Abstract
Isomerization of amino acids in proteins has recently been identified as a part of the aging process. Increases in D-amino acids as a consequence of isomerization influence the function and structure of proteins. Senescence-related pulmonary diseases, such as chronic obstructive pulmonary disease, are thought to be caused by reductions of lung function with age. We hypothesized that changes of protein structure in lung tissue induced by the isomerization of amino acids could result in decreased lung function. Therefore, we examined whether isomerization of amino acids takes place in the lungs of rats as they age. We measured the content of L- and D-amino acids in collagen 1 by HPLC using a chiral column. We found that collagen 1 was increasingly racemized with age, so that significantly higher proportions of D-Ser were present in 12- and 24-month-old rats than in 8-week-old rats. D-Asp increased slightly but not significantly. We also investigated the localization of collagen 1 in lung tissue. Stacks of collagen 1 were observed in the parenchyma and airway wall, and age-dependent changes were especially prominent in the airway wall. Racemization of collagen 1 could therefore influence lung function and contribute to pulmonary diseases.
Introduction
Lung function decreases with age.1–3 Prominent changes in aged lung are a decrease of static elastic recoils, compliance of the chest wall and an increase in airway resistance. 1 Chronic obstructive pulmonary disease (COPD) and pulmonary fibrosis occur partly in an age-dependent fashion,4–6 and asthma, an immunological and inflammatory disease, is also influenced by age-related changes of the lung.7–9 Elastin is a component of connective tissue in the lung and is associated with the passive recoils of lung function, and age-related changes and diseases related to the remodeling of elastin have been reported.1,10,11 Collagen and laminin, which are major components of the extracellular matrix in lung, expect elastin, are also reported to increase with age. 12
Isomerization of l-amino acids into d-amino acids and the resulting racemization of structural proteins, including elastin, were recently found to be markers of elderly tissues.13–15 An increase in d-amino acids changes the structure and function of proteins and is related to the pathophysiology of several diseases.13,16 Collagen 1 is a high-molecular-weight protein, about 300 kDa, that forms a triple-helix composed of two alpha-1 chains (about 96 kDa) and a single alpha-2 chain (about 96 kDa). In bone, the Asp1211 in collagen 1 is racemized in vivo with age.17,18 This can be attributed to decreased turnover rates of collagen17,19,20 because collagen synthesis and degradation both decrease with age. 21 In cataracts, isomerization of Asp increases in A-crystallin and B-crystallin,22,23 and racemization of the Asp23 residue accelerates fibril formation of amyloidal β sheets in vitro. 16
Racemization of collagen 1 may induce collagen 1 deposition, resulting in the suppression of lung function and the induction of pulmonary diseases. Collagen 1 deposition is increased in the aged lung, but the racemization of collagen 1 in the lung has not been examined. In this study, we investigated collagen 1 racemization and deposition in rats of 3 ages. The molecular characteristics of Ser and Asp make them particularly susceptible to isomerization from the l-amino acid to the d-amino acid, and there are many reports that describe biological functions and diseases related to d-Ser and d-Asp.18,22,24–28 Therefore, we examined the isomerization of Ser and Asp in particular. We found that isomerization of Ser and Asp in collagen 1 increases in the lung with age.
Materials and Methods
Animals
Wistar rats, male, were purchased from SLC Japan and were raised in Charles River Japan (Kanagawa, Japan) until they became 8 weeks, 12 months, or 24 months old. Then, the rats were moved into the animal facilities of Musashino University School of Medicine under specific pathogen–free conditions. Care and use of the animals followed the guidelines of the “Principles of Laboratory Animal Care” formulated by the National Society for Medical Research.
