Abstract
The current study assessed the acute effects of pacemaker optimization (PMO) on cardiac function using echocardiographic (ECHO) tissue Doppler imaging (TDI) in the post CRT setting.
Data were analyzed from 50 consecutive patients clinically referred for PMO. Patients underwent a sequential ECHO/TDI-guided PMO study to determine optimal pacemaker settings.
In 34 of 50 patients a change in pacemaker settings was made because of an objective improvement in ECHO/TDI findings. Overall, significant improvements were observed for ECHO/TDI measures of systolic function (global systolic contraction score, p < 0.001; ejection time, p < 0.05), diastolic function (diastolic filling period, p < 0.01; mitral velocity-time integral, p < 0.05) and left ventricular (LV) dyssynchrony (standard deviation of time to peak displacement, p < 0.05). In most patients referred for chronic PMO, ECHO/TDI-guided PMO can be used to objectively improve cardiac systolic function, diastolic function and/or LV dyssynchrony.
Introduction
Cardiac resynchronization therapy (CRT) is a safe and effective device therapy for patients with systolic heart failure (HF). This treatment has been shown to improve symptoms, increase exercise capacity, beneficially affect left ventricular (LV) remodeling and reduce hospitalization and mortality in patients with HF (Abraham et al. 2002; Aurricchio et al. 2002; Bristow et al. 2004; Cazeau et al. 2001; Cleland et al. 2005; Gras et al. 1998; Gras et al. 2002). However, approximately 25%-35% of patients receiving this therapy do not significantly respond. Potential reasons for non-response to CRT include lack of mechanical dyssynchrony at baseline and/or failure to resynchronize with CRT because of suboptimal lead placement, extensive disease, or suboptimal pacemaker settings. There are some studies investigating the acute effects of optimizing atrio-ventricular (A-V) delay (Kerlan et al. 2006; Sawhney et al. 2004; Aurricchio et al. 1999) or left ventricle to right ventricular (V-V) timing (Sogaard et al. 2002) in CRT patients and other studies assessing the chronic effects of A-V delay (Porciani et al. 2006) and V-V timing (Naqvi et al. 2006). However, there are no studies to our knowledge evaluating a comprehensive approach (i.e. adjusting lower rate limit, A-V delay, V-V timing together in one protocol) to pacemaker optimization (PMO) as a therapy designed to address chronic sub-optimal response to CRT.
The present study is a retrospective analysis of the acute echocardiographic effects of applying a PMO protocol to 50 consecutive patients who were deemed by their cardiologist to be chronic sub-optimal responders to CRT. This approach may be one of many potential strategies for improving the response rate to CRT therapy.
Methods
Patient Population
Fifty consecutive patients who were clinically referred for PMO over a two-year period were studied. Patients were referred by their cardiologist for echocardiogram (ECHO), tissue Doppler imaging (TDI)-guided PMO because the physician felt that the clinical response to CRT was either suboptimal or was absent. All patients met standard indications for CRT. Patients had A-V delays set at the time of their pacemaker implantation either based on the implanting physician's judgment or per protocol if the patient was enrolled in a CRT study. All patients had LV leads placed via the coronary sinus (i.e. no epicardial leads) with all attempts to target the lateral and/or posterolateral section of the LV All LV leads were capturing appropriately and all patients in the study had greater than 90% bi-ventricular pacing as determined by device interrogation. All patients were at least two months out from their device placement. The protocol was reviewed and approved by the local institutional review board for the analysis of existing clinical data.
Study Protocol
Patients initially underwent an ECHO/TDI study with their current pacemaker settings (including any V-V timing offsets). The following settings were then adjusted and the acute hemodynamic effects were measured by ECHO/TDI: lower rate limit, A-V delay, ventricular lead configuration [right ventricle (RV) only, LV only, RV and LV], and V-V timing. A flow diagram indicating the protocol used during the PMO procedure is shown in Figure 1. These pacemaker settings were changed in a sequential and additive manner in the order listed. ECHO/TDI images were collected approximately 1 minute after a given device setting change was made. Once a pacemaker change was found to result in improved heart function, the pacemaker was left at that new setting and an additional parameter was studied. For example, once the optimal lower rate was determined, A-V delays were studied at the new optimal lower rate. In this manner, patients may have ended up with zero (non-optimized patients), one, or several parameters changed to obtain the final (so-called optimal) pacemaker settings. Patients presented with different clinical scenarios (atrial sensed vs. atrial paced, for example) and multiple different devices with various programming capabilities (V-V timing options present or absent).
