Abstract
Objectives
Curcumin has antioxidant and antiproliferative properties, and its therapeutic effect must be considered. Nanocurcumin capsules showed a potential increase against in vitro biological cancer. This study sought to determine how curcumin nanoparticles and nanocapsules affected the expression of p53, Bcl-2, Bax, and Bax in a liver cancer cell line (Hep-G2). Mechanisms of apoptosis were also examined in this cell line.
Methods
This study used quantitative real-time polymerase chain reaction (qRT-PCR) to analyze the p53, Bcl-2, Bax, and Caspase-3 gene pathways and to evaluate the molecular mechanisms responsible for the efficacy of curcumin nanoparticles (CNPs) and curcumin nanocapsules (CNCs) against liver cell lines. Flow cytometry was used to check for signs of apoptosis and the cell cycle.
Results
Curcumin nanocapsules produced by the ball milling process at 90 min significantly boosted the populations of apoptotic cells in a dose- and time-dependent manner. The mRNA expression analysis revealed that the proapoptotic Bax, Caspase-3, and the tumor suppressor gene p53 were upregulated throughout the process started by curcumin nanocapsules and decreased in the Bcl-2/Bax ratio.
Conclusion
This research provides a fresh understanding of the molecular mechanisms behind the liver cancer-fighting abilities of curcumin nanoparticles. Curcumin nanocapsules produced through a unique mechanical technique can be used as an anticancer agent.
Introduction
Future research in nanomedicine will have a significant influence on human health. 1 In medicine, nanoparticles (NPs) with one dimension less than 100 nm are used as an innovative method of delivering medicines, 2 proteins, DNA, and monoclonal antibodies. 3 Curcumin, berberine, quercetin, and hesperidin are just a few examples of the many natural polyphenolic chemicals that have been successfully delivered in the form of nanoemulsions, nanoparticles, liposomes, and films; thanks to recent developments in drug delivery methods, making them anticancer, antimicrobial, and antioxidant agents.4,5
The molecule of curcumin, or 1-(1E, 6E)-1,7-bis(4-hydroxy-3-methoxyphenyl)-1,6-heptadiene-3,5-dione, was symmetric. The research was done on diferuloylmethane (MW 368.38 gmol−1). 6 Curcumin’s chemical makeup is elucidated as 2-o-methoxyphenol linked to α, β-unsaturated diketone moiety through the 7-carbon linker. The hydroxyl and methoxy groups work as antioxidants and anticancer agents, respectively.
Malignant tumors develop when abnormal cells proliferate quickly and spread throughout the body. 7 Dysregulation of apoptosis and growth factor abnormalities cause cancer. 8 Hepatocellular carcinoma (HCC) is one of the most familiar diseases in the world, the third leading cause of cancer-related death, and accounts for more than 80,000 fatalities annually. 9 Several potential drugs were used as promising therapeutic targets for HCC, with varying degrees of inhibition of HCC.10,11
Curcumin, the brilliant yellow substance, is the principal curcuminoid found in turmeric (Curcuma longa). It is a dietary herbal supplement, a cosmetic component, and a food flavoring and coloring. 12 Curcumin has gained attention as an antibacterial, antioxidant, antiaging, and scavenger of free radicals. It was revealed that curcumin suppresses the proliferation of cancerous cells, including those that cause breast, stomach, colon, liver, lung, and cervical cancer. 13
In addition, Bcl-2 (B-cell lymphoma 2), Caspase-3 (cysteine-aspartic proteases), and Bax (Bcl-2-associated X) regulate both cell proliferation and apoptosis. 16 The most common mutation in human HCC cancers is in the tumor suppressor gene p53 (protein p53).
