Abstract
Infection is one of the most devastating outcomes of cardiovascular implantable electronic device (CIED) implantation and is related to significant morbidity and mortality. In our country, there is no evaluation about CIED infection. Therefore, our aim was to investigate clinical characteristics and outcome of patients who had infection related to CIED implantation or replacement. The study included 144 consecutive patients with CIED infection treated at 11 major hospitals in Turkey from 2005 to 2014 retrospectively. We analyzed the medical files of all patients hospitalized with the diagnosis of CIED infection. Inclusion criteria were definite infection related to CIED implantation, replacement, or revision. Generator pocket infection, with or without bacteremia, was the most common clinical presentation, followed by CIED-related endocarditis. Coagulase-negative staphylococci and Staphylococcus aureus were the leading causative agents of CIED infection. Multivariate analysis showed that infective endocarditis and ejection fraction were the strongest predictors of in-hospital mortality.
Keywords
Introduction
The use of cardiovascular implantable electronic devices (CIEDs) has increased worldwide and now includes implantable cardiac defibrillators (ICDs) and cardiac resynchronization therapy devices in addition to permanent pacemakers (PPMs). Infection is one of the most devastating outcomes of CIED implantation and is related to significant morbidity and mortality. 1 –4 It can manifest as infection of the generator pocket, the leads, and can also involve endocardial structures; CIED infections now comprise approximately 10% of all endocarditis cases. 5 Recently, the indications for treatment with CIEDs have expanded so that patients with more various cardiovascular diseases receive these devices. Studies have suggested an increasing rate of infection over the past 2 decades which could not be explained from the increasing rate of device implantation. 6,7 The causes of this are unknown, but one explantation likely is ascending comorbidities of CIED recipients. 8
In our country, there is no evaluation about CIED infection. The procedural complications are unknown. Therefore, our aim was to investigate clinical characteristics and outcome of patients who had infection related to CIED implantation or replacement. Various centers from all over the country were invited to collaborate in order to identify contributing clinical factors, infection prevention practices, and practice site differences associated with CIED infections and its complications.
Materials and Methods
Study Design
The study was designed as a retrospective cross-sectional multicenter registry.
Study Population
The study population consisted of 144 consecutive patients with CIED infection treated at 11 major hospitals in Turkey from 2005 to 2014, respectively. All hospitals are tertiary referral centers (Dicle University, Mevlana University, Harran University, Necmettin Erbakan University, Siyami Ersek Education and Research Hospital, Abant Izzet Baysal University, Osman Gazi University, Selçuk University, Adana Education and Research Hospital, and Duzce University), which receive patients from surrounding hospitals. These hospitals were located in different cities throughout Turkey. We analyzed the medical files of all patients hospitalized with the diagnosis of CIED infection. Inclusion criteria were definite infection related to CIED implantation, replacement, or revision. All device implantations that occurred within the time frame of the investigation were included from all the institutions. Also, all variables were recorded to standard data sheet at all institutions.
Definition
The CIED infection was defined as previously described. 3,9 –11 Clinical evidence of CIED infection included local signs of inflammation at the generator pocket (eg, erythema, warmth, fluctuance, wound dehiscence, tenderness, purulent drainage, or erosion of generator or lead through skin). The CIED-related endocarditis was clinically confirmed when valvular or lead vegetations were detected by echocardiography or if the Duke criteria for infective endocarditis (IE) were met. 12 The CIED infection was microbiologically confirmed on the basis of positive culture results using samples obtained from the generator pocket, leads, or blood (in the presence of local inflammatory signs at generator pocket or absence of another source of bacteremia and resolution of bloodstream infection after device explantation). The study was approved by the local Medical Ethics Committee.
Data Collection
Data on demographic characteristics, age, sex, underlying heart disease, presenting signs and symptoms, diabetes mellitus and other comorbidities, results of laboratory and microbiological investigations, echocardiographic findings, treatment given during hospitalization, surgical requirements, and cardiac and extracardiac complications were collected. The patients were also analyzed for factors associated with recurrent episodes of IE. The antibiotic regimen, aspects related to the surgical approach, and in-hospital outcome were also recorded. Complete blood count, C-reactive protein, erythrocyte sedimentation rates, serum chemistry, and urine analysis comprised the routine laboratory investigations that were recorded.
