Abstract
Introduction
Leukocytoclastic vasculitis (LV) is a necrotising vasculitis of the small dermal blood vessels, clinically presented as palpable purpura. It is a heterogeneous disorder often limited to the skin but which may involve other organs. LV might be a serious drug reaction, caused by bacterial and viral infections, or less commonly a manifestation of systemic vasculitic syndromes.
Case Reports
Three patients were admitted to our institution with petechiae and palpable purpura. The cutanous lesions were affecting the lower limbs and in one patient also the upper extremities and the trunk. The diagnosis of leukocytoclastic vasculitis was made based on clinical and histopathological findings. Systemic involvement was excluded, as was connective tissue disease. Clinical examination revealed ulcers on the legs of each patient. Smears from those ulcers were taken and investigated for micro organisms. Culture results showed infection with coagulase negative staphylococci. Systemic signs of sepsis were absent; therefore the infections were treated locally. Two patients developed necrotic blisters during the first week of hospitalisation. To avoid further vasculitic complications steroids were administered parenterally and LV lesions diminished in all patients within ten days.
Conclusion
Drugs and connective tissue disease were ruled out as triggering factors of LV in the patients reported on. Therefore, it was concluded that superantigens produced by the coagulase negative staphylococci were responsible for LV.
Introduction
Leukocytoclastic vasculitis (LV) is a necrotizing vasculitis of the small dermal and subcutaneous blood vessels, clinically presenting as petechiae and palpable purpura. LV is a heterogeneous disorder often limited to the skin especially of the lower extremities, because of hydrostatic forces and turbulence (Claudy, 1998), but may also be present as a systemic disorder and involve other organ systems. Histologically LV is characterised by a dense neutrophil infiltrate, leukocytoclasia, fibrin deposition, necroses and haemorrhage. After the acute phase the disease may present with a chronic evolution and recurrent episodes. Usually it is a self limiting disease, but steroids may be used to prevent complications. LV might be a serious drug reaction, or caused by bacterial or viral infections (Fan et al. 1980; Garcia-Porrua and Gonzalez-Gay, 1999; Hellmich, 2001; Jessop, 1995; Koutkia et al. 2001; Lee et al. 1998; Tai et al. 2006). Less commonly this condition is a manifestation of systemic vasculitic syndromes (Gonzalez-Gay and Garcia-Porrua, 1999; Jessop, 1995; Tai et al. 2006) We report on three patients admitted to our institution with acute evolving LV.
Patient and Methods
All relevant diagnostic and therapeutic procedures during the reported observation were discussed with the affected patients and written informed consents were obtained for the invasive diagnostic tests.
Relevant laboratory parameters of the reported patients at the time of admission are summarised in Table 1.
Relevant laboratory parameters of each patient at admission.
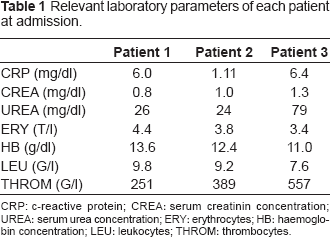
CRP: c-reactive protein; CREA: serum creatinin concentration; UREA: serum urea concentration; ERY: erythrocytes; HB: haemoglobin concentration; LEU: leukocytes; THROM: thrombocytes.
Case 1
A 65 year old man with prediagnosed diabetes mellitus, coexistent polyneuropathia and atherosclerosis developed multiple petechiae and purpura affecting the upper and lower legs bilaterally. The diagnoses of LV was suspected as a cause of the clinical appearance and confirmed by skin biopsy. Four weeks prior two ulcers located on the second toe of the right foot were documented. Both ulcers were caused by occlusion of the supplying arterial blood vessels. For several years there had been stable medication including metformin and lisinopril. Other drugs were refused by the patient. The cultures obtained from the ulcers were positive for staphylococcus (staph) simulans and candida glabrata. No signs of sepsis or involvement of internal organs were detected and the infection therefore was treated locally. Within three days the LV lesions worsened and necrotic blisters occurred. Steroids (1 mg per kilogram bodyweight of prednisone equivalent) were administered intravenously and continuously tapered during the next weeks. Ten days later the petechiae and purpura had completely diminished. The necrotic blisters evolved to ulcers and were treated locally.
