Abstract
Atrial fibrillation is the commonest arrhythmia worldwide and is a growing problem. AF is responsible for 25% of all strokes, and these patients suffer greater mortality and disability. Warfarin has traditionally been the only successful therapy for stroke prevention, but its limitations have resulted in underutilisation. Major progress has been made in AF research, leading to improved management strategies. Better risk stratification permits identification of truly low-risk patients who do not require anticoagulation and we are able to simplify ourevaluation of a patient's bleeding risk.
The advent of novel anticoagulants means warfarin is no longer the only choice for stroke prophylaxis. These drugs circumvent many of warfarin's inconveniences, but only long-term study and use will conclusively demonstrate how they compare to warfarin. The landscape of stroke prevention in AF has changed with effective alternatives to warfarin available for the first time in 60 years—but each new option brings new considerations.
Introduction
Atrial fibrillation (AF) is the most common arrhythmia encountered worldwide, affecting 1% of the general population 1 with a lifetime risk of 25% for adults. 2 The prevalence of AF increases with age,3,4 with almost one in ten patients over 80 affected. 1 AF is a growing problem and it is estimated that between 6 million and 16 million people in the US will suffer with the rhythm disorder by 2050.1,5 These projected increases are owed largely to the ageing population, and the increased survival of patients with chronic cardiac disorders which predispose to AF—such as ischaemic heart disease and heart failure.6,7 The presence of AF in these patients with underlying heart disease is associated with a worse outcome, 8 and increased healthcare costs: AF is responsible for an estimated 1% of the United Kingdom National Health Service expenditure in the UK. 9
The most feared and devastating complication of AF is stroke. The presence of AF is an independent risk factor for stroke, conferring a five-fold increase in the stroke risk. 10 AF is held responsible for up to a quarter of all strokes, 11 and these patients have more severe strokes: greater mortality, longer hospital stay, increased disability.12–14 AF is also a significant risk factor for stroke recurrence. 15 There is substantial evidence to suggest that most thromboembolic complications could be prevented with adequate pharmacological anticoagulation.16,17
Traditionally, vitamin K antagonists (eg, warfarin) were the only available therapeutic option for oral anticoagulation. Warfarin provides highly effective prophylaxis against thromembolism. 18 Unfortunately, warfarin has many limitations and inconveniences 19 which have contributed to reluctance amongst both patients and physicians to use this therapy. 20 This variability in the management of stroke prevention in AF has seen renewed efforts in the past decade to establish validated risk stratification models and novel alternative antithrombotic therapies. The progress made in this sphere has offered increased guidance and choice to both clinicians and patients to improve the prevention of stroke in atrial fibrillation.
This article reviews the current treatment options available for stroke prevention in AF, their evidence base, safety and efficacy. Attention is given to the novel oral anticoagulants which are available now or soon to become available, and their potential place in therapy.
Risk Stratification
The risk of stroke in AF is dependent upon the presence or absence of several risk factors.21,22 Traditionally these risk factors were used to stratify patients into “low”, “intermediate”, or “high” risk for stroke. Older guidelines used this grouping to recommend oral anticoagulation to high-risk patients, aspirin for low-risk patients, and a choice of either anticoagulation or aspirin for the intermediate grouping. This had the potential of introducing confusion (for example, for intermediate-risk patients no definitive guidance was given as to whether to offer anticoagulation or aspirin) and also undertreating a cohort of patients at substantial risk of stroke.
There is evidence that aspirin does not reduce the risk of stroke in low-risk patients, 23 and that warfarin is superior to aspirin for patients at intermediate risk of stroke.24,25 The CHADS2 score 26 also classified a large number of patients into the intermediate group. These limitations spurred on the development of a risk stratification system that more reliably identifies truly low-risk patients, and minimises patients being denied oral anticoagulation when they would derive significant benefit from it.