Statistical Analysis
Statistical Package for Social Sciences software (SPSS 12, Chicago, Illinois) was used for analysis. Descriptive statistics are presented as mean ± standard deviation or percentages. Logistic regression analysis was performed in order to provide independent predictors of mortality in patients with CIED infections. Covariates were age, sex, ejection fraction, diabetes mellitus, Staphylococcus aureus infection, and IE.
Results
Baseline characteristics of the patients are shown in Table 1. The mean age of patients was 63 ± 17 years (range, 13-95); 87 of the patients were male. In a total of 9207 device implantation, CIED infection was detected in 144 (1.6%) of them. The most common implanted device type was dual chamber pacemaker (34%) followed by single chamber pacemaker (30%). Table 2 shows CIED infection proportion according to different types of procedure and device. The time of implant to infection for the first procedure was 1.18 ± 2.21 years and the time of implant to infection for reimplantation was 1.16 ± 1.25 years, respectively. Antibiotic prophylaxis rate was 45%. Intravenous antibiotic prophylaxis with cefazolin was used prior to CIED implantation and continued 3 times a day during in-hospital period in 95% of the total cohort. Oral amoxicillin-clavulanic acid was given twice a day for 10 days after discharge. The most common symptom at presentation was purulent discharge (36%). Heart block (51%) and primary and secondary prevention of ventricular arrhythmias (23%) were the most common indications for CIED implantation. In 105 (73%) patients, infection developed after implantation of the original device; in 35 (24%) patients, infection developed after replacement; and in 4 (3%) patients, infection developed after revision. Mean left ventricular ejection fraction was 46% ± 14%. Generator pocket infection, with or without bacteremia, was the most common clinical presentation (69%), followed by CIED-related endocarditis (19%), and pocket infection with bacteremia was 12%. Vegetation was found on device lead in 17 patients, valvular vegetation in 2 patients, and both in 6 patients on transesophageal echocardiography (TEE). Coagulase-negative staphylococci (41%) and S aureus (24%) were the leading causative agents of CIED infection (Table 3).
Baseline Characteristics.a,b
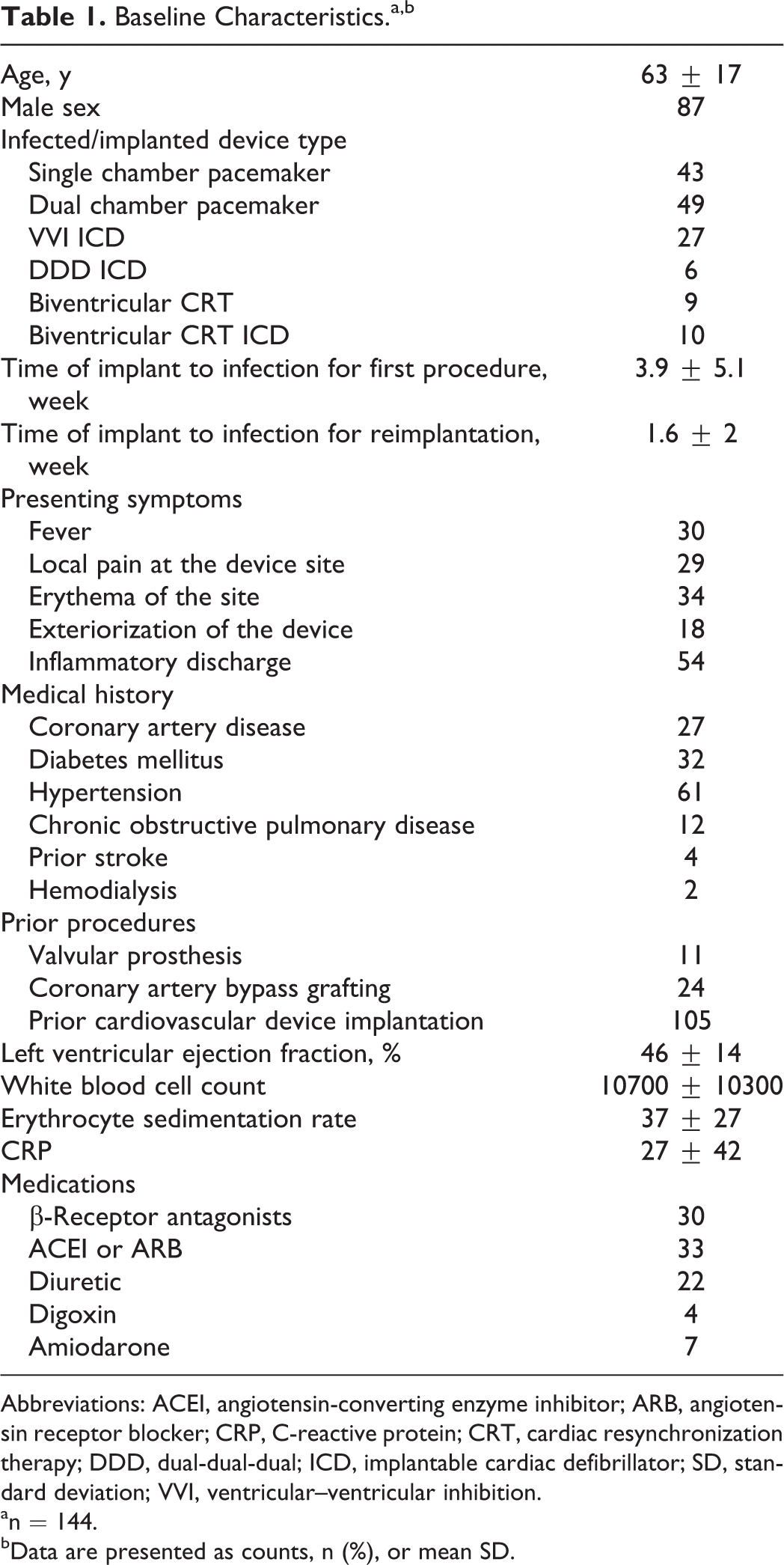
Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; CRP, C-reactive protein; CRT, cardiac resynchronization therapy; DDD, dual-dual-dual; ICD, implantable cardiac defibrillator; SD, standard deviation; VVI, ventricular–ventricular inhibition.
a n = 144.
b Data are presented as counts, n (%), or mean SD.
The Proportion of CIED in Total Device Implantation According to the Procedure Type.a
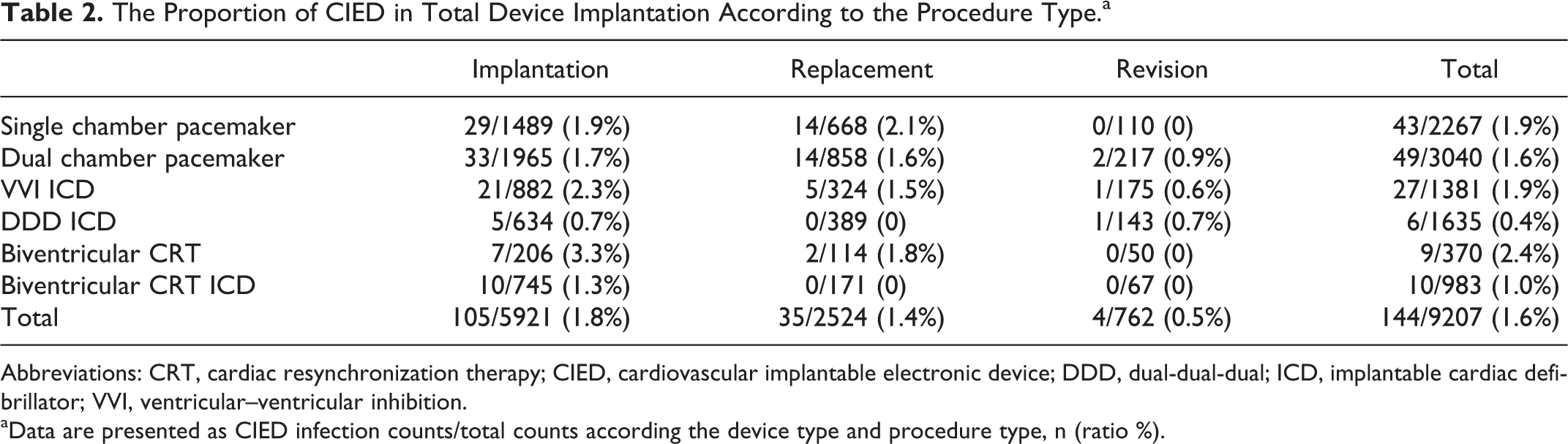
Abbreviations: CRT, cardiac resynchronization therapy; CIED, cardiovascular implantable electronic device; DDD, dual-dual-dual; ICD, implantable cardiac defibrillator; VVI, ventricular–ventricular inhibition.
