Abstract
Introduction
Idiopathic pulmonary fibrosis (IPF) is a progressive interstitial lung disease with an unknown etiology and usually happens in elderly people. Its radiological and pathological features are consistent with usual interstitial pneumonia (UIP). 1 Generally, the patients present as dyspnea and gradual deterioration of lung function with acute exacerbation occurring during the course of disease. The prognosis of IPF is poor and median survival time was only 3.8 years since diagnosis in people over 65 years old.1–3 Epithelial-Mesenchymal Transition (EMT) plays an important role during dysfunctional epithelial regeneration and may be major pathogenic driver in IPF.4,5 Besides, a mendelian randomization study showed that high level of IL-14 had a causal relationship with the increased risk of IPF. 6 In addition, the treatments for IPF were limited, up to now only two drugs, pirfenidone and nintedanib, have evidence-based medical support that they can slow down the rate of decay of lung function but cannot improve the mortality of IPF.7,8 Therefore, explore potential risk of IPF is important, which could possibly improve the prognosis of this tricky disease.
Gastroesophageal reflux disease (GERD) is a prevalent condition especially in European and North American populations. Bretagne et al 9 reported that about 31.3% French used to have typical symptoms of GERD. The prevalence of GERD in the U.S population is estimated to range from 18.1% to 27.8%. 10 Asian people also have high morbidity of GERD, Fujiwara et al 11 showed that about 17.9% Japanese had GERD. In IPF patients, there has been reported to have higher morbidity of GERD. Mays et al 12 found 44% IPF patients were diagnosed with GERD, while age matching group was only 19%. Therefore, we speculated there were correlation between GERD and IPF, persistent existing of GERD could contribute to the occurrence of IPF. In vivo studies, Chen et al 13 found that in GERD rat models expression level of TGF-β1, Type I collagen and connective tissue growth factor were up-regulated in lung tissues and the rats usually had more severe fibrosis extent. Kouki et al 14 revealed that dogs afflicted with IPF exhibited elevated levels of total bile acid in both saliva and bronchoalveolar lavage fluid, surpassing those observed in their healthy counterparts. Hence, micro-aspiration may potentially exert a pivotal role in the occurrence and progression of GERD, while mitigating micro-aspiration could potentially attenuate the incidence rate and development of IPF, so the correlation between GERD and IPF has confirmed, but the causal relationship between need further explored 15
Exploring etiology of a disease is usually associated with its prognosis. The key treatment of GERD is to block-up micro-aspiration, which includes lifestyle modifications, intervention with anti-acid drugs (AAT) and laparoscopic anti-reflux surgery. There were several studies focusing on whether AAT or laparoscopic anti-reflux surgery could slow down progress and improve the prognosis of IPF, but the results were controversial. Retrospective studies 16 and a meta-analysis 17 both revealed that treatment of GERD can improve prognosis in patients with IPF. However, the study of Kreuter et al 18 suggested that use of AAT in patients with IPF could not improve prognosis and in the contrary increased the risk of severe infection. Raghu et al 19 revealed that laparoscopic anti-reflux surgery could not decrease mortality in IPF patients. However, in observational studies, research results are often interfered by various confounding factors so as to make etiological explanations unreliable. Therefore, the exploration of the relationship between disease risk and outcome necessitates the implementation of novel methodological studies.
Mendelian randomization (MR) was first described by Katan,
20
The underlying principle of this concept is that distinct genotypes give rise to diverse phenotypes (Figure 1).
21
By simulating the impact of exposure characteristics on a disease, the association between genotype and disease can be elucidated. The estimated effect between genotype and disease remains untainted by confounding factors that could potentially induce reverse causal associations, as alleles adhere to the principle of random allocation during gamete formation. Therefore, this method has an incomparable advantage over observational studies in causal inference. Mendelian randomization design, adopted from Jareebi (2024).
21

We conducted this study to investigate the impact of GERD on the risk of developing IPF in a MR design. By utilizing genetic variants associated with GERD as instrumental variables, we aimed to clarify if there is potential causal relationship between GERD and the development of IPF, ultimately providing novel insights into this hypothesis.
Methods
GWAS summary data collection and IVs selection
Mendelian randomization (MR) analysis with R software (version 4.2.2) and the TwoSampleMR package was applied in our original research. It encompassed collecting genetic data for the exposures and their corresponding outcomes. To ensure compatibility, we performed data harmonization to align alleles across two distinct datasets to facilitate MR analysis. We conducted MR analyses from both the IEU OpenGWAS and FINNGEN datasets. A significance threshold of p < .05 was applied, with a primary emphasis on employing the inverse variance weighted (IVW) method, weighted median approach, and MR-Egger methods. Additionally, we employed MR-PRESSO and leave-one-out analysis to identify and address any potential bias caused by horizontal pleiotropy or a single SNP.
