Abstract
Current treatment guidelines for acid-related diseases (ARDs) recommend first-line treatment with a proton pump inhibitor (PPI) to reduce gastric acid production. PPIs are indicated in the management of gastroesophageal reflux disease (reflux esophagitis, nonerosive reflux disease), peptic ulcer (gastric and duodenal ulcer, non-steroidal anti-inflammatory drug (NSAID)-associated ulcer, bleeding ulcer), functional dyspepsia, and in association with Helicobacter pylori eradication therapy when needed. Currently, PPIs (omeprazole, lansoprazole, pantoprazole, rabeprazole and esomeprazole) are widely used for the treatment of ARDs. All 5 PPIs are effective. However, there are differences in PPI pharmacokinetic and pharmacodynamic profiles that might influence their clinical utility. Rabeprazole is a useful option for the treatment of acid-related diseases due to its rapid onset of acid inhibition and few drug interactions.
Introduction
Proton pump inhibitors (PPIs) are indicated in the management of acid-related diseases (ARDs) and in association with Helicobacter pylori (H. pylori) eradication therapy when needed. PPIs represent the most important recent advance in the treatment of ARDs. With efficacy profiles superior to those of histamine H2-receptor antagonists (H2RA), and prokinetics, PPIs are now considered the drugs of choice in managing patients with ARDs. Currently, PPIs (omeprazole, lansoprazole, pantoprazole, rabeprazole and esomeprazole) are widely used for the treatment of ARDs. All 5 PPIs are effective and safe, however, there are differences in PPI pharmacokinetic and pharmacodynamic profiles that might influence their clinical utility. 1 This article provides an update on the clinical efficacy and safety of rabeprazole when used to treat ARDs.
Onset and power of Acid Inhibition
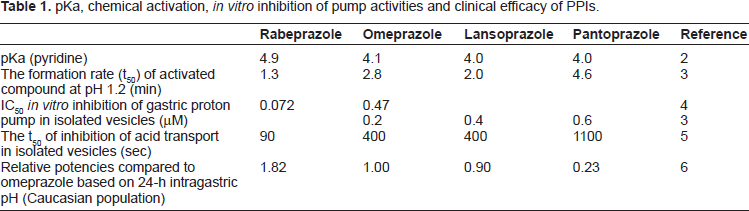
pKa, chemical activation, in vitro inhibition of pump activities and clinical efficacy of PPIs
Median pH monitoring values before and on the first day of each proton pump inhibitor treatment. Values in parentheses are 10%-90% confidence intervals.

P < 0.03 vs. LAN (Wilcoxon signed rank test for paired data);
P < 0.03 vs. PAN (Wilcoxon signed rank test for paired data);
P < 0.02 vs. OME CAPS (Wilcoxon signed rank test for paired data);
P < 0.04 vs. OME MUPS (Wilcoxon signed rank test for paired data);
P < 0.04 vs. PBO (Wilcoxon signed rank test for paired data).
Metabolic pathways
The primary metabolic pathway of rabeprazole is non-enzymatic conversion to rabeprazole-thioether. A minor metabolic pathway is via the cytochrome P450 liver isoenzymes CYP2C19 and CYP3A4. Rabeprazole undergoes extensive hepatic metabolism, predominantly nonenzymatic reduction to the thioether and to a lesser extent via CYP2C19 (to desmethyl rabeprazole) and CYP3A4 (to a sulphone metabolite). No metabolites of rabeprazole have any significant antisecretory activity.
11
In comparison with omeprazole, lansoprazole and pantoprazole, the pharmacodynamics of rabeprazole are the least affected by cytochrome P450 (CYP)2C19 genotype (Fig. 1).
12
Multiple-dose studies in healthy volunteers showed no significant effect of CYP2C19 genotype on the effects of rabeprazole on intragastric pH.13,14 For example, with subjects administered lansoprazole and omeprazole, but not rabeprazole, poor metabolizers had significantly (P < 0.005) higher intragastric pH measurements after 7
13
or 8
14
days treatment than homozygous and heterozygous extensive metabolizers. The largely non-enzymatic metabolism of rabeprazole partially accounts for fewer drug–drug interactions between this PPI and other P450 isoenzyme dependent medications. Co-administration of rabeprazole with P450 metabolized medications such as theophylline, diazepam, warfarin and phenytoin does not interfere with their pharmacokinetics.