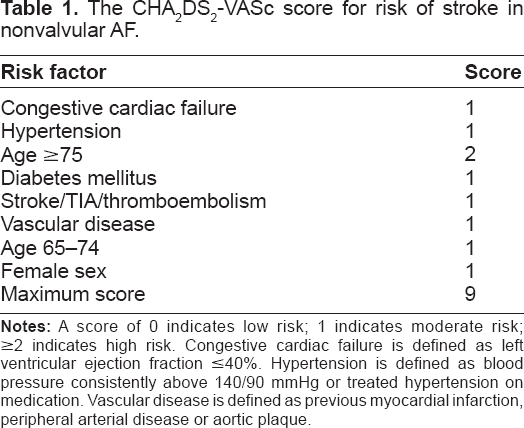
The CHA2DS2VASc score [Table 1] was suggested as such a scheme to improve risk stratification for stroke, to focus more on the identification of such ‘truly low risk’ patients. 27 The CHA2DS2VASc scoreis better at identifying truly low-risk patients, and categorises fewer patients as intermediate risk. 28 It has now been validated in various large real-world cohort of patients 29 and may even perform better than CHADS2 in identifying patients at high-risk of stroke. The CHA2DS2VASc score is now included in European guidelines on the management of atrial fibrillation. 30
The CHA2DS2-VASc score for risk of stroke in nonvalvular AF.
Bleeding is the most important and feared complication of anticoagulant therapy among clinicians and patients. Bleeding risk is a limiting factor in the prescription of antithrombotic therapy, and leaves a substantial number of patients untreated when they have clear indications for anticoagulation. 31 Clinicians should undertake an assessment of a patient's risk for bleeding before initiating anticoagulant therapy. 32
The novel HAS-BLED score 33 was developed to allow clinicians to assess simply and practically assess the individual risk of bleeding in their patients before initiating antithrombotic therapy, and makes clinicians think about the common correctable risk factors for bleeding, for example, uncontrolled blood pressure, concomitant aspirin/NSAID use with oral anticoagulation, labile INRs, etc [Table 2]. It allows periodic reassessment of a patient's bleeding risk considers the quality of the anticoagulation control. 34 This risk score has been validated in a large cohort of real-world patients, 35 and performs favourably when compared to other scoring schemes. 36 The HAS-BLED score has also been included in European guidelines, 30 and when used in conjunction with the CHA2DS2VASc score it allows clinicians to make a simple and informed judgment as to the relative benefits and risks of anticoagulation.
The HAS-BLED score for bleeding risk.

The Ideal Anticoagulant
The efficacy of warfarin as prophylaxis against stroke is established and unequivocal.18,37 Unfortunately, there are many limitations associated with warfarin [Table 3]: its narrow therapeutic window, slow onset and offset of action, unpredictable pharmacokinetics and pharmacodynamics leading to variability in dose response amongst individuals and multiple drug and food interactions. Due to these factors, warfarin requires close laboratory monitoring of coagulation via the INR and subsequent dose adjustments. These regular clinic attendances bring an increased financial burden and inconvenience to patients. Thus many patients who are eligible for warfarin choose not to use it. 38
Limitations of warfarin.

A clinically viable alternative to warfarin will need to possess several key characteristics.39,40 Novel agents [Table 4] need to be proven to be predictably at least as effective as warfarin in clinical trials. Other key features include: oral administration, fixed dose regimens, wide therapeutic windows, low propensity for food and drug interactions, predictable pharmacokinetics and pharmacodynamics with little inter and intra patient variability [Table 5]. New therapies would of course need to be safe and well-tolerated, with low frequency and severity of adverse effects. They should also obviate the need for regular coagulation monitoring.
Pharmacokinetic and pharmacodynamics properties of the novel anticoagulants.

Characteristics of the ideal anticoagulant.

Mechanism of Action and Pharmacokinetic Profile
Warfarin
Warfarin is a vitamin-K antagonist that produces its anticoagulant effect by interfering with the cyclic interconversion of vitamin K and its epoxide. Vitamin K is a cofactor for the posttranslational carboxylation of glutamate residues of vitamin K-dependent clotting factors(II, VII, IX, X).41,42 These coagulation factors require carboxylation to be biologically active, therefore when warfarin inhibits the vitamin K conversion cycle it leads to hepatic synthesis of decarboxylated (or partially carboxylated) proteins with reduced coagulant activity. 43 The effect of warfarin can be counteracted by vitamin K1 (either ingested in food or administered therapeutically) and this effect may persist for up to a week as vitamin K accumulates in the liver.
