Abstract
The utilization of artificial intelligence (AI) in gastrointestinal (GI) endoscopy has witnessed significant progress and promising results in recent years worldwide. From 2019 to 2023, the European Society of Gastrointestinal Endoscopy has released multiple guidelines/consensus with recommendations on integrating AI for detecting and classifying lesions in practical endoscopy. In Vietnam, since 2019, several preliminary studies have been conducted to develop AI algorithms for GI endoscopy, focusing on lesion detection. These studies have yielded high accuracy results ranging from 86% to 92%. For upper GI endoscopy, ongoing research directions comprise image quality assessment, detection of anatomical landmarks, simulating image-enhanced endoscopy, and semi-automated tools supporting the delineation of GI lesions on endoscopic images. For lower GI endoscopy, most studies focus on developing AI algorithms for colorectal polyps’ detection and classification based on the risk of malignancy. In conclusion, the application of AI in this field represents a promising research direction, presenting challenges and opportunities for real-world implementation within the Vietnamese healthcare context.
Introduction
Gastrointestinal (GI) diseases are associated with high morbidity, mortality, and economic burden, with the total expenditure for GI-related conditions reaching up to $119.6 billion. 1 According to GLOBOCAN 2020, GI cancers, including those of the esophagus, stomach, and colon, rank among the top 10 malignant diseases in terms of both prevalence and mortality rates. 2 However, if detected at an early stage, the 5-year survival rate of patients can be as high as 80%–90%.3,4
Endoscopy is essential for diagnosing, screening, and treating various GI diseases, especially GI cancers.5 –7 However, precancerous or high-risk lesions can be missed during endoscopy. For example, the missed rate of gastric cancer is reported to be 9.4%. 8 A meta-analysis of 45 studies involving 15,000 colonoscopies reported miss rates of 26% for adenomas, 9% for advanced adenomas, and 27% for serrated polyps. 9 Therefore, technological solutions are needed to narrow the experience gap between endoscopists and limit medical errors.
Artificial intelligence (AI) has been increasingly applied to various fields of medicine, including endoscopy. 10 Studies on AI applications in endoscopy have shown that computer-aided endoscopy has good accuracy in diagnosing and classifying GI lesions such as colorectal polyps, Barrett’s esophagus, gastric cancer, and esophageal cancers.11,12 The accuracy is markedly high on still images and videos. In 2022, The European Gastrointestinal Endoscopy Society (ESGE) published scopes for different applications of AI in endoscopy, including detecting and treating neoplastic lesions, diagnosing atrophy, dysplasia, or precancerous conditions. ESGE also emphasized that an AI system can be used in real practice if it achieves a threshold equivalent to experienced endoscopists. 13
In Vietnam, GI cancers also pose a significant health burden, with colorectal and gastric cancers ranking fourth and fifth, respectively, with over 16,000 new cases in 2022.2,14 Additionally, many factors may lead to an increased miss rate of GI lesions in Vietnam: overcrowding—the number of daily procedures in major endoscopy centers can be up to 300 cases—and variations in the quality of endoscopy systems across different healthcare facilities. 15 A survey conducted among healthcare professionals specializing in internal medicine, gastroenterology, and GI endoscopy showed that 98% of respondents expressed their need for AI support applications during endoscopy, with a preference for automated lesion detection and delineation. 15 These findings underscore the necessity of developing big data and implementing AI applications in the field of endoscopy in Vietnam.
