β = 0.004065) and with moderate association with the changes HDL (p < 0.07, β = –0.00027) when controlled for age, sex and race, time and baseline. These changes were significantly different between races. Blacks were more affected by the changes in the A1C over time with changes in the TC (blacks vs. whites β = 0.007358, p = 0.04), TC/HDL ratio (blacks vs. whites β = 0.00856, p = 0.01), non-HDL (blacks vs. whites β = 0.00755, p = 0.027) and HDL (blacks vs. whites β = 0.00820, p = 0.03). Elevations in TC and HDL persisted in Blacks even when controlled for A1C. Longitudinal analysis using mixed model, the changes of TC, HDL, and Non-HDL with time are strongly correlated with the changes of A1C (all p < 0.0001), when controlled for age, sex and race.

Mean A1C by race and duration of diabetes. Even though there was no significant difference in A1C at diagnosis between black and white children, over time Black children had higher A1C.

Mean changes in TC and HDL by race and duration of diagnosis of diabetes. Black children had higher HDL and total cholesterol with time.
Descriptive statistics of lipid variables and A1C with duration of diabetes.
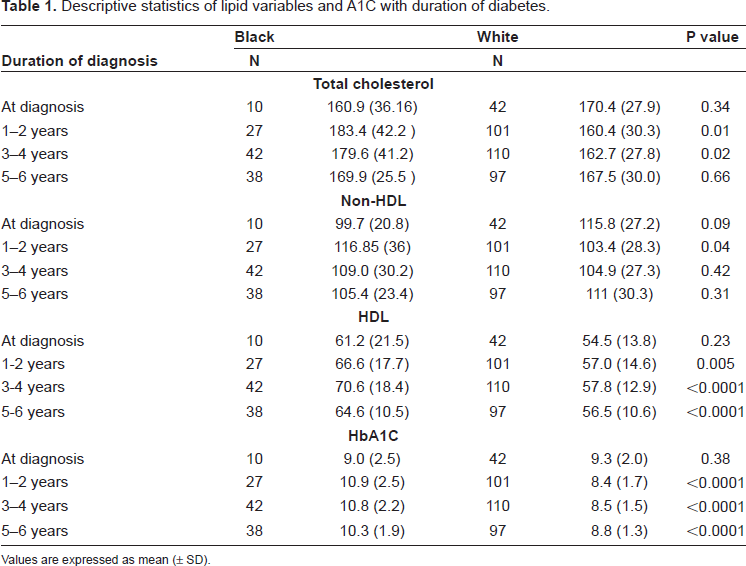
Values are expressed as mean (± SD).
Discussion
In agreement with other researchers3,12,19 our data clearly indicate that TC, non-HDL and HDL are significantly affected by chronic glycemic control, as shown by the strong statistical correlation with A1C. In accordance with previous literature,9,12,20 we found that A1C also has a significant negative correlation with HDL. Other factors affecting the lipid profile in children with T1DM in our study were sex, duration of diabetes, BMI, and race. The effect of BMI on non-HDL and HDL has been previously reported.3,12,21
Even though the A1C is negatively correlated with HDL levels, the Black children had higher concentration of HDL at baseline and at the end of the study period. Historically Black children are known to have higher HDL.22–24 Insulin therapy is reported to increase antiatherogenic particles. 25 It is a very important observation that inspite of the poor glycemic control, there were enhanced levels of HDL concentrations in Black children.
Further studies are required to identify the cause for these racial disparities in glycemic control outcome. Delamater et al. has previously reported that A1C was higher in black than white children after statistically adjusting for the effects of insulin dose, diabetes duration, and socioeconomic status. 26 Hanson et al. has previously reported poor glycemic control in Black females with T1DM. They reported that the poor glycemic control was not associated with a deficiency in the girls’ knowledge about diabetes, adherence to treatment, self-concept, coping patterns, family functioning, stress, social support, or involvement in the healthcare system. 27 Libman et al. has reported that Black children and adolescents, with type 1 diabetes frequently have features associated with insulin resistance and type 2 diabetes and suggest a role for heterogeneous pathogenesis in Black and White children with diabetes. 28 All the children in the present study received the standard diabetic education provided by the Division of Pediatric Endocrinology. After the initial insulin treatment all families were instructed to contact the endocrinology department to report their blood sugars and their insulin dosages were adjusted according to blood sugars. It is also important to note that there was no BMI difference between Black and White children with type 1 diabetes in our population. Hence, one will have to attribute the reasons for disparities in the outcome to greater burden of disease among minorities or differences in access or utilization of diabetes care, socioeconomic status, and genetic predisposition to insulin resistance18,28 or other factors.
Limitations of the study: Familial factors are important determinants of lipid levels. 29 Hence lack of reliable family history to exclude an underlying genetic cause of hyperlipidemia. As there is significant fall off in the numbers of patients reported having lipid measurements by the fourth time point, potential bias in the longitudinal data is possible.
Conclusions
The results of this study illustrate that Black children with type 1 diabetes have a higher degree of chronic hyperglycemia as determined by the surrogate A1C. The main factors determining the lipid profile in children with T1DM are age, glycemic control, BMI, duration of diabetes, sex and race.
Disclosure
The authors report no conflicts of interest.