Warfarin has a high bioavailability, 44 is absorbed quickly and reaches maximal plasma concentrations within 90 minutes. 45 Warfarin has a half-life of 36-hours and predominantly circulates bound to albumin. Warfarin accumulates in the liver where it is metabolised by two pathways. The dose-response of warfarin is impacted on by environmental and genetic factors. Polymorphisms of genes that encode for the vitamin-K epoxide reductase enzyme and CYP2C9 enzyme have been identified as the most important contributors to the wide inter-individual variations in dose requirements.46–48 Drugs may influence the pharmacokinetics of warfarin by reducing GI absorption or interfering with metabolic clearance; 49 drugs may also disrupt the pharmacodynamics of warfarin by inhibiting synthesis or increasing clearance of vitamin K-dependent clotting factors. Dietary intake of vitamin K can also impact on the anticoagulant effect of warfarin. 50
Direct Thrombin Inhibitors
The final step of the coagulation pathway requires thrombin to convert fibrinogen to fibrin. Direct thrombin inhibitors bind to thrombin and prevent its interaction with substrates; this inhibits fibrin production. 51 The effect of this class of drugs also prevents thrombin-mediated activation of activation of Factors V, VIII, XI, and XIII, and thrombin-induced platelet-aggregation. 52 Direct thrombin inhibitors can inhibit clot-bound and free thrombin, owing to the fact they bind directly to the active catalytic site. 53 Numerous parenteral direct thrombin inhibitors are available (such as bivalirudin, hirudin and lepirudin) but the lack of an oral preparation does not lend them to use in lifelong stroke prevention for patients with AF.
Ximelegatran was the first available oral direct thrombin inhibitor. 54 It is a prodrug that is rapidly converted to melegatran (its active form). 55 Ximelegatran had twice daily fixed dosing with a fast onset and offset of action. There were no food interactions, 56 little potential for drug interactions, 57 and low variability in the dose-response relationship. 58 Ximelegatran was withdrawn from the market in 2004 due to its potential to cause raised liver enzymes and some reported cases of fulminant hepatic failure. 59
Dabigatran etexilate is an oral prodrug which is converted in the liver to its active compound, dabigatran. 60 Dabigatran is a competitive, direct and reversible inhibitor of thrombin. 52 As detailed above, dabigatran exerts an effect on both clot-bound and free thrombin. Dabigatran has a fast onset of action (peak 0.5-4 hours), a half-life of 17 hours with multiple doses (7-9 hours with single doses), 62 and reaches clinical steady state within 2.5 days of initiation. 61 Dabigatran is predominantly (80%) cleared by the kidneys. 61 Neither the prodrug nor its metabolite exert an effect on the CYP system, thus dabigatran lends itself to fewer food and drug interactions than warfarin. Absorption of dabigatran may be delayed by food, 63 and there is an age effect on pharmacokinetic parameters 64 although no reported gender effect. 65
Factor Xa Inhibitors
Factor Xa plays a key role in the common pathway of the clotting cascade. This protein is located at the junction of the intrinsic and extrinsic pathway, leading to the conversion of prothrombin to factor IIa (thrombin). Thrombin then converts fibrinogen to fibrin, leading to clot formation. Parenterally administered heparin-based anticoagulants act by binding to antithrombin and producing a conformational change to inhibit both thrombin and factor X. 66 Fondaparinux inhibits factor Xa indirectly, utilising antithrombin as a cofactor and producing a conformational change similar to heparin to inhibit factor Xa—but it can only inhibit factor Xa in its free form. 67 Trials have demonstrated the efficacy of heparin-based anticoagulants improves with greater selectivity for FXa.68,69
The emerging direct factor Xa inhibitors do not require antithrombin as a cofactor, and bind directly to the active site of factor Xa. 70 Factor Xa has fewer effects outside of the clotting cascade, so may cause fewer side-effects than direct thrombin inhibitors 71 (although direct thrombin inhibitors may also have beneficial effects outside the coagulation pathway 35 ).