Overview of AI in gastrointestinal endoscopy worldwide
AI refers to computer systems that perform tasks normally associated with human intelligence. One branch of AI, Machine Learning (ML), focuses on developing algorithms capable of making predictions from large datasets (“Big Data”). Deep learning (DL), a further specialization of ML, utilizes deep neural network structures to replicate the functionality of the human brain. This technology enables the interpretation of large and complex datasets, especially image data. In the endoscopy field, DL is often used in analyzing endoscopic images to create algorithms for detecting and classifying lesions. 16
Building databases
The development of effective AI algorithms for detecting anatomical landmarks and lesions in GI endoscopy depends on the quality of the training dataset. The requirements for the dataset include a large number of images with diversity in aspects including anatomical landmarks; lesions shapes, types, and sizes 17 ; light modes (white light and images-enhanced modes); image quality (brightness and resolution); and endoscopy systems.18 –20 Experience in the implementation of AI shows that ensuring a standard resolution (ideally more than 512 × 512 pixels for optimal performance) will facilitate the training of high-quality algorithms for clinical deployment.18,19,21 Furthermore, the images in the dataset need to be annotated, labeled, and validated by endoscopy experts. These steps are vital to ensure the accuracy and reliability of the dataset, which in turn directly impacts the performance of the AI algorithms developed from it.17,22,23
Currently, numerous GI endoscopic databases have been published. Most of these databases are open-access, rendering them valuable resources for algorithm training and development. A recent study has summarized the existence of 40 published still image databases, with 34 being open access. Of these, 16 databases focus on polyps, while six focus on small intestinal lesions. 24
For the upper GI tract, published endoscopic image datasets encompass various lesion types and anatomical landmarks.25 –27 These databases mainly consist of still images with varying characteristics for cleanliness, light modes, image quality, and resolutions. However, certain databases still exhibit limitations, such as partially obscured images due to endoscopic instruments, low image resolution, and a lack of lesion annotation and labeling.26,28,29
As for the lower GI tract, researchers focus on building image and video datasets of colorectal polyps classified and labeled by experts.30 –35 However, these datasets are limited in diversity and polyp size and lack images/videos captured in enhanced light mode. Some datasets have relatively low image resolutions ranging from 288 × 384 pixels to 480 × 409 pixels.32,35,36
AI algorithms developed in gastrointestinal endoscopy
According to the ESGE 2022 guidelines, AI algorithms in GI endoscopy include automated detection of anatomical landmarks, quality assessment of endoscopy, and detection and classification of GI lesions. 13
For the upper GI tract, the research direction of AI focuses on the detection of anatomical landmarks, enhancement of image quality, and detection of precancerous or early-stage lesions. 37 First, algorithms for detecting anatomical landmarks of upper GI endoscopy play a vital role in lesion identification, particularly in regions such as the pylorus, Z-line, and cecum. 38 Some algorithms categorized the anatomical landmarks of upper GI endoscopy into seven to eleven main zones according to guidelines issued by the World Endoscopy Organization.39 –41 Moreover, researchers have further divided the stomach into 26 zones to reduce blind spots during endoscopy. 42 Notably, the accuracy of these AI algorithms consistently exceeds 90%.38 –42 Several clinical trials have shown that AI-integrated endoscopy systems have significantly lower rates of blind spots (ranging from 2.8 to 5.5%) than conventional endoscopy (ranging from 3.92 to 22.46%).42 –44 Second, enhancing image quality is crucial in effective AI training. Techniques such as flipping, cropping, resizing, and blurring existing images have been used to diversify and enrich input datasets for AI model optimization. 20 Third, for lesion detection, researchers have shown that AI has high accuracy in detecting gastric neoplastic lesions, Barrett’s esophagus, esophageal squamous cell carcinoma, chronic atrophic gastritis, H. pylori infection, and gastric cancer. While most studies were conducted with still images,37,45 –47 clinical trials have been conducted to assess the application of AI in real-world scenarios. In two clinical trials evaluating AI application in the upper GI, tract, the rates of missed neoplasm gastric lesions (6.1%) and cancer lesions (1.7%) in the AI group were significantly lower than the conventional endoscopy group (27.3% and 6.7%, respectively).48,49
For the lower GI tract, the principal research directions of AI are centered on cleanliness assessment, detection, and characterization of colonic adenomas/polyps. Studies on using AI to support the assessment of bowel preparation and mucosal cleanliness have reported accuracy ranging from 85% to 95%, with no significant difference between the assessment of endoscopy experts and AI.50 –52 In clinical trials, AI-supported colonoscopy achieved a lower adenoma miss rate and a higher adenomas/polyps detection rate than the conventional group, especially for small, flat lesions.53 –58 Regarding polyp classification, a marked increasing number of clinical trials provide evidence of AI’s advantages in detecting small lesions (<5 mm).59,60 Several AI systems (such as CAD-EYE, EndoBrain, and GI Genius) have been commercialized in the market. 61 (Table 1)
Parameters for evaluating some AI algorithms in GI endoscopy.

AUC, Area Under the Receiver Operating Curve; BLI, Blue Light Imaging; GI, gastrointestinal; LCI, Linked Color Imaging; ME, Magnification endoscopy; NBI, Narrow Band Imaging; WLI, White Light Imaging.