We searched for relevant genome-wide association studies (GWAS) data on FINNGEN (https://www.finngen.fi/en)
22
and IEU OpenGWAS (https://gwas.mrcieu.ac.uk/),23–25 selecting genetic variants significantly associated with GERD as instrumental variables (IVs) in our MR design (see Figure 2). Two representative GERD-related GWASs were included in our study. The first GWAS was downloaded from IEU OpenGWAS(ebi-a-GCST90000514) as test set, and the second was extracted from FINNGEN (K11_REFLUX) as validation set. The IVs identified in the test set are referred to as IV-1, while those identified in the validation set are referred to as IV-2 (Table 1). Workflow of Mendelian randomization study revealing causality from GERD on IPF. Association of the instrumental SNPs with GRED and IPF among IV1 and IV2.

Statistical Analyses
Summary of the causal effects of each trait on IPF via different MR methods.

The validation set extracted from FINNGEN (K11_REFLUX), using data from 22867 GERD and 292,256 control cases. To increase the number of SNPs associated with GERD, we used a more lenient threshold (p < 5 × 10−7) and evaluated LD (r2 < 0.001). This resulted in the identification of three SNPs (rs79348626, rs12759463 and rs4269485). The average F-statistics was 26.52 (Table 1-2).
Two-sample MR
After standardizing the effect alleles in the IVs and FINNGEN(IPF), we used IVW, weighted median and MR-Egger to calculate MR estimates. If these approaches yielded inconsistent results, we would set a stricter instrument p value threshold. 26 We used the IVW approach to detect heterogeneity (Cochran Q-derived p < .05) and checked for directional pleiotropy using the MR-Egger regression intercept (p < .05 indicated its presence). 27 In addition, we utilized MR-PRESSO and leave-one-out analysis to detect and correct for horizontal pleiotropy or any potential bias caused by a single SNP.28,29 These analyses were conducted in R (version 4.2.2).
Outcomes
The findings are concisely summarized and visually presented (Figure 3). Odds ratio plot for causal effect of GERD on IPF.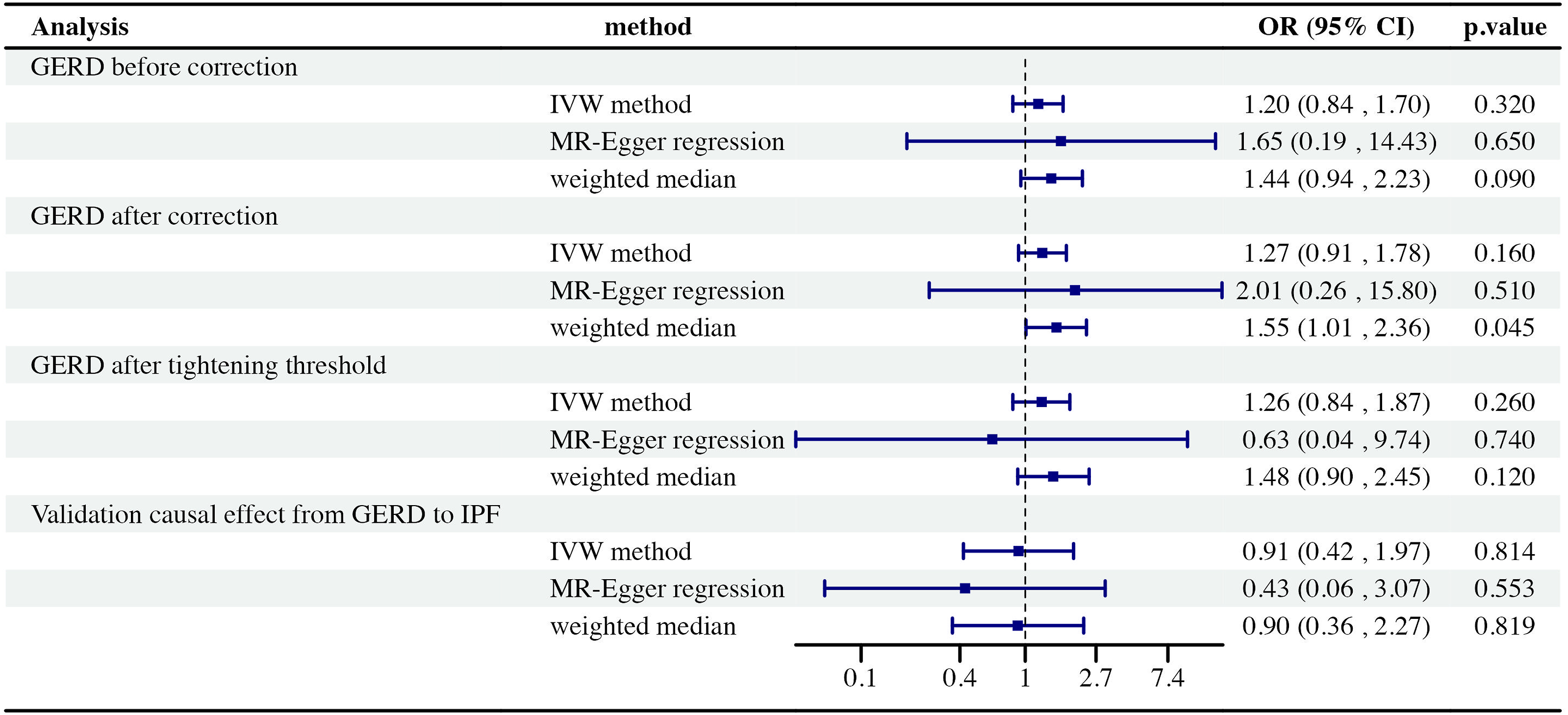
Causal effect from GERD to IPF
