Abstract
We examined angiogenesis-related gene expression profiles using collagen gel culture and a DNA chip. After isolation of total RNA from cultures before and after capillary tube formation, a mouse whole-genome array study was performed. Seventy-three out of over 35,000 transcripts were expressed after capillary tube formation. The majority of genes did not show any significant differences between before and after capillary tube formation. However, there were 7 up-regulated genes; tumor necrosis factor alpha-induced protein (Tnfaip) 2, vascular endothelial growth factor b (Vegfb), corticotropin releasing hormone receptor (Crhr) 2, vascular endothelial growth factor C (Vegfc), fibroblast growth factor (Fgf) 6, protein O-fucosyltransferase (Pofut) 1, hypoxia inducible factor 1 alpha subunit (Hif-1a). Eleven down-regulated genes were identified; ras homolog gene family member B (RhoB), Fgf-2, Thrombospondin (Thbs) 1, Angiogenin (Ang) 3, guanine nucleotide binding protein alpha (Gna) 13, tumor necrosis factor receptor superfamily member 12a (Tnfrsf12a), plasminogen (Plg), Elk3, upstream binding protein (Ubp) 1, integrin alpha V (Itgav), mitogen activated protein kinase (Mapk) 14. These up- and down-regulated genes identified in this study offer the strategies which improve the myocardial substrate during and following a myocardial infarction, such as the regrowth of functional blood vessels to the ischemic myocardium.
Introduction
Revascularization of injured, ischemic and regenerating organs is essential to restore organ function. Neovascularization resulting from the proliferation, migration and remodeling of terminally differentiated endothelial cells (ECs) from preexisting blood vessels is called angiogenesis (Risau, 1997).
Several techniques have been developed for in vitro studies on angiogenesis. The culture of aortic explant in collagen gels has been used widely and effectively for analyzing the biological process of angiogenesis (Mori et al. 1988; Nicosia and Ottinetti, 1990; Akita et al. 1997a; Akita et al. 1997b; Fujita et al. 2002; Nagatoro et al. 2003; Fujita et al. 2004; Suda et al. 2004; Fujita et al. 2006). Our previous paper showed that the capillary tubes formed in the collagen gel had architecture similar to that of capillaries in vivo observed by electron microscopy (Akita et al. 1997a; Akita et al. 1997b). In this collagen gel culture, RT-PCR and immunohistochemical investigations detected the expression of fibroblast growth factor (Fgf) -2 (Nagatoro et al. 2003) and integrin ≈3 (Suda et al. 2004). Capillary-like tubes formed in the collagen gel were strongly positive for endothelial cell-specific tomato lectin (Fujita et al. 2006).
A number of genes are known to be involved in angiogenesis, including vascular endothelial growth factors (Vegfs) and Vegf receptors (Roy et al. 2006), Fgf-2 and matrix metalloproteinases as proangiogenic factors (Schweigerer et al. 1987; Haas and Madri, 1999). In rheumatoid arthritis, platelet-derived growth factor (PDGF), Fgf-2, epidermal growth factor (EGF), insulin-like growth factor (IGF), hepatocyte growth factor (HGF), transforming growth factor (TGF)-β, tumor necrosis factor (TNF)-≈, interleukin-1 (IL-1), IL-6, IL-8, IL-15, IL-18, angiogenin, platelet-activating factor (PAF), angiopoietin, soluble adhesion molecules and endglin play an important role in angiogenesis (Maruotti et al. 2006).
On the other hand, thrombospondins and tissue inhibitor of metalloproteinases are known as antiangiogenic factors (Taraboletii et al. 1990; Moses and Langer, 1991). Despite the numerous reports of angiogenesis, researchers have found it difficult to study the great variety of genes related with angiogenesis. The DNA chip (micro-array) has been used as a powerful tool to study the genetic reprogramming of cells and its link to cellular phenotype such as angiogenesis (Shim et al. 2005). In this study, we examined angiogenesis-related gene expression profiles using collagen gel culture and a DNA chip.