AI research in endoscopy in Vietnam
Since 2019, studies regarding AI applications in the field of GI endoscopy in Vietnam have been conducted using various methodologies. The primary focuses have been developing comprehensive databases of morphology and light modes and constructing AI algorithms for lesion detection using these databases.
Building databases for AI in Vietnam
In Vietnam, the development of image databases for AI training in endoscopy remains limited. To our knowledge, three databases have been established. Two of these databases are data collected from Vietnamese subjects, while the remaining one is a database that employs AI algorithms to annotate lesions within pre-existing datasets.
In 2019, Hoang Trung Hieu and colleagues utilized AI algorithms to annotate features such as polyps, dyed polyps, dyed-resection polyps, and instruments for 4470 unclassified images from the KVASIR and BioMedia 2019 Development datasets. This database has been made publicly accessible and available at https://endoscopy.selab.hcmus.edu.vn (Figure 1). 65 However, this study did not provide details on the role and contribution of expert knowledge in the review and approval of the AI-generated labels, which may potentially affect the accuracy of the labeled images.

Endoscopy images of colorectal polyps with AI-generated labels. 65
To establish an endoscopic image database specifically for Vietnam, serving as educational materials for endoscopy and a foundation for AI training, Dao Viet Hang and colleagues have built two databases of endoscopy images for upper and lower GI tracts since 2020.
For lower GI endoscopy, the database comprises 8190 colonoscopy images with polyps and 4000 images without polyps in white light and flexible spectral imaging color enhancement (FICE) modes. The polyps in these images are delineated and labeled by endoscopists with >5 years of experience. 66 A portion of the dataset has been published with 1200 polyp images classified and labeled for neoplastic and non-neoplastic lesions, namely BKAI-IGH NeoPolyp. 67 (Figure 2)

The BKAI-IGH NeoPolyp dataset and classification labels. 67
For upper GI endoscopy, in 2022, Dao Viet Hang’s research team developed a database of 8207 endoscopy images in four different light modes, including white light imaging and three image-enhanced endoscopy (IEE) modes: FICE, Blue Light Imaging (BLI), and Linked-Colored Imaging. Among these, 5546 images are upper GI images without lesions of 10 anatomical landmarks (from the pharynx to the duodenum). The remaining images have at least one of five important upper GI lesions including erosive esophagitis, esophageal cancer, gastric cancer, gastritis, and duodenal ulcers (Figure 3). 68 All images with lesions have been classified, delineated, and labeled by endoscopy experts. The distinguishing features of these upper and lower GI datasets,66,68 when compared to existing datasets worldwide, are the addition of advanced IEE modes (BLI and LCI) and the expert delineation and labeling of lesions.
