Abstract
Various autoantibodies are seen in idiopathic inflammatory myopathies. Among myositis-specific antibodies, anti-aminoacyl-tRNA synthetase and anti-melanoma differentiation-associated protein 5 (MDA5) antibodies are associated with interstitial lung disease (ILD). Anti-MDA5 antibodies are associated with dermatomyositis (DM) or clinically amyopathic DM complicated with rapidly progressive ILD. In anti-MDA5-positive patients, a random ground-glass attenuation pattern is a characteristic finding of ILD in chest high-resolution computed tomography. Conversely, anti-aminoacyl-tRNA synthetase antibodies are not associated with rapidly progressive ILD but with chronic ILD. DM or clinically amyopathic DM patients with anti-MDA5, and characteristic high-resolution computed tomography findings are highly likely to have devastating ILD and need aggressive treatment.
Keywords
Introduction
The term idiopathic inflammatory myopathy (IIM) appeared in Bohan and Peter's 1 review in 1975. They classified IIM into the following five subgroups: polymyositis (PM), dermatomyositis (DM), cancer-associated myositis (CAM), juvenile DM, and overlap syndrome (OS); these terms are not identical to the original names given by Bohan and Peter because they were subsequently modified by Euwer and Sontheimer. 2 Because various autoantibodies have been detected, IIM is considered an autoimmune disease whose main target organs are muscles and skin. In 1991, Dalakas 3 suggested incorporating inclusion body myositis (IBM) in the IIM group. However, it is still controversial whether IBM is an autoimmune disease. Pluk et al. 4 reported anti-cytosolic 5′-nucleotidase 1A antibodies found in 33% of sporadic IBM patients. However, recently, Herbert et al. 5 reported that anti-cytosolic 5′-nucleotidase 1A antibodies are detected in other autoimmune diseases and are not specific to IBM. In 1993, Euwer and Sontheimer 2 suggested adding amyopathic DM (ADM) to the IIM group. ADM lacks muscle symptoms but is characterized by a pathognomonic skin rash of DM and is considered an established category associated with DM.
Approximately 50% of IIM patients are complicated with interstitial lung disease (ILD), and having ILD is related to poor prognosis. In this review, several biomarkers and autoantibodies of ILD complicated with IIM are introduced, and their application to diagnosis and treatments is discussed.
Autoantibodies in IIM
Autoantibodies detected in IIM are divided into two categories, myositis-specific antibodies (MSA) and myositis-associated antibodies (MAA). MSA and MAA are associated with specific symptoms and clinical features (Table 1).6–8 The association between MSA and symptoms was reported by Love et al. 9 They investigated the association between MSA and clinical features in 212 adult IIM patients and found that antiaminoacyl-tRNA synthetase (ARS) antibodies are associated with anti-synthetase syndrome, while anti-signal recognition particle (SRP) antibodies are associated with refractory PM, and anti-Mi-2 antibodies are associated with a certain skin rash of DM. They suggested a new approach to classify IIM using MSA. The concept of classification of IIM using MSA has been supported by the discoveries of anti-melanoma differentiation-associated protein 5 (MDA5) antibodies associated with ADM 10 and anti-transcription intermediary factor 1-γ/a (TIF1-γ/a) antibodies associated with CAM. 11 When we consider the classification of IIM using MSA, it is notable that MSA are mutually exclusive and generally do not overlap. In the analysis of IIM patients positive for MSA by Love et al. 9 , each patient had a single type of MSA.
Autoantibodies detected in idiopathic inflammatory myopathies.
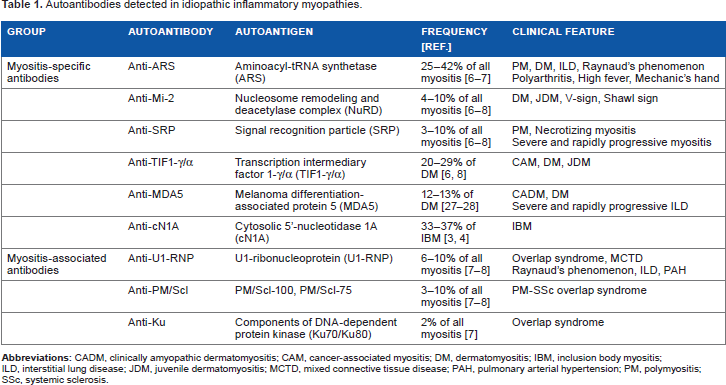
Distinct from MSA, MAA are not specific to IIM patients. Anti-U1-RNP antibodies are major MAA and detected in patients with systemic lupus erythematosus (SLE), systemic sclerosis (SSc), PM, and mixed connective tissue disease. Anti-PM/Scl antibodies 12 are also seen in various connective tissue diseases, such as PM-SSc OS, PM, DM, SSc, SLE, and undifferentiated connective tissue disease (UCTD). In our analysis of six anti-Ku-positive patients, two had PM-SLE OS, two had SLE, one had PM, and one had UCTD. Taken together, MAA can be seen in patients with myopathy complicated with other connective tissue diseases.