15
Unlike other PPIs, the hepatic metabolism of rabeprazole is predominantly via nonenzymatic processes and only to a small extent via CYP mediated reactions.
11
Metabolic pathways for omeprazole and esomeprazole, lansoprazole, rabeprazole and pantoprazole. The thickness of the arrows indicates the approximate contribution of each cytochrome P450 (CYP) isoform to each of the metabolic pathways. Ishizaki T, Horai Y. Review article: cytochrome P450 and the metabolism of proton pump inhibitors-emphasis on rabeprazole. Aliment Pharmacol Ther. 1999 Aug;13 Suppl 3:27-36; Horai Y, Kimura M, Furuie H, et al. Pharmacodynamic effects and kinetic disposition of rabeprazole in relation to CYP2C19 genotypes.
Drug Interactions
Drug interactions mediated by the P450 enzyme family are considered unlikely with rabeprazole. 16 For example, studies have shown no clinically significant interactions between rabeprazole and single oral doses of warfarin or theophylline, nor with single intravenous doses of diazepam or phenytoin. 17 The American Food and Drug Administration has indicated that proton pump inhibitors increase the international normalized ratio (INR) when used concomitantly with warfarin, due to metabolization by cytochrome P450 2C19. We therefore reviewed patients taking warfarin. Two hundred and forty patients administered warfarin after surgery were divided into two groups: Group I (n = 114) administered rabeprazole (10 mg/day) and Group II (n = 126) administered lansoprazole (15 mg/day). The initial dose of warfarin was 3 mg and INR was initially measured on postoperative day 4. The initial INR was significantly lower in Group I (1.66 ± 0.87) than in Group II (2.06 ± 1.03, P = 0.0011). Complications of delayed cardiac tamponade and hemothorax were seen in 6 and 1 patients, respectively, in Group II from 5 days to 3 months postoperatively. In contrast, no patients in Group I experienced delayed hemorrhagic complications (P = 0.015). Fisher's exact test showed that only lansoprazole was associated with bleeding events (P = 0.0151). These results suggest that lansoprazole enhances the effects of warfarin. Rabeprazole can be safely used concomitantly with warfarin. 18 Recent evidence has shown that clopidogrel and PPIs are metabolized by the same pathway, and that patients taking both drugs have greater levels of platelet reactivity and more adverse outcomes than patients taking only clopidogrel. 19 Clopidogrel is a pro-drug that is metabolized to its active ingredient by hepatic cytochrome P-450 isoenzymes, with isoenzyme CYP2C19 playing a major role. 20 PPIs may also competitively inhibit CYP2C19 metabolism. In vitro testing of a model substrate showed that lansoprazole and omeprazole were the most potent inhibitors, whereas pantoprazole and rabeprazole were the least potent, with inhibition constants (Ki) for the latter PPIs above their pharmacologically relevant concentrations generally achieved in vivo 21 Patients taking aspirin 100 mg plus clopidogrel 75 mg/day were assigned to groups administered rabeprazole 10-20 mg/day or without rabeprazole, prospectively. The decision to administer RPZ or not was at the discretion of the treating physician. The pharmacodynamic endpoint, platelet aggregation, and the clinical endpoint, cardiovascular adverse events, were followed up at 1 year. Platelet aggregation was higher with than without rabeprazole, although the difference was not statistically significant. No differences were seen in cardiovascular adverse events between groups. No significant interaction was detected between rabeprazole and clopidogrel. 22 When concomitant use of a PPI and clopidogrel is considered, rabeprazole may be safer than other PPIs.