p53 is involved in cell cycle control, apoptosis, and maintaining genome integrity. The protein encoded by this gene acts as a tumor suppressor, activating transcription, binding DNA, and forming oligomers. In response to various stimuli, the encoded protein controls the expression of target genes, triggering events such as cell cycle arrest, apoptosis, senescence, DNA repair, and metabolic alterations. 17 As a result of its function in preserving stability by avoiding genome mutation, it is known as “the guardian of the genome.” However, p53 content in cells needs to be controlled. High levels of p53 may speed up aging by causing excessive apoptosis while suppressing tumors. Mdm2 (Mouse double minute 2 homolog), which may cause the ubiquitin system to start degrading p53, is the primary regulator of p53. The tumor suppressor p53 relies on Mdm2 as an essential negative regulator. 18
Curcumin’s gastrointestinal digestion was improved by the encapsulation process, which improved bioavailability, showed outstanding stability, and increased superoxide dismutase, catalase, and glutathione peroxidase activities. 19 In the current study, CNPs and CNCs were tested in a human liver cancer cell line to see how they affected gene expression for Bax, Bcl-2, p53, and Caspase-3 (Hep-G2). We have also investigated the possible mechanistic function of apoptosis in this cell line.
Materials and methods
Curcumin nanocapsules preparation
Curcumin nanoparticles (CNPs) were prepared using a mechanical ball milling process (Model: PQ-N2 Planetary Ball Mill, Gear Drive 4-Station Planetary Ball mill, 220 v) 20 at a spin speed of 40,000 r/min for 90 min. Powder of ground curcuma was stirred and extracted with ethanol alcohol 95% using a magnetic stirrer at room temperature for 3 hours 21 using the homogenization model (Homogenizer PRO 400 PC, Germany) in a matrix comprising sodium alginate and Tween 20 (T20), for CNC preparation. One gram of curcumin extract was added to a sodium alginate gel and T20 solution. The emulsion was produced in a high-pressure homogenizer by mixing the solution at 18.000 r/min for 30 min.
Transmission electron microscopy (TEM)
Transmission electron microscopy assessed the morphology and characterization of CNC. Specimens were then observed at an accelerating voltage and viewed under the microscope, and the diameter was obtained from micrographs.
Cell culture and treatments
This research used the Hep-G2 cell line (ATCC® HB-8065TM) established to investigate hepatocellular carcinoma. The cells were grown in RPMI-1640 L-glutamine medium with 10% fetal bovine serum and penicillin–streptomycin antibiotic in a humidified incubator with 5% CO2 and 37°C. Trypsin/EDTA sub-culturing was performed as standard procedure twice in a week. A stock solution of CNPs and CNC was diluted with a complete RPMI-1640 medium to provide the following concentrations: 50, 100, 150, 200, and 250 μmol/L. On a 96-well tissue culture plate with full RPMI-1640 growth media, 10 × 103 Hep-G2 cells were planted per well, and the plate was incubated at 37°C for 24 h to allow the cells to adhere. Forty-eight hours of development in a humidified 5% CO2 environment were followed by cell attachment. At that point, the culture medium was aspirated and substituted with 200 μl of new, complete growth media with varied NPs and CNC concentrations (3 wells per dosage). The MTT test was used to determine cell viability. 20 μl MTT (5 mg/mL in PBS) was applied to each well, and the plate was incubated at 37°C for 3 hours. 22 After 48 h in a humidified 37°C 5% CO2 incubator, Hep-G2 cells were either left untreated (control) or treated with curcumin nanoparticles and capsules (100 μmol/L).
The Hep-G2 cell line was seeded in 96-well E-plates and treated with a range of concentrations (100 μg/mL) of either CNPs or CNCs to test their cytotoxic effects on the cell lines.
This was accompanied by photometric detection of the absorbance at 590 nm utilizing the microplate ELISA reader (FLUOstar OPTIMA; BMG Labtech GmbH, Offenburg, Germany). The program was used to standardize the cell index, and sigmoidal dose-response analysis was used to calculate the IC50 values and activity.