Biomarkers Associated with ILD in IIM
Serum Lactate Dehydrogenase, KL-6, and Surfactant Protein D
Serum lactate dehydrogenase (LDH), KL-6, and serum surfactant protein D (SP-D) reflect lung damage and are universal activity markers of ILD and can also be biomarkers of ILD complicated with IIM. Serum LDH is a classical activity marker of ILD, although it is less specific for lung lesions than serum KL-6 and SP-D. KL-6 is a mucinlike high molecular weight glycoprotein expressed in type II alveolar pneumocytes and bronchiolar epithelial cells. Fathi et al. 13 reported that the serum KL-6 level was significantly higher in PM/DM patients with ILD than in PM/DM patients without ILD (sensitivity: 83% and specificity: 100%) and serum KL-6 level reflected pulmonary functional reserve in PM/DM patients with ILD. Ihn et al. 14 reported that serum SP-D level was also significantly higher in PM/DM patients with ILD than in PM/DM patients without ILD and the serum SP-D level was inversely correlated with percent vital capacity and percent diffusing capacity of the lungs for carbon monoxide.
Serum Ferritin and Interleukin-18
Gono et al reported that the serum ferritin level was elevated in anti-MDA5-positive clinically ADM (CADM) or DM patients 15 and correlated with severity (A-aDO2 and P/F ratio), 15 prognosis, 15 and disease activity of ILD in anti-MDA5-positive CADM or DM patients. In addition, serum interleukin-18 level was significantly correlated with response to therapy and prognosis of ILD in those patients. 16 These findings imply that macrophage activation plays an important role in the pathophysiology of ILD in anti-MDA5-positive patients.
Autoantibodies Associated with ILD in IIM
Anti-ARs Antibodies
Anti-ARS antibodies are seen in 25%-42% of all myositis patients6,7 and are also detected in patients with IIM or idiopathic interstitial pneumonia (IIP). 17 ARS is essential when messenger RNA is translated to amino acids. The definition of anti-ARS antibodies includes not only their binding ability to ARS but also their blocking ability against the function of ARS.18–21 There have been reports of anti-Jo-1, 18 anti-PL-7, 19 anti-PL-12, 20 anti-EJ, 21 anti-OJ, 21 and anti-KS antibodies, which correspond to histidyl-, threonyl-, alanyl-, glycyl-, isoleucyl-, and asparaginyl-tRNA synthetases, respectively. Love et al. 9 described five common symptoms, namely ILD, Raynaud's phenomenon, polyarthritis, high fever, and mechanic's hand, seen in anti-ARS-positive IIM patients and named them as anti-synthetase syndrome.
Anti-ARS antibodies may have stronger connections with ILD than with myopathy. A significant association of anti-ARS antibodies with ILD-preceding IIM patients has been reported (Fig. 1). 22 Moreover, anti-ARS antibodies have been found in ILD patients without myopathy.17,22 Watanabe et al. 23 examined the histopathology of lungs sampled from eight anti-ARS-positive ILD-alone patients. Three patients showed cellular nonspecific interstitial pneumonia, three patients showed fibrous nonspecific interstitial pneumonia, and two patients showed usual interstitial pneumonia with cellular infiltration. 23 They also reported that chest highresolution computed tomography (HRCT) revealed traction bronchiectasis, ground-glass opacity, lower reticulation, and subpleural sparing (Fig. 2), while honeycombing was not observed. 23 Additionally, shrinking lung is also a common finding of chest X-ray in anti-ARS-positive ILD patients.