Clinical Efficacy in the Treatment of ARDs
Gastroesophageal reflux disease (GERD)
GERD is a common condition with an increasing prevalence in Western nations. In Japan, the prevalence of GERD has been on the increase since the end of the 1990s. The reasons for the increase in the reported prevalence of GERD may involve increased gastric acid secretion, a decrease in the H. pylori infection rate, increased awareness of GERD, and advances in the concept of GERD. 23 Furthermore, although many GERD patients may present with typical symptoms such as heartburn and acid regurgitation, others may complain mainly of atypical symptoms such as epigastric pain or pressure, nausea/vomiting, hoarseness, chest pain, or wheeze. 24 Symptom assessment, management and resolution remain the primary goals of medical intervention for both patients and physicians. Evaluation of the response of GERD symptoms to treatment, however, has been hampered by the lack of a valid, reliable, highly responsive, and easy to use assessment tool. 25
We conducted a survey of the actual symptoms of Japanese GERD patients. A total of 124 patients with an endoscopic diagnosis of GERD completed a 50 part questionnaire (requiring only ‘yes’ or ‘no’ answers) that covered various symptoms related to the upper gastrointestinal tract, including psychosomatic symptoms. We extracted the 12 questions to which patients most often answered ‘yes’ and produced a multiple choice questionnaire that graded the frequency of each symptom (never = 0, occasionally = 1, sometimes = 2, often = 3, and always = 4), that we named the Frequency Scale for Symptoms of GERD (FSSG, Fig. 2). A significant reduction in the FSSG score occurs in patients with both mild and severe GERD after PPI therapy.
26
The FSSG contains the 12 symptoms most commonly experienced by GERD patients, with 7 of the 12 related to reflux symptoms and the remaining 5 to dyspeptic symptoms. Sixty-eight GERD patients receiving proton pump inhibitor therapy completed the questionnaire before and after treatment for 8 weeks. A significant positive correlation was seen between reflux symptoms and acid-related dyspepsia before and after therapy (r = 0.569 and r = 0.569; both P's < 0.001), and acid-related dyspepsia in patients with both NERD and RE. We concluded that GERD patients suffer not only from reflux symptoms, but also from acid-related dyspepsia, and PPIs improve both types of symptoms.
27
The clinical efficacy of rabeprazole in the management of GERD has been evaluated in terms of symptom relief,
28
33
healing
34
37
and maintenance therapy
34
37
of reflux esophagitis (RE), and in the symptomatic relief of non-erosive reflux disease (NERD)
38
40
or uninvestigated GERD.41,42
The FSSG (Frequency Scale for the Symptoms of GERD) asks questions about the 12 symptoms most commonly experienced by GERD patients, with 7 of the 12 related to reflux symptoms and the remaining 5 to dyspeptic symptoms. Kusano M, et al. J Gastroenterol. 2004 Sep;39(9):888-91; Kusano M, et al. Dig Dis Sci. 2007 Jul;52(7):1673-7. Questions with significant differences in improvement rates between groups for gastroesophageal reflux symptoms (Q1 Black bars) and dysmotility-like symptoms (Q3 Gray bars).

Reflux esophagitis (RE)
Participants in studies evaluating rabeprazole for the acute treatment of erosive or ulcerative GERD28–33,43 were required to be symptomatic, with endoscopically confirmed lesions at (or prior to) study entry. Patients were administered rabeprazole 10 or 20 mg once daily or a comparator PPI or placebo for 1 28 or 429–33,43 weeks, with endoscopic assessment performed at baseline and after 429,30,32 and/or 829,43 weeks of study treatment in all23–27,37 but the week-long trial. 22 Rabeprazole was effective in the acute symptomatic treatment and healing of erosive or ulcerative GERD in well-designed trials of 1 28 or 4-839–34 weeks’ duration. Studies of RE have aimed to identify differences in the speed of symptom relief and mucosal healing. In one study, RE patients were randomized to receive omeprazole 20 mg/day, lansoprazole 30 mg/day or rabeprazole 20 mg/day for 8 weeks. 