Abstract
Curcumin 4‘-O-glucooligosaccharides were synthesized by a two step-enzymatic method using almond β-glucosidase and cyclodextrin glucanotransferase (CGTase). Curcumin was glucosylated to curcumin 4‘-O-β-D-glucopyranoside by almond β-glucosidase in 19% yield. Curcumin 4‘-O-β-D-glucopyranoside was converted into curcumin 4‘-O-β-glucooligosaccharides, i.e. 4‘-O-β-maltoside (51%) and 4‘-O-β-maltotrioside (25%), by further CGTase-catalyzed glycosylation. Curcumin 4‘-O-β-glycosides showed suppressive action on IgE antibody formation and inhibitory effects on histamine release from rat peritoneal mast cells.
Keywords
Introduction
Curcumin [1,7-bis(4-hydroxy-3-methoxyphenyl)-1, 6-heptadien-3,5-dione] is an important principle among curcuminoids found in Curcuma longa Linn., which has been used for centuries as a spice. Recently, much attention has been paid to pharmacological properties of curcumin because of its potential to be a very useful drug. Curcumin has been widely studied for its antioxidative, anticancer, and antiinflammatory effects.1–8 Irrespective of its pharmacological properties, curcumin is insoluble in water and poorly absorbed after oral administration.9,10
Biological glycosylation allows water-insoluble and unstable organic compounds to be converted into the corresponding water-soluble and stable compounds through convenient one-step enzymatic glycosylation.11–14 Furthermore, glycosides of physiologically active compounds, i.e. vitamin glycosides, have been reported to be useful anti-allegic agents.11,15,16 The therapeutic value of curcumin glycosides is of pharmacological interest. Here we report enzymatic synthesis of curcumin 4‘-O-β-glucooligosaccharides and their anti-allegic properties.
Materials and Methods General
Curcumin was purchased from Tokyo Kasei Kogyo Co. Ltd. and was purified by silica gel column chromatography before use for this experiment. Almond β-glucosidase was purchased from Wako Pure Chemicals Int. Ltd. and cyclodexrin glucanotransferase (CGTase) was from Amano Pharmaceutical Co. Ltd. The 1 H NMR (nuclear magnetic resonance) (400 MHz), 13 C NMR (100 MHz), H-H COSY (correlation spectroscopy), C-H COSY, and HMBC (heterobuclear multiple-bond correlation) spectra were measured using a Varian XL-400 spectrometer in CD3OD solution and the chemical shifts were expressed in δ (ppm) referring to tetramethylsilane. The HRFABMS (high resolution fast atom bombardment mass spectrometry) spectra were recorded using a JEOL MStation JMS-700 spectrometer.
Synthesis of Curcumin 4‘-O-β-D-Glucopyranoside
Curcumin 4‘-O-β-D-glucopyranoside was prepared as follows. To the mixture (20 ml) of acetonitrile and water (9:1 v/v) containing 5 mmol of D-glucose and 500 U of almond β-D-glucosidase was added 5 mmol of curcumin. The mixture was incubated with stirring for 48 h at 40 °C. The reaction mixture was concentrated by evaporation under reduced pressure, and the residue was purified by column chromatography on silica gel to give curcumin 4‘-O-β-D-glucopyranoside.
Synthesis of Curcumin 4‘-O-β-Glucooligosaccharides
To a solution containing 1 mmol of curcumin 4‘-O-β-D-glucopyranoside and 5 g of starch in 25 mM of sodium phosphate buffer (pH 7.0) was added 100 U of CGTase. After stirring of the reaction mixture at 40 °C for 24 h, the mixture was centrifuged at 3000 g for 10 min. The supernatant was subjected on to a Sephadex G-25 column equilibrated with water to remove CGTase. The fractions containing glycosides were purified by preparative HPLC to give two curcumin 4‘-O-β-glucooligosaccharides, i.e. curcumin 4‘-O-β-maltoside and curcumin 4‘-O-β-maltotrioside.
Anti-Allergic Activity
Suppressive action on IgE antibody formation
The inhibitory action of curcumin 4‘-O-β-glycosides on IgE antibody formation was examined as follows. Ovalbumin was used as the antigen (1 mg/rat), and Al(OH)3 and pertissius vaccine were used as the adjuvants (20 mg and 0.6 ml/rat, respectively). Sensitization was made by injection of a mixture (0.6 ml) of the antigen and the adjuvant into the paws of each rat (male, ca. 200 g). Paw edema was measured 24 h after injection and the treated rats were divided in groups with an equal average swelling volume. Each sample was dissolved in physiological saline containing 10% Nikkol and the solution was injected daily into the rat for 11 d starting on the day of grouping. Hydrocortisone was used as the positive control. The amount of IgE was measured by the passive cutaneous anaphylaxis method on the 15th day. 17 The results were expressed as average of plasma IgE level of 5 rats administered a total of 10 mg/kg of each test compound.
Inhibition on histamine release from rat peritoneal mast cells
Effects of curcumin 4‘-O-β-glycosides on histamine release from rat peritoneal mast cells were examined as follows. Peritoneal mast cells were collected from the abdominal cavity of rats (Male Wistar rats, Nippon SLC) and purified to a level higher than 95% according to the method previously described. 18 The purified mast cells were suspended in a physiological buffered solution containing 145 mM NaCl, 2.7 mM KCl, 1.0 mM CaCl2, 5.6 mM glucose, and 20 mM HEPES (pH 7.4) to give approximately 104 mast cells/mL. Cell viability was always greater than 90% as judged by the trypan blue exclusion test. Mast cells were preincubated with the test compound (1 μM) for 15 min at 37 °C, and subsequently exposed to compound 48/8018 at 0.35 μg/ml. Histamine release was determined by a fluorometric assay according to the previously reported method, 18 and was expressed as a percentage of total histamine.
