Abstract
Unsedated upper gastrointestinal endoscopy (UGE) can induce patient discomfort, mainly due to a strong gag reflex. The aim was to assess the effect of a bupivacaine lozenge as topical pharyngeal anesthetic compared with standard treatment with a lidocaine spray before UGE. Ninety-nine adult outpatients undergoing unsedated diagnostic UGE were randomized to receive either a bupivacaine lozenge (L-group, n = 51) or lidocaine spray (S-group, n = 42). Primary objective was assessment of patient discomfort including acceptance of the gag reflex during UGE. The L-group assessed the discomfort significantly lower on a visual analog scale compared with the S-group (P = 0.02). There was also a significant difference in the four-point scale assessment of the gag reflex (P = 0.03). It was evaluated as acceptable by 49% in the L-group compared with 31% in the S-group. A bupivacaine lozenge compared with a lidocaine spray proved to be a superior option as topical pharyngeal anesthetic before an UGE.
Introduction
Upper gastrointestinal endoscopy (UGE) is a diagnostic procedure commonly performed worldwide.1–3 Unfortunately, some patients experience discomfort while undergoing UGE, mostly due to a strong gag reflex. 4 To increase patient tolerance, UGE is generally performed using topical pharyngeal anesthesia and/or sedation.1,5 Studies have shown that unsedated UGEs are well tolerated, and local anesthetics are frequently used to anesthetize the oral cavity and pharynx to reduce the gag reflex during UGE.1,6,7 Currently, different formulations of lidocaine are used as local pharyngeal anesthesia and are administrated as a gel, inhaler, or spray. 8 Several studies have found the lidocaine pharyngeal anesthesia to be beneficial.9–11 In a study by our research group Mogensen et al, 12 a lidocaine lozenge was found to be superior to lidocaine solution. In order to ease administration, improve the lozenge, and increase the time of anesthesia during UGE, a smaller lozenge with taste masking was formulated and bupivacaine was chosen as the active pharmaceutical ingredient (API). Bupivacaine is an amide like lidocaine, but has a longer anesthetic effect and is four times as potent as lidocaine. 13
The aim of this study was therefore to assess the effect of a new topical, pharyngeal, anesthetic treatment with a bupivacaine lozenge compared with standard anesthetic treatment with lidocaine spray (Xylocaine®) before UGE with respect to discomfort and patient acceptance.
Methods
The study was approved by the Regional Ethics Committee, Copenhagen, Denmark, and the Danish Medicine Agency. The study was registered at the Danish Data Protection Agency and ClinicalTrials.gov (NCT01832675) and was performed in accordance with the Declaration of Helsinki. The Good Clinical Practice unit of the Copenhagen University Hospitals monitored the data throughout the study.
Study medicine
A lozenge with a weight of 800 mg (12-mm diameter) containing 25 mg of bupivacaine was manufactured by direct compression at the Pharmacy of the Capital Region, Denmark. The lozenge consisted of bupivacaine hydrochloride as API, aspartame as a sweetening agent, magnesium stearate with talc as glidans, and perlitol SD 200 as filler and binder. The bitter taste of bupivacaine was disguised by liquorice powder.
Xylocaine® 10% Pump Spray (AstraZeneca, Södertälje, Sweden), consists of 10 mg of lidocaine per puff with banana essence, ethanol, macrogol, menthol, and saccharin.
Patients
This study was conducted from March 2012 to August 2012 at a private clinic in Hvidovre, Denmark. Consecutive patients undergoing an unsedated UGE were eligible to participate. The inclusion criteria were an age between 18 and 80 years; ability to speak, read, and understand Danish; and the ability to provide oral and written consent. Exclusion criteria were known allergy to bupivacaine or other local anesthetics of the amide type, pregnancy, breastfeeding, or use of other medication than the study drug before UGE. Informed consent was obtained from all participants.
Study design
The study was nonblinded and was conducted as a randomized study comparing a bupivacaine lozenge with standard treatment. A research assistant carried out the randomization by opening a sealed opaque envelope. The patients received either one lozenge containing 25 mg of bupivacaine 10-15 minutes before the UGE or three puffs of the lidocaine spray equivalent to 30 mg of lidocaine just before the UGE. The lozenge was to be sucked until it was completely dissolved (takes approximately 10-15 minutes), whereas the solution from the spray was applied to the pharynx. Two experienced endoscopists performed the UGE procedures. The first endoscopist examined patients 1-38 and the second endoscopist examined patients 39-99.
Patient assessments
Immediately after the UGE, the research assistant gave the patients a questionnaire, where the patients assessed the discomfort experienced during the procedure on a visual analog scale (VAS) from 0 to 100 mm, where 0 was no discomfort and 100 was the worst discomfort imaginable, and on a four-point verbal rating scale, where 1 = no discomfort; 2 = acceptable discomfort; 3 = moderate discomfort; and 4 = unacceptable discomfort. The patient acceptance of the gag reflex was assessed on a four-point verbal rating scale, where 1 = no gag reflex; 2 = acceptable gag reflex; 3 = moderate gag reflex; and 4 = unacceptable gag reflex. The patients’ perception of taste of the local anesthetics was evaluated on a three-point verbal rating scale, where 1 = good taste; 2 = moderate taste; and 3 = bad taste.
