Abstract
In order to capture the extent of exposure to polycyclic aromatic hydrocarbons (PAHs), various biomarkers have been employed. The biomarkers employed for PAHs include PAHs genetoxic end points in lymphocytes, urinary metabolites, PAH-DNA adducts, and PAH-Protein adducts. Of these, excretory 1-hydroxypyene, a metabolite of pyrene, has been used extensively as a biological monitoring indicator of exposure to PAHs. This study attempts to assess the level of this biomarker in the body fluid of 68 exposed subjects using high performance liquid chromatography HPLC. The subjects screened included auto mechanics, drivers, and fuel attendants. 1-hydroxypyrene was extracted from the urine of the subjects using solid phase extraction method. The HPLC analysis was done in isocratic mode using water:methanol (12:88 v/v) mobile phase. The stationary phase was XBridge C18 (150 × 4.6 mm) 5 μm column. The wavelength was 250 nm at a flow rate of 1.2 mL/min. The oven temperature was 30 °C and the injection volume was 20 μL. The run time was 3 minutes. The level of urinary 1-hydroxypyrene detected varied for the different categories of occupation studied. About 27% of sampled fuel attendants and 22% of auto mechanics had detectable 1-hydroxypyrene in their urine samples. There was no detectable 1-hydroxypyene in the urine samples of commercial drivers or in the urine samples of students used as controls. The results of this study showed that fuel attendants and auto mechanics have significant exposures to PAHs. So far, there is no established benchmark for level of PAHs in urine, but our findings indicate the possibility of future cancer cases in this population as a result of their occupational exposure. The study was not able to link the level of 1-hydroxypyene with the smoking habits of the subjects.
Introduction
Cancer is a group of diseases characterized by irrepressible growth and spread of abnormal cells that can infiltrate other parts of the body mainly through the blood and lymphatic systems. Cancer kills more people than AIDS, tuberculosis, and malaria combined; thus, it is the world's single leading cause of death.1,2 Cancer accounted for 13% of all deaths in 2007, 14% in 2008, and about 25% in 2009. 3 The trend of cancer occurrence is shifting rapidly from First World countries to Third World countries. In First World countries, some 50% of cancer patients die of the disease, while in Third World countries, 80% of cancer victims already have late-stage incurable tumors when they are diagnosed.4,5 About 72% of all cancer deaths in 2007 occurred in low- and middle-income countries. 3
Cancer arises as a result of the interaction between a person's genetic factors and three categories of external agents, namely, physical, chemical, and biological carcinogens. Physical carcinogens include ultraviolet and ionizing radiation. Chemical carcinogens include heavy metals, dioxns, polycyclic aromatic hydrocarbons (PAHs), aflatoxin, and others. Biological carcinogens include infections from certain viruses, bacteria, or parasites. Among the chemical carcinogens, PAHs are of special importance. PAHs are the largest class of chemical compounds known to be cancer-causing agents and were ranked the ninth most threatening chemical compounds to human health in 2001.6,7 Many of these compounds are genotoxic, mutagenic, teratogenic and carcinogenic. They tend to bioaccumulate in the soft tissues of living organisms.8,9 Various studies have focused on the carcinogenicity of PAHs.10–14 Despite the overwhelming literature on the carcinogenicity of PAHs, avoidable exposure to these compounds is on the rise, especially in Third World countries. With the unsophisticated health care system in these countries, most deaths that occur as a result of certain occupational exposure to PAHs may not be captured, and this informs the need to capture the level of PAH exposure in order to extrapolate the carcinogenic and other health effects of PAHs.
In order to capture the extent of exposure to PAHs, various biomarkers have been employed. A biomarker is a measurable biochemical, physiological, behavioral, or other alteration within an organism that can be recognized as associated with an established or possible health impairment or disease. The biomarkers employed for PAHs include PAHs gene-toxic end points in lymphocytes, urinary metabolites, PAH-DNA adducts, and PAH-protein adducts.15–17 Of all these, excretory 1-hydroxypyene (Fig. 1), a metabolite of pyrene, has been used extensively as a biological monitoring indicator of exposure to PAHs. 18
The chemical structure of 1-hydroxypyene.