Case 2
A man aged 87 was referred to our hospital with palpable purpura on both feet and lower legs, accompanied by arthralgia. Skin biopsy was conducted and consistent with LV. Two weeks prior the patient had had a trauma resulting in an about 20 mm large wound located on the right dorsum pedis. Smears were taken from the wound and cultures showed the growth of staph saprophyticus and corynebacteria. The traumatic lesion was treated locally, while 25 mg of prednisone equivalent were orally administered to control the LV. The purpura diminished within six days and no necrotic lesions occurred. No drugs were taken by the patient prior to the purpura occurrence.
Case 3
An 89 year old man was admitted to our institution with massive pain caused by ulcerations on both ankles. Additionally, numerous purpura and petechiae were found on the upper and lower extremities, the whole trunk and even the neck. LV was suspected and confirmed by skin biopsies. Systemic involvement and connective tissue disease were excluded. Smears and cultures obtained from the ulcers revealed the presence of staph epidermidis. Due to the extensive skin ulcerations an antibiotic treatment with vancomycin was initiated. Additionally, 50 mg of prednisone equivalent were administered intravenously. Steroids were tapered continuously as the LV lesions decreased. As comorbidities atherosclerosis, hypertension and combined hyperlipidemia were documented. For fourteen months constant medication including losartan, bisoprolol, simvastatin and acetyl salicylic acid was established.
Discussion
In the three patients being reported on the diagnoses of LV was made based on clinical findings and the histopathological evaluation of skin biopsies obtained from each patient. In smears taken from the skin lesions of all patients coagulase negative staph were confirmed by cultivation.
No signs of sepsis or coexisting connective tissue disease occurred during the overseen time of 8 to 24 months. Immunological investigations were negative for antinuclear antibodies, complement factors, anti-DNA antibodies, antineutrophil cytoplasmic antibodies, anticardiolipin antibodies, cryoglobulinaemia and rheumatoid factor. Renal function tests (including serum creatinin and serum urea concentrations), were compared with previous test results and showed stable values. The microscopic evaluation of urine sediment in each of our three patients revealed no evidence of renal involvement. Active and recent viral infections and other systemic bacterial infections, as well as malignancies were excluded. No temporal relationship or causative association of LV with the pre-existing drug therapy could be established.
Various infectious micro organisms such as bacteria, viruses, helminthes and fungi are associated with vasculitic syndromes. While staph aureus infections have already been linked to Wegener's granulomatosis (Popa et al. 2002; Popa et al. 2003) and Henoch-Schonlein purpura (Eftychiou et al. 2006; Samarkos and Vaiopoulos, 2005) coagulase negative staph to the best of our knowledge had only once been reported to cause a cutaneous small vessel vasculitis (Cantwell and Kelso, 1982) in a severely immunocompromised patient.
Coagulase negative staph colonise the human skin and mucous membranes and these micro organisms were considered to be apathogen for a long time. Medical progress, especially the use of implants, proved that these micro organisms are responsible for nosocomial infections. Systemic infections caused by coagulase negative staph already were reported to occur in immunocompromised patients. The three patients being reported on surely were not fully immunocompetent because of their age and comorbidities but only localised infections with different coagulase negative staph were detected. Blood cultures obtained in all patients and a careful clinical examination ruled out bacteraemia and generalised infection. With no signs of accompanying connective tissue disease or systemic infections the LV in the reported patients might be interpreted as hypersensitivity angiitis caused by staph molecules acting as immunomodulators.
During follow up, specimens of the primary lesions in each patient and the primary and secondary lesions of patient 1 were cultured. None of these tests showed evidence for coagulase negative staph after local treatment (and additional intravenous vancomycin in patient 3) regimes. Of course, we cannot rule out that the initially identified micro organisms only reflect normal dermal flora. However, the documented skin lesions represent areas with a severely compromised barrier function. Therefore, it might be possible for staph toxins and molecules to reach deeper tissue and even the blood stream.
Staph produce numerous toxins which may act as superantigens and are able to cause various syndromes (Eftychiou et al. 2006; Samarkos and Vaiopoulos, 2005; Savige et al. 2002). LV is probably a multi-factorial disease (Claudy, 1998) and the pathogenicity of staph superantigens still needs to be debated as a factor responsible for the induction of vasculitis. The causative and temporal relationship between the infection and the occurrence of the LV seems to support this hypothesis in our patients. Coagulase negative staph might be responsible for LV and therefore deserve further investigations in the context of LV and other vasculitic syndromes.