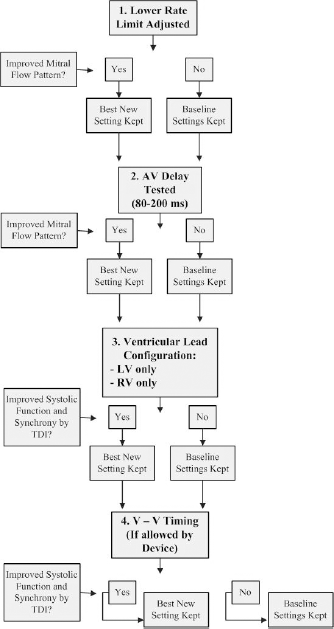
Flow-diagram depicting the steps taken in each pacemaker optimization study.
Lower rate limit and A-V delay were optimized based on mitral filling velocities in order to obtain the best separation of mitral E and A waves without truncation of the A wave. Ventricular lead configuration and ventricular lead activation sequence (V-V timing) were optimized based on tissue-tracking displacement curve morphology and timing (dyssynchrony measures) and tissue-tracking displacement amplitude at end-systole: global systolic contraction score (GSCS) (Sogaard et al. 2002). In patients whose devices allowed for changes in V-V timing, the general protocol consisted of pre-activating the RV wall in incremental steps of 10 msecs each followed by pre-activation of the LV wall in the same stepwise fashion. In general, cardiovascular medications were stable before and after PMO.
Standard Echocardiography
ECHO studies were obtained using standard American Society of Echocardiography criteria on the Vivid 7 ultrasound machine (General Electric Co, Milwaukee, WI) and analyzed on the GE ECHOPAC reading station. Ejection Fraction (EF) was measured by blinded tracing of end-diastolic and end-systolic volumes on apical 4- and 2-chamber views and was calculated using the modified Simpson's method. Mitral regurgitation was assessed using a semi-quantitative scale of 0 to 6 (i.e. 0 = none, 1 = trace, 2 = mild, 3 = mild-moderate, 4 = moderate, 5 = moderate-severe, 6 = severe). Complete ECHO studies were performed at baseline. After changing pacemaker settings, only apical 2-D images, mitral inflow and mitral regurgitation were serially assessed.
Tissue Doppler
Tissue-tracking mode was used for all TDI analyses except for the measure of early diastolic relaxation velocity (E'), which was obtained from the tissue velocity curves. Lateral E/E', a measure of diastolic function, was defined as the ratio of peak early left ventricular filling velocity (E wave; via standard pulsed-Doppler) to TDI-derived peak early diastolic velocity of the lateral mitral annulus (E' wave). With tissue tracking the longitudinal magnitude of movement and timing of movement can be analyzed for any regional myocardial segments (Bank and Kelly, 2006). Apical 4-chamber, 2-chamber, and long axis TDI data were acquired. Tissue-tracking displacement curves were measured at six basal (anteroseptal, anterior, lateral, posterior, inferior and septal) and six mid-ventricular segments. The starting point for all tissue-tracking curves (the beginning of each heartbeat) was standardized and defined as the time of mitral valve closure. Timing events (i.e. mitral valve closure, aortic valve closure, etc.) were obtained by identifying these events using standard pulsed Doppler echocardiography of transmitral and transaortic blood flow patterns. The events were marked on the pulsed Doppler images and then automatically transferred to TDI images by the ultrasound machine. GSCS was defined as the average longitudinal displacement at end-systole (aortic valve closure) of the 12 segments. This measure has been shown by Sogaard et al. (2002) to be a sensitive marker of systolic LV function that correlates well with EF. Delayed longitudinal contraction (DLC) was defined as peak longitudinal systolic displacement that occurred more than 30 ms after aortic valve closure. Delayed onset of activation (DOA) was defined as an initial negative longitudinal tissue-tracking displacement greater than 0.5 mm. Standard deviation of time to peak displacement (SD-TTPD) was also used as a measure of dyssynchrony.