39 Symptoms were assessed daily for the first 7 days of administration. Whereas there was a decrease in the mean heartburn score with rabeprazole therapy at the initial study interval of 1 week, the 8 week comparison showed no difference in the endoscopic rate of RE healing. Although pharmacokinetic differences are seen with CYP2C19 polymorphism, the CYP2C19 genotype did not affect healing rates at 4 and 8 weeks in 103 Japanese patients with erosive GERD. Oesophageal healing rates in patients identified as homozygous-extensive metabolizers, heterozygous-extensive metabolizers and poor metabolizers after 4 weeks’ treatment with rabeprazole 10 mg were 86.1%, 92.0% and 82.4%, respectively. The corresponding healing rates at 8 weeks were 86.1%, 92.0% and 82.4%, showing no significant differences between genotypes in terms of healing rate at either time point. 43 The study endpoints included time to endoscopically confirmed GERD relapse 37 and frequency of heartburn relapse.34,36 Patient discontinuations during the 5 years were more common in the placebo group than in the rabeprazole groups. Clinical relapse was the primary reason for early treatment termination in 59% of the placebo group, compared with 15% and 7% of the rabeprazole 10 and 20 mg/day groups, respectively. The median time to relapse was 1848 days in the rabeprazole 10 mg/day group (no relapse in the 20 mg/day group) compared with 32 days with placebo (P = 0.0001 for both rabeprazole dosages vs. placebo; P = 0.007 for rabeprazole 20 vs. 10 mg/day). Relapse rates for heartburn frequency were also significantly lower in the rabeprazole than placebo groups.34,36 Heartburn frequency relapse rates differed significantly between rabeprazole and placebo groups at all timepoints during the 5 year study. From weeks 4 to 260, the heartburn frequency relapse rates were 21%-39% in the rabeprazole 20 mg group, 22%-48% in the rabeprazole 10 mg group, and 68%-78% in the placebo group. 36
Non-erosive reflux disease (NERD)
Symptom relief with 4 weeks of rabeprazole treatment in patients with NERD was rapid. 44 We combined data from two similarly designed, double-blind, placebo-controlled, multicenter US trials in which 261 patients were asked to record daily their acid reflux symptoms. The median time to 24 h heartburn-free status was significantly shorter in the rabeprazole group (3.5 versus 19 days in the placebo group), and complete heartburn relief at 4 weeks was 32% in the rabeprazole group vs. 3.8% in the placebo group (P < 0.0002). The median time to the first 24 h interval without heartburn symptoms was significantly shorter in the rabeprazole 10 and 20 mg/day groups (2.5 and 4.5 days) than in the placebo group (21.5 days; P = 0.004) in a 4 week, placebo-controlled trial with patients with NERD. Approximately 30% of patients administered rabeprazole reported complete heartburn relief at week 4 (compared with 3.4% of those administered placebo), and both rabeprazole dosages significantly reduced daytime and nighttime heartburn scores over the first 2 days of treatment (P < 0.01). 40 Symptoms of regurgitation, belching, bloating, nausea and early satiety were also significantly ameliorated with rabeprazole treatment (P < 0.01). 40 Rabeprazole was effective as on-demand, long-term maintenance therapy in patients with NERD or uninvestigated GERD, and was an acceptable alternative to continuous once-daily rabeprazole in patients with NERD.28,42
In another study, Japanese patients with NERD were classified as grade N (endoscopically normal), M (minimal change), or erosive GERD, and answered a 51 item, yes or no questionnaire pre and post-treatment. Compared to erosive GERD, clear differences existed in pretreatment prevalence of symptoms and responsiveness to 4 weeks of rabeprazole 10 mg/day in grades N and M. These results suggest that stomach-aches (especially at night) were significant symptoms in patients with grade N disease, and dysmotility-like symptoms such as abdominal bloating were significant in those with grade M disease, whereas gastroesophageal reflux symptoms were significant in those with erosive GERD. The clinical significance of classifying NERD was indicated from the different symptoms and responsiveness to rabeprazole. 38 Rabeprazole provides early symptom control in patients with NERD, and is therefore appropriate for both continuous and on-demand therapy, as demonstrated in clinical trials. Further trials evaluating on-demand PPI therapy, including rabeprazole, are needed in patients with erosive GERD. 45
Supraesophageal manifestations of gastroesophageal reflux disease
Supraesophageal manifestations of GERD include asthma, cough, laryngopharyngeal reflux (LPR), non-cardiac chest pain (NCCP) and sleep disorders.