1-hydroxypyene is preferred for various reasons. It is the principal product of pyrene metabolism, representing 90% of its metabolites. It has been found to be a good short-term measure of exposure to PAHs. The half-lives of 1-hydroxypyrene reported in literature range from 6 to 35 hours.19–22 On average, the half-life of 1-hydroxypyrene is about 18 to 20 hours; hence, urinary 1-hydroxypyrene represents the last 24 hours of cumulative PAH exposure. Pyrene is the only known precursor of this metabolite, and it forms a consistent proportion of higher molecular weight PAHs in the environment. Also, 1-hydroxypyrene is found to be clearly elevated in occupational settings with high PAH exposure.21,23,24 PAH exposure occurs as a mixture of compounds, and pyrene is almost always found in this mixture in reasonably high proportion. Urinary 1-hydroypyrene (1-OHPY) has strongly been linked with an increased risk of cancer.
PAHs are highly stable and have multiple sources but prominent among them are petroleum-related sources. Many people are occupationally exposed to PAHs. This study attempts to assess the level of this biomarker in the body fluid of 68 exposed subjects using high performance liquid chromatography (HPLC).
Methods
Materials
All the chemical and reagents were of highest purity possible. HPLC grade methanol, acetonitrile, and water were obtained from Sigma-Aldrich, Germany. Other reagents used included 1-hydroxypyrene standard (from Sigma-Aldrich, Germany), β-glucuronidase enzyme (from Sigma-Aldrich, Germany), creatinine picric acid reagent, and creatinine buffer (10 mM sodium borate, 240 mM sodium hydroxide and surfactant). For accurate measurement, a Metier Toledo weighing balance was employed. Also used in the study were a 100 mg CI8, 96- well plate, an oven, a UV spectrophotometer, and Agilent 1100 HPLC.
Sample collection
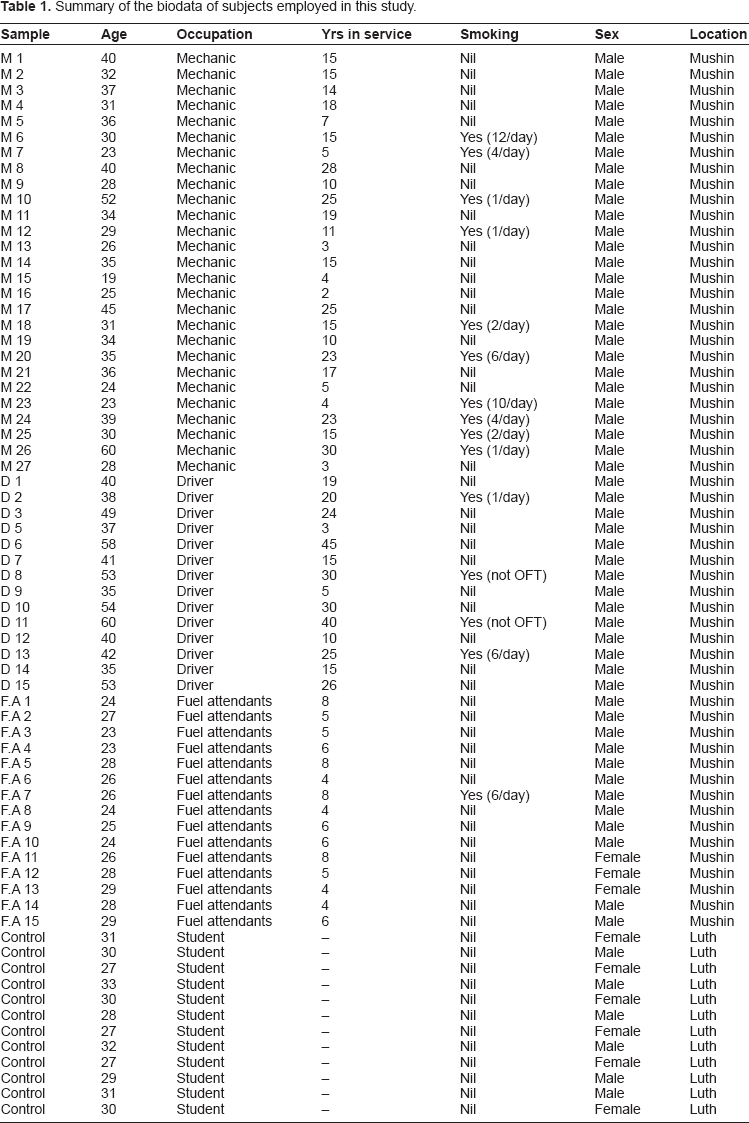
From the subjects, 24-hoururine samples were collected. Table 1 shows the different groups and characteristics of the subjects involved in this study. The urine samples were collected in a 3 L specimen collection jar with volumes ranging from 1450 mL to 2600 mL.
