Abstract

Acute pulmonary embolism (PE) displays a heterogeneous clinical presentation, and patients often present with nonspecific symptoms.1,2 The common clinical features of PE include chest pain and dyspnea, 3 which resemble the features of acute coronary syndrome (ACS), thereby creating a diagnostic dilemma. Moreover, an elevated serum troponin level and electrocardiographic (ECG) changes can lead physicians to an erroneous diagnosis of ACS instead of PE. These diagnostic challenges might delay effective treatment of PE and result in poor prognosis. 4 This retrospective study was conducted to investigate the clinico-radiological features of patients with PE in whom ACS was initially suspected.
Data pertaining to all patients hospitalized with PE from January 2007 to August 2018 at the Kyungpook National University Hospital (KNUH), a tertiary referral center in Daegu, South Korea, were collected. Information regarding patients with PE, who had undergone coronary angiography (CAG) during the same time period, was obtained from electronic databases at KNUH and two other centers. This study was approved by the Institutional Review Board of each institution, and requirements for informed written consent were waived. The subjects were divided into two groups: patients from the three institutions who underwent CAG for the suspicion of ACS prior to PE diagnosis (CAG group) and patients with PE from the KNUH who did not undergo CAG (control group).
Results are presented in Table 1. Overall, 32 patients with PE were initially suspected to have ACS and underwent CAG prior to PE diagnosis. Twenty-nine patients were diagnosed with PE by computed tomography (CT) scan; two by ventilation-perfusion scan; and one by conventional pulmonary angiography. The CAG group had a significantly higher incidence of unprovoked PE than the control group (p < 0.001) (Table 1). Chest pain was significantly more common in the CAG group than the control group (p < 0.001). Clinical prediction rules, including Wells score 5 (p < 0.001) and revised Geneva score 6 (p = 0.001), were significantly lower in the CAG group compared to the control group. The CAG group included significantly higher proportions of high-risk (p = 0.001) or intermediate–high-risk patients with PE (p = 0.009) according to the European Society of Cardiology (ESC) risk stratification, 7 adverse outcomes (p = 0.008), and PE-related in-hospital deaths (p = 0.024) compared to the control group. ECG changes suggestive of right ventricular (RV) strain (p = 0.002) and a higher level of troponin I (p < 0.001) were more frequently observed in the CAG group than in the control group. On CT scan, the CAG group was characterized by a significantly higher incidence of central PE (p = 0.040) and RV dilation (p = 0.018) than the control group.
Clinical characteristics, blood biomarkers, and CT findings.
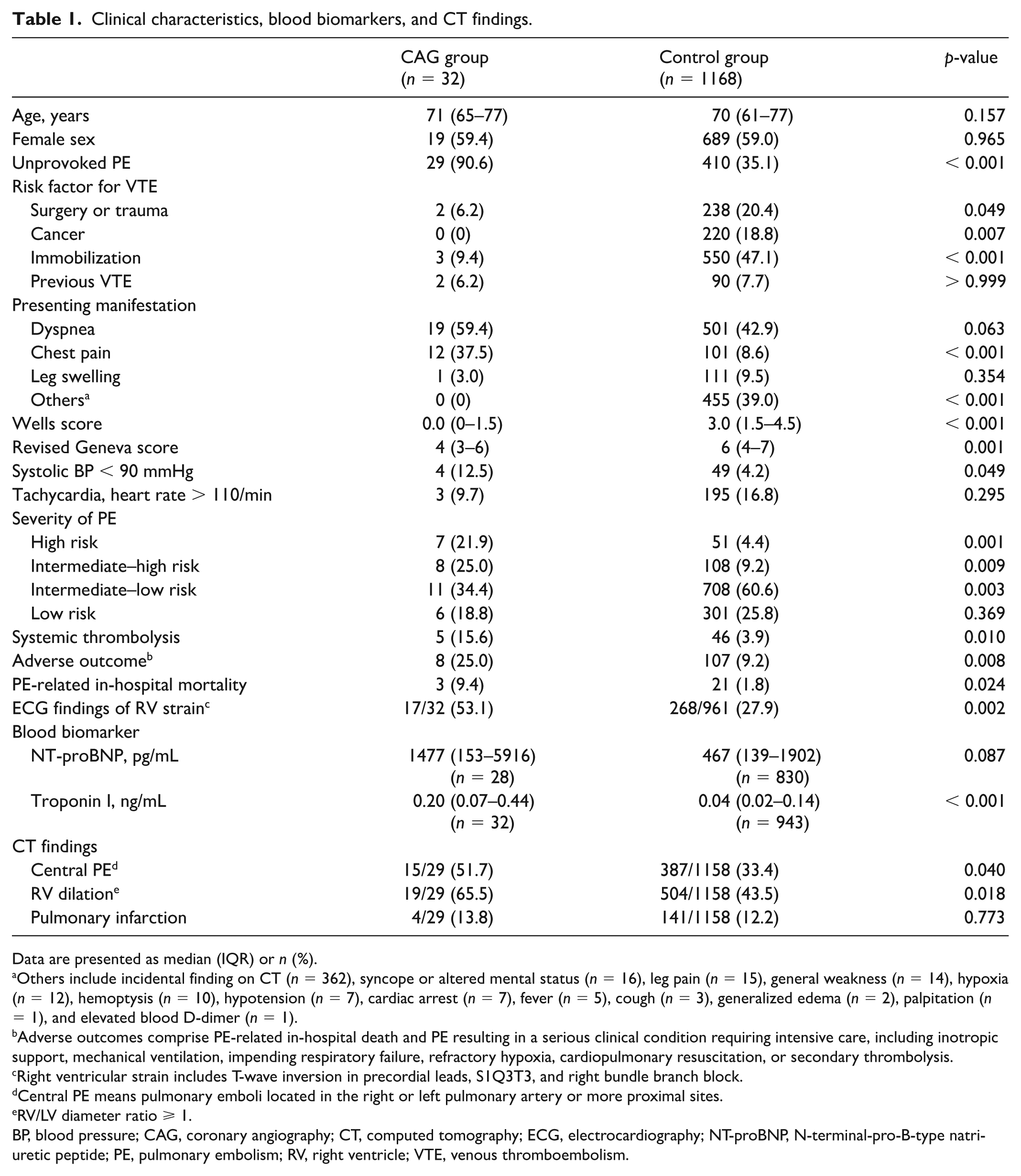
Data are presented as median (IQR) or n (%).