a Data are presented as CIED infection counts/total counts according the device type and procedure type, n (ratio %).
Diagnostic and Therapeutic Characteristics and In-hospital Outcome of Patients.a
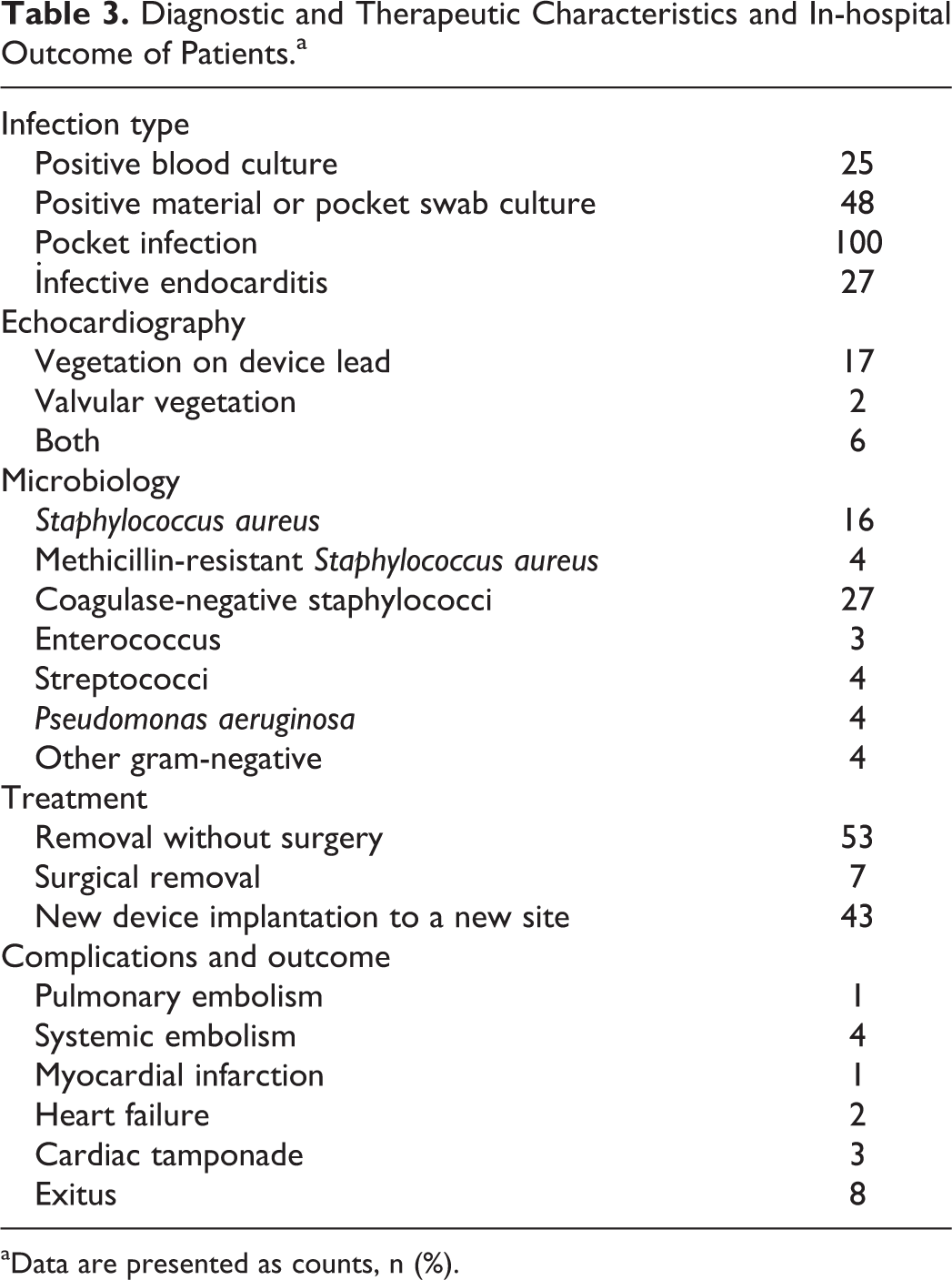
a Data are presented as counts, n (%).