RNA extraction and cDNA synthesis
Following treatment, the medium was discarded, and the cells were rinsed in ice-cold PBS. The mRNA expression of p53, Bcl-2, Bax, Caspase-3, and the housekeeping (control) gene βeta-actin was analyzed in the human liver cancer cell line (Hep-G2 ATCCHB-8065).
Homogenates were prepared from liver cell lines, and total RNA was extracted using Trizol reagent (Invitrogen, Carlsbad, CA) per the manufacturer’s protocol. After being diluted in RNase-free water, the RNA was measured spectrophotometrically.
Gel electrophoresis on an ethidium bromide–stained 1% agarose gel was used to evaluate the RNA’s integrity. The SuperScript Choice System (Life Technologies, Breda, Netherlands) synthesized first-strand cDNA from 2 mg total RNA and 0.5 mg of oligo(dT)12e18 primer in a final volume of 12 mL. After heating the mixture to 70°C for 10 min, 20.5 μl of a solution comprising 50 mmol/L Tris-EHCl (pH 8.3), 75 mmol/L KCl, 3 mmol/L MgCl2, 10 mmol/L DTT, 0.5 mmol/L dNTPs, 0.5 μl RNase inhibitor, and 200u Superscript Reverse transcriptase were added. After 1 h of incubation at 42°C, the mixture was stored at −80°C for later use.
Real-time PCR
The gene expression was investigated in the cells treated with 100 μg/mL for the CNP sample and CNC sample.
Primers sequence for all studied genes.

To standardize the fold change in gene expression between treated and untreated cells, an assay was developed to measure β-actin expression levels; no-template controls were put on the same plate to ensure no reagent contamination. Relative gene expression (ie ΔΔ Ct) was used to analyze the RT-PCR results. Each gene’s ΔCt was normalized to the average ΔCt of Hep-G2 cells that had not been treated. By utilizing the following equation, we calculated the fold change in target gene expression between treated and untreated cells, normalized to the expression of β-actin: fold change = 2−Δ(ΔCt), where ΔCt = Ct(target) −Ct(β-actin) and Δ(ΔCt) = ΔCt (treated) −ΔCt (untreated). The Wilcoxon rank-sum test was used to compare ΔCt values (cycle counts at the threshold level of log-based fluorescence adjusted to the β-actin control gene) with a two-sided p < 0.05 denoting statistical significance from the real-time PCR data. Results were represented as mean and standard deviation (SD). Fold changes in gene expression were calculated using the mean differences in ΔCt (ΔΔCt) using the following formula: fold = 2−ΔΔC.23,24
Analysis of Apoptosis by flow cytometry
The mode of cell death was investigated in the cells treated with the samples at 100 μg/mL for the CNP and CNC samples. Cell death was determined following the manufacturer’s procedure using Annexin V and propidium iodide (PI) staining (Exbio, Vestec, Czech Republic). According to Taghi et al. (2018), the cells were examined using a flow cytometer (Coulter Epics, Navios, USA) to determine the apoptosis percentage. 25
The flow cytometric cell cycle analysis
The cell cycle arrest was investigated in the cells treated with 100 μg/mL for the CNP sample and CNC. The DNA PREP Kit was used to analyze cell cycle distribution in the treated samples (Beckman Coulter USA, Inc., Brea, CA 92821). Using the IC50 concentration of curcumin, monolayer cells of the Hep-G2 cell line were tested for 5 minutes. The ultimate cell concentration was between 3–5 × 106 cells/ml, and it was achieved by adding 100 μL of single-cell suspension to 100 L μL of LPR. The final step was adding 2 mL of DNA PREP Stain and incubating it for 30 min (Beckman Coulter Epics XL).