Asthma, cough
Sugawa et al established a novel rat model and examined the pathogenic interaction between RE and bronchial asthma. RE and bronchial asthma were induced in Brown-Norway rats by ligating the transitional region between the forestomach and glandular stomach and wrapping the duodenum near the pylorus, and by ovalbumin sensitization and challenge with ovalbumin aerosol. Significant increases in the number of cells, especially eosinophils, and IL-13 but not IFN-gamma concentration, were observed in bronchoalveolar lavage fluid (BALF) in the RE + bronchial asthma group compared with the bronchial asthma group. These augmentations of ovalbumin-induced airway inflammation were prevented by treatment with rabeprazole. 46 Improvement in peak expiratory flow (PEF: a measure of the severity of obstructive airways disease) has been demonstrated in patients suffering from concomitant asthma and GERD. Treatment with rabeprazole 20 mg/day was found to improve PEF by >20% in 40% of non-steroid dependent asthma patients. 47
Laryngopharyngeal reflux (LPR)
LPR has also been reported to be a major cause of laryngeal inflammation, and presents with a constellation of symptoms that differ from those of GERD. 48 Koufman was the first to clearly distinguish LPR from GERD, noting that patients with LPR rarely complain of typical GERD symptoms, such as heartburn and acid reflux. 49 Acid-suppression therapy with a PPI is considered the mainstay of treatment for LPR. In a study with 100 patients with LPR symptoms, upper abdominal and LPR symptoms were evaluated using both the FSSG and the LPR symptom scoring system. In the 52 subjects with LPR, changes in these symptoms with rabeprazole 20 mg/day for at least 4 weeks were evaluated. There were significant reductions in the frequency of acid reflux-related symptoms, dysmotility-like symptoms, and laryngopharyngeal symptoms after rabeprazole therapy. The LPR symptom score decreased to less than half the pretreatment score in 25 subjects (therapeutic response group). The pretreatment frequency of dysmotility-like symptoms appeared higher in the nonresponse group than in the response group, although the difference was not significant. No significant difference was seen between the two groups in the pretreatment frequency of acid reflux-related symptoms. The frequency of dysmotility-like symptoms was similar to that of acid reflux-related symptoms. The pretreatment frequency of dysmotility -like symptoms, but not that of acid reflux-related symptoms, might be a predictor of patient response to rabeprazole. 50
Non-cardiac chest pain (NCCP)
The use of rabeprazole in the empirical diagnosis of GERD-related chest-pain (the so called PPI test) was assessed in a recent double-blind, randomized, placebo-controlled, crossover trial, that demonstrated a sensitivity of 75% and a specificity of 90% for rabeprazole 20 mg bid, administered for 7 days as a therapeutic trial. 51 Kim et al compared the efficacy of one-week and two-week PPI trial in patients with NCCP at least weekly, with the aim of determining the optimal duration for the diagnosis of GERD-related NCCP. There was no significant difference in the rate of positive PPI tests between the GERD-related NCCP group and the non GERD-related NCCP group during the first week of the PPI test. However, during the second week, the GERD-related NCCP group had a higher positive PPI test rate (n = 13, 81%) than the non GERD-related NCCP group (n = 7, 27%) (P = 0.001) with a sensitivity and specificity of 81% and 62%, respectively. The rabeprazole empirical trial was diagnostic for patients with GERD-related NCCP, with an optimal duration of at least two weeks. 52
Sleep disorders
Sleep dysfunction, defined as a PSQI score > 5.5, was detected in 70 (52.2%) of patients with GERD. NERD was significantly associated with sleep dysfunction compared to erosive reflux disease (OR 2.18, 95% CI: 1.05-4.53). Rabeprazole 10 mg/day for 8 weeks led to an effective decrease in sleep dysfunction. Acid plays an important role in sleep dysfunction in all patients with GERD. 53
Peptic ulcer
NSAID use and H. pylori infection are both well recognized as contributors to the aetiology of peptic ulcer disease.