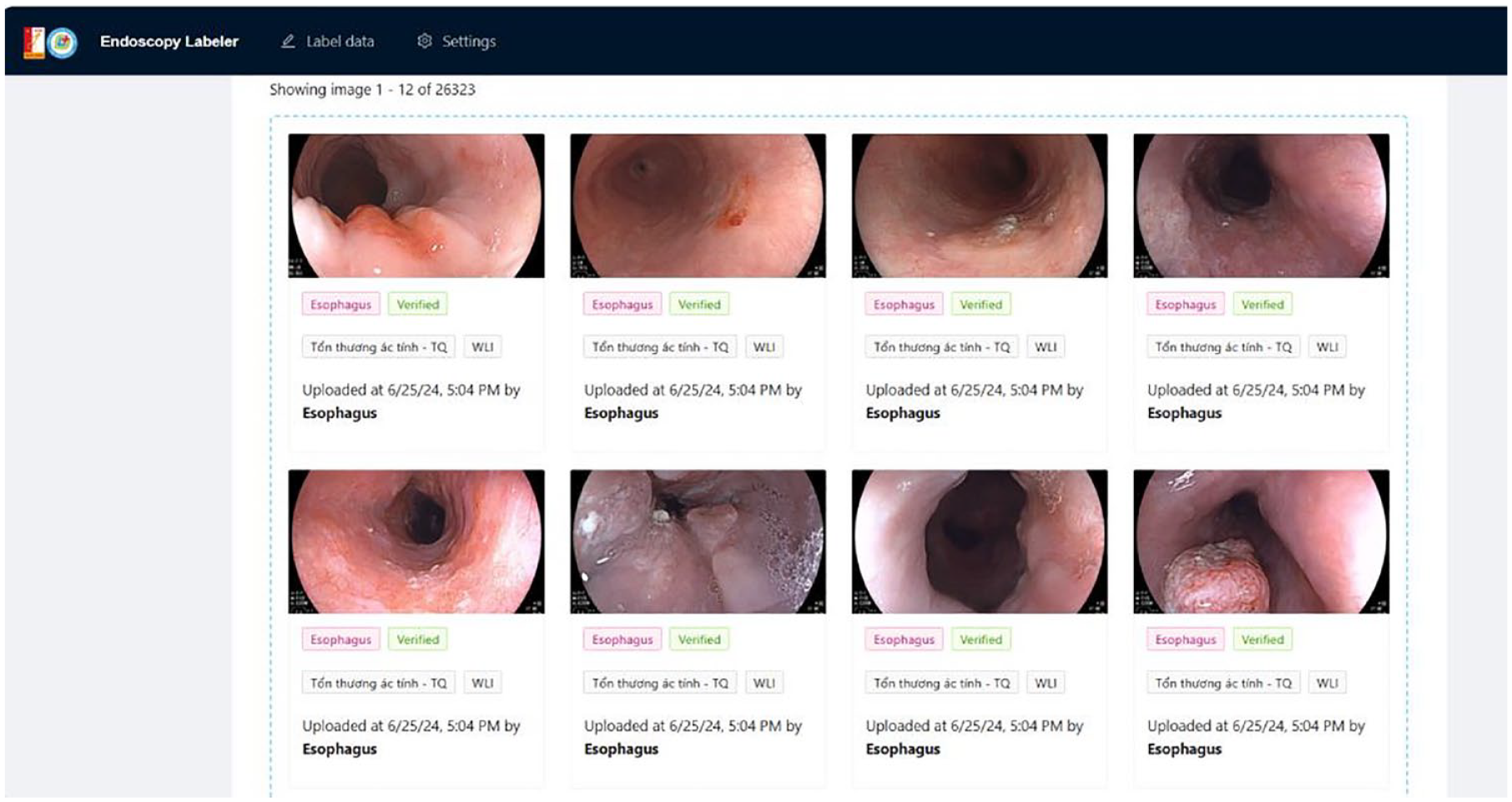
The upper GI endoscopy image database storage system has been labeled.
Initial research of developing AI algorithms in gastrointestinal endoscopy
Researchers in Vietnam have so far implemented studies to develop AI algorithms to improve image quality, identify anatomical locations, detect and delineate lesions, and classify lesions on endoscopy.
Upper GI endoscopy
Bubbles, mucus, or residual food debris in the GI tract are common issues during endoscopy, as these factors can obscure lesions and hinder observation and lesion identification. An algorithm has been developed to assess gastric cleanliness by delineating bubbles during endoscopy. The accuracy of this algorithm was 90% on still images. 69
To enhance the algorithm training quality and save resources, AI algorithms for simulating IEE modes have been tested and shown promising results. To evaluate the similarity between the images in authentic IEE modes and simulated-IEE modes, we mixed these two types of images and asked the endoscopists to detect which IEE image was simulated. The detection rates of new (<5 years of experience) and experienced endoscopists (⩾5 years of experience) were relatively low (54.7% and 52.7%, respectively). 70
Identifying anatomical landmarks is a critical task when implementing ML systems to support endoscopy procedures. For anatomical landmark identification, the accuracy of the algorithms developed in Vietnam for detecting 10 anatomical locations from the pharynx to the duodenum is very high, ranging from 97.0% to 98.6% when run on different datasets and network systems.68,71,72 This algorithm has met the minimum required for anatomical landmarks detection during GI endoscopy procedures, similar to the ESGE guidelines. 73 Algorithms detecting anatomical landmarks in upper GI endoscopy have been widely developed and clinically tested on patients with promising results (accuracy > 90%). Some algorithms have also been enhanced with a feature that detects blind spots.38 –42 Compared to the algorithms developed in Vietnam, the algorithm by Qi He et al. has similar accuracy in anatomical landmarks detection. The WISENSE algorithm classifies and detects blind spots in upper GI endoscopy with a more detailed dataset, up to 26 separate regions of the stomach. However, the dataset lacks data in other positions such as the esophagus and the duodenum. 42 Other algorithms detect fewer anatomical landmarks. The algorithm by Cogan et al. was designed to detect three basic locations (pylorus, Z-line, and cecum). The algorithm of Takiyama et al. detected seven locations (larynx, esophagus, stomach, duodenum, etc.) while the study by Seong Ji Choi et al. detected eight locations (proximal esophagus; Z-line; stomach cardia and fundus on retroflexed view; stomach body; stomach angle; stomach antrum; duodenal bulb; the second part of duodenum).39,40 In the future, researchers in Vietnam may refine algorithms for detecting anatomical landmarks by integrating additional features such as blind spot detection.