Apixaban is an oral, selective, reversible 72 direct factor Xa inhibitor. 73 It has high oral bioavailability 74 and onset of action is within three hours. 60 The drug has a half-life of 12 hours 75 and is cleared via multiple pathways: 25% renal, 75% in faeces. 74 Data indicate that apixaban does not inhibit or induce CYP enzymes. 77 Its absorption is not impacted by food. 77
Rivaroxaban is an oral, reversible, direct factor Xa inhibitor. 78 It inhibits prothrombinase activity, as well as free and clot-associated FXa activity in plasma. 79 It has high oral bioavailability 80 and is rapidly absorbed with a half-life of 9 hours in healthy subjects 81 and 12 hours in those aged over 75. 82 It has a rapid onset of action with maximal concentrations reached between 2 and 4 hours. 80 Rivaroxaban has a dual mode of elimination with no identified active circulating metabolite: one-third of the dose is renally cleared, the remainder being metabolized by the liver. 83 The pharmacokinetics of rivaroxaban are dose-proportional 84 and generally unaffected by gender, body weight or extremes of age. 85 Although rivaroxaban can theoretically be affected by drugs that interact with CYP3A4, 76 a low potential for clinically significant drug or food interactions has been reported.86–88
Clinical Studies: Efficacy and Safety
Warfarin
It was conclusively proved in a series of randomised control trials in the late eighties and early nineties that warfarin was effective prophylaxis for stroke prevention in atrial fibrillation when compared to placebo.88–93 In 1994 pooled data from five studies demonstrated that warfarin conferred a 68% risk reduction in stroke compared with no therapy, and with no increased risk of bleeding. 94 A meta-analysis in 2002 showed that warfarin significantly reduced ischaemic stroke when compared to aspirin, 95 and this was confirmed in 2007 when a further analysis of almost 30,000 patients revealed that warfarin reduced strokes by 40% compared to aspirin. 96 The benefit of warfarin over aspirin was preserved when trials were confined to an elderly population. 97
The ACTIVE-W trial 98 in 2006 sought to identify whether a combination of aspirin and clopidogrel would be non-inferior when compared to warfarin. The study showed clearly that warfarin was superior to aspirin and clopidogrel, and was stopped early due to the clear benefit of oral anticoagulation. The rates of major bleeding in the two study groups were comparable (2.4% per annum for clopidogrel and aspirin vs. 2.2% per annum for warfarin). In the ACTIVE-A trial 99 patients deemed unsuitable for warfarin were given either aspirin alone or a combination of aspirin and clopidogrel. Aspirin plus clopidogrel reduced the rate of ischaemic stroke by 28% compared with aspirin alone. However, it is worth noting that the rates of major bleeding with aspirin and clopidogrel were 2.0% in the ACTIVE-A study group. This figure is similar to the major bleeding rate of dual antiplatelet therapy observed in ACTIVE-W, and also to the rate of major bleeding with warfarin. Therefore, aspirin and clopidogrel in combination would not be a suitable alternative to warfarin in patients deemed high-risk for haemorrhage. Dual antiplatelet therapy may, however, be a therapeutic option for patients truly ineligible for warfarin for other reasons 100 (inability or unwillingness to undergo regular monitoring, for example).
Dabigatran
Dabigatran was initially evaluated in 2007 in the phase II trial PETRO: 101 in this trial, 502 patients with nonvalvular AF were randomized to dabigatran 50, 150, or 300 mg twice/day alone or combined with aspirin 81 or 325 mg/day versus open-label warfarin in patients with a CHADS2 score of 1 or higher. Major bleeding was more common in patients taking dabigatran 300 mg with aspirin (6.25%) compared with dabigatran 300 mg alone (0%, P < 0.02). Thromboembolism was only observed in patients randomised to dabigatran 50 mg.
The RE-LY trial was a large randomised controlled trial comparing dabigatran with warfarin. 102 It was a phase III, blinded, noninferiority trial in 18,113 patients with nonvalvular AF with a CHADS2 score of 1 or higher or who were older than 65 years with coronary artery disease. 103 Patients were randomised to either dabigatran, at a dosage of 110 or 150 mg twice daily or warfarin titrated to a goal INR of 2-3. The primary efficacy outcomes of the study included stroke or systemic embolism. Efficacy outcomes occurred at 1.69% per year in patients assigned to warfarin compared with 1.53% in the dabigatran 110-mg group (RR 0.91, CI 0.74-1.11, P < 0.001 for noninferiority, P = 0.34 for superiority compared with warfarin) and 1.11% in the dabigatran 150-mg group (RR 0.66, 95% CI 0.53-0.82, P < 0.001 for noninferiority and superiority compared with warfarin). This difference in effect between dabigatran 150 mg and warfarin was found to occur at 2 months into the trial and was carried throughout until trial completion. Thus low-dose dabigatran was shown to be non-inferior to warfarin and high-dose dabigatran was shown to be superior to warfarin. No statistically significant difference was demonstrated between the groups for the secondary outcome of all-cause mortality (4.13% for warfarin vs. 3.75% for dabigatran 110 mg; RR 0.91, 95% CI 0.80-1.03). There was, however, a numeric decrease in both dabigatran groups that approached significance for those receiving dabigatran 150 mg.