The CIED infection was complicated by distant abscess formation in 4 patients (1 patient with lung abscess and 3 patients with systemic embolism), myocardial infarction in 1 patient, heart failure in 2 patients, cardiac tamponade in 3 patients, and in-hospital death in 8 patients.
Management and Outcome of Cases
Eighty four patients were treated with only antibiotics. Pulse generator and pacing leads were removed from 60 patients at the time of their initial presentation with a CIED infection. Leads were removed percutaneously in 53 (37%) patients using manual traction in 41 patients and extractor (Evolution mechanical dilator sheath; Cook Medical, Bloomington, IN, USA) in 12 patients. Seven (5%) patients underwent thoracotomy for lead removal. There was no complication related to percutaneous lead extraction. The mean duration of antibiotic therapy for CIED infection was 20 days (median, 14 days). Patients with pocket infection were treated with 7 to 14 days of antibiotic therapy, and those who had device-related endocarditis received up to 6 weeks of parenteral antibiotics. Reimplantation of a new CIED to a new site was deemed to be necessary in 43 (30%) of the cases. The mean time from removal of an infected device to placement of new system was 22 days. Eight patients died of infection-related causes during hospitalization. Five CIEDs couldn’t be removed due to manual traction failure and 2 of them died while waiting for extraction device. Removal with extraction device was failed in 3 patients. Characteristics of patients who died during hospital stay are shown in Table 4.
Characteristics of Patients who Died During Hospital Stay.
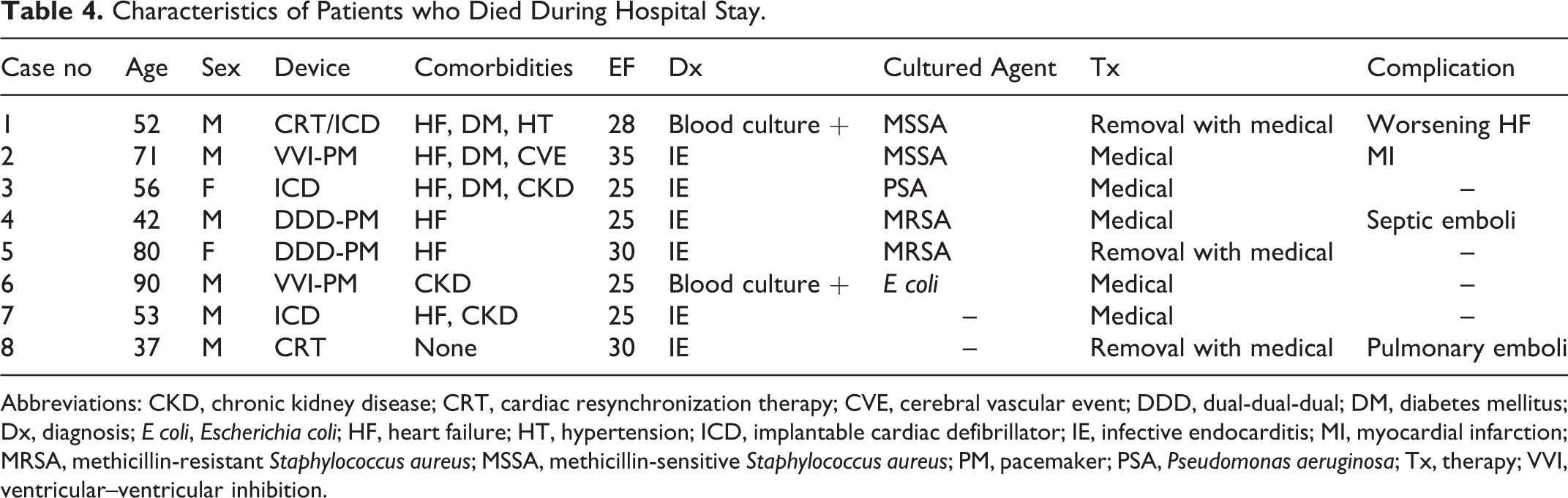
Abbreviations: CKD, chronic kidney disease; CRT, cardiac resynchronization therapy; CVE, cerebral vascular event; DDD, dual-dual-dual; DM, diabetes mellitus; Dx, diagnosis; E coli, Escherichia coli; HF, heart failure; HT, hypertension; ICD, implantable cardiac defibrillator; IE, infective endocarditis; MI, myocardial infarction; MRSA, methicillin-resistant Staphylococcus aureus; MSSA, methicillin-sensitive Staphylococcus aureus; PM, pacemaker; PSA, Pseudomonas aeruginosa; Tx, therapy; VVI, ventricular–ventricular inhibition.