Using 65 GERD-related SNPs, we found the potential causal effect of GERD on the risk of IPF was not statistically significant from the IVW approach (OR = 1.20, 95% CI = 0.84-1.70, p = .32), MR-Egger regression (OR = 1.65, 95% CI = 0.19-14.43, p = .65) and weighted median approaches (OR = 1.44, 95% CI = 0.94-2.23, p = .09) (Figure 4). However, there was heterogeneity observed using MR-Egger regression (p value derived from Cochran Q-test = 0.001) and IVW methods (p = .001). A similar result was also obtained with MR-PRESSO (global heterogeneity test p value <.01). After removing one outlier (rs9636202), we re-applied MR methods to assess the relationship between GRED and IPF. With weighted median method, we found that GERD significantly increased the risk of IPF (OR 1.55, 95% CI: 1.01-2.36, p = .045). However, when using the IVW (OR: 1.27, 95% CI: 0.91-1.78, p = .16) and MR-Egger approach (OR: 2.01, 95% CI: 0.26-15.8, p = .51), opposing results were observed which showed inconsistency with our findings from the Weighted median method. To address this inconsistency, we tightened the instrument p value threshold to a more stringent level of <1 × 10−8 and used a set of only forty-two SNPs as instrumental tools. As a result, the MR estimates did not reach statistical significance using IVW (OR = 1.26, 95% CI = 0.84-1.87, p = .26), MR-Egger regression (OR = 0.63, 95% CI = 0.04-9.74, p = .74) and weighted median approaches (OR = 1.48, 95% CI = 0.90-2.45, p = .12), indicating that a genetically predicted increase in GERD was not significantly associated with IPF risk. Additionally, there was no directional pleiotropy observed (intercept = −0.01; SE = 0.036. p = .772). The leave-one-out analysis showed that no single SNP against the overall effect (Figure S1). Furthermore, the funnel plot indicating no pleiotropy (Figure S2). Scatter plots of the association of GERD and IPF.
Validation causal effect from GERD to IPF
We identified three SNPs (rs79348626, rs12759463 and 4269485) that were significantly associated with GERD independently. However, our analysis showed no evidence of causality between GERD and IPF using the IVW method (OR = 0.91, 95% CI = 0.42-1.97, p = .814), MR-Egger regression (OR = 0.43, 95% CI = 0.06-3.07, p = .553) and weighted median approaches (OR = 0.90, 95% CI = 0.36-2.27, p = 0 0.819) (Figure S3). There is no significant heterogeneity present via analysis with MR-Egger (p = .545) and IVW methods (p = .595).