Reproducibility of TDI measurements
Twenty-four HF patients were studied to assess reproducibility of TDI measurements. Intra-observer variability demonstrated a mean difference of 0.06 ± 0.36 mm with a coefficient of variation (CV) of 5.4% for GSCS and a mean difference of 0.4 ± 9.3 ms with a CV of 12.1% for SD-TTPD. Inter-observer variability demonstrated a mean difference of −0.21 ± 0.25 mm with a CV of 7.2% for GSCS and a mean difference of −3.3 ± 13.0 ms with a CV of 11.8% for SD-TTPD. Short-term (5 minutes apart) reproducibility demonstrated a mean difference of −0.17 ± 0.71 mm with a CV of 9.3% for GSCS and a mean difference of 2.6 ± 16.7 ms with a CV of 20.0% for SD-TTPD. Medium-term (2—4 weeks apart) reproducibility demonstrated a mean difference of 0.004 ± 0.8 mm with a CV of12.1% for GSCS and a mean difference of −1.5 ± 22.2 ms with a CV of 27.1% for SD-TTPD. Bland-Altman analyses (Bland and Altman, 1986) showed no effect of the magnitude of the measure on the variability of the data.
Statistical Analysis
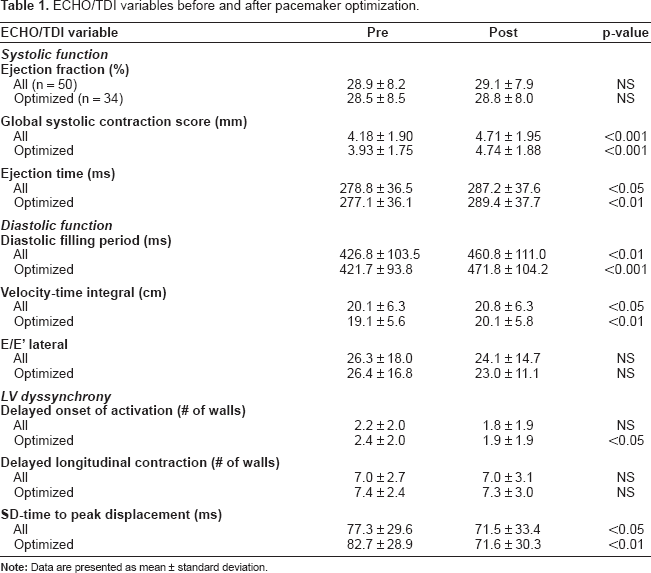
Statistical analysis and graphing was performed using Microsoft Excel. The main analysis focused on the acute change in ECHO/TDI with PMO. Selected ECHO/TDI variables measured before and after PMO were analyzed using paired t-tests. These a priori selected ECHO/TDI variables included measures of systolic function: EF, GSCS and ejection time (EJT); diastolic function: diastolic filling period (DFP), E/E' lateral and mitral flow velocity time integral (VTI); and dyssynchrony: number of segments demonstrating DOA, number of segments demonstrating DLC and the SD-TTPD. Statistical analysis of ECHO/TDI findings before and after PMO was performed on all 50 patients and separately on the subset of 34 patients who had pacemaker settings altered. Because of the small number of patients in this study, adjustments were not made for multiple comparisons. Statistical significance was defined as p < 0.05. Data are presented as mean ± standard deviation.
Results
In the overall group, the baseline demographics were as follows: mean age = 69.6 ± 11.1 yrs, gender = 72% male, HF etiology = 68% ischemic and 32% non-ischemic, mean duration of HF = 6.0 ± 3.9 yrs. Patients were on the following HF medications: beta-blockers (76%), ACE-inhibitors/ARBs, (76%), diuretics (74%) and aldosterone receptor antagonists (28%). RV lead location was septal (80%) and apical (20%). LV lead location was anterior (8%), posterior (2%), posterior-lateral (20%) and lateral (70%). Three patients were in atrial fibrillation at the time of PMO. Twenty four percent of the patients in this study had lower rate limits set at >60 beats per minute and were pacing the atria over 50% of the time. Prior to CRT implantation, mean visually estimated EF for all patients was 23.3 ± 7.3%. At presentation for PMO, mean traced EF in all patients was 28.9 ± 8.2%. The average time period between CRT placement and PMO was 1.1 ± 0.9 years.