Results and Discussion Synthesis of Curcumin 4‘-O-β-Glucooligosaccharides
Curcumin 4‘-O-β-D-glucopyranoside (2) was synthesized by almond β-D-glucosidase-catalyzed reverse-hydrolysis. Incubation of curcumin (1) with almond β-D-glucosidase in the presence of glucose in acetonitrile/water (9:1) solvent gave curcumin 4‘-O-β-D-glucopyranoside (2) in 19% yield.
Enzymatic glycosylation of curcumin 4‘-O-β-D-glucopyranoside (2) with CGTase afforded curcumin 4‘-O-β-glucooligosaccharides (Fig. 1). Soluble starch was used as a glucose-donor. Two glycosylation products 3 and 4 were isolated by the preparative HPLC on a YMC-Pack R&D ODS column. The compounds 3 and 4 were identified as curcumin 4‘-O-β-maltoside (3, 51%) and curcumin 4‘-O-β-maltotrioside (4, 25%) on the basis of HRFABMS, 1 H and 13 C NMR, H-H COSY, C-H COSY, and HMBC-spectra. 14
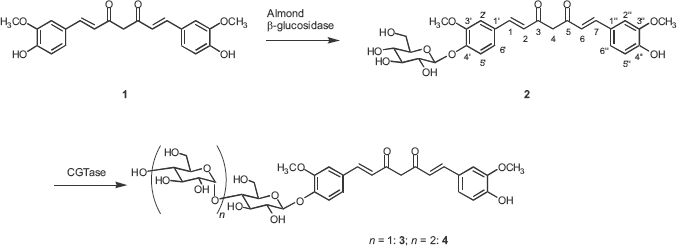
Synthetic route of curcumin 4‘-O-β-glycosides 2–4 through enzymatic glycosylations.
Anti-Allergic Activity
The suppressive action of curcumin 4‘-O-β-glycosides 2–4 on IgE antibody formation was investigated by in vivo bioassay using ovalbumin as an antigen. The average of rat plasma IgE level after treatment of ovalbumin with or without test compounds was examined and was summarized in Table 1. The inhibitory activity of curcumin 4‘-O-β-glycosides 2–4 was higher than that of hydrocortisone. Curcumin 4‘-O-β-D-glucopyranoside (2) exhibited the highest suppressive activity among the three curcumin 4‘-O-β-glycosides; the inhibitory activity decreased in the order of 2, curcumin 4‘-O-β-maltotrioside (4), and curcumin 4‘-O-β-maltoside (3). These results suggested that curcumin 4‘-O-β-glycosides 2–4 would act as useful anti-allergic agents.
Inhibitory effects of curcumin 4‘-O-β-glycosides 2–4 on IgE antibody formation.

Average of plasma IgE level of five rats treated with test sample.
Effects of curcumin 4‘-O-β-glycosides 2–4 on compound 48/80-induced histamine release from rat peritoneal mast cells were examined. A high level of histamine (control, 41%) was released from rat peritoneal mast cells, which had been stimulated with 0.35 μg/ml of compound 48/80 (Table 2). Curcumin 4‘-O-β-D-glucopyranoside (2) effectively inhibited the compound 48/80-induced histamine release from rat peritoneal mast cells;%inhibition of 1 was 70%. The inhibitory activities of curcumin 4‘-O-β-maltoside (3) and 4‘-O-β-maltotrioside (4) were 46 and 58%, respectively. These findings suggested that curcumin glycosides having β-glucoside and β-glucooligosides at its C-4’ position possess potent inhibitory activity toward histamine release from mast cells.
Inhibitory effects of curcumin 4‘-O-β-glycosides 2–4 on histamine release from rat mast cells.

Compound 48/80-induced histamine release from rat mast cells after treatment with or without test compound.
Recently, we reported that enzymatic glycosylation of physiologically active compounds drastically improved their water-solubility and that the glycosides acted as potential anti-allergic agents.11,19 The glycosides of vitamin E showed suppressive action on IgE antibody formation 11 and those of polyphenols exhibited inhibitory effects on histamine release from rat peritoneal mast cells. 19 The results obtained here demonstrate that two-step enzymatic glycosylation by almond β-D-glucosidase and CGTase is useful to prepare curcumin 4‘-O-β-glycosides and that curcumin 4‘-O-β-glycosides have potent anti-allergic activities.
Conclusions
Curcumin 4‘-O-β-glycosides, i.e. 4‘-O-β-glucoside, 4‘-O-β-maltoside, and 4‘-O-β-maltotrioside, were successfully synthesized through enzymatic glycosylation by almond β-D-glucosidase and CGTase. Three curcumin 4‘-O-β-glycosides showed suppressive action on IgE antibody formation and exhibited significant inhibitory effects on histamine release from mast cells. Curcumin 4‘-O-β-glucoside showed the highest inhibitory activity for both IgE antibody formation and histamine release among the curcumin 4‘-O-β-glycosides synthesized here.
Disclosures
The authors report no conflicts of interest.