Sample extraction
The extraction of collagen 1 was performed according to the method described by Lafuma et al. 29 Animals were decapitated at 8 weeks, 12 months, or 24 months (n = 9 of each) of age and lung tissues were removed immediately. Tissues were cut into pieces and homogenized in 1 M NaCl; this and all liquids used in this method contained Proteinase Inhibitor Cocktail (Sigma, Saint Louis, MO, USA). Incubation of tissues for 30 min at 4 °C was followed by centrifugation for 5 min at 4 °C and 8,000 x g. Pellets were suspended into 2 M MgCl2 and incubated for 30 min at 4 °C. After centrifugation for 15 min at 4 °C and 14,000 x g, the supernatant was removed and the pellets were washed with water until they were salt-free. Dilapidation and dehydration were carried out with acetone and chloroform/methanol. Lung samples were dried with an evaporator, resuspended in guanidine buffer (5 M guanidine hydrochloride, 0.05 M dithiothreitol, 0.1% EDTA, 0.1 M Tris, pH 8.5), and incubated for 24 h at 20 °C. Finally, the collagen 1 extract obtained from the guanidine buffer was filtered with a MICROCON YM-100 centrifugal filter (Millipore, Bedford, MA, USA) for desalting and size selection.
SDS-PAGE
To verify the purity of the collagen 1 samples purified with the MICROCON YM-100, we applied them to SDS-PAGE. The SDS-PAGE was performed on a 3%–10% gradient polyacrylamide gel, Super Sep™ 3%–10% (Wako, Japan), as described by Laemmli. 53 Gels were stained with Coomassie Blue G250, and the gel was photographed with a LAS-3000 (FUJIFILM, Japan).
HPLC analysis
Sample preparation for high performance liquid chromatography (HPLC) analysis
To improve the precision of the HPLC analysis, collagen extracts were filtered and desalted with Zip-Tip C4 pipette tips (Millipore, Bedford, MA, USA). The solutions obtained after ZipTip C4 filtering were eluted with 75% acetonitrile (ACN) in 0.1% trifluoro-acetic acid (TFA). The purified samples were hydrolyzed with HCl on a Pico-Tag Workstation (Waters, Tokyo, Japan) at 108 °C for 7 h. The hydrolyzed samples were dissolved in 15 μl of 200 mM borate buffer, and 10 μl out of the 15-μl sample were mixed with 20 μl of 4-Fluoro-7-nitrobenzofurazan (NBD-F) (Dojindo, Kumamoto, Japan). The samples were protected from light for the remainder of the procedure. The samples were heated to 60 °C for 5 min for the NBD-F amine labeling. The sample solutions were cooled, and the reaction was stopped by the addition of 70 μl of 1% acetic acid in methanol. Then 50 μl of each sample solution was applied to HPLC.
We also examined elastin, which is a major component of lung. After extraction by immunoprecipitation with Seize X Protein G (Pierce, IL, USA), elastin was analyzed by HPLC after filtration with a ZipTip C4 (Millipore, MA, USA).
HPLC experiments
Chromatographic analysis was carried out on a HPLC system (Shimadzu, Kyoto, Japan) consisting of SIL-10AD, LC-10ATVP, FCV-10ALVP, DGU-12A, CTO-10ASVP, RF-10AXL, and SCL-10AVP. The LC solution software controlled both the HPLC system and the data analysis. HPLC analysis method was as described previously. 31 To determine the Ser and Asp content, mobile phase A (1% tetrahydrofuran, 0.02% TFA, 10% ACN in MilliQ-purified water) and mobile phase B (1% tetrahydrofuran, 0.02% TFA in ACN) were used. Then, d/l-amino acid mixtures were eluted with a gradient from 0% B in A to 25% B in A over 50 min and then 25% B in A to 75% B in A over 40 min through a reverse-phase column, TSKgel ODS-80TsQA (Tosoh, Tokyo, Japan). After the mobile buffer was replaced with methanol, each amino acid fraction was separated into d- and l-amino acids with a chiral column, OA2500-S (Sumica Chemical Analysis Service, Osaka, Japan). The mobile phase for the chiral HPLC was 5 mM citric acid in methanol. The concentration of d/l-Ser was calculated as follows: the enantiomeric ratio of NBD–d- or –l-Ser was quantified as a percentage of the total (D+L)-Ser concentration, which was determined by the peak area ratio of the Ser derivative on the chromatogram recorded by LCsolution (Shimadzu, Kyoto, Japan). The d/l-Asp ratio was calculated with the same method as the Ser ratio.