Gastric and duodenal ulcer
The efficacy of rabeprazole in the treatment of peptic ulcer has been evaluated in three randomized, double-blind, multicenter comparisons with omeprazole. 54 56 In two trials comparing rabeprazole 20 mg/day with omeprazole 20 mg/day, there were no significant differences between rabeprazole and omeprazole in terms of symptom resolution based on most of the 12 symptom-related efficacy indices.55,56 However, in one of the studies, significant differences were seen between treatment groups in terms of the proportion of patients with improvement (defined as any decrease from baseline) in gastric ulcer pain frequency after 6 weeks’ treatment (82% for rabeprazole vs. 65% for omeprazole; P = 0.006), day pain severity after 3 weeks (88% vs. 75%; P = 0.023) and resolution of night pain after 6 weeks (84% vs. 68%; P = 0.022). 54
Ando et al 57 demonstrated that the healing efficacy of rabeprazole is less influenced by CYP2C19 genetic polymorphism than that of omeprazole and that this advantage can result in earlier repair of gastric mucosal damage. Eighty patients with active gastric ulcer were administered rabeprazole 10 mg or omeprazole 20 mg once daily for 8 weeks. Reuction in gastric ulcer size and ulcer healing rates were stratified according to CYP2C19 metabolizer subgroup after 2 and 8 weeks of treatment. Percentage ulcer area healing ratios were more consistent across subgroups in the rabeprazole group at 2 weeks (80.7 ± 17.4% vs. 89.3 ± 12.5% vs. 84.3 ± 13.2%, respectively, for homozygous extensive metabolizers, heterozygous extensive metabolizers, and poor metabolizers), than in the omeprazole group (63.4 ± 24.6% vs. 85.2 ± 12.2% vs. 84.0 ± 16.4%, P = 0.0347 rabeprazole vs. omeprazole in homozygous extensive metabolizers), resulting in an overall significant difference (85.9 ± 14.4% vs. 76.5 ± 21.0%, P = 0.0210 rabeprazole vs. omeprazole total healing rate).
Non-steroidal anti-inflammatory drug (NSAID) ulcer
An alternative strategy is to protect the GI mucosa from damage through acid suppression. It is known that maintaining an intragastric pH > 4 heals gastric ulcers, and that in patients taking NSAIDs, the extent of NSAID-associated gastric damage is pH dependent. 58 The level of acid suppression provided by H2RAs at traditional dosages does not prevent most NSAID-related gastric ulcers. Sugimoto et al investigated whether aspirin induces esophageal mucosal injury, and whether rabeprazole can prevent such injury in relation to CYP2C19 genotypes. Fifteen healthy Japanese volunteers underwent a 7 day randomized 5-way crossover trial: placebo, aspirin 100 mg, rabeprazole 10 mg, and aspirin 100 mg plus rabeprazole 10 mg either once daily or 4 times per day. All subjects underwent endoscopy and 24 h intragastric pH monitoring on day 7. With the aspirin regimen, esophageal mucosal damage was seen in 7 subjects (46.7%). The median pH in grade A GERD was significantly lower (1.5) than in patients without GERD (5.6, P = 0.04). Rabeprazole significantly inhibits acid secretion irrespective of CYP2C19 genotype, and decreases the incidence of aspirin-related esophageal injury and symptoms according to the increase in pH. 59
Nishino et al investigated whether aspirin-induced gastric mucosal injury might have any association with the intragastric pH. Fifteen healthy, H. pylori-negative volunteers randomly underwent the four different 7-day regimens: (1) aspirin 100 mg, (2) rabeprazole 10 mg, (3) aspirin 100 mg + rabeprazole 10 mg, and (4) aspirin 100 mg + rabeprazole 40 mg. Gastric mucosal injury was evaluated using the modified Lanza score (MLS), 24 h intragastric pH monitoring, and histopathology of the gastric mucosa, prior to commencement and on day 7 of each regimen. The median MLS was 0 at baseline and for the rabeprazole 10 mg regimen. The median MLS in the aspirin regimen was 3, and 0 for both the aspirin + rabeprazole 10 mg and aspirin + rabeprazole 40 mg regimens. Rabeprazole significantly prevented aspirin-induced gastric mucosal injury (P = 0.001 for rabeprazole 10 mg and P = 0.005 for rabeprazole 40 mg). The MLS negatively correlated with 24 h intragastric pH (r = -0.711, P < 0.001), whereas aspirin had no effect on intragastric pH. Aspirin increased the mean gastric mucosa microvessel diameter, which in turn correlated negatively with intragastric pH. 60 In Japan, Mizokami investigated the efficacy and safety of rabeprazole coadministered with continuous non-steroidal anti-inflammatory drug (NSAID) in