Summary of the biodata of subjects employed in this study.
Extraction efficiency of the solid phase extraction (SPE) sorbent used
Prior to the extraction of 1-hydroxypyrene from urine samples, the extraction efficiency of C18 SPE cartridge with different solvents was determined. The different solvents used included methanol, acetonitrile, and a mixture of both.
Preparation of β-glucuronidase
Dissolved in 68.00 mL of distilled water was 36.8 mg of β-glucuronidase enzyme. Following this, 1 mL (2000 units of β-glucuronidase) was added to each of the 68 samples, and these were incubated overnight.
Preparation of creatinine working reagent for the UV analysis of urinary creatinine concentration
To constitute the working reagent, 40 mL each of creatinine picric acid reagent and creatinine buffer (10 mM sodium borate, 240 mM sodium hydroxide and surfactant) reagent were properly mixed. The spectrophotometer was zeroed with the reagent blank at 510 nm. Added to 0.5 mL of each sample was 1 mL of the working reagent. The samples were shaken and immediately transferred into the cuvette at 37 °C and the absorbance recorded.
Sample preparation for urinary 1-hydroxypyene
Measured into 68 clean test-tubes was 1 mL of each of the urine samples. Added to each of the samples was 1 mL of beta Glucoronidase (2000 units) in order to hydrolyze the conjugated 1-OHPY. The resulting mixtures were incubated overnight (for about 15 hours) at 38 °C. The incubated samples were thoroughly shaken to remix the precipitate with the clear solution making them homogenous solutions. The C18 SPE cartridges were activated with 2 mL of methanol to condition and open up the SPE pores. The cartridges were washed with 2 mL of water to allow for easy passage of liquid denser than methanol through the SPE pores. Next, 1 mL of the homogenized incubated urine samples were transferred to the SPE sorbents. The sorbents were washed with 2 mL of water to remove water-soluble compounds from the sample matrix. The SPE cartridges were then desorbed with 2 mL of methanol. The eluates were made up to 2 mL each with the eluting solvent.
Sample analysis
HPLC analysis was carried out on an Agilent 1100 HPLC system. The mobile phase was water:methanol (12:88 v/v). The stationary phase was an XBridge C18 (150 × 4.6 mm) 5 μm column. The wavelength was 250 nm at a flow rate of 1.2 mL/min. The temperature of the column was 30 °C. The injection volume was 20 μL, and the run time was 3 minutes.