Discussion
We used two-sample MR to evaluate the association between GERD and IPF. Overall, we did not find strong evidence to support that GERD increases IPF risk, which was opposite to previous studies.30–32
Until now, the relationship between GERD and IPF has remained controversial.15,33,34 Most published studies were retrospective, making it difficult to determine causality. Some diagnosed cases of GERD and undiagnosed subclinical GERD may potentially contribute to the development of IPF; however, it remains uncertain whether GERD can accurately predict the onset or prognosis of IPF. Moreover, previous observational studies were susceptible to confounding risk factors.33,35 In our study, we established causality using MR methods and avoided bias through improved study design. The utilization of two sets of genetic instruments unveiled no causal association between GERD and IPF incidence, in line with recent high-quality investigations. Khor et al 36 indicated that the use of antacid medication or anti-reflux surgery did not improve prognosis in patients with IPF, even if they did not have confirmed diagnosis of GERD. Similarly, a study conducted by the Australia IPF Registry found no significant impact on longer-term outcomes from either antacid therapy or GERD symptoms. 37 In a study based on large populations, it showed that using PPIs did not lower the risk of death or hospitalization for patients with IPF. 38 These studies suggest that treating GERD may not be as helpful for IPF as previously thought and is only conditionally recommended in treatment guidelines.
Although our findings suggest that there is no direct link between GERD and IPF incidence, whether GERD contributes to the progression of IPF remains unknown. The presence of gastric contents in the lungs has been demonstrated in mouse studies to be associated with the development of pulmonary fibrosis. This intricate process involves a multitude of molecules that actively contribute to the progression of fibrosis, encompassing TGF-β, basic fibroblast growth factor, vascular endothelial growth factor, and connective tissue growth factor.39,40 In IPF patients, several studies found that pepsin was exist in the bronchoalveolar lavage fluid (BALF). This could result in acute exacerbation of IPF.41–44 Additionally, children with GERD had more pepsin compare to healthy controls in their tracheal fluid.45,46 Furthermore, pepsin was not present in the tracheal fluid of children who did not have GERD or respiratory symptoms. 47
Our study possesses several notable strengths. Firstly, we were able to emulate randomized controlled trials within observational settings through the utilization of MR. MR studies effectively circumvent confounding bias as SNPs are randomly assigned at conception and offer a cost-effective approach. In comparison to other observational studies, MR holds the advantage of mitigating reverse causation. Using MR method. Besides, our findings suggest that AAT and laparoscopic anti-reflux surgery may not be necessary for IPF treatment. Instead, more attention should be given to exploring the pathogenesis of IPF in relation to GERD.
Meanwhile, there are also some limitations. The data used in all GWASs came from European populations, it remains to be seen whether our findings apply to other populations. Secondly, it is important to recognize the diversity among IPF patients, it is possible that GERD may be a causal factor for a specific type of IPF. More comprehensive studies including subgroups of IPF should be considered in the future. In addition, we have tested the MR assumptions using different methods, although the results were broadly consistent, there was inevitably some residual uncertainty. Besides, the calculation and justification of the sample size selected for this study were not done in this study. Finally, it should be noted that the SNP size of the validation data set was small in our study.
Conclusion
Findings of our MR study not support a causal effect between GERD and IPF, which highlight we need to search and control for other candidate risk factors of IPF.
Supplemental Material
Supplemental Material - Gastroesophageal reflux disease and idiopathic pulmonary fibrosis risk: A mendelian randomization study
Supplemental Material for Gastroesophageal reflux disease and idiopathic pulmonary fibrosis risk: A mendelian randomization study by Minjie Lin, Junjie Wang, Jie Wei, Yu Yao, Cheng Tang, Wenfang Jin, Weihong Yuan and Yanling Lv in European Journal of Inflammation.
Supplemental Material
Supplemental Material - Gastroesophageal reflux disease and idiopathic pulmonary fibrosis risk: A mendelian randomization study
Supplemental Material for Gastroesophageal reflux disease and idiopathic pulmonary fibrosis risk: A mendelian randomization study by Minjie Lin, Junjie Wang, Jie Wei, Yu Yao, Cheng Tang, Wenfang Jin, Weihong Yuan and Yanling Lv in European Journal of Inflammation.
Supplemental Material
Supplemental Material - Gastroesophageal reflux disease and idiopathic pulmonary fibrosis risk: A mendelian randomization study
Supplemental Material for Gastroesophageal reflux disease and idiopathic pulmonary fibrosis risk: A mendelian randomization study by Minjie Lin, Junjie Wang, Jie Wei, Yu Yao, Cheng Tang, Wenfang Jin, Weihong Yuan and Yanling Lv in European Journal of Inflammation.
Footnotes
Authors’ contribution
YLL, WHY and MJL contributed to the conception, methodology design, and writing of the manuscript. JW, YY, CT, JJW and WFJ took responsibilities for data abstraction and accuracy of data analysis. All authors are guarantors of this work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by National Natural Science Foundation of China, grant number 82203561.
Data availability statement
All data generated or analyses in this study are included in this article. Further enquiries can be directed to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Abbreviations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