Statistical analysis
The sample size was determined with the following data; a minimum relevant clinical difference of 15 mm on a VAS of discomfort, with a standard deviation of 25, at a significance level of 0.05 and a power of 80%. This led to a sample size of 45 patients in each of the two groups, and 99 patients were included to account for dropouts. The bupivacaine lozenge (L-group) versus the lidocaine spray (S-group) was compared for the continuous and normally distributed residuals using a t-test. The normality has been investigated with Q-Q plots. The Kruskal–Wallis test was used for continuous nonnormally distributed response variables to compare the two groups. For categorical response variables, a χ 2 test was applied. In cases with an expected less than five in each cell, we used an exact test instead. In the post hoc analysis, multiple linear regression was used to adjust the effect of groups for endoscopist on patient assessment on VAS discomfort. All statistical analyses were performed with SAS statistical software version 9.1 (SAS Institute Inc., Cary, NC, USA).
Results
A total of 145 consecutive patients scheduled for an UGE were screened. Eighteen patients did not fulfill the inclusion criteria, and 28 patients refused the invitation to participate in the study. Five patients were inconveniently included twice, but only the first inclusion was included in the analysis. None were excluded due to the exclusion criteria. In total, 94 patients were enrolled and randomized; 51 patients were randomized to the L-group and 43 patients to the S-group. One patient from the S-group dropped out because of unacceptable gagging. A total of 51 patients in the L-group and 42 patients in the S-group completed the study (see Fig. 1).
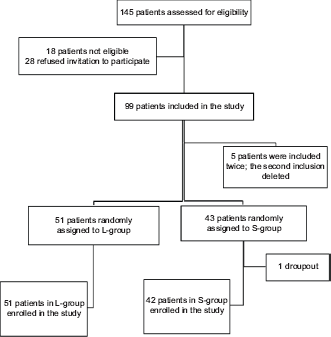
Flow chart showing the patient allocation. Lozenge group (L-group) and spray group (S-group).
The demographic data for the two treatment groups were similar with regard to age, gender, and body mass index (BMI) (see Table 1).
Demographic data and number of previous UGE. Lozenge group (L-group) and spray group (S-group).

Patient assessment
The patients in the L-group experienced significantly less discomfort when assessed using VAS compared to the S-group (t-test: 33.7 vs. 45.3, P = 0.02, SD (L-group) = 21.3, SD (S-group) = 26.8) (see Fig. 2).
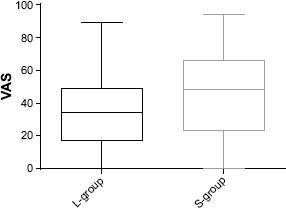
Patient assessment of discomfort during UGE using VAS showed in a box-plot with maximum and minimum values, upper and lower quartiles, and median (P = 0.02). L-group median (IQR): 34.0 (17.0, 49.0), S-group median (IQR): 48.5 (23.0, 66.0). Lozenge group (L-group) and spray group (S-group).
There was a significant difference in the four-point scale assessment of discomfort (exact test: P = 0.002). It was evaluated as acceptable by 60% in the L-group compared with 31% in the S-group (see Fig. 3A). There was also a significant difference in the four-point scale assessment of the gag reflex (exact test: P = 0.03). It was evaluated as acceptable by 49% in the L-group compared with 31% in the S-group (see Fig. 3B). The three-point scale assessment for taste of the anesthetics was significantly different (χ2 test: P < 0.0001), and 62% in the L-group evaluated it as good compared with 12% in the S-group (see Fig. 3C).

(
Post hoc analysis of the patient assessment of discomfort on VAS showed that when adjusting for the endoscopist who had examined the patient, no difference was found on the effect of the groups compared to the univariate analysis (univariate β = -11.5, P = 0.02; adjusted β = -12.6, P = 0.01). None of the patients experienced any side effects. An unpublished pilot study with 10 healthy subjects showed no signs of pulmonary aspiration after administration of a 25 mg bupivacaine lozenge (Mogensen et al, in preparation).
Discussion
Topical pharyngeal anesthetics have been widely used for several years as premedication to UGE procedures in many countries,1–3 as it reduces the patient discomfort and improves the patient tolerance.1,2,8,14 As pharyngeal anesthesia, a lidocaine oral solution or pharyngeal spray is often administered.2,14 However, these formulations are swallowed immediately after administration, which leads to a minimal pharyngeal mucosal contact time, and therefore a reduced effectiveness of the local anesthesia. 14 Furthermore, these formulations leave an unpleasant bitter taste in the mouth and pharynx, due to poor taste masking of lidocaine. 2 The development of new formulations focus on the patient comfort and therapeutic benefits. 15 To improve oromucosal drug delivery, new dosage forms have been developed such as lozenges or lollipops.2,8,15 The use of a bupivacaine lozenge as topical anesthetic before UGE is a novel clinical approach.