Results
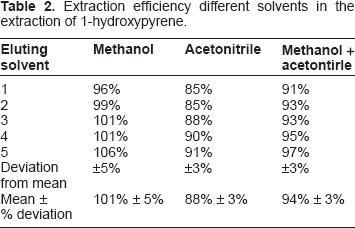
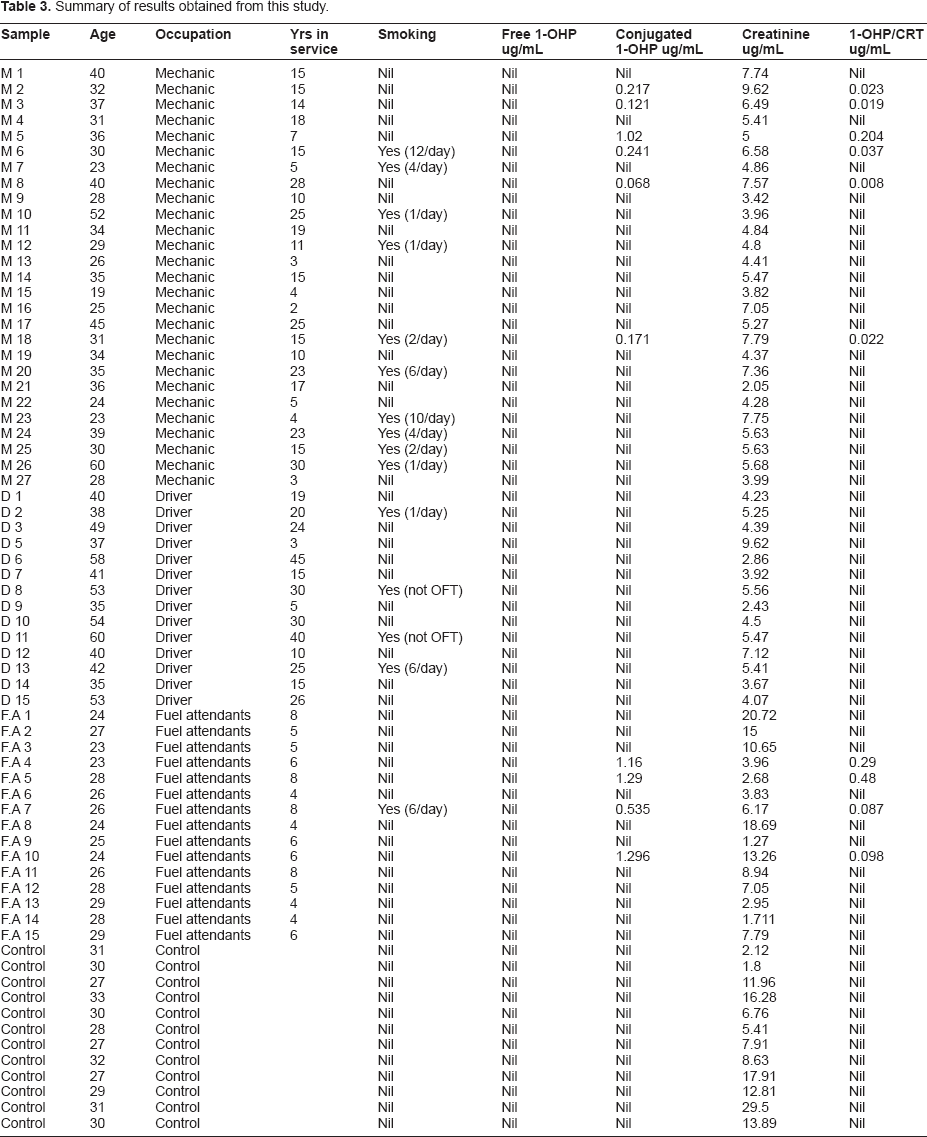
Prior to the study, three different solvent systems were screened for their extraction efficiency and their effect on the percentage recoveries of 1-hydroxypyrene. Table 2 shows the summary of the results obtained. Methanol was found to be the best eluent with a percentage recovery of 101.5%, followed by a mixture of methanol and acetonitrile (1:1) with a percentage recovery of 94.3%. This was followed by acetonitrile with a percentage recovery of 88.3%. Methanol is more polar and interacts better with 1-hydroxypyrene making it more soluble. 1-OHPY is a representative lipophyl metabolite of PAH compounds and many other studies have established 100% methanol as a good eluent. 25 Table 1 gives the characteristics of the subjects employed in this study, which include age, years of exposure, smoking history, and lifestyle. The calibration plot was linear with a correlation coefficient of 0.99 (Fig. 2). Figure 3 shows a representative chromatogram for 1-hydroxypyrene using the conditions described in the methods. Table 3 gives the results obtained in the study. The level of urinary 1-hydroxypyrene detected varied for the different categories of occupations studied. About 27% of sampled fuel attendants had detectable 1-hydroxypyrene in their urine samples, but for auto mechanics, it was 22%. There was no detectable 1-hydroxypyene in the urine samples of commercial drivers and students used as controls. Of the 15 smokers captured, only 3 had detectable urinary 1-hydroxypyrene. The detected hydroxypyrene concentrations among smokers ranged from 0.01 μg/mol creatinine to 0.092 μg/mol creatinine. Among the creatinine concentrations of the 15 fuel attendants, the highest detected concentration was 0.48 μg/mol creatinine. This represents the highest urinary hydroxypyrene concentration observed in this study. Of subjects with detectable urinary 1-hydroxypyrene, 40% were fuel attendants. Table 4 gives the summary of the results obtained in this study.
