Abstract
Left main coronary artery (LMCA) stenosis is a high risk subset of coronary artery disease; however, its occurrence may be misjudged by coronary angiography. Coronary CT angiography (CCTA) was performed on 5 patients with clinically highly suspicious and angiographically borderline LMCA disease (ostial: 3 patients, mid: 1 patient and distal: 1 patient). A cross-sectional CCTA image was used to calculate the diameter stenosis. The quantitative coronary angiography (QCA) reference diameter (RD) correlated well with CCTA RD in all 5 patients. However, the lesion site minimal lumen diameter (MLD) by QCA correlated less well with that obtained by CCTA. The grade of stenosis measured by CCTA was significantly lower than that measured by QCA. Morphologically, three patients had LMCA ostial angulation (with minimal atheromatous changes), one patient had a biconcave configuration of the LMCA (with normal vessel wall) and one other had a tapering-morphology of the LMCA (with minimal atheromatous changes). In conclusion, CCTA may have a complementary diagnostic role in patients with angiographically ambiguous or inconclusive LMCA lesions and therefore it should be considered in the assessment of selected patients before they undergo coronary bypass surgery. In order to validate CCTA for main stem stenosis a proper gold standard, such as IVUS, is required.
Keywords
Introduction
Coronary angiography (CA) has been considered as the gold standard for the quantification of coronary artery disease; however, several studies have reported an unsatisfactory correlation between histologic and angiographic findings, particularly in the evaluation of the left main coronary artery (LMCA) [1–3]. Furthermore, ostial LMCA lesions can be misjudged by CA most markedly in patients whose LMCA is moderately diseased [1–4].
The presence of LMCA disease has important prognostic and therapeutic implications [5–7]. In patients with significant (≥50% diameter stenosis) LMCA disease, coronary bypass surgery improves survival significantly [8–12]. Unnecessary bypass surgery may reveal inappropriate use of available grafts and premature occlusion of either the native vessel or the graft.
Coronary computed tomography angiography (CCTA), using a multidetector CT (MDCT) scanner, is a non-invasive 3D imaging technique that provides an excellent overview of the cardiac and coronary arteries in vivo [13]. Unlike CA, the cross-sectional nature of CCTA may enable accurate assessment of the vessel wall, depiction of plaque, and the precise lesion site in the LMCA. Furthermore, CCTA can assess without difficulty the reference diameter (RD) and minimal lumen diameter (MLD) necessary for calculating the grade of stenosis. Recently, Caussin et al. [14] assessed the ability and accuracy of CCTA (using 64- row MDCT) to quantify minimal lumen area (MLA) and determine lesion severity in intermediate stenosis by CA compared with intravascular ultrasound (IVUS). Based on their results, CCTA was able to quantify coronary stenosis with good correlation compared with IVUS and determine lesion severity in patients with intermediate lesions by CA.
In this paper we describe the CCTA findings in 5 patients with clinically highly suspicious yet angiographically borderline LMCA disease, which were referred to us to fully evaluate the LMCA involvement and to assist in making therapeutic decisions.
Materials and Methods
Patient Population
For a period of four months, 5 patients (age range: 46 to 70 years) underwent angiographic and CCTA evaluation of the LMCA. These patients had ischemic symptoms before diagnostic angiography and were referred because the angiographic assessment of LMCA lesion severity was inconclusive (ostial: 3 patients, mid: 1 patient and distal: 1 patient). Of these, 4 were women. One patient (No. 1) underwent coronary bypass surgery 2 years previously for ostial LMCA disease. In 4 patients CABG was the default therapeutic option. The time interval between CA and CCTA was 5-10 days. Written informed consent was obtained from all subjects included in the study.
Multidetector CT scan protocol
ECG-gated CCTA was performed using a multi-detector computed tomography (MDCT) scanner (VCT General Electric Medical systems, Milwaukee, WI). The following scanning parameters were applied: detector collimation 64 × 0.625 mm, gantry rotation time 0.35 second, slice thickness 0.6 mm, tube voltage of 120 kV, with pitch range 0.2-0.29. Subjects received a β-blocker two hours prior to the examination (atenolol 50 to 100 mg orally) if resting heart rate exceeded 70 beats per minutes (bpm). Sublingual nitroglycerin was given to all subjects before scanning. A bolus of 70-80 ml Iomeron 400 (iomeprol) [(400 mgl/ml, Bracco, Milan, Italy)] was intravenously injected (4 ml/s) via an 18-gauge catheter placed in the antecubital vein, followed by a bolus of 40 ml of saline. Scan delay was determined according to the Smart Prep program (automatic bolus test; the region of interest was placed on the ascending aorta). The subjects were instructed to maintain an inspiratory breath hold during which the CT data and ECG trace were acquired. Datasets were acquired at phases 40%, 70% and 80% of the R-R cycle. The image data sets were processed on a separate workstation (ADW 4.2, GE Healthcare).