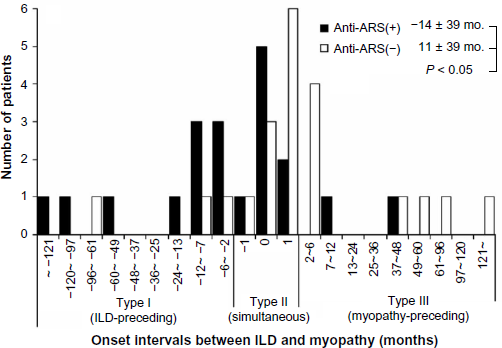
Onset intervals between ILD and myopathy in anti-ARS-positive and anti-ARS-negative patients.

Chest HRCT image of an anti-ARS-positive patient.
ILD complicated by anti-ARS-positivity responds to immunosuppressive therapy but tends to show a recurrent clinical course. ILD in anti-ARS-positive patients showed a more favorable response to glucocorticoid therapy than in anti-ARS-negative patients, 22 and a recurrent course of ILD was significantly more frequent in the anti-ARS-positive patients than in the anti-ARS-negative patients. 22 These results imply that anti-ARS antibodies can be markers for clinical course prediction of ILD complicated with IIM. Indication of immunosuppressants, such as oral tacrolimus and intravenous cyclophosphamide, should be considered in patients with recurrent ILD complicated with IIM.
Hamaguchi et al. 24 investigated the difference in clinical features among patients positive for each kind of anti-ARS antibodies. ILD-alone cases were frequent in patients with anti-PL-12, anti-OJ, and anti-KS, while myopathy and rash were frequent in patients with anti-Jo-1, anti-PL-7, and anti-EJ. Polyarthritis is more strongly associated with anti-Jo-1 than with non-Jo-1 anti-ARS antibodies.24,25 Aggarwal et al. 25 reported that five-year survival was lower in patients with non-Jo-1 anti-ARS antibodies than in patients with anti-Jo-1 antibodies.
Recently, we established a new ELISA system (MESA-CUP™ Anti-ARS Test; MBL Co., Ltd.) that covers anti-Jo-1, anti-PL-7, anti-PL-12, anti-EJ, and anti-KS antibodies simultaneously, 17 and it is used daily in Japan. To diagnose IIM or idiopathic interstitial pneumonia, its sensitivity is 22.7% (95/418), and its specificity is 97.7% (299/306), using a cut-off level of 25 U/mL. The limitation of the system is that it cannot identify the kind of anti-ARS antibodies or anti-OJ.
Anti-MDA5 Antibodies
Sontheimer 26 enlarged the category of ADM and referred the entity of CADM that includes hypomyopathic DM with typical rashes and subclinical myopathy. The study did not mention patients with rapidly progressive and fatal ILD and required a two-year observation with no evident myopathy to diagnose CADM. However, several cases of CADM complicated with devastating ILD have been reported from Asia and also from the USA 27 and Europe. 28 Those patients were refractory to immunosuppressive therapy leading to a fatal clinical course, and histopathology of the autopsied lungs showed diffuse alveolar damage. In 2005, Sato et al found anti-140 kDa antibodies in Japanese CADM patients, 29 and finally, MDA5 was identified as its target antigen. 10
Anti-MDA5 antibodies are detected in CADM or DM patients.10,15,27,28,30 ILD is seen in 50%-100% of the anti-MDA5-positive CADM or DM patients.10,27,28,30 Tanizawa et al. 31 reported that the findings of chest HRCT were dominated by (i) lower consolidation or ground-glass attenuation pattern and (ii) a random ground-glass attenuation pattern (Fig. 3). Mortality ranged from 36% to 46% in anti-MDA5-positive CADM or DM patients.10,15 If an anti-MDA5-positive patient shows even slowly progressive ILD, the most intensive immunosuppressive regimen should be considered to prevent ILD from advancing to a devastating status. Importantly, Teruya et al. 32 reported that an anti-MDA5-positive ADM patient with rapidly progressive ILD was successfully treated with polymyxin-B direct hemoperfusion.
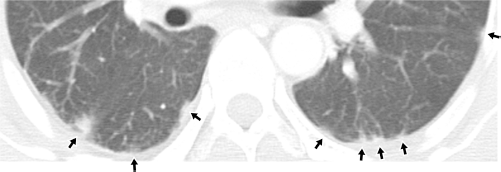
Chest HRCT image of an anti-MDA5-positive patient.