Materials and Methods
Collagen Gel Culture
This collagen culture technique is a modification of our previous work (Nagatoro et al. 2003; Fujita et al. 2004). The thoracic aorta samples were obtained from 1 month old ICR male mice (n = 16). After separating the tunica adventitia from the aorta under a stereoscopic microscope, the blood vessel specimens were cut into pieces of about 2 mm in length. Four pieces were placed at the bottom of each tissue culture dish (35 mm; n = 32), overlaid with an even layer of reconstituted collagen solution (0.3% Cellmatrix type IA, Nitta Gelatin, Tokyo, Japan) and allowed to gel at 37 °C for about 10 min. After the gels had formed, they were overlaid with Ham's F-12 medium (Invitrogen Corp., Carlsbad, CA, USA) containing 20% fetal bovine serum (FBS), 1% non-essential amino acids, 100 units/ml of penicillin and 100 mg/ml of streptomycin (Invitrogen Corp., Carlsbad, CA, USA) and cultured for 5 to 10 days in an incubator (95% air/5% CO2). Capillary-like tube formation was observed with a phase contrast microscope during the culture period.
Gene Array Analysis
Total RNA was extracted by TRIZOL® (Invitrogen Corp., Carlsbad, CA, USA) from each culture plate (day 5 and day 10) without the aortic explant. For micro-array analysis, RNA was purified using an RNeasy® MinEluteTM Cleanup Kit (Qiagen, Hilden, Germany). Gene array analysis was done as a ‘custom order’ by Kurabo Industries (Osaka, Japan) with CodeLinkTM Bioarray Mouse Whole Genome Array (Amersham Biosciences), which displays probe DNAs for about 35,000 mouse genes and expressed sequence tags. Double-stranded cDNA was prepared using RNA from the culture (day 5 and day 10) and was transcribed in vitro with biotin-labeled dUTP as a substrate. The complementary RNA was used as probe for hybridization of the array and the hybridized RNA was detected with indodicarbocyanine-conjugated streptavidin. The arrays were scanned with a Bio-chip Reader (Applied Precision) and the array image was analyzed with CodeLink System software (Amersham Biosciences). DNA micro-array analysis was performed in duplicate for each sample and the average signal intensities of day 5 and day 10 were calculated. The manufacturer's protocol (Amersham Biosciences) recommends that a ratio of at least 2 is 4 standard for the selection of genes showing significant differences in expression.
Results
Phase Contrast Microscopy
At day 5 of the culture, numerous cells migrated from the aortic explant into the collagen gel, but the capillary tubes had not yet formed. The newly formed capillary tubes began appearing after 7 days of culture in the collagen gels. Figure 1 shows phase contrast micrographs before tube formation (day 5) and after tube formation (day 10).
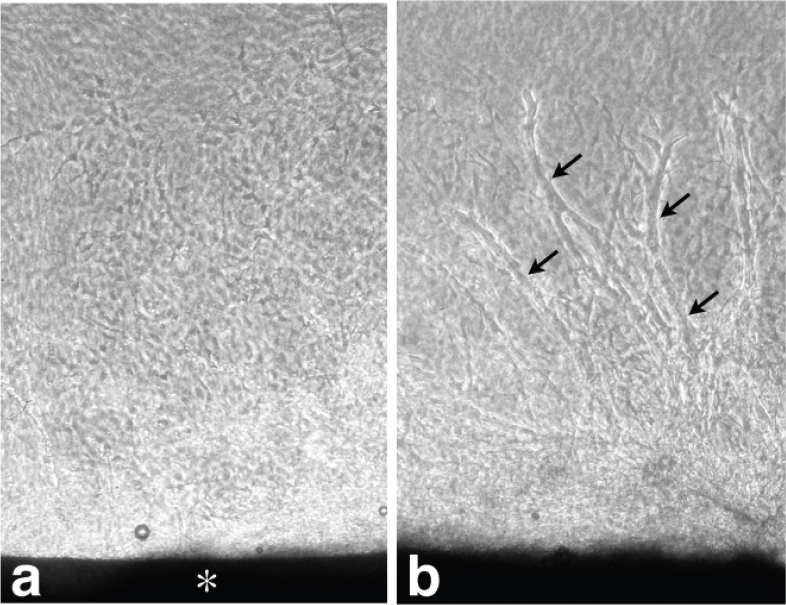
(
Gene Array Analysis
The CodeLink® Mouse Whole Genome array consists of probe sets representing over 35,000 transcripts. The Gene Ontology terms associated with angiogenesis (GO:0001525) and negative regulation of angiogenesis (GO:0016525) were examined. Seventy-three out of over 35,000 transcripts were expressed after tube formation. The expression of 43 transcripts was high, while 30 showed lower expression. Table 1 showed the highly expressed genes. Comparing day 10 with day 5, Table 2 showed a scatter of all the genes of Gene Ontology terms associated with angiogenesis and negative regulation of angiogenesis. The majority of genes examined showed only small differences, with ratios ranging between 2.0 and −2.0. However, there were 7 up-regulated genes (those with ratios of more than 2.0). These up-regulated genes (Tnfaip2, Vegfb, Crhr2, Vegfc, Fgf-6, Pofut1 and Hif1a) showed good levels of expression (sufficient signal intensity in the micro-array system).