Histological examination
For histological analysis, the lungs were inflated and fixed by intratracheal administration of 10% formalin. 32 3 μm sections were prepared by microtome and stained with Hematoxylin eosin (HE) to analyze the mean linear intercept (MLI).30,32 MLI was determined on 20 photomicroscopic images per animal.
Immunohistochemistry
Paraffin sections (3 μm) were treated with H2O2, incubated with Protein Block (Dako Cytomatin, Glostrup, Denmark), and probed with rabbit anti-collagen 1 (abcam, Tokyo, Japan) for 1 h at room temperature. The sections were then incubated with the secondary antibody, Alexa Fluor 488–conjugated goat anti-rabbit IgG (Invitrogen, CA, USA), for 1.5 h at room temperature, protected from light. Samples were mounted in Vectashield Mounting Medium (Vector Laboratories, CA, USA) and detected using a confocal microscope, FV1000 (Olympus, Tokyo, Japan). The diameters of alveoli were estimated from confocal microscopic images.
Results
SDS-PAGE for validation of sample purity
We examined whether the proportion of racemized amino acids varies depending on age. We obtained collagen 1 from the lung tissues of rats at 3 ages (8 weeks, 12 months, and 24 months) and dissociated the alpha chain with reductants in SDS-sample buffer. The dissociated chains were also detected as multiple bands, beta-chain and gamma-chain, which are complex tangles of alpha-chains, on SDS-PAGE (Fig. 1).
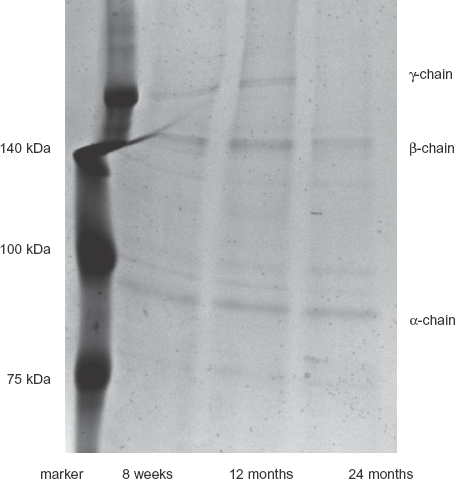
SDS-PAGE analysis of collagen 1 extracted from lungs of rats at each indicated age. Collagen 1 obtained from rat lungs was detected as bands of about 95 kDa and 140 kDa in an SDS-PAGE gel stained with Coomassie Brilliant Blue. Proteins in complex tangles may migrate as bands of irregular size. Lane 1, Molecular weight markers; lane 2, extract from 8-week-old rat lung; lane 3, from 12-month-old rat lung; lane 4, from 24-month-old rat lung.
Ratios of D-Ser and D-Asp to total Ser and Asp
Ser and Asp have molecular characteristics, which has tendency to isomerize from L-amino acid to D-amino acid easily than other amino acids. Besides, there are many reports described about biological functions and diseases which are related to D-Ser and D-Asp. Because of these facts, we examined particularly isomerization of Ser and Asp.
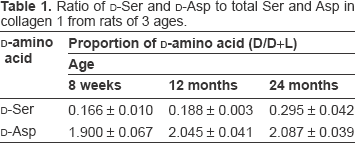
We used a reverse-phase column to separate the Ser and Asp amino acid fractions from hydrolysates of collagen 1 obtained from lungs of rats at 8 weeks, 12 months, and 24 months of age (Fig. 2). Then the D/L-mixtures of the Ser fraction and Asp fraction were separated into D- and L-amino acid fractions with a chiral column, and the ratio of each D-amino acid to the D/L-mixture was calculated. The ratio of D-Ser increased significantly between 8 weeks and 12 months (P < 0.05) and between 12 months and 24 months (P < 0.05) of age (Table 1, Fig. 3A). D-Asp increased slightly but not significantly with age (Fig. 3B). On the other hand, the percentage of D-amino acids in elastin, which is also predominant in lung tissue, did not increase with age (Fig. 3C, D).