In 2022, the EndoUnet algorithm, equipped with features for lesion classification, delineation, and labeling, was developed using the upper GI dataset by Hang et al. The overall accuracy of the algorithm was 98.6%. For each of the five important upper GI lesions, the accuracy was 95.1% for erosive esophagitis, 96.9% for esophageal cancer, 88.1% for gastric cancer, 96.9% for gastritis, and 81.9%) for duodenal ulcer. 74 For the lesion delineating and labeling task, test results on three convolutional neural networks including ResNet-50, DenseNet-121, and VGG-19 showed high accuracy. The two lesions with the most accurate delineation were gastric cancer and esophageal cancer (Dice scores of 87% and 83%, respectively). 68 Specifically for erosive esophagitis, the author team of Dao Viet Hang and Tran Thanh Hai developed lesion delineation algorithms based on differences in the color and structure of the lesions and the surrounding normal mucosa. The delineation performance of the algorithm needs improvement for erosive esophagitis grades A and M due to the similar characteristics between these lesions and surrounding mucosa.75,76 Factors related to missed lesions include small lesion sizes and poor mucosal cleanliness. Images with poor cleanliness and those diagnosed with gastric metaplasia or Barrett’s esophagus increase the false-positive rate. 77 Study results showed that algorithms developed in Vietnam had similar accuracy to previous studies in detecting upper GI lesions (Table 2).37,45 –47 However, the datasets and algorithms in Vietnam still need to be improved in the detection of other lesions such as Barrett’s esophagus, esophageal neoplasia, and gastric neoplasia, especially lesions of small sizes and at early stages. Besides lesion detection, developing algorithms to classify lesions according to international standards is a potential direction to comprehensively support endoscopists.
Some results of AI applications in GI endoscopy in Vietnam.

BLI, Blue Light Imaging; DICE, DICE Score/DICE Similarity Coefficient; FICE, Flexible spectral Imaging Color Enhancement; IoU, Intersection over Union; LCI, Linked Color Imaging; px, pixels; Se, Sensitivity; Sp, Specificity; WLI, White Light Imaging.
Lower GI endoscopy
Based on a database of colorectal polyps delineated and labeled by experts, Dao Viet Hang and colleagues developed the EfficientNet algorithm for detecting colorectal polyps. When tested on still images, initial results showed the algorithm’s sensitivity and positive predictive value to be 96.4% and 94.6%, respectively. 66 This algorithm continued to be evaluated for accuracy on a different dataset of still images, including 2000 images with polyps and 2000 without polyps. Trong Hieu et al. used the Kvasir-SEG dataset and developed an algorithm for detecting and delineating colorectal polyps that achieved high accuracy when identifying polyps, with Dice and IoU scores of 88.2% and 81.6%, respectively. 66 For polyp delineation and classification, the accuracy of the AI algorithm using the BlazeNeo and NeoUnet networks on non-neoplastic polyps were 71.7% and 72.0%, respectively. As for neoplastic polyps, the accuracy of BlazeNeo and NeoUnet were 88.5% and 88.9%. When compared to algorithms using other deep learning networks worldwide, the accuracy of these two algorithms was superior, with a true delineation rate of 90.4% and 91.1%. The processing speed of the algorithm using the BlazeNeo network was slower than that of the NeoUnet network and other deep learning networks.81,82
To enhance the accuracy of polyp delineation, researchers have adopted various approaches. The methods include using data from WLI images to train polyp segmentation models for BLI images or consistently updating the AI algorithm while collecting images for the dataset.85,88,89 Some algorithms and deep learning networks have been developed and improved, such as AG-CUResNeSt, MetaFormer (M2UNet), ConvTransNet, and ColonFormer, to improve the accuracy of polyp delineation and describing polyp characteristics.83,86,87,91,90 These algorithms and deep learning networks have all demonstrated high accuracy when tested on different polyp datasets.