Highly expressed genes associated with angiogenesis after tube formation.

Comparative gene expression profiles before and after tube formation.

There were 11 down-regulated genes (those with ratios of less than −2.0). These down-regulated genes were as follows; Rhob, Fgf-2, Thbs1, Ang3, Gna13, Tnfrsf12a, Plg, Elk3, Ubp1, Itgav, Mapk14. The up-regulated and the down-regulated genes are summarized in Table 2.
Discussion
Up-regulated genes
Seven up-regulated genes were identified. These genes play an important role for capillary tube formation and growth.
TNF α-induced protein 2 (Tnfαip2)
Tnfαip2 was originally described as a TNFα-inducible primary response gene in endothelial cells. During vasculogenesis in the mouse embryo, Tnfaip2 was expressed in the myocardium, aortic arch and aortic endothelium (Wolf et al. 1994). In our previous study using collagen gel culture, capillary tube growth was induced by TNF-α in a dose-dependent manner (Fujita et al. 2002). TNF-α play important roles in angiogenesis, including proliferation of endothelial cells and capillary sprouts (Yoshida et al. 1997). Tnfip2 as well as TNF-α could be involved in capillary tube formation and growth.
Vegf-B, Vegf-C and hypoxia inducible factor 1 alpha subunit (Hif-1a)
Vegfs are considered as key molecules in the process of angiogenesis. The Vegf family consists of seven members; Vegf-A, Vegf-B, Vegf-C, Vegf-D, Vegf-E, Vegf-F and placental growth factor (PGF). Vegf-A is a key molecule in induction of angiogenesis and vasculogenesis it causes proliferation, sprouting, migration and tube formation of endothelial cells (Ferrara et al. 2003). Hao et al. 2007 reported that gene transfer of plasmid encoding Vegf-A was applicable for therapeutic angiogenesis in a rat myocardial infarction model.

In the present study, Vegf-A, -B, -C and PGF were highly expressed, especially Vegf-B and -C were up-regulated. The precise role of Vegf-B is not known. Vegf-B has been reported to be weakly angiogenic after adenoviral delivery to periadventitial tissue (Bhardwaj et al. 2003). Reduced synovial angiogenesis in Vegf-B knockout arthritis models suggest a role of Vegf-B in inflammatory angiogenesis (Mould et al. 2003). Vegf-C induces mitogenesis, migration and survival of endothelial cells. Developmental studies, knockout models and gene transfer experiments suggests that Vegf-C is primarily a lymphangiogenic growth factor and its lymphangiogenic effects are mediated by VegfR-3 (Enholm et al. 2001; Karkkainen et al. 2004). Vegf-A mediates its responses primarily by activating VegfR-1 (Flt-1) and VegfR-2 and it binds to neuropilin (Nrp) -1 and Nrp-2 (Klagsbrun et al. 2002). Vegf-A mRNA expression is induced by hypoxia, which induces binding of the hypoxia-inducible factor-1a (Hif-1a) to the hypoxia-responsive element in the Vegf-A gene promoter region, which in turn increases Vegf-A transcription (Roy et al. 2006). In the present study, VegfR-1, -2, Nrp-1 and Hif-1a were highly expressed. In this culture, Vegfs and its receptors play an important role for the formation and growth of capillary tubes.