Coronary arteries were reviewed by two experienced radiologists blinded to the QCA results. LMCA was assessed for morphology, diameter, and concomitant atheromatous wall changes. Maximal intensity projection and multiplanar reconstruction were used to select the tightest site. Measurements were then made on the cross-sectional arterial reconstruction. MLD and RD were manually drawn on the cross-sectional reconstruction. MLD and RD were measured in the diastolic phase. The contrast window was manually adjusted to visualize optimally arterial contour. The target lesion was compared with the reference segment. Clinical examples are shown in Figures 1 and 2.

This case example illustrates the discrepancy between angiographic (
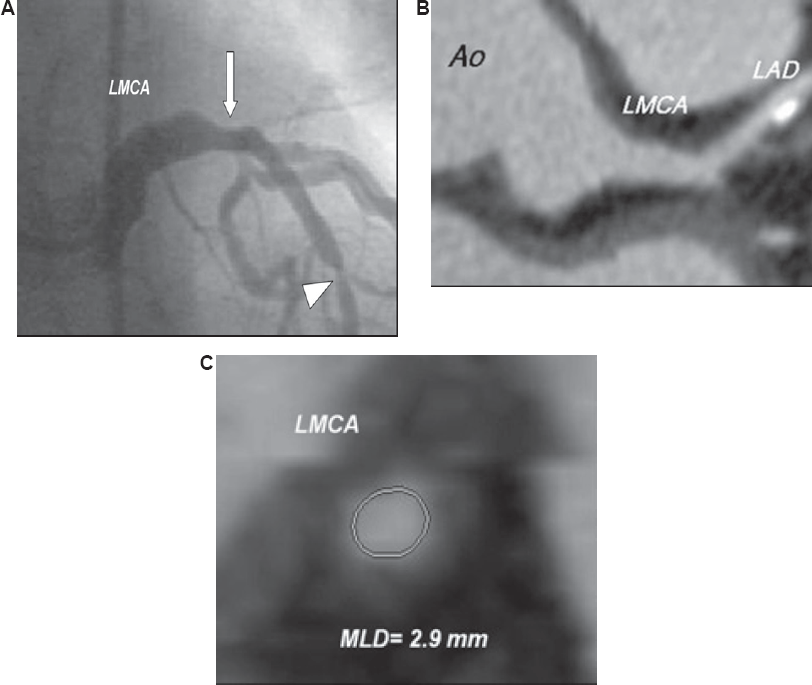
A 55-year-old patient with angiographically intermediate distal LMCA stenosis (arrow) (
Coronary Angiography
Coronary angiography was performed in a standard fashion. Intracoronary nitroglycerin (200 μg) was injected into all patients prior to CA. Pressure ventricularizzation was assessed during the CA. Quantitative coronary angiography (QCA) was performed by 2 independent, experienced technicians blinded to the results of CCTA, who used validated, automated, edge-detection software (MDQM-QCA), according to previously validated and published protocol [5]. The LMCA was visualized in at least 5 projections in each patient. The MLD in the endiastolic frame was measured from angiographic projections revealing the “worst” view. The RD was measured from an angiographically normal segment.
Results
All LMCA segments were considered assessable. The CA and CCTA characteristics are summarized in Table 1. The data from the table indicate a good correlation between QCA RD and CCTA RD, and a large variation between CA and CCTA regarding MLD and grade of stenosis. Notably, in the three patients with ostial LMCA disease and pressure ventricularization, no significant ostial stenosis was found by CCTA. However, morphologically, each patient had ostial angulation (acute take-off angle at aortic-coronary junction) of the LMCA and minimal eccentric atheromatous wall changes (Fig. 1).
Characteristics of patients and LMCA angiographic and CCTA findings.

The patient after CABG (No. 1) had complete occlusion of the left internal mammary artery (LIMA) graft to the left anterior descending artery (LAD). In the patient with angiographically mid LMCA disease (No. 4), CCTA revealed biconcave appearance of the left main without vessel wall abnormality. The patient with angiographically intermediate distal LMCA stenosis (No. 5) had mild stenosis according to CCTA. The LMCA had a tapering morphology with minimal vessel wall atheromatous changes at the lesion site (Fig. 2).
As a result of the analysis by CCTA, all patients were treated medically. One patient (No. 5) underwent percutaneous coronary intervention (PCI) for significant mid LAD stenosis. The lesion was detected by both diagnostic modalities. There was no death or myocardial infarction during the follow-up period (range16-24 months).
Discussion
Angiographic determination of LMCA stenosis has obvious limitations in terms of assessing the degree of narrowing and quantification of vessel wall atheromatous changes [1–4]. Furthermore, autopsies studied have shown that in many situations in which LMCA was mildly diseased, these cases were often reported by angiography as significantly stenosed [1]. Several studies showed a limited correlation between IVUS and CA lumen dimensions [5]. Abizaid et al. [5] reported that in patients with an LMCA stenosis, IVUS MLD and DS were correlated poorly with QCA MLD and DS. Interestingly, in our evaluated patients, three ostial LMCA lesions and one midvessel and one bifurcation lesions were overestimated by CA.