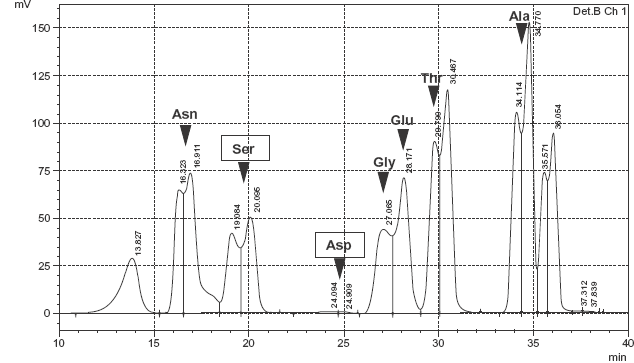
Reverse-phase chromatogram of collagen 1 hydrolyzed into amino acids. Amino acids were derivatized with NBD-F.
Ratio of D-Ser and D-Asp to total Ser and Asp in collagen 1 from rats of 3 ages.
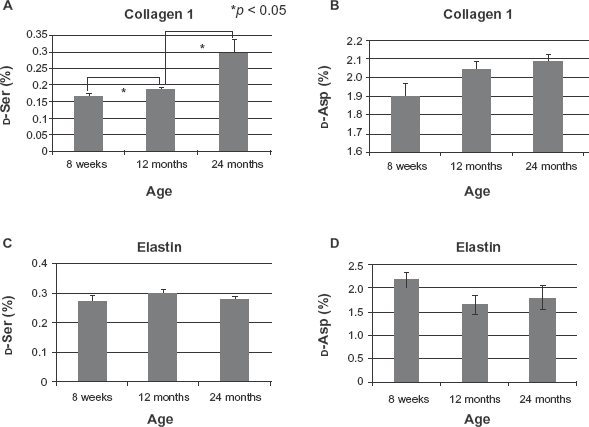
The ratio of D-Ser and D-Asp to total Ser and Asp, respectively, in collagen 1 and in elastin in lungs of rats at each indicated age (*P < 0.05, student t test).
Immunohistochemistry
We hypothesized that racemized proteins would undergo structural changes, resulting in suppression of the protein function and changes in organ morphology. We therefore investigated the morphology of the lungs from rats at the 3 different ages. There were no macroscopic differences in the morphology of the lungs from rats of each age. However, MLI increased significantly with age (P < 0.01; Fig. 4). Increased MLI is characteristic of pulmonary diseases, such as emphysema. 33 Immunohistochemical and confocal microscopic analysis of the lungs showed alveolar septal destruction and irregular size of alveoli developing with age (Fig. 5B–D). Next, we investigated morphological changes of the bronchi. Collagen 1 is present in the connective tissue matrix that ensures the resilience of the bronchiolar wall. Collagen 1 detected in bronchioles from 8-week-old rats was uniformly but thinly stacked in the connective tissue (Fig. 5F). Collagen 1 was unambiguously stacked in the bronchioles of 12-month-old rats (Fig. 5G) and was thickly stacked in the connective tissue of 24-month-old rats (Fig. 5H).
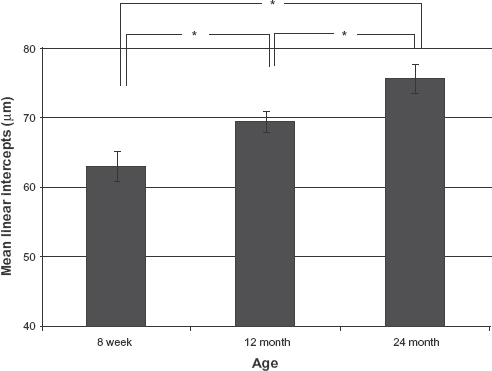
Quantification by mean linear intercept (MLI). To assess the alveolar wall destruction, we calculated MLI (mean ± SD) in rats of different ages (*P < 0.05, student t test).