Fibroblast growth factor-6 (Fgf-6)
Fgfs are a family of heparin-binding proteins involved in many biological processes including angiogenesis (Cancilla et al. 2001). Pizette et al. 1991 noted that Fgf-6 displays a strong mitogenic activity on fibroblastic cells and is able to morphologically transform these cells. Fgf-6 showed only a limited mitogenic response on adult bovine aortic endothelial cells. It is suggest that Fgf-6 relate mainly to proliferation of fibroblasts than that of endothelial cells.
Corticotropin-releasing hormone receptor 2 (Crhr2)
According to Kokkotou et al. 2006, Crhr2-deficient mice display cardiovascular abnormalities. The significance of Crhr2 up-regulation is obscure.
Protein O-fucosyltransferase 1 (Pofut1)
Pofut1 has been shown to be essential in Notch signaling, which has an important role in vascular development, differentiation, proliferation, apop-tosis and tumorigenesis (Luo et al. 2006; Shi and Harris, 2006; Rehman and Wang, 2006). Delta-like 4 (Dll4), a Notch signaling molecule, was also expressed in this study. Dll4, a membrane-bound ligand for Notch1 and Notch4, is expressed selectively in the developing endothelium and it is induced by Vegf-A and hypoxia (Williams et al. 2006). Up-regulation of Pofut1 may relate to the up-regulation of Hif-1a.
Down-regulated genes
Eleven down-regulated genes were identified in the present study. These genes were more strongly expressed before tube formation. They act at the early stage of angiogenesis.
The ras homolog gene family, member B (RhoB)
Rho GTPases have been shown to have an important role in cancer development and angiogenesis. Adini et al. 2003 demonstrated a novel and critical role for RhoB in vascular development. RhoB null mice have retarded vascular development in the retina characterized by altered sprout morphology. Moreover, pharmaceutical means to deplete RhoB in neonatal rats is associated with apoptosis in the sprouting endothelial cells of newly forming vessels. Similarly, acute depletion of RhoB by antisense or dominant-negative strategies in primary endothelial cell culture models led to apoptosis and failures in tube formation. As Adini et al. 2003 noted, the function of RhoB appears to only be rate limiting for endo-thelial cell sprouting.
Fibroblast growth factor-2 (Fgf-2)
Fgf-2 is one of the most potent angiogenic factors, primarily targeting vascular endothelial cells as a mitogenic and survival factor (Klint and Claesson-Welsh, 1999). Intramyocardial administration of Fgf-2 microspheres can promote the growth of microvessels and improve left ventricular function and myocardial viability in the early period of acute myocardial infarction (Liu et al. 2006). However, Fgf-2 was down-regulated in this study. In our previous study, Fgf-2 expression was observed predominantly before tube formation, when fibroblastic cells were migrating actively from the aortic explant, but the expression grew indistinct with time (Nagatoro et al. 2003). Take account the data including our previous study, Fgf-2 relate mainly to proliferation of fibro-blasts and endothelial cells than tube formation and/or growth.
Thrombospondin 1 (Thbs-1)
Thbs-1 is a potent inhibitor of angiogenesis and activator of transforming growth factor (TGF-β). Frangogiannis et al. 2005 hypothesized that Thbs-1 is induced in healing myocardial infarcts and plays a role in suppressing the postinfarction inflammatory response, inhibiting local angiogen-esis and limiting expansion of granulation tissue into the noninfarcted area. Thbs-1 mRNA was markedly induced by Fgf-2 (Frangogiannis et al. 2005). Thbs-1 binds the vβ3, 3β1 and 5β1 integrins (Bornstein et al. 2001). In the present study, Fgf-2 and integrin av were also down-regulated.
Angiogenin, ribonuclease a family, member 3 (Ang3)
Angiogenins are proteins in the pancreatic ribonuclease superfamily that utilize their ribo-nuclease activity to induce formation of new blood vessels. Fu et al. 1999 identified a new member of the angiogenin gene family, mouse angiogenin-3. According to Holloway et al. 2001, mice possess genes encoding one ortholog and three homologs of Angiogenin, designated angiogenin-related protein (mAngrp), angiogenin-3 (mAng-3) and angiogenin-4 (mAng-4). Structural and functional differences of these homologs have not been well-known. Angiogenin is required for endothelial cell proliferation induced by various other angiogenic proteins including Fgf-1 and -2, EGF and Vegf. Down-regulation of angiogenin in endothelial cells by small interfering RNA (siRNA) and antisense results in a decrease in rRNA transcription, ribosome biogenesis and cell proliferation induced by these angiogenic factors. Inhibitors of the nuclear translocation of angiogenin abolish the angiogenic activities of these factors. Stable angio-genin antisense transfection in HeLa cells reduces tumor angiogenesis in athymic mice despite the elevated expression level of bFgf and Vegf. Thus, nuclear angiogenin assumes an essential role in endothelial cell proliferation and is necessary for angiogenesis induced by other angiogenic factors. Angiogenin-stimulated rRNA transcription in endothelial cells may thus serve as a crossroad in the process of angiogenesis induced by various angiogenic factors (Kishimoto et al. 2005).