The titer of anti-MDA5 antibodies reflects the severity of ILD and skin ulcerations. 30 Cao et al. 30 reported that the titer of anti-MDA5 antibodies reflected the HRCT score of lung legions, and the titer was decreased after treatment.
Anti-U1-RNP Antibodies
Anti-U1-RNP antibodies are found in major MAA associated with OS or mixed connective tissue disease and are associated with both myopathy and ILD. 33 The frequency ranges from 6% to 10% in all myositis.7,8 In general, ILD progresses chronically in anti-U1-RNP-positive patients. Glucocorticoids are effective in treating ILD in anti-U1-RNP-positive patients. 34
Anti-PM/Scl Antibodies
The prognosis of ILD in anti-PM/Scl-positive patients cannot be determined currently because of the limited number of studies published. However, Muro et al. 35 reported that 4 of the 11 patients positive for anti-PM/Scl antibodies (two DM, one CADM, and one UCTD) had ILD. All of them required immunosuppressive therapy, although they did not show fatal outcomes.
Anti-Ku Antibodies
The clinical course of ILD in anti-Ku-positive patients is also uncertain because the number of reports is limited. Rigolet et al. 36 reported that 11 of the 30 anti-Ku-positive patients (mostly with IIM) had ILD. Eight of the 11 anti-Ku-positive patients with ILD received immunosuppressive therapy; however, their ILD was resistant to the therapy in six of the eight patients.
Application of Biomarkers and Autoantibodies to Treatments
According to the profiles of autoantibodies, ILD complicated with IIM can be divided into three groups: high-, moderate-, and low-risk groups.
High-Risk Group: Anti-MDA5 Antibodies
Decisions for Treatment
In the patients with anti-MDA5 antibodies, treatment should begin before respiratory symptoms or impaired pulmonary function develops. When an anti-MDA5-positive patient shows characteristic chest HRCT findings and elevated biomarkers, such as serum LDH, KL-6, SP-D, and ferritin, the most aggressive immunosuppressive treatments should commence.
Follow-up
Symptoms, oxygenation status, image findings, serum LDH, KL-6, and SP-D will be measures of therapeutic effects. Serum ferritin, interleukin-18, and the titer of anti-MDA5 antibodies may reflect the activity of ILD in anti-MDA5-positive patients.
Moderate-Risk Group: Anti-ARS, Anti-U1-RNP, Anti-PM/SCL, and Anti-Ku Antibodies
Decisions for Treatment
Respiratory symptoms, speed of reduction in pulmonary function, and speed of expansion of chest HRCT findings should be taken into consideration to determine when to start immunosuppressive treatments.
Follow-up
Symptoms, pulmonary function, image findings, and universal activity markers of ILD (serum LDH, KL-6, and SP-D) are helpful to evaluate the potential outcomes of treatment. Anti-ARS-positive patients occasionally show recurrent ILD; therefore, careful attention is needed to detect any disease recurrence. Immunosuppressants should be considered in patients with recurrent ILD complicated with IIM.
Low-Risk Group: Anti-Mi-2, Anti-SRP, and Anti-TIF1-γ/α Antibodies
Decisions for Treatment
Treatment should follow the same course as the moderate-risk group. However, because ILD is less common in patients with anti-Mi-2, 37 anti-SRP, 38 or anti-TIF1-γ/α antibodies, 39 careful consideration is needed before treating ILD in these patients. Possible overlap with other conditions including infectious or drug-induced pneumonia should be examined.
Conclusion
Serum LDH, KL-6, and SP-D reflect lung damage and are the activity markers of ILD complicated with IIM. Anti-ARS and anti-MDA5 antibodies are associated with ILD, while anti-Mi-2, SRP, and TIF1-γ/α antibodies are less so. Anti-MDA5 antibodies are associated with rapidly progressive ILD, while anti-ARS antibodies are not associated with rapidly progressive ILD but with a recurrent course of ILD. DM or CADM patients with anti-MDA5 and characteristic chest HRCT findings are highly likely to have devastating ILD and require aggressive treatment.
Author Contributions
Conceived the concepts: HY. Analyzed the data: HY. Agree with manuscript results and conclusions: HY. Developed the structure and arguments for the paper: HY. Made critical revisions: HY. The author reviewed and approved of the final manuscript.