treating NSAID-induced ulcer. Upper gastrointestinal endoscopy of patients on continuous NSAID treatment revealed an ulcerous lesion (open ulcer) with diameter ≥3 mm. Endoscopies were performed at the start of treatment, during the treatment period, and at the conclusion (or discontinuation) of treatment. Findings were evaluated in terms of size (maximum diameter) and stage based on the Sakita-Miwa classification. An ulcer was regarded as cured when the ‘white coating’ was seen to have disappeared at endoscopy. In the safety evaluation, all medically untoward symptoms and signs (adverse events, laboratory abnormalities, incidental symptoms, etc.) occurring after the commencement of rabeprazole treatment were considered adverse events. The endoscopic cure rate in 38 patients in the efficacy analysis group (endoscopic evaluation) was 71.1% (27/38). Of those 38 patients, 35 had a gastric ulcer, with a cure rate of 71.4% (25/35), and 3 a duodenal ulcer, with a cure rate of 66.7% (2/3). The treatment efficacy of rabeprazole for NSAID-induced ulcer under continuous NSAID administration was confirmed. 61
Bleeding ulcer
Clinicians have debated for many years concerning the role of antisecretory therapy in the management of gastrointestinal (GI) bleeding from peptic ulcers. The theoretical basis for use of these agents is that (1) gastric acid and pepsin inhibit clot formation and cause clot lysis, and (2) gastric acid impairs ulcer healing. Unfortunately, early studies assessing the efficacy of H2RAs in the management of GI bleeding were disappointing. Many researchers hypothesized that the high failure rate was related to the inability of H2RAs to provide gastric pH values above 6, the level required to achieve hemostasis. 62 64 Uedo et al conducted a prospective randomized controlled trial at a tertiary cancer center, investigating whether PPIs more effectively prevents bleeding than H2RAs following the recently developed technique of endoscopic submucosal dissection (ESD) for early gastric cancer (EGC). A total of 143 patients with EGC who underwent ESD were randomly assigned on the day before ESD to treatment with either rabeprazole 20 mg (PPI group) or cimetidine 800 mg (H2RA group), which was continued for 8 weeks. The primary end point was the incidence of bleeding, defined as hematemesis or melena that decreased the patient's hemoglobin by more than 2 g/dL and required endoscopic hemostasis. Bleeding occurred in 6.1% (4/66) of patients in the PPI group and 17.2% (11/64) of the H2RA group (P = 0.057). Multivariate analysis revealed that PPI treatment significantly reduced the risk of bleeding (adjusted hazard ratio 0.47, 95% confidence intervals 0.22-0.92, P = 0.028). Rabeprazole prevented delayed bleeding from the ulcer created at ESD more effectively than did cimetidine. 65
Zollinger Ellison syndrome
The key to successfully reducing acid output in patients with Zollinger Ellison syndrome (ZES) is to adjust the PPI dosage regimen to meet the individual patient's needs. The dosage of rabeprazole in patients with a pathological hypersecretory condition varies with the individual patient. The recommended adult oral starting dosage for ZES is 60 mg once daily. The dosage should be adjusted to the individual patient's needs, and treatment should continue for as long as clinically indicated. 66
Helicobacter pylori eradication
The benefits of H. pylori eradication in patients with peptic ulcer include more rapid ulcer healing, reduced ulcer recurrence, and a reduction in hemorrhagic complications. 67 Combination therapies with a PPI plus two or three antibacterial agents are well established for this purpose, with the most widely used antibacterial combination being clarithromycin and amoxicillin. Rabeprazole has demonstrated good in vitro antibacterial activity against H. pylori. In an in vitro study using 133 strains of H. pylori isolated from patients with gastric and/or duodenal ulcer, the minimum inhibitory concentration at which 90% of the tested isolates were inhibited (MIC90) was 0.5 mg/mL for rabeprazole and 0.25 mg/mL for the rabeprazole thioether metabolite, whereas the MIC90 values for lansoprazole and omeprazole were 1 mg/mL and 16 mg/mL, respectively. 