Besides initial studies with algorithms based on Vietnamese patient databases, Dao Viet Hang et al. conducted a clinical trial on an AI-integrated support system for detecting colorectal polyps developed by the University of Hong Kong. The study participants underwent tandem endoscopy to evaluate the adenoma/polyp detection and missed rates. The research results showed that endoscopy with AI support increased the adenoma detection rate but did not reduce the miss rate compared to the traditional group. 92
Almost all studies on developing AI algorithms for endoscopy in Vietnam are currently at the stages of development, calibration, and evaluation on still-image datasets. To be commercialized, an AI algorithm must undergo a comprehensive evaluation of performance and accuracy in real-time settings using endoscopy video datasets. 23 After integration into a device, the algorithm must be examined in clinical trials to compare its effectiveness against novice and expert endoscopists.
Challenges in applying AI in endoscopy
Datasets
The most crucial aspect of developing an AI model with good performance is building a large-scale, high-quality dataset of endoscopy images, annotated and labeled by expert endoscopists experts. 20 However, to guarantee the diversity of images, endoscopic images in Vietnam are collected from multiple centers across the country, resulting in several challenges when constructing a comprehensive dataset from multiple healthcare facilities in Vietnam, such as including inconsistency in image quality, difficulties in data extraction, and significant resources required for data cleaning and labeling. 15
First, the healthcare facilities in Vietnam are not uniform in their data storage processes. Most healthcare workers face difficulties in retrieving endoscopy results. The data storage system in Vietnam is still limited, which does not allow for bulk extraction of images and endoscopy results. 15 Additionally, implementing data collection at multiple healthcare facilities requires strict adherence to patient confidentiality policies and approvals from ethical committees and data management authorities.93 –95 If the data from healthcare facilities is accessible to multiple individuals and organizations, it can increase the risk of privacy and security breaches. Therefore, researchers must establish robust and transparent management systems and procedures to ensure privacy and data security.96,97 Developing a large dataset for scientific research and AI development research, it is necessary to establish and clarify clear regulations and policies on data security and sharing among collaborating units, and to create an open database among healthcare facilities. Technical mechanisms for sharing, such as cloud servers and access granting, also need to be established.
Secondly, focusing on the data cleaning and labeling process is crucial to developing a high-quality input dataset for training AI algorithms. Image quality can be influenced by various factors, including doctor expertise, available endoscopic systems (generation, type of endoscopes, light modes, resolutions, etc.), level of inflation, cleanliness, artifacts, capture angles, and experiences of endoscopists, etc.18,98 Therefore, the image dataset must be thoroughly cleaned to maintain patient confidentiality and meet quality standards before proceeding to the labeling process. Another common issue is the potential for bias due to the heterogeneity in physicians’ perspectives, which can lead to incorrect classification and annotation during the labeling stage.20,21 Furthermore, manual labeling is also time-consuming for doctors. Therefore, developing and sharing standardized annotation tools and common reference standards with clinicians before labeling is important to ensure data quality and save time.20,99,100 However, establishing common standards may also lead to “overfitting” on the training when the dataset is used for training, resulting in poor performance on validation datasets or actual patients. 99 To our knowledge, the criteria to collect endoscopic datasets for AI training in Vietnam to ensure diversity include images with both high and low resolution, diverse in lesions (types and morphology), and light modes (WLI and enhance-light mode) (Table 2). To ensure the quality of the input dataset, some studies also collect images that met cleanliness criteria (according to commonly used scales in endoscopy such as BBPS 101 and MVS 102 ) and annotations reviewed and verified by experts before training77,81,80 (Supplemental 1).
Resources
Developing AI systems in endoscopy requires highly skilled AI engineers with the knowledge and practical experience in the latest technology advancements. The application of AI in healthcare, particularly in endoscopy, necessitates close collaboration between IT and healthcare professionals to optimize performance. 103 Vietnam currently has 140 IT training programs, including 57 courses related to AI applications. However, the number of IT professionals and experts in medical technology and big data development remains limited due to inadequate incentives.103,104
To date, several products have been developed to support real-time lesion detection and characterization in endoscopy, such as Fujifilm’s CADEYE, Olympus’s EndoBrain, and Medtronic’s GI Genius. However, these products require the most advanced endoscopy systems, high resolution input, and high cost.105,106 While this is a major barrier to the application of AI in healthcare in Vietnam, especially in low-resource centers, it also presents opportunities for researchers in Vietnam to develop AI systems that can be integrated into endoscopic systems with various specifications and image qualities, so that AI technologies can be available to most healthcare facilities.