Major bleeding was the primary safety outcome, defined as a reduction in haemoglobin level of 2 g/dL, transfusion requiring at least 2 units of blood, or symptomatic bleeding in a critical area or organ. Major haemorrhage occurred in 3.36% per year in patients taking warfarin, 2.71% in low-dose dabigatran (RR 0.8, 95% CI 0.69-0.93, P = 0.003 vs. warfarin), and 3.11%/year in high-dose dabigatran 150-mg group (RR 0.93, 95% CI 0.81-1.07, P = 0.031 vs warfarin). Thus major bleeding was less with 110 mg of dabigatran when compared to warfarin, and rates of major haemorrhage are similar with 150 mg dabigatran and warfarin. High-dose dabigatran was associated with a significantly increased risk of major gastrointestinal haemorrhage (1.51%) compared with dabigatran 110 mg (1.12%) or warfarin (1.02%). However, all composite major bleeding rates were found to be similar between dabigatran 150 mg and warfarin.
Discontinuation rates were 15% for dabigatran 110 mg, 16% for dabigatran 150 mg, and 10% for warfarin after the first year of the trial; and 21% for dabigatran 110 mg, 21% for dabigatran 150 mg, and 17% for warfarin at the end of the second year of the trial (P < 0.001 for dabigatran vs warfarin). The primary driver for this increased discontinuation of dabigatran was its propensity to cause dyspepsia: 11.8% for 110 mg and 11.3% for 150 mg compared to 5.8% for warfarin (P < 0.001). Thus, warfarin was better tolerated than dabigatran.
Dabigatran 150-mg was found to have an increased rate of myocardial infarction (0.74%) when compared with warfarin (0.53%/year). This effect that trended towards, but did not reach, statistical significance (RR 1.38, 95% CI 1-1.91, P = 0.04). It is possible that the increased occurrence of myocardial infarction observed in patients taking dabigatran in this trial owes more to the protective effects of warfarin rather than an inherent risk associated with dabigatran treatment. A meta-analysis comparing warfarin and other treatment regimes showed that warfarin was associated with significant reduction in myocardial infarction (relative risk, 0.77; 95% CI, 0.63-0.95; P < 0.01). 104
A subgroup analysis of the RE-LY trial investigated the safety and efficacy of dabigatran compared to warfarin with differing achievements in INR control. 105 The study found that the time in therapeutic range did not impact on the original trial's findings with regard to efficacy or intracranial haemorrhage. A further subgroup analysis was undertaken in patients with a history of previous stroke or TIA. 106 The effects of dabigatran compared with warfarin were not significantly different in patients with a previous stroke or TIA in any other outcomes compared with other patients—confirming dabigatran's role in secondary prevention and supporting the findings of the original RE-LY trial. An analysis of patients undergoing cardioversion 107 showed the risk of stroke and major haemorrhage on dabigatran was similar to warfarin.
A network meta-analysis compared dabigatran favourably to antiplatelet therapy: 108 dabigatran 150 mg reduced stroke risk by 63% compared to aspirin alone and 61% compared to dual antiplatelet therapy, as well as 77% when compared to placebo.
Rivaroxaban
The oral direct factor Xa inhibitor rivaroxaban was compared to warfarin in the ROCKET-AF study. 109 This trial was a phase III, randomised, double-blind, event-driven noninferiority trial with over 14,000 patients comparing rivaroxaban with warfarin in non-valvular AF (at least two documented episodes) and a history of stroke, TIA, or non-CNS embolism or at least two independent risk factors for future stroke. Enrolment of patients without stroke, TIA, or systemic embolism and only two risk factors was capped at 10% of the overall study population; all subsequently enrolled patients were required to have at least three stroke risk factors or a history of stroke, TIA, or systemic embolism. 86% of the total population had a CHADS2 score of 3 or higher.
Patients were randomised to rivaroxaban 20 mg once daily (or 15 mg once daily in patients with moderate renal impairment), or dose-adjusted warfarin titrated to a target INR of 2.5. The per-protocol, as-treated primary analysis was designed to determine whether rivaroxaban was noninferior to warfarin for the primary end point of stroke or systemic embolism; if the noninferiority criteria were satisfied, then superiority was analysed in the intent-to-treat population.