Flow cytometric analysis of cell cycle arrest
Once the cells were grown, processed, and collected, the monolayers were washed and preserved in 90% ethanol at −20°C overnight. Cells were stained for 15 min at room temperature, out of direct sunlight, using PI/RNase staining solution (BD Biosciences, CA, USA). Using instrument software, the percentages of cells in each cell cycle phase were determined from a sample of 10,000 events acquired by flow cytometry (FACS, BD FACS CaliburTM, BD Biosciences, CA, USA). 26
Statistical analysis
For analyzing real-time PCR data, we used the Wilcoxon rank-sum test to compare ΔCt values (cycle counts at the threshold level of log-based fluorescence adjusted to the
Results
Characterization of curcumin nanocapsules
Transmission electron microscopy of the prepared curcumin nanocapsules proved that the new milling technique was fulfilled to enroll CNC in the nanorange. TEM image (Figure 1) showed pictures of spherical micelles that have the size of nanocapsules ranging between 7 and 42 nm according to the size distribution. Transmission electron microscopy (TEM) images of curcumin nanocapsules CNC.
Cytotoxicity assay (MTT)
The derivative CNC was the best cytotoxic compound with an IC50 of 1.3 μM compared to CNP with an IC50 of 1.8 μM against Hep-G2 cells.
Treatment with CNP and CNC nanocomposite triggers apoptosis in Hep-G2 cells
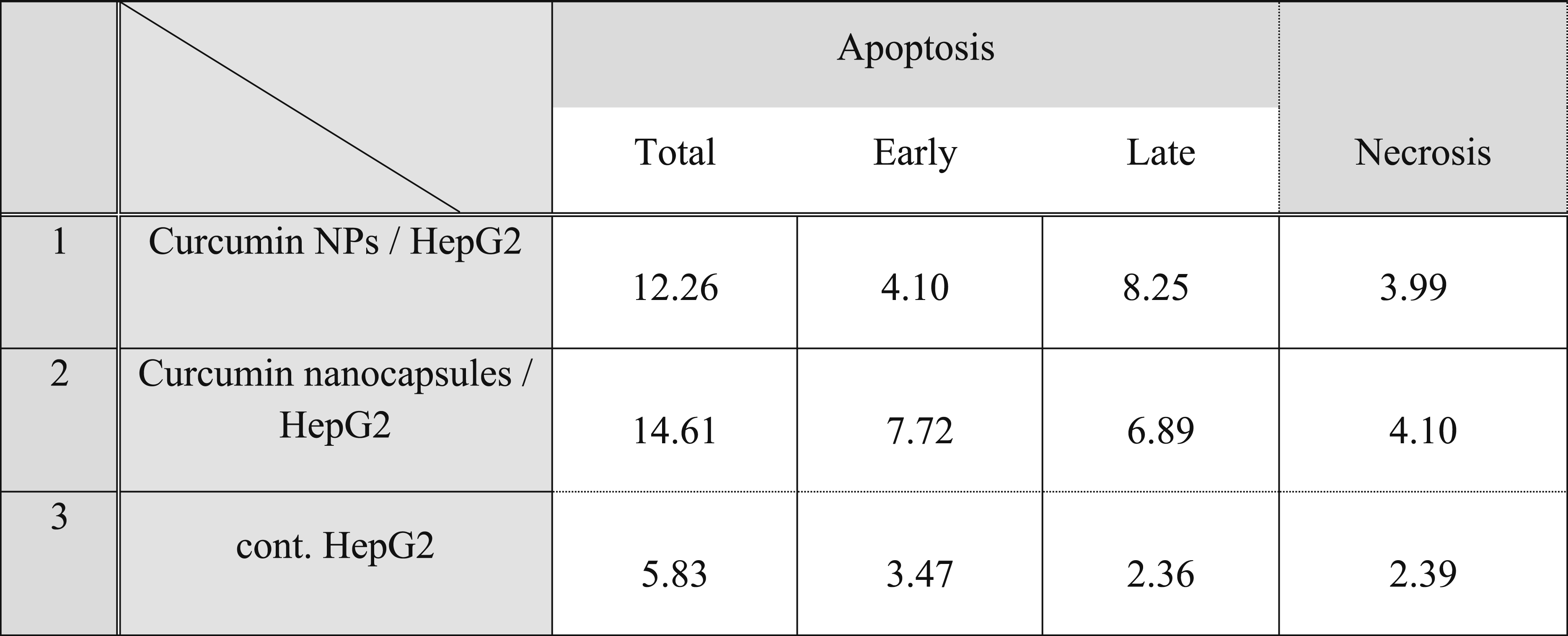
The proportion of cells experiencing early and late apoptosis and necrosis.


