Abstract

Adolescence is a challenging time for managing diabetes, with recent data from the type 1 diabetes (T1D) exchange highlighting teenagers have the highest risk of poor glycemic control. 1 Insulin pump therapy (CSII) is a standard treatment modality for patients with type 1 diabetes but caution in the use of CSII therapy in this group stems from the potential for metabolic decompensation to diabetic ketoacidosis because of the sole use of rapid acting insulin, or secondary to experience bias that is in conflict with published data supporting the use of CSII.2,3 To evaluate the safety and efficacy of CSII in adolescents (aged 14-17 years) with T1D for > 1 year, insulin pump naïve and with very poor glycemic control (defined for this study as an average HbA1c >10.0% in the preceding 6 months) we conducted a prospective cohort study over a 12-month period (ACTRN12614001198617, ethics approved by Princess Margaret Hospital Health Research Ethics Committee [201407EP]).
Before pump start (MiniMed Paradigm™ 722, Medtronic, USA) a comprehensive education package was administered. Ten participants were enrolled (7 females, mean age 16.1 ± 1 years, mean diabetes duration 8.0 ± 3.9 years). All but one participant remained on CSII for the 12-month study duration. HbA1c improvement was maximal at 3 months (HbA1c reduction 3.0%, P = .001), before rising and plateauing at 12 months (HbA1c reduction at 12 months: 1.5%, P = .016) (Figure 1). There were 3 events of diabetic ketoacidosis, all in one participant. Study participants had improved diabetes-specific quality of life at 6 months (73.2 ± 11.5) compared to baseline (59.7 ± 15.1, P = .03), but not at 12 months, and parents had improved diabetes-specific quality for the study duration. On the visual analogue scale of the EQ-5D-Y, the low baseline score (57 ± 8.2) showed improvement at 6 weeks (74 ± 6.0, P = .01) and 6 months (77 ± 4.7, P = .02), but was no longer significantly different at 12 months (68 ± 6.6, P = .29). Treatment satisfaction was high and unchanged through the study: 137 ± 11 at 6 weeks, 137 ± 13 at 6 months, and 134 ± 10 at 12 months.
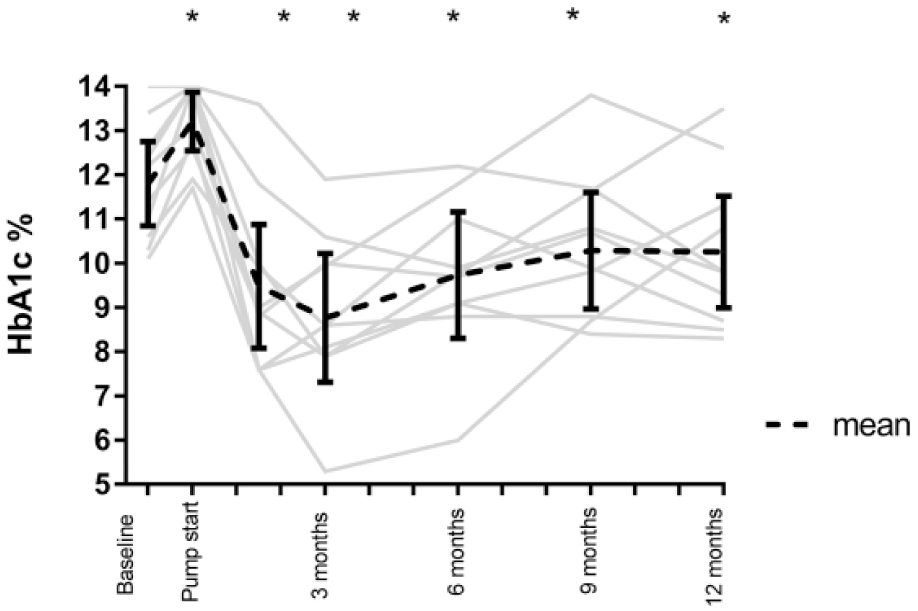
HbA1c over study duration, individual participants, and mean with 95% confidence interval. The repeated measures ANOVA showed that within the intervention cohort, HbA1c differed significantly as a function of time, F(6, 54) = 13.2, P < .001. HbA1c trajectory illustrates interindividual variability in response.
Our results support that of others; DeVries et al investigated CSII in adults with long standing poor glycemic control, and while CSII improved HbA1c by 0.84%, the study was limited by a short duration of 16 weeks. 4 Over 2 decades ago, Steindel et al conducted a 24-month retrospective review of 6 children (aged 12-16.5 years) with baseline poor control (mean HbA1c 9.02% for the previous 12 months), who initiated CSII, and found that the intervention was safe and cost effective, without reducing HbA1c. 5
We acknowledge this study is limited by its small size, and lack of a control arm. Outcomes may be biased because the participants nominated themselves to be included in the trial, and were therefore already motivated for change. Furthermore, as these patients received significant education in the initial part of the study, this may have affected the results independently of the insulin pump.
We conclude that CSII, combined with high-quality and flexible education and supportive framework, can be an effective tool to improve glycemic control in adolescents with long-standing treatment resistance.
Footnotes
Abbreviations
CGM, continuous glucose monitoring; CSII, continuous subcutaneous Insulin infusion, or insulin pump therapy; T1D, type 1 diabetes; WACDD, Western Australian Children’s Diabetes Database.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