Others include incidental finding on CT (n = 362), syncope or altered mental status (n = 16), leg pain (n = 15), general weakness (n = 14), hypoxia (n = 12), hemoptysis (n = 10), hypotension (n = 7), cardiac arrest (n = 7), fever (n = 5), cough (n = 3), generalized edema (n = 2), palpitation (n = 1), and elevated blood D-dimer (n = 1).
Adverse outcomes comprise PE-related in-hospital death and PE resulting in a serious clinical condition requiring intensive care, including inotropic support, mechanical ventilation, impending respiratory failure, refractory hypoxia, cardiopulmonary resuscitation, or secondary thrombolysis.
Right ventricular strain includes T-wave inversion in precordial leads, S1Q3T3, and right bundle branch block.
Central PE means pulmonary emboli located in the right or left pulmonary artery or more proximal sites.
RV/LV diameter ratio ⩾ 1.
BP, blood pressure; CAG, coronary angiography; CT, computed tomography; ECG, electrocardiography; NT-proBNP, N-terminal-pro-B-type natriuretic peptide; PE, pulmonary embolism; RV, right ventricle; VTE, venous thromboembolism.
Compared to patients in the control group, most patients in the CAG group had unprovoked PE and lower clinical prediction rule scores, suggesting that PE can occur under unpredictable circumstances. The lower incidence of PE compared to ACS8,9 might also be one of the reasons for not considering PE as a differential diagnosis. Chest pain, a common symptom of PE, is usually caused by pleural irritation due to small emboli causing pulmonary infarction; moreover, angina-like chest pain may occur due to RV ischemia in patients with central PE. 7 CAG should be performed immediately, for diagnostic purposes, in patients at high risk and in whom the differential diagnosis is not certain. 10 In the CAG group, all patients were symptomatic at presentation, and the lobar or more proximal pulmonary artery was the largest pulmonary vasculature involved by emboli in most patients. In terms of prognosis, these patients were characterized by a relatively more severe form of PE, which manifested as higher levels of blood biomarkers and higher incidences of RV dilation and central PE. Consequently, patients in the CAG group were more likely to exhibit high- or intermediate–high-risk PE (classified according to the 2014 ESC risk stratification model), adverse outcomes, and PE-related in-hospital deaths compared to patients in the control group.
Selection bias was inevitable in this study, given that patients with PE in the control group were from a single study center and that the study is of a retrospective nature. Another limitation is the small number of patients who underwent CAG.
In conclusion, patients with PE undergoing coronary angiography before diagnosis of PE are characterized by a higher incidence of unprovoked PE and a low clinical prediction score, often presenting with chest pain and more severe forms of PE, compared to patients in the non-CAG group. In such cases, keeping PE in the differential diagnosis is essential to make an accurate diagnosis and thereby potentially reduce long-term complications of otherwise undiagnosed high-risk PE.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