Flow chart of the effect of CNPs and CNC capsules on the apoptotic percentage of Hep-G2 cells.
In contrast, to control, CNP considerably raised the proportion of cells going through early apoptosis (to 4.10%) and late apoptosis (to around 8.25%) in the overall cell population. For the overall cell population, 12.26% became apoptotic when treated with CNP, compared to 14.61% when treated with curcumin capsules nanocomposite.
CNP and CNC treatment induce Hep-G2 cell cycle arrest
We use flow cytometry to analyze the effect of CNP and CNC on the cell cycle profile of Hep-G2 cell proliferation. Our cell cycle analysis revealed that untreated cells showed a predictable cell cycle pattern for continuously growing cells. In contrast, treatment with CNPs for 24 h caused an accumulation of cells’ percentage at pre-G1 compared to untreated Hep-G2 cells. In the pre-G1 phase, there was also an accumulation of cells treated with CNCs compared to the control. Treatment with CNC significantly increased the cell populations at G0/G1 phase compared to the CNPs-treated cell. While treatment with CNPs and CNCs significantly reduced the cell populations at G0/G1 phase compared to the control. Concomitant decreasing value had been shown from CNPs toward CNCs in pre-G1 and G2/M cycle growth arrest Figure 3. The influence of curcumin NPs and capsules on cell cycle analysis. Bars are standard errors.
After being treated with nanocapsules, several cells were seen to be accumulated in the G2/M phase. For curcumin NPs, G2/M scored an accumulation of 31.18%, while curcumin nanocapsules proved that 28.94% of the cells were in the G2/M phase. The S-phase population had no significant difference between control and curcumin NPs or nanocapsules Figure 3.
Expression analysis of Bax, Bcl-2, Caspase-3, and p53 genes using real-time PCR