Rivaroxaban was similar to warfarin for the primary efficacy endpoint of prevention of stroke and systemic embolism (event rate 1.71 versus 2.16 per 100 patient years for rivaroxaban versus warfarin; hazard ratio [HR] 0.79, 95% confidence interval [CI] 0.66-0.96, P < 0.001 for noninferiority). The stricter intention-to-treat analysis also showed rivaroxaban was similar to warfarin but did not reach statistical significance for superiority: event rate 2.12 versus 2.42 per 100 patient years for rivaroxaban versus warfarin; HR 0.88, 95% CI 0.74-1.03, P = 0.117 for superiority. Superiority was only demonstrated in the per-protocol analysis of patients who continued to receive treatment for the 40-month trial period: event rate 1.70 versus 2.15 per 100 patient years for rivaroxaban versus warfarin; HR 0.79, 95% CI 0.65-0.95, P = 0.015 for superiority.
Major and nonmajor clinically relevant bleeding was similar with rivaroxaban and warfarin: event rate 14.91 versus 14.52 per 100 patient years for rivaroxaban versus warfarin; HR 1.03, 95% CI 0.96-1.11, P = 0.442. The rivaroxaban group demonstrated significantly less fatal bleeding (0.2 versus 0.5 per 100 patient years, HR 0.50, 95% CI 0.31-0.79, P = 0.003), intracranial haemorrhage (0.5 versus 0.7 per 100 patient years; P = 0.02). However, significantly more patients receiving rivaroxaban had a haemoglobin decrease of 2 g/dL or more (event rate 2.77 versus 2.26 per 100 patient years, HR 1.22, 95% CI 1.03-1.44, P = 0.019) and required a blood transfusion (1.65 versus 1.32 per 100 patient years, HR 1.25, 95% CI 1.01-1.55, P = 0.044).
The number of patients experiencing a serious adverse event was similar in the two groups (rivaroxaban 37.3% versus warfarin 38.2%) as was the documentation of an adverse event requiring discontinuation of the study drug (rivaroxaban 15.7% versus warfarin 15.2%). Premature discontinuation rates were also comparable, at approximately 23%. A higher percentage of patients taking rivaroxaban experienced epistaxis (10.1% versus 8.6%), and the rates of ALT elevation were the same in both groups (2.9%).
Apixaban
The AVERROES study was designed to evaluate the use of apixaban for stroke prophylaxis by comparing it to aspirin in patients unsuitable for warfarin. 111 The study enrolled 5600 patients with AF who were either intolerant of or unsuitable for warfarin and compared apixaban 5 mg twice daily (2.5 mg twice daily for patients aged over 80 years, weighing under 60 kg, or with renal impairment) with aspirin 81-324 mg/day. The study was prematurely because of an acceptable safety profile and benefit in favour of apixaban. After a year, patients taking apixaban were found to have a 55% reduction in the primary endpoint of stroke or systemic embolism (1.6% versus 3.7% per year, HR 0.45, 95% CI 0.32-0.62, P < 0.001). The rate of major bleeding was similar in both groups: 1.4% per year for apixaban and 1.2% per year for aspirin (HR 1.13, 95% CI 0.74-1.75, P = 0.57). Aspirin was the less well-tolerated therapy. 112
The ARISTOTLE trial has compared apixaban to warfarin in patients with atrial fibrillation. 113 It is a randomised phase III, double-blind, international trial comparing apixaban 5 mg twice/day versus warfarin titrated to an INR between 2 and 3 in over 18,000 patients. 114 The primary outcome was stroke (either ischaemic or haemorrhagic) or systemic embolism, and the trial was designed to test for noninferiority. Secondary objectives included an analysis for superiority with respect to the primary outcome and to the rates of major bleeding and all-cause mortality. The follow-up period was 1.8 years.