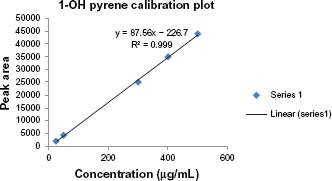
Calibration curve for 1-Hydroxypyrene standard.
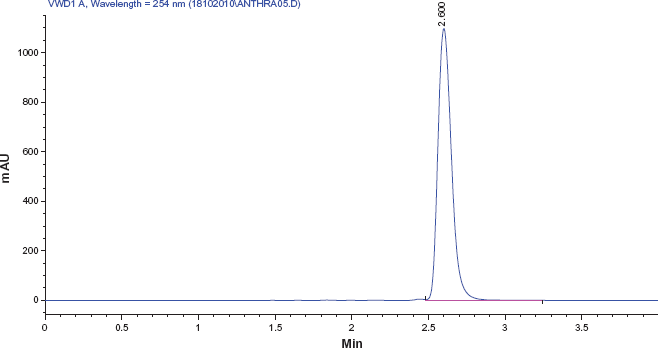
Representative Chromatogram for 1-Hydroxypyrene Standard with the following chromatographic conditions: Mobile Phase [water: methanol (12:88v/v)]; Stationary phase [X-Bridge C18 (150 × 4.6 mm) 5 μm column]; Wavelength [250 nm]; Flow rate [1.2 mL/min]; Column temperature [30 °C]; Injection volume [20 μL].
Extraction efficiency different solvents in the extraction of 1-hydroxypyrene.
Summary of results obtained from this study.
Discussion
The results from this study are in line with various other findings. A comprehensive review by Hansen et al 26 surveyed 101 studies among workers in various occupations. The subjects surveyed included those who worked in the petrochemical industry, foundries, and asphalt work. In all, the highest concentrations of urinary 1-hydroxypyrene were found among workers in the petrochemical industry at work places in Taiwan and China with reported levels as high as 1696 mmol/mol creatinine. This review revealed that workers in the petrochemical industry and in foundries had urinary 1-OHP concentrations four times higher than the concentration levels in the other studied occupations. 26 High levels of benzene in the breathing zone of fuel station attendants during refueling of automobiles has been reported. 27 In their study Tsai et al 28 found that vehicle exhaust significantly influenced the total PAH exposure (11.4 μg/m3). Urinary 1-hydroxypyrene levels in both mechanics and fuel attendants (3.02 ug/mol creatinine) at a Taiwan highway toll station were significantly higher than those of controls (0.41 ug/mol creatinine). 28 In 2003, Bartimaeus and Jacobs showed that considerable exposure to petrol or its products over a long period of time could cause nephrotoxicity in motor mechanics. 29
Kuusimaki et al 30 found high concentrations of 1-hydroxypyrene in bus-garage workers and waste collectors (0.125 μg mol/mol creatinine) when compared with controls (0.055 μg mol/mol creatinine). 31 Autrup et al reported higher urinary 1-hydroxypyrene level for suburban/rural bus drivers (0.25 μg mol/mol creatinine). 32 Burgaz et al have reported a higher urinary 1-hydroxypyrene levels in taxi drivers (0.57 μg mol/mol creatinine). 33 Other researchers have also reported higher urinary 1-hydroxypyrene in professional drivers (0.181 μg mol/mol creatinine) and commercial drivers (0.263 μg mol/mol creatinine). 33
From our findings, none of the three personal factors (age, work experience, and smoking habit) had a significant effect on predicting urinary 1-hydroxypyrene level in the different occupations. The highest concentration of 1-OHP was recorded in a 28-year-old fuel attendant with just 8 years working experience and no smoking history (Table 3) while a 60-year-old subject with 40 years exposure had no trace of urinary 1-hydroxypyrene. Smoking is the most widely studied confounder of environmental and occupational PAH exposure studies and the most preventable risk factor for lung cancer, 34 coronary heart disease, 35 and chronic obstructive pulmonary disease. 36 Overall, our study showed no significant correlation between cigarette smoking and increased urinary 1-hydroxypyrene of all three occupations. In this study, 1-hydroxypyrene was detected in only 20% of smokers. Several researchers found cigarette smoking to be correlated with an elevated urinary 1-hydroxypyrene. Merlo et al 37 found that the average urinary 1-hydroxypyrene for smoking traffic officers (0.201 μg mol/mol creatinine) was higher than their non-smoking counterparts (0.102 μg mol/mol creatinine) and Chuang et al reported that smoking office employees also had higher 1-OHPY levels (0.179 μg mol/mol creatinine) when compared with their non-smoking counterparts (0.067 μg mol/mol creatinine). 