The use of a lozenge or lollipop as topical anesthesia before UGE has been evaluated in a few randomized studies. A study determined that topical anesthesia administrated as benzocaine/tyrothricin lozenges with conscious sedation had no advantages over conscious sedation alone. 2 However, there are several disadvantages associated with sedation. The patient has to remain at the hospital until recovery is satisfactory and must be escorted home and is unable to work. This increases the cost as it requires more staff pre- and postprocedure. 16 A placebo-controlled trial evaluated the effectiveness of a lidocaine spray (60 mg of lidocaine) compared with a lidocaine lozenge (20 mg of lidocaine) in unsedated patients. 17 The spray was found to be superior to the lozenge, with regards to patient tolerance score, a greater intensity of numbness, less gag reflex, and less discomfort. However, the lozenge was found to have a better taste. The superiority of the spray may well be due to the difference in total dosage of lidocaine administrated in the two groups. A single-blinded, randomized study evaluated the effect of a lidocaine lollipop (300 mg of lidocaine) compared with lidocaine spray (300 mg of lidocaine). 8 The study showed a superior effect of the lollipop, as the patients had less gag reflex, better tolerance for the procedure, and required less sedation. The lollipop had a good taste and was well tolerated by the majority of patients. 8 A newer study by our research group Mogensen et al 12 evaluated the effect and acceptance of a lidocaine lozenge (100 mg of lidocaine) compared with a lidocaine viscous oral solution (100 mg of lidocaine). The lozenge significantly improved the patient's acceptance of the discomfort, the gag reflex, and the UGE. Furthermore, the lidocaine lozenge as topical pharyngeal anesthesia seemed to be both effective and palatable.12,18
The variations in methods, design, API, dose, dosage form, and route of administration limits the generalizability of the studies. As some of the studies included conscious sedated patients, this can introduce bias due to impaired judgment following sedation. 12 To avoid this bias, the present study solely included unsedated patients. Despite the use of conscious sedation, similar results to our study were achieved by Ayoub et al 8 and Mogensen et al, 12 showing a superior effect for the lidocaine lollipop and the lidocaine lozenge. In our study the bupivacaine lozenge decreased the discomfort the patients experienced during an UGE compared with the lidocaine spray. Furthermore, it led to a higher patient acceptance of the gag reflex, and the lozenge had a significantly better taste than the lidocaine spray.
The superior effect of the lozenge may be attributed to the slow release of the anesthetic. As the lozenge dissolves, it is mixed with saliva, which increases the distribution of the anesthetic thus improving its performance. In contrast to this, when administrating the spray the contact time with the pharynx is comparatively short, as the patient swallows the spray solution quickly after administration. 8
The discomfort associated with UGE procedures is primarily due to a strong gag reflex. 7 These results are supported by our findings as the patients receiving a pharyngeal anesthesia experienced lower discomfort and increased acceptance of the gag reflex. As comfort is an important predictor of patient acceptance, the results from our study indicate that the bupivacaine lozenge can improve the patients’ acceptance of the UGE.
Drug delivery systems such as lozenges and solutions are easy to use, but an unpleasant taste can decrease the patient acceptance. 19 It has been suggested that the patients’ taste perception of the API could influence the tolerance and satisfaction of the UGE. 19 The present study showed that the lozenge had a significantly better taste than the spray formulation. A positive evaluation of the bupivacaine lozenge's taste was expected, as its taste was masked in the same way as the 100 mg lidocaine lozenge in the study by Mogensen et al.12,18 Results from our study indicate that the bitter taste of bupivacaine can be disguised with a liquorice flavor.
This study design does have some limitations. The UGE was performed by two experienced endoscopists. The results may have been more consistent if only one endoscopist had performed the procedure. The post hoc analysis did not show any bias. Furthermore, our study was not blinded. Blinding of the patients was not feasible as we compared a new treatment with, in regard to the patients, a well-known standard treatment. Indeed, approximately 40% of the patients in each group had previously undergone UGE. Blinding of the endoscopists would also be difficult as the lozenge gives the tongue a distinct brown color caused by the liquorice powder.
In conclusion, a bupivacaine lozenge compared with a lidocaine spray proved to be an improved treatment option as topical pharyngeal anesthesia before an UGE owing to reduced patient discomfort, a higher acceptance of the gag reflex, and improved taste. These results suggest that the lozenge can be an option to alleviate the patients’ discomfort during UGE. A large double-blinded study including a placebo lozenge and a placebo spray including objective measures (e.g. the duration of gagging during the UGE) is warranted to verify the results from this study.
Author Contributions
All authors contributed to the trial design. NS included patients and collected data. CT, SM and JJ supervised the project. NS, CT and JP analyzed the data. NS wrote the first manuscript draft and CT finalized the manuscript. SM, MR, JP, OA and JJ edited the manuscript. All authors reviewed and approved the final manuscript.
Footnotes
Acknowledgments
We are grateful to Kaare Nielsen, MD, and André Wettergren, MD, for performing the UGEs and thank the nursing staff at Surgical Clinic Hvidovre for enthusiastic cooperation. The study was registered at ClinicalTrials.gov (NCT01832675).