The body of evidence on AI in healthcare in Vietnam is growing. Most studies were funded by commercial organizations, academic institutions, and international grants, and only a few were funded by national grants. 104 In recent years, the Vietnamese government has begun to prioritize AI development and its applications across various sectors through key national projects. The expansion of international partnerships has offered Vietnamese researchers valuable opportunities to align their works with global AI advancements. 107
Legal and ethical policies
Despite a relatively young field of science, AI application and digitalization in healthcare have been rapidly growing, prompting the establishment of policies and regulations. In other countries, guidelines for digitalization and AI application in clinical practice have been developed in the general context and specific areas of healthcare.108,109 In Vietnam, the Prime Minister has issued the “National Strategy for Research, Development, and Application of Artificial Intelligence until 2030” to promote AI research, development, and application. In 2020, the Ministry of Health approved the “Digital Transformation Program in Healthcare until 2025” with a vision to establish a smart healthcare system.107,110 Despite these efforts, Vietnam currently lacks official documentation providing guidelines for the development, evaluation, and implementation of AI algorithms in healthcare. The World Endoscopy Organization has identified challenges in deploying AI-integrated medical devices in clinical practice. These include insufficient data on clinical benefits and cost-effectiveness, lack of reliable guidelines, unclear indications, AI usage costs, and training requirements. 111 Additionally, the integration of AI in clinical settings raises ethical concerns that demand careful consideration, such as potential harm, legal responsibilities, bias in clinical decisions, impacts on doctor-patient relationships, and workforce implications. According to a recent survey in Vietnam, only 59,3% of endoscopists showed high acceptance of AI-integrated endoscopy systems. 112 Endoscopy experts in Asia have also emphasized the need for guidelines on conducting research and applying AI in the field of endoscopy.113,114
Conclusion and recommendation
Although still in the early stages of applying AI in endoscopy, Vietnamese researchers have applied already developed algorithms and have also started developing Vietnamese databases and algorithms. Some notable accomplishments include building endoscopic image databases that meet the qualified standards and diversity (lesions, light mode, resolution, etc.), and labeled and delineated by endoscopy experts. Algorithms for identifying, detecting, and classifying characteristic lesions of the GI tract have also been developed, showing high sensitivity, specificity, and accuracy with potential for commercialization in endoscopy. This progress presents an opportunity to establish a strong network and collaboration among multiple medical sites, as well as between medical and IT sectors. However, the algorithm development process faces many challenges, such as the lack of rich input database algorithms developed and tested mainly on still image sets with a limited variety of training images. Clear regulations on data security and sharing between collaborating institutions are crucial for the development of large datasets for AI development. Additionally, the government needs to establish a legal framework with detailed guidelines on regulations for developing and applying AI in clinical practice, ensuring adherence to medical ethics principles and protecting patient rights. Addressing these challenges could significantly influence the policymaking decisions regarding the integration of AI in the medical field in Vietnam. For researchers, the next approach is to design clinical trials appropriate to the national healthcare system to evaluate the effectiveness of the newly developed AI algorithms; evaluate the cost-effectiveness, and acceptance of patients, physicians, and endoscopists; and explore the potential of the application of AI in endoscopy training.
Supplemental Material
sj-docx-1-cmg-10.1177_26317745241306562 – Supplemental material for Application of artificial intelligence in gastrointestinal endoscopy in Vietnam: a narrative review
Supplemental material, sj-docx-1-cmg-10.1177_26317745241306562 for Application of artificial intelligence in gastrointestinal endoscopy in Vietnam: a narrative review by Hang Viet Dao, Binh Phuc Nguyen, Tung Thanh Nguyen, Hoa Ngoc Lam, Trang Thi Huyen Nguyen, Thao Thi Dang, Long Bao Hoang, Hung Quang Le and Long Van Dao in Therapeutic Advances in Gastrointestinal Endoscopy
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