31 Ichiba et al 38 reported a decrease in urinary 1-hydroxypyrene and 2-naphthol after smoking cessation. A study by Carmella and co-workers in 2009 39 also showed that smoking cessation among 17 smokers at various times corresponded to a decrease in their 1-hydroxypyrene concentrations. In addition, cigarette smoking has been reported to significantly increase urinary 1-hydroxypyrene concentrations 40 and has been linked to lung and head and neck cancers.41,42 Kidney diseases have also been found to be higher in smokers than in ex-, and non-smokers. 43 The contrast between our findings and these studies could be due to the level of smoking among the subjects we studied.
Summary of results.

Several epidemiological studies have linked exposure to high PAH concentrations to cancer. In Xuan Wei in China, mortality rate from lung cancer was found to be five times the Chinese national average especially among women. This was attributed to the high atmospheric PAHs arising from the use of smoky coal as fuel. In addition, increased risk of lung cancer was observed among subjects working on aluminum smelters.49–50 Boffetta and co-workers 51 also observed increased risk of lung tumors in both pavers and roofers. Tumors of the stomach, bladder, and skin and leukemia were also observed. An association between PAH-DNA adducts and breast cancer incidences have also been reported.52–54 While several studies have reported the carcinogenicity of PAHs,55,56 its developmental toxicities (embroyolethality, reduced fetal weight, etc.) have also been widely reported in Ukraine, 57 the United States, 58 Czech Republic, 55 and Poland. 59
Conclusion
The findings of this study clearly show that fuel attendants and auto mechanics have significant exposures to PAHs as manifested by the level of urinary 1-hydroxypyrene detected. So far, there is no established benchmark for the level of PAHs in urine, but our findings indicate the possibility of future cancer cases in this population as a result of their occupational exposure. Even though various studies have linked cigarette smoking to increased 1-hydroypyrene levels, the current study was not able to link the level of 1-hydroxypyene with the smoking habits of the subjects.
Author Contributions
Conceived and designed the experiments: CA. Sample collection: CI, KO, FF, KOk, and AA. Sample Analysis: CA, CI, KO, FF, KOk and AA. Wrote the first draft of the manuscript: CI. Contributed to the writing of the manuscript: CA. Agree with manuscript results and conclusion: CA, CI, KO, FF, KOk and AA. Jointly developed the structure and argument for the paper: CA and CI. Made critical revision and approved final version: CA. All authors reviewed and approved of the final manuscript.
Funding
Author(s) disclose no funding sources.
Competing Interests
Author(s) disclose no potential conflicts of interest.
Disclosures and Ethics
As a requirement of publication author(s) have provided to the publisher signed confirmation of compliance with legal and ethical obligations including but not limited to the following: authorship and contributorship, conflicts of interest, privacy and confidentiality and (where applicable) protection of human and animal research subjects. The authors have read and confirmed their agreement with the ICMJE authorship and conflict of interest criteria. The authors have also confirmed that this article is unique and not under consideration or published in any other publication, and that they have permission from rights holders to reproduce any copyrighted material. Any disclosures are made in this section. The external blind peer reviewers report no conflicts of interest.