In 16 of the 50 (32%) patients referred for PMO, there were no pacemaker setting changes that resulted in improvement in systolic function, dia-stolic function, or dyssynchrony that was apparent at the time of optimization (non-optimized patients). In these patients, the pacemaker was left at baseline settings. In 34 of the patients (68%), one or more of the new settings studied resulted in an improvement in systolic function, diastolic function and/or dyssynchrony (optimized patients). Choices for potential changes in pacemaker settings varied greatly among the patients depending upon their device, current settings (e.g. atrial paced or not), and underlying pathology (e.g. complete heart block or not). Lower rate limit was decreased in 11 patients and A-V delay was changed in 20 patients. The patients who had their lower rate limits reduced to ≤60 beats per minute demonstrated a significant increase in DFP of 128 msecs (p < 0.0001), and a significant increase in GSCS of 1.2 mm (p < 0.0001). Fourteen patients had A-V delay lengthened by an average of 38 msecs and 6 patients had A-V delay shortened by an average of 27 msecs. V-V timing changes were allowable in only 13 patients (based on programming capabilities of the specific devices) and were modified in six patients. Three patients had V-V timing offsets at baseline (all 3 with LV pre-activation). The RV lead was turned off in nine patients who were all paced in the mid-septum. In one patient, CRT was discontinued by turning off both ventricular leads. In 21 patients only one setting was changed, in 12 patients two settings were changed and in one patient three settings were changed.
In one patient, there were no changes in pacemaker settings that resulted in improved ECHO/ TDI parameters except turning off biventricular pacing. In this patient five of the six basal segments demonstrated DLC at baseline and GSCS was very low at 2.1 mm. After turning off CRT only two of the six basal segments had DLC and GSCS improved to 3.0 mm.
Table 1 shows ECHO/TDI parameters before and after PMO for all 50 patients and in the subset of 34 patients whose pacemaker settings were changed (optimized patients). For the overall group and in the subset of patients whose settings were changed, there were statistically significant improvements in measures of systolic function (GSCS, EJT), diastolic function (DFP, VTI), and dyssynchrony (SD-TTPD). In the optimized group, significant reductions were also noted for number of segments with DOA. There were no significant changes in mitral regurgitation as a result of PMO. Figure 2 displays the percent improvements for ECHO/TDI parameters in the patients where pacemaker modifications were made.
ECHO/TDI variables before and after pacemaker optimization.

Summary of ECHO/TDI improvements in the subset of patients whose pacemaker settings were optimized. *denotes p < 0.05; ***denotes p < 0.01; ***p < 0.001.
Discussion
The main finding of this study is that PMO in the chronic post-CRT setting is a practical and effective therapy for acutely improving ECHO/TDI measures. Approximately two-thirds of patients referred for this treatment due to unsatisfactory response to CRT have an acute ECHO/TDI improvement in systolic function, diastolic function, dyssynchrony or a combination of these measures.
Although CRT is a successful and potentially life-saving procedure for many patients, an unacceptably large percentage of patients receive little or no demonstrable benefit. There are a number of potential reasons for non-response to CRT. The major mechanism of action of CRT is likely improvement in left ventricular wall contraction synchrony. Therefore, patients without LV mechanical dyssynchrony at baseline or without significant improvement in LV mechanical dyssynchrony after CRT are more likely to be non-responders. One reason for lack of clinical improvement post-CRT is sub-optimal pacemaker programming. Settings of lower rate limit, A-V delay, ventricular lead pacing configuration and V-V timing can be programmed variably (depending on the pacemaker model) to alter systolic function, diastolic function and/or dyssynchrony.