Multivariate analysis that was conducted to examine the clinical factors associated with in-hospital mortality among patients with CIED showed that IE and ejection fraction were the strongest predictors of death (odds ratio [OR]: 13.3; 95% confidence interval [CI]: 2.072-85.328, P = .006 and OR = 0.925; 95% CI: 0.874-0.980; P = .008; respectively).
Discussion
The present study showed that the most common clinical presentation was pocket infection in patients with CIED infection. Main causative pathogen was coagulase-negative staphylococci. The IE and ejection fraction were the strongest predictors of in-hospital mortality.
Indications for the use of CIED have expanded recently, so there is an increase in the number of new device implantations. 13 Due to the increasing number of CIED, the CIED infection incidence has increased. The increased infection rates differed for patients with PPM infection (2.8-fold) compared with that of patients with ICD infections (6-fold). 13,14 A population-based investigation demonstrated a difference in device infection rates. The cumulative probability of device infection was greater among patients who had an ICD than those who had a PPM. 14 In our study, ICD-related infection ratio was 30%.
The mechanisms that are operative in prompting a dramatic increase in the rate of CIED infections remain largely undefined. Findings of a population-based investigation, however, may offer some explanation for the apparent rate increase in device infections. 15 Risk factors identified in a mixed (PPM and ICD) cohort included renal insufficiency, male sex, warfarin use, congestive heart failure, and generator exchange. Long-term corticosteroid use, however, was identified as a risk factor for PPM infection. In our study, the most common comorbidities were hypertension (42%), diabetes mellitus (22%), coronary artery disease (19%), chronic obstructive pulmonary disease (8%), prior stroke (3%), and hemodialysis (1%). In a large series, the mean age-adjusted Charlson comorbidity index was increased from 3.15 to 4.60 over the period 1975 to 2004, which may be a driving force behind the rapid increase in rate of device infections. 15 However, in our study, Charlson comorbidity index was relatively low (1.19). Thus, in our country, CIED infection rate will probably increase in the near future due to aging and increased comorbidities.
The CIED infections can either be superficial or deep, infecting the transvenous lead or epicardial electrode. Pocket infections are the most common. 9,16 Delayed erosion of the leads and/or device through the skin may result from a chronic infection, but may also be related to weight loss, a superficial pocket, or trauma at the site. These late infections may have minimal local inflammatory changes and may lack systemic symptoms. Infection of transvenous electrodes primarily involves the intracardiac lead and is essentially a right-sided endocarditis. 17,18 Coexistent left-sided endocarditis is occasionally seen. 16,19 Rarely, left-sided endocarditis occurs with malpositioning of the lead into the left ventricle. 20 The presentation is usually subacute, although occasional patients present with sepsis syndrome and shock. In our study, we found vegetation on device lead in 17 patients, valvular vegetation in 2 patients, and both in 6 patients on TEE. In our study, the rate of IE was 19%, isolated pocket infection was 69%, and pocket infection with bacteremia was 12%. Mortality of IE-related CIED without removed CIED is 31% to 66%, with removed CIED and aggressive medical treatment is 18%. 17,21 Our data are concordant with this finding. In our cohort, 8 patients died from CIED IE. Infections involving the generator pocket typically develop soon after CIED implantation or battery exchange. These infections present as acute or subacute wound infections; however, fever and systemic symptoms are often absent when infection is localized to the generator pocket. 3 The most common symptom at presentation was inflammatory discharge (36%).