Plasminogen (Plg)
Plg, by virtue of its role in the degradation of extracellular matrix proteins and by facilitation of cell migration, may contribute to angiogenesis. Angiogenesis was assessed in gene-targeted mice with deficiencies of plasminogen in a mouse corneal model. A significantly decreased angiogenic response of new vessel formation to both Vegf and Fgf-2 was observed in Plg deficient (Plg-/-) mice (Oh et al. 2003).
Integrin alpha v (Itgav)
The αvβ3 integrin heterodimer has been thought to play an important role in angiogenesis (Eliceiri and Cheresh, 1999). This notion has been called into question, however, as several groups have shown that mice lacking either the αvβ3 integrin or both the αvβ3 and αvβ5 integrins develop normally (Bader et al. 1998; Reynolds et al. 2002). Moreover, mice lacking both the αvβ3 and αvβ5 integrins develop more extensive tumors with richer vascular supplies than their litter-mate controls (Reynolds et al. 2002).
Mapk14 (formerly p38)
According to Yoshino et al. 2006, inhibitors of p38 Mapk and/or JNK suppressed Vegf secretion significantly. Fgf-1/p38 Map kinase inhibitor therapy induces cardiomyocyte mitosis, reduces scarring and rescues function after myocardial infarction. In contrast, p38 Map kinase inhibition alone fails to rescue heart function despite increased cardiomyocyte mitosis. This therapy combines the effects of p38 Map kinase inhibition on apoptosis and proliferation with the effects of Fgf-1 on apoptosis, proliferation and angiogenesis to enhance cardiac regeneration (Engel et al. 2006). It is suggested that down-regulation of p38 Map kinase has no direct effect on angiogenesis.
Tnfrsf12, Elk3, Gna13 and Ubp1
The precise angiogenic or anti-angiogenic role of Tnfrsf12a, Elk3, Gna13 and Ubp1 is unknown.
We need more direct analyses using RT-PCR and /or immunohistochemical study on the protein level for the up- and down-regulated genes identified in this study. The DNA chip (micro-array) technique could be used as a powerful tool to study the evaluation of gene expression profiles of angiogenesis. Take account our data, the following conclusion can be proposed. The up-regulated genes after tube formation act for formation and growth of capillary tube and they also relate to maturation and act as a stabilizer of the newly formed capillary tube. On the other hand, the down-regulated genes relate cell migration and sprouting at the early stage of angiogen-esis. The up- and down-regulated genes identified in this study offer the possibility of therapeutic angiogenesis for the ischemic disease such as myocardial infarction. Establishment of functional and stable collaterals in the ischemic myocardium is crucial to restoring cardiac function after myocardial infarction. Lu et al. 2007 shown that dual delivery of a combination of Fgf-2 with PDGF-BB to the ischemic myocardium could significantly reestablish stable collateral networks and improve myocardial perfusion and function. This study suggests that single angiogenic factor is unable to accomplish the complex process of angiogenesis. A combination of the up- and down-regulated genes identified in this study promotes myocardial collateral growth and stabilizes the newly formed collateral networks. Otherwise, after applying a combination of the down-regulated genes, then we can follow a combination of the up-regulated genes. Our findings provide conceptual guidelines for the clinical development of angiogenic factors for the treatment of ischemic heart disease.
Footnotes
Acknowledgment
This work was supported in part by a Grant-in-Aid for Scientific Research (C) (No. 16591796 and No. 18591958) from the Japan Society for the Promotion of Science and by a grant from The Shimabara Science Promotion Foundation.