Lower Rate Limit
The appropriate setting for lower rate limit in patients with biventricular pacemakers is not known. The vast majority of the medium-sized and large studies of CRT set the lower rate limit at ≤ 60 beats per minute. This allowed most patients to sense rather than pace the atria. Patients with bradycardia indication for pacing were typically excluded from enrollment in CRT studies. In clinical practice, many CRT patients are programmed to a lower rate limit of 70 or 80 beats per minute in an attempt to pace the atria and presumably increase cardiac output. Figure 3 shows mitral inflow Doppler velocities in a patient prior to CRT and at presentation for PMO. Decreasing the patient's lower rate limit from 80 to 60 beats per minute resulted in significant increases in E and A wave separation and in DFP. Adjusting V-V timing further improved diastolic filling. Approximately one-fourth of the patients in this study had lower rate limits set at >60 beats per minute and were pacing the atria over 50% of the time. One of these patients was left at a lower rate limit of 80 beats per minute because of significant ventricular ectopy including ventricular tachycardia that occurred more frequently at slower heart rates. The patients who had their lower rate limits reduced to ≤60 beats per minute demonstrated significant improvements in DFP and GSCS. The longer DFP likely allowed for increased LV preload and hence a more forceful LV contraction with increased stroke volume.

Mitral inflow doppler velocities in a 64 year old patient under 4 conditions. During A-V sequential pacing for complete heart block (panel
Atrio-ventricular delay
Setting the appropriate A-V delay is important in order to obtain the maximum benefit from CRT (Auricchio et al. 1999). A number of methods have been used to optimize A-V delay (Auricchio et al. 1999; Ishikawa et al. 2002; Kindermann et al. 1997). We chose to use a fast and effective iterative ECHO method (Kindermann et al. 1997) that has been used in multi-center trials (MUSTIC) to set A-V delay acutely after CRT implantation. In the current study, 14 patients had A-V delay lengthened and 6 patients had A-V delay shortened. Most studies of A-V delay optimization have been performed shortly after device implantation in all patients. The use of ECHO/ TDI to chronically adjust A-V delay in a subset of patients who did not have a satisfactory clinical response to CRT has not previously been studied.
Ventricular Pacing Lead Configuration
In nine patients, turning off the RV lead and pacing the ventricle only from the LV lead produced a significant decrease in dyssynchrony and a significant increase in GSCS. In the majority of these patients there was initial negative displacement and significant DOA of the basal (and usually mid) segments of the septal, inferior and/or posterior walls of the left ventricle. Figure 4 shows TDI images in a patient before and after discontinuation of the RV pacing lead. LV only pacing markedly improved the DOA and the GSCS in this patient. The initial abnormality may have resulted from retrograde activation of the septal, inferior, and posterior walls of the LV such that the basal portions of these walls started movement late and demonstrated peak apical motion after aortic valve closure and well into diastole. Turning off the RV lead and pacing the ventricle only from the LV lead in these patients may have improved dyssynchrony and thus systolic function by avoiding retrograde septal activation. There is currently significant debate regarding the advantages and disadvantages of biventricular vs. LV-only pacing in patients with systolic HF and dyssynchrony (Auricchio et al. 2003; Etienne et al. 1999). Our study suggests that in some patients receiving biventricular pacing, the RV lead can promote dyssynchrony and adversely affect systolic function.

Tissue tracking curves (apical 4-chamber view) from a patient during bi-ventricular pacing (panel
V-V timing
Only 26% of the patients referred for this study had pacemakers capable of V-V timing adjustments. In six of these 13 patients we were able to improve systolic function and/or dyssynchrony by adjusting ventricular activation timing. Although optimizing V-V timing in unselected patients shortly after CRT has proven effective (Sogaard et al. 2002), there are few studies assessing this strategy in clinical non-responders. Naqvi et al. (2006) studied eight clinical non-responder CRT patients and found that individualized tailoring of A-V and V-V timing resulted in improved cardiac output via variable improvements in intra and inter-ventricular synchrony, strain, and/or mitral regurgitation reduction. It is difficult to make conclusions on the benefit of this approach in clinical non-responders due to the small numbers in our study.