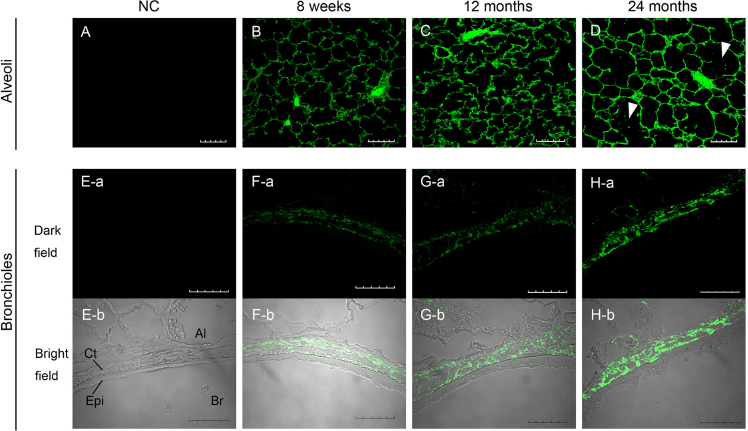
Immunohistochemical and confocal microscopic analysis of collagen 1 in rat lungs. Upper panels show alveoli (
Discussion
In this study, the proportion of D-Ser in rat collagen 1 was significantly higher at 24 months of age than at 8 weeks, suggesting that racemized collagen 1 increases in the lung with age. Thus, collagen undergoes qualitative changes with age in addition to increases in its quantity.12,34
In the lung, collagen is the main component of the extracellular matrix, and its deposition is related to interstitial pneumonitis and airway diseases.35–37 Changes in lung tissue with age include decreased turnover of elastin and collagen as well as a decrease in elastic recoil.38,39 Elastin fiber content decreases and collagen content increases in the parenchyma of the murine lung. 34 Our results are also consistent with the increased collagen deposition in the peribronchial regions of aged rats found by Calabresi C et al. 12 The increased deposition of collagen appears to be related to susceptibility to and the progression of lung diseases, such as COPD, interstitial pneumonitis, and asthma.40–42
Recently, racemization of structural proteins was found to be an indicator of elderly tissues.13–15 Racemization is a spontaneous chemical reaction, resulting in the alteration of L-amino acids to D-amino acids. 13 Although D-amino acids can be formed from 19 amino acids, isomerization of Asp and Ser are predominant in mammals.13,43 The alteration of protein structure and function by racemized Asp was first reported in synthetic peptides in vitro.16,43,44 D-Ser is present at high levels in the forebrain 45 and functions mostly in the maintenance of neuronal activity through the NMDA receptors. 46 Moreover, β-amyloid protein, which is increased in and thought to be a causative protein of Alzheimer's disease, is racemized at both Ser and Asp, 47 and D-Ser influences the aggregate form of β-amyloid. 47 Accumulation of racemized Asp is increased in Down's syndrome, 48 in crystallin in cataracts, and in aged skin.23,49
It is not clear that what amount of protein alterations would be necessary to cause structural changes in lung function. Meanwhile, in onsets of some diseases, accumulations of the denatured protein are reported, such as collagen in pulmonary fibrosis. 50 The denaturation of a very small amount of protein by racemization might be useful as a mark of diseases.
Until now, it is revealed that the domain including Ser residues are necessary to keep its conformation and/or trimerization of collagen. 51 Isomerized Ser increasing with age may affect deposition of collagen through not only enhancing structural alteration, but also influence such regulatory regions. It is also predicted that isomerized Ser may inhibit function of collagenase against collagen 1.
We found that D-Ser was present in lung collagen 1 and increased significantly with age. Similar to the effects of racemized proteins in cataracts23,24 and Alzheimer's disease, 50 collagen 1 racemization due to Ser isomerization could influence the onset and progression of pulmonary diseases.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors report no conflicts of interest.
Footnotes
Acknowledgment
This work was supported in part by the Ministry of Education, Culture, Sports, Science and Technology (MEXT), HAITEKU (2004–2008).