68 In general, shorter courses (7 days vs. 10-14 days) of H. pylori eradication triple therapy are common in Europe. In the US, lansoprazole and esomeprazole are currently indicated for H. pylori eradication, having demonstrated high eradication rates in combination with amoxicillin + clarithromycin over 10-14 days and 10 days, respectively. However, rabeprazole has recently been approved by the US FDA for H. pylori eradication as part of a 7 day regimen, also in combination with amoxicillin + clarithromycin. H. pylori eradication rates with rabeprazole plus clarithromycin + amoxicillin triple therapy do not differ significantly from those observed with triple therapy with omeprazole,69,70 esomeprazole69,71 or pantoprazole, 69 but are greater than with lansoprazole 72 containing regimens. The reported eradication rates in the rabeprazole + amoxicillin + clarithromycin (RAC) regimen were 88% (130/147) based on intention-to-treat analysis conducted in all subjects, and 91% (130/143) based on per protocol analysis conducted after excluding dropouts. For the lansoprazole + amoxicillin + clarithromycin (LAC) regimen, the eradication rates were 78% (116/148) according to intention-to-treat analysis and 81% (116/144) according to per protocol analysis. Both intention-to-treat (P < 0.05) and per protocol analyses (P < 0.01) showed significantly higher eradication rates for RAC than LAC. 72 It has been suggested that the greater direct antibacterial activity of rabeprazole, its fast onset of action and its predominantly nonenzymatic route of metabolism, and consistent acid inhibition across CYP2C19 genotypes, may result in increased antimicrobial efficacy allowing shorter triple therapy regimens. This warrants further investigation. 73
Functional dyspepsia (FD)
The consensus guidelines recommend PPIs for the treatment of FD. 74 Empiric PPI treatment would be expected to provide symptom relief to the majority of dyspepsia sufferers who present in clinical practice. PPIs represent the best currently available therapy for ARDs and should be considered the first-line management approach in patients with uninvestigated dyspepsia. 75 In healthy volunteers, rabeprazole 20 mg bid did not significantly alter gastric emptying, but reduced symptoms and had a borderline effect on postprandial gastric volume. The mechanism of reduction of postprandial symptoms by rabeprazole requires further study. 76 In another study, 97 Japanese patients diagnosed with FD according to the Rome III criteria were randomized to either rabeprazole 10 mg once daily (N = 46) or itopride 50 mg tid (N = 51) for 4 weeks. As reported in Western patients, faster onset of action was seen with rabeprazole compared to itopride in Japanese FD patients. For epigastric pain syndrome (EPS), rabeprazole showed faster onset and greater improvement than Itopride. However, for postprandial distress syndrome (PDS), itopride showed faster onset of action but not significantly more symptomatic improvement, than rabeprazole.77,78 Tominaga et al conducted the CAESAR study (Clinical Assessment to Establish the Symptomatic Advantage of Rabeprazole: a double-blind randomized placebo-controlled study) to examine the efficacy of rabeprazole in improving the symptoms of FD. They randomly assigned 98 subjects with FD, diagnosed according to the Rome III criteria, to 4 weeks of either rabeprazole 10 mg/day or placebo. The severity of gastrointestinal symptoms was assessed using the Gastrointestinal Symptoms Rating Scale (GSRS) at baseline and at weekly intervals during the treatment period. The primary endpoint was change in scores for each item of the GSRS following treatment. Four weeks’ treatment with rabeprazole decreased reflux domain scores (2.5 ± 0.2 to 1.5 ± 0.1) compared to placebo (2.5 ± 0.2 to 1.9 ± 0.1, P = 0.0232). Significant differences were seen in the changes from baseline in the regurgitation and nausea scores between rabeprazole and placebo. Rabeprazole is clinically beneficial in ameliorating FD symptoms in Japanese patients, in particular for overlapping regurgitation and nausea symptoms. 79
Summary and Conclusion
The physicochemical and pharmacokinetic profiles of PPIs can influence their safety and efficacy in clinical practice. Rapid symptom relief is a key feature of therapy for patients with ARDs. Rabeprazole is a useful option for the treatment of ARDs, and provides a valid alternative to the other currently available PPIs, with the added benefits of having a favorable efficacy profile and low incidence of drug interactions.
Footnotes
Acknowledgements
The authors would like to express their gratitude to Ryoichi Nishiki, B.S., of Eisai Co., Ltd. for his kind provision of data related to rabeprazole.
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.