Advantages of tissue tracking methodology for PMO
We used tissue tracking to perform PMO in this study. This methodology offers several advantages. Detailed calculations, which are needed with some methods for measuring dyssynchrony, are not required. Dyssynchrony can be determined on-line quickly and easily by looking for walls that demonstrate DOA and/or DLC, making it a feasible approach for this type of clinical protocol. Adjustments in ventricular lead pacing configuration or timing can then be made based on which walls are dyssynchronous and dyssynchrony then can be reassessed. In addition, tissue tracking allows for fast, sensitive and reproducible measurement of longitudinal systolic function that can also be assessed on-line under different pacemaker settings. We did not find any significant changes in EF following PMO despite improvement in our measure of longitudinal systolic function, GSCS. This is not surprising since measurement of EF by tracing endocardial borders is more variable than GSCS and less sensitive at picking up modest changes in systolic function.
Limitations
This study was retrospective, non-randomized and assessed only the acute effects of changes in pacemaker settings. Therefore, this report serves mainly as a proof of concept trial and future longer-term randomized studies need to be performed to assess this and other chronic PMO strategies. However, despite not having long-term clinical follow-up data after the PMO procedure, the results of this study are promising in that we showed significant improvements in multiple aspects of cardiac function. Although speculative, these types of acute changes could aid in promoting reverse remodeling (via improved synchrony) which is important since it has previously been shown that reverse remodeling is a better predictor of long-term survival after CRT than clinical improvement (Yu et al. 2005). Further chronic follow-up studies are needed to clarify the effect of PMO on ECHO and clinical status. The classification of responders and non-responders to initial CRT was subjective and was based on the clinical judgment of individual patient's cardiologist; we were unable to include objective measures such as LV dimensions, NYHA class, or other clinical surrogates in this retrospective study. All combinations of reasonable or feasible pacemaker settings were not assessed. It is possible that changing one pacemaker setting, such as ventricular lead configuration, could alter the previously measured and fixed best setting for a different parameter, such as A-V delay. We designed our optimization protocol to be practical for the patients and feasible for the providers as part of routine clinical care. Therefore, we designed our optimization protocol to allow for completion in approximately one hour once the initial ECHO was obtained. The protocol requires the presence of an echocardiographer, a device technician, and a physician together during the study so decisions can be made, analyzed and acted upon immediately. We studied all patients referred for optimization regardless of their device type, underlying rhythm (including atrial fibrillation), symptoms, or their ECHO response to CRT. Therefore the current results should be generalizable since the patients are representative of typical patients seen in a cardiology clinic with clinical incomplete or non-response to CRT.
Conclusions
This report demonstrates that a sequential ECHO/ TDI-guided pacemaker optimization protocol results in significant acute improvements in systolic function, diastolic function and/or dyssynchrony in a majority (2/3) of patients who were deemed by their cardiologists to be chronic CRT non- or suboptimal-responders. This may prove to be a practical and efficient strategy to improve cardiac function in this group of patients. However, future randomized, controlled trials will be needed to assess whether this approach leads to improvements in clinical response.
Disclosures
Drs. Bank and Adler receive research grant support and honoraria from Medtronic, Inc. and Boston Scientific. Dr. Bank receives honoraria from General Electric (ultrasound division). Drs. Kelly and Kaufman receive research grant support from Medtronic, Inc. and Boston Scientific. Kevin Burns and Andrea Thelen have no disclosures.
Glossary of Abbreviations
Atrio-Ventricular;
Cardiac Resynchronization Therapy;
Diastolic Filling Period;
Delayed Longitudinal Contraction;
Delayed Onset of Activation;
Echocardiogram;
Ejection Fraction;
Ejection Time;
Global Systolic Contraction Score;
Heart Failure;
Left Ventricle;
Pacemaker Optimization;
Right Ventricle;
Standard Deviation of Time to Peak Displacement;
Tissue Doppler Imaging;
Ventricular to Ventricular.
Funding Support
None.
Footnotes
Acknowledgments
The authors would like to thank Thomas Rector, Ph.D. for statistical assistance and Michael Stuckey, Ph.D. and Allison Carland for assistance with chart review/data retrieval.