The rate of the primary outcome in ARISTOTLE was 1.27% per year in the apixaban group versus 1.60% per year in the warfarin group (hazard ratio with apixaban, 0.79; 95% confidence interval [CI], 0.66 to 0.95; P < 0.001 for noninferiority; P = 0.01 for superiority). This was primarily driven by a reduction in haemorrhagic stroke, as the rates of ischaemic stroke were comparable with warfarin: 0.97% per year in the apixaban group versus 1.05% per year in the warfarin group (hazard ratio, 0.92; 95% CI, 0.74 to 1.13; P = 0.42). Conversely, rate of haemorrhagic stroke was 0.24% per year in the apixaban group versus 0.47% per year in the warfarin group (hazard ratio, 0.51; 95% CI, 0.35 to 0.75; P < 0.001). Apixaban demonstrated a benefit with regards to all-cause mortality compared to warfarin: rates of death from any cause were 3.52% in the apixaban group versus 3.94% in the warfarin group (hazard ratio, 0.89; 95% CI, 0.80 to 0.99; P = 0.047). Apixaban was found to be safer than warfarin in regard to major bleeding: 2.13% per year in the apixaban group versus 3.09% per year in the warfarin group (hazard ratio, 0.69; 95% CI, 0.60 to 0.80; P < 0.001). Drug discontinuation occurred less frequently with apixaban compared to warfarin: 25.3% versus 27.5% (P = 0.001). The average time spent in therapeutic INR was 62.2% for the warfarin-treated patients. The reported adverse and serious adverse effects were similar in both groups of patients.
Patient Values and Preferences
An important consideration when deciding on a therapeutic strategy for stroke prophylaxis in patients with AF is that of patient preference. Patients will, generally speaking, be taking the prescribed therapies for the duration of their lives so it is crucial that they are adequately informed. Evidence suggests that well-informed patients are more compliant with therapy 115 and have better outcomes. 116 The predominant concern of patients is that of stroke, 117 and many are willing to accept slightly increased bleeding risks to avoid a stroke. Physicians tend to be more concerned with hospital admissions, whereas patients are ultimately worried about death. 118 The AF-AWARE study also found that physicians tended to overestimate the burden of anticoagulant treatment. 118 By and large, patients are willing to accept the inconveniences of anticoagulation to avoid serious adverse outcomes. 119 However, the use of decision-making aids leads to fewer patients opting for anticoagulation. 120
The advent of novel anticoagulant therapies is changing the landscape of stroke prevention in atrial fibrillation, and will significantly impact on patient preference. The new agents circumvent many of the inconveniences of warfarin: regular INR checks, dietary restrictions, drug interactions. They also, however, bring with them their own considerations and caveats [Table 6].
Potential limitations of new anticoagulants.

There are no known antidotes currently available for dabigatran, rivaroxaban or apixaban (although preliminary work is being done on a potential factor Xa inhibitor antidote, 121 and a phase IV study is investigating various reversal strategies for dabigatran). 122 The benefit of not requiring regular INR monitoring is offset by the fact that there is no validated way to assess the anticoagulant effect or level of the drug. We are also yet to establish how successful anticoagulant bridging prior to surgery can be achieved with the new agents [Table 7].
Anticoagulant bridging.

Dabigatran and apixaban require twice daily dosing, which is not an issue for rivaroxaban. Patients with GI dysfunction must be counselled regarding dabigatran's propensity to cause dyspepsia and increased rates of gastrointestinal bleeding. Dabigatran and rivaroxaban must be used with caution in patients with renal insufficiency, and the dose of dabigatran recommended by the FDA for renal impairment 123 was not studied in the RE-LY trial. 124 Concerns were raised following RE-LY of the increased (but small excess) number of myocardial infarction events in the dabigatran-treated group, but this finding has not been seen in the trials for apixaban or rivaroxaban. Furthermore, supplementary findings from the RE-LY trial 125 reporting newly identified events in the dabigatran group found the difference in the myocardial infarction rates was less pronounced.
The efficacy and safety of warfarin has been established over the last two decades, and it is readily reversed by vitamin K. Patients must be fully aware that, by definition, little is known regarding the long-term safety and efficacy profiles of novel agents. Further research ought to enhance our knowledge of and confidence in the new agents available for stroke prophylaxis in AF (such as the RELY-ABLE trial, 126 an extension of the RE-LY trial focussing on patient safety), and future work must emphasise patient preference.