In ostial and bifurcation lesions, the stenosis may be obscured by overlapping contrast-filled structures. Detection of ostial LMCA disease is confounded by the tendency for catheters to be “seated” beyond the lesion, thus requiring reflux of contrast into the aorta for adequate visualization; refluxing contrast fills the aortic cusp, which may obscure the lesion. Intermediate stenoses (with an angiographic severity ranging from 40% to 75%) are particularly problematic in patients whose symptomatic status is difficult to assess. Some of these patients, and based on ventricularization of pressure observed during CA, may be referred to unnecessary surgical revascularization by the attending cardiologist. In fact, such an operation performed too early in one of our patients (case 1) resulted in premature closure of the LIMA-LAD graft, further highlighting the insignificance of the LMCA angiographic findings. Morphologically, the LMCA in this patient had an ostial angulation on CCTA; such an anatomical factor may impair angiographic left main evaluation. A similar finding was also observed in the other two patients with angiographically ostial LMCA disease (cases 2 and 3). This angiographic limitation was previously described by several studies [2–4]. Furthermore, the differences between CA and CCTA might be due to the fact that the appearance of an eccentric stenosis in CA depends on the availability of a projection showing the real MLD. Therefore, a true 3D standard of reference is needed. On the other hand, proximal LMCA stenosis is sometimes caused by thin ostial membrane. This membrane can be too thin to be accurately depicted by CCTA, whereas CA can detect them. This may explain some of the discrepancies between CCTA and CA.
Recently, Zeina et al. [15] described the 3D configurations of the aortocoronary junction and the LMCA ostium in normal subjects who underwent CCTA. In this study, the normal LMCA had various anatomical configurations: biconcave-shape appearance (type 1), tapering morphology (type 2), combined morphology (type 3) and funnel-shape appearance (type 4). Ostial angulation was considered as a normal variant usually associated with the posterior position of the LMCA orifice of origin in the aortic sinus.
Angiographically, the distal LMCA is often obscured by the confluence of shadows from the overlapping left anterior descending, left circumflex, and ramus medianus arteries. The overlap can explain inaccuracy of stenosis assessment by CA. In addition, the tapering morphology of the LMCA observed in the patient with distal LMCA disease (case 5) may result in inaccurate calculation of the DS by CA using the “very wide” proximal portion as the normal reference diameter. Atheromatous plaques responsible for LMCA lesions, as well as the adjacent normal vessel, can be visualized well by CCTA. Thus, accurate identification of the normal reference diameter is not difficult using this diagnostic modality.
Mid LMCA lesion was a constant angiographic finding in case 4, despite an intracoronary nitro-glycerin injection; therefore, the mid left main stenosis was a convincing angiographic finding for the attending cardiologist. We think that, the anatomic biconcave configuration of the LMCA on 3D MDCT reconstruction images was wrongly interpreted as a mid LMCA disease on CA. The completely normal vessel wall and dimensions, as assessed by CCTA, was an important diagnostic result for the attending cardiologist in assisting therapeutic decisions and follow-up.
Other possible limitations of CA in assessing LMCA disease appear to be the following: 1) A short LMCA also makes identification of a normal reference segment difficult. 2) There is compensatory enlargement (positive remodeling) of the vessel as the plaque burden increases to preserve the lumen size, as shown by Glagov et al. [16]. 3) The two-dimensional silhouette method of angiography depicting contrast-filled lumen cannot detect diffuse, concentric plaque because of the lack of an unselected reference segment for comparison.
With the newer generations of MDCT scanners, the diagnostic performance for the assessment of coronary arterial stenoses has significantly improved, and the proportion of nonassessable segments has decreased [17–19]. Of note, previous studies showed good correlation of CCTA with reference standards [14]. The use of cross-sectional reconstruction for measurement allows calcification localization with respect to the lumen. Thus, the lumen and plaque are well defined for acceptable contour tracing. However, some difficulties may be encountered when calcification and the contrast agent are in the same density range.
By providing a cross-sectional, tomographic perspective with direct visualization of the vessel wall, CCTA can overcome the limitations of CA. This noninvasive 3-D technique, which allows accurate assessment of lumen size, plaque location, and composition (calcified or non-calcified), has resulted in excellent diagnostics for the analysis of coronary artery disease.
Limitations
We studied only a small number of participants; therefore, additional large prospective, unbiased studies should be performed including wider range of LMCA stenosis. Unfortunately, none of the patients with angiographically borderline LMCA disease underwent IVUS; therefore, comparisons between the CCTA and IVUS results were not performed.
The ability of CCTA to delineate calcified and non-calcified lesions within the coronary artery wall that may or may not cause luminal stenosis has been demonstrated [20, 21]; however lipid-rich soft plaque can not be differentiated from fibrous plaque using this diagnostic modality.
Conclusion
Based on our observations, we think that CCTA may have a complementary diagnostic role in patients with angiographically ambiguous or inconclusive LMCA lesions and therefore it should be considered in the assessment of selected patients before they undergo coronary bypass surgery. In order to validate CCTA for main stem stenosis a proper gold standard, such as IVUS, is required.