An earlier meta-analysis of 7 randomized trials showed that antibiotic prophylaxis prior to PPM placement was beneficial. 22 In our study, antibiotic prophylaxis rate was 45%.
Perioperative contamination of the pacemaker pocket with skin flora appears to be the most common source of subcutaneous infection. This was illustrated in a prospective study including 103 patients undergoing elective pacemaker implantation. 23 Erosion through the skin of a pacemaker or ICD system component can be a portal for contamination. Alternatively, such erosions can occur as a secondary phenomenon due to pressure that develops on the overlying skin from a preexisting infection. 23 Infection of the intravascular component of a pacemaker or ICD system occurs primarily on the intracardiac portion of the lead along the right atrium, the tricuspid valve, or the right ventricular contact point. These infections may track intravascularly from infection of a subcutaneous pace component or may arise by bacteremic seeding from a remote site. 17,21,24,25
Staphylococcus aureus and coagulase-negative staphylococci, often Staphylococcus epidermidis, cause 65 to 75 percentage of generator pocket infections and up to 89 percentage of device-related endocarditis. 9,26 –28 Episodes arising within 2 weeks of implantation are more likely to be due to S aureus. 29,30 Seeding of the device from systemic bacteremia primarily occurs with S aureus infections. Among staphylococci causing device infection, methicillin resistance should be assumed until tests demonstrate methicillin susceptibility. Streptococci, Corynebacterium spp, Propionibacterium acnes, gram-negative bacilli, and Candida spp have caused occasional pulse generator pocket infections and device-related endocarditis. Among 162 episodes of precisely defined pacemaker endocarditis, coagulase-negative staphylococci caused 61% and S aureus caused 30%. 12,13,16,24 Methicillin resistance was common among all of these staphylococci. In our study, coagulase-negative staphylococci (41%) and S aureus (24%) were the leading causative agents of CIED infection.
The presence of multiple pacing wires is a potential cause of central venous thrombosis (in the area of the leads) and has been thought to increase the risk of device infection by serving as a nidus for secondary seeding of microorganisms. 31 Data regarding the infection rates during initial implantation and replacement are contradictory. 14,32 Replace registry showed that infection due to replacement procedure was lower than the initial implantation after 6 months. 33 Hematoma following CIED placement has been previously identified as increasing infection risk. 34 The 2010 American Heart Association guidelines on CIED infection, therefore, recommend strategies for prevention of hematoma following CIED placement. 4 These strategies include meticulous cautery of bleeding sites, consideration of packing the pocket to provide tamponade while leads are being placed, use of a pressure dressing for the first 12 to 24 hours, and evacuation only with impending dehiscence. Forty-seven percent of our cohort had multiple wires. We found that the replacement ratio was 24% and hematoma formation after implantation of CIED was 19% (in 28 patients), which was more excessive than the literature data that showed an occurrence of pocket hematoma formation of 2% to 5%. 35,36
Conclusion
In conclusion, CIED infection is an important health issue in our country. The most common clinical presentation was pocket infection. Main causative pathogen was coagulase-negative staphylococci. The IE and ejection fraction were the strongest predictors of in-hospital mortality. Subsequent work should be dedicated to the development of strategies to minimize the modifiable risk variables and to determine whether such modification impacts future infectious complications of CIED implantation.
Limitations of the Study
Main limitation of our study was its retrospective design with a potential for referral bias. There were variations in generator sizes, lead diameters, and lead materials between various pacemaker devices that depend on the manufacturing company and year of device availability. This variation in surface area of generator and lead material could affect the adherence properties of bacteria and the host inflammatory response to the device and subsequent risk of CIED infection. The specific device characteristics were not readily available.
Footnotes
Acknowledgments
This research received no grant from any funding agency in the public, commercial, or not-for-profit sectors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