Place in Therapy
Warfarin has a clearly defined place in therapy, as the established gold standard antithrombotic for stroke prevention in atrial fibrillation. The optimal INR for AF patients is 2.0-3.0, 127 with increased risk of thromboembolism and haemorrhage outside this range at either end. The benefit of warfarin is strongly linked to the proportion of time spent in the therapeutic INR range (time in therapeutic range, TTR). 128 A string of outcome measures in AF are all linked to the quality of the INR control: stroke and systemic embolism, myocardial infarction, major bleeding and death. 129 Even modest TTR improvements of 5%-10% have profound beneficial effects on clinical outcomes. 130 TTR in clinical trials is typically 60%-65%, but this exceeds that routinely achieved in clinical practice. 131 Very low TTR may completely obliterate the potential benefit of warfarin. It has been demonstrated that self-monitoring improves the quality of INR control and therefore outcome measures. 132 Despite its efficacy, the limitations of warfarin mean that a large group of patients with AF are not receiving effective prophylaxis against stroke.
The ultimate place in therapy of the novel oral anticoagulants is yet to be established. Currently, only dabigatran has been improved by the FDA and incorporated into guidelines. The US guidelines 133 recommend dabigatran 150 mg BD as an alternative to warfarin (or 75 mg for patients with renal impairment). The European guidelines 30 currently recommend 150 mg dabigatran twice a day for patients at low bleeding risk (HAS-BLED score 0-2) and 110 mg dabigatran twice a day for those at high risk of bleeding (HAS-BLED score 3 or greater). The Canadian guidelines 134 also recommend dabigatran as an alternative to warfarin.
Rivaroxaban and apixaban have completed phase III trials and will now undergo analysis and approval before their inclusion in guidelines. These two factor Xa inhibitors have not been shown to cause significant GI upset, so may represent an appealing treatment option for those patients unsuited to warfarin and unable to tolerate dabigatran due to dyspepsia. It is difficult to offer speculative comparisons between the new agents based on their study designs. For example, it may be tempting to infer that rivaroxaban is has more proven efficacy in high-risk patients as ROCKET-AF included few low-risk patients whereas RE-LY had significantly more (32% of patients in RE-LY had a CHADS2 score of 0-1, compared to < 1% for ROCKET-AF). Given the results of the ATLAS-ACS2 trial 138 (where rivaroxaban reduced the risk of the composite end point of death from cardiovascular causes, myocardial infarction, or stroke in patients with a recent acute coronary syndrome), rivaroxaban may find favour with clinicians treating patients following acute coronary syndromes. Conclusive comparisons between the new and emerging agents cannot be made until they have been evaluated against each other in trials.
As new agents are becoming available to clinicians for prevention of stroke in AF, new considerations must be undertaken [Table 8]. Patients who are well-established on warfarin with good quality INR control are unlikely to derive sufficient benefit to warrant switching to a new drug. The safety data available for the novel anticoagulants is reassuring, but long-term data is mandatory as patients will mostly be maintained on thromboprophylaxis for the duration of their lives. Emphasis must be given to individual patient characteristics, and patient preferences.
Cost-effectiveness of new agents.

Conclusions
For six decades, warfarin has been the only available therapeutic strategy for prophylaxis against stroke in patients with AF. Its limitations have led to its underutilisation and wide variability in AF management. Major progress has been made in AF research, providing clinicians with improved management strategies. Better risk stratification schemes permits accurate identification of truly low-risk patients who do not require anticoagulation, and those patients who ought to be receiving antithrombotic therapy. We are also able to simply and practically evaluate a patient's risk in relation to bleeding, enabling risk-benefit decisions to be made in a more straightforward manner.
The advent of novel anticoagulants means that warfarin is no longer the only choice for effective stroke prophylaxis. Clinicians will be tasked with coming to terms with the strengths and weaknesses of each new therapeutic option and employing them in appropriate settings. Only long-term study and use of novel anticoagulants will conclusively demonstrate how these drugs measure up to warfarin in terms of efficacy, safety and cost-effectiveness (see Table 8). The nature of AF thromboprophylaxis means that patients will likely be receiving a drug lifelong, so the importance of long-term data for new agents and an emphasis on patient values and preference cannot be overstated. The landscape of stroke prevention in atrial fibrillation has changed and will continue evolve as research continues into newer drugs 135 —and with each new management option comes new considerations.
Disclosures
GL has served as a consultant for Bayer, Astellas, Merck, AstraZeneca, Sanofi, BMS/Pfizer, Daiichi-Sankyo, Biotronik, Portola and BoehringerIngelheim and has been on the speakers bureau for Bayer, BMS/Pfizer, BoehringerIngelheim, and Sanofi Aventis. YA-none declared.
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