The human hepatic cancer cell line tested using real-time technology showed amplification of Bax, Bcl-2, Caspase-3, and p53 genes. Standard curves and the PCR amplification efficiencies for the housekeeping gene, βeta-actin, were successfully obtained. Linear regression for standard curve analysis showed a high correlation for βeta-actin. The number of targeted genes was extra plotted from a standard curve equation, and the relative quantification of human cell lines examined was calculated.
Fold change in the expression of Bax, Bcl-2, Caspase-3, and p53 genes using real-time PCR.

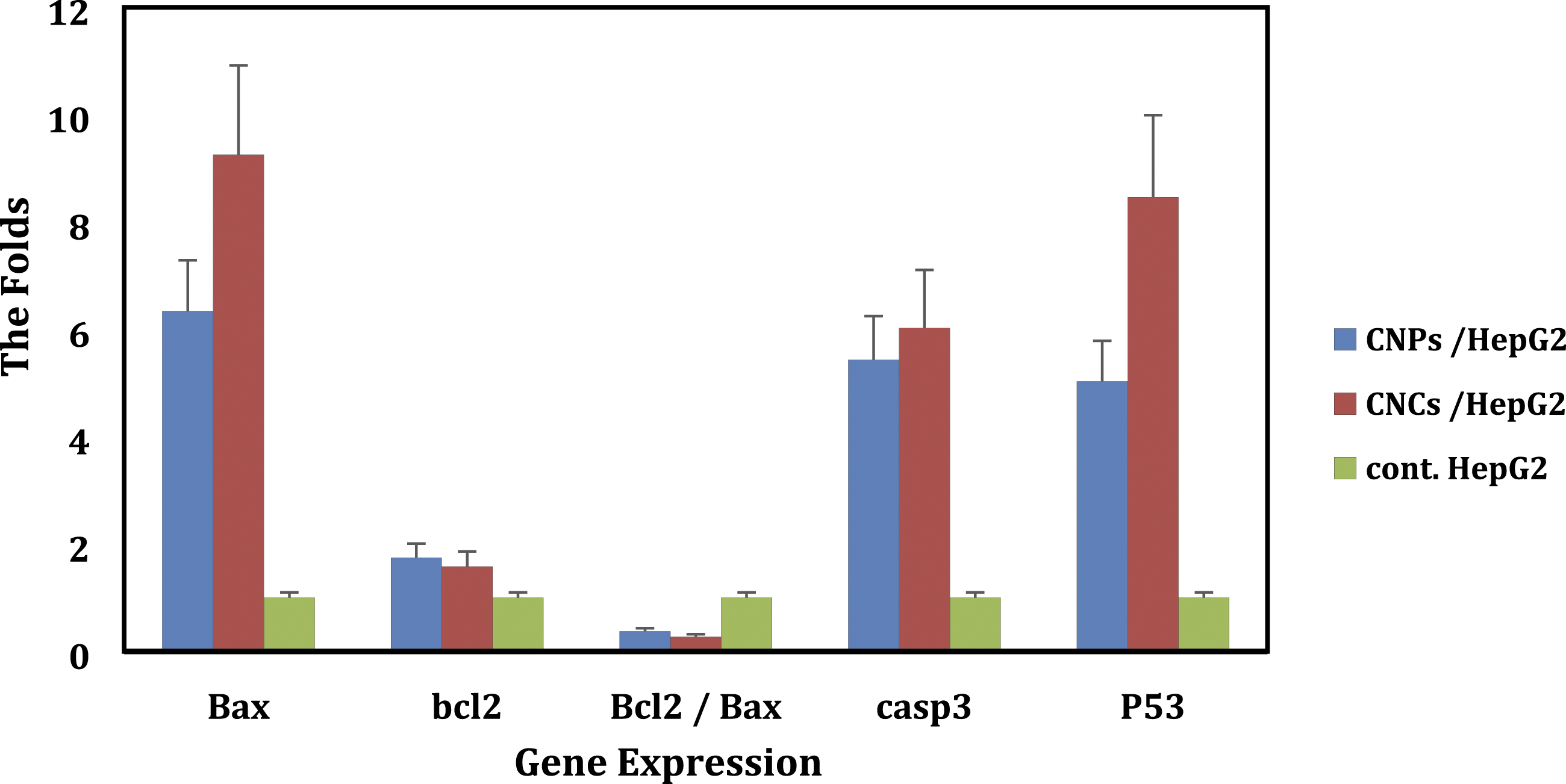
Influence of curcumin NPs and NCs on gene expression. Bars are standard errors.
The tumor suppressor gene, p53, controls apoptosis, slowing cell growth. This research studied the effect of CNPs and nanocapsules on p53 gene expression. Figure 4 revealed that curcumin NPs and nanocapsules significantly increased p53 mRNA expression by 5.03- and 8.47-fold, respectively. There was an increase in fold change of p53 mRNA expression followed by curcumin nanocapsule treatment compared to curcumin NPs (Table 3 and Figure 4). Alternatively, p53 can produce these effects through transcription-independent apoptosis, mainly achieved by the interaction between the antiapoptotic Bcl-2 proteins and p53. This response, which occurs in response to apoptotic stressors, has the following effect: As cytoplasmic p53 rushes to the mitochondria, it binds to antiapoptotic Bcl-2 proteins and releases proapoptotic Bax from the antiapoptotic protein complex.
Discussion
Using natural products does not result in a rise in the toxicity of cancer medications as could be anticipated. In any event, they might increase their anticancer activity; as a result, using natural ingredients enables existing anticancer drugs to be used in lower quantities and raises their therapeutic index. 27 The precise and effective delivery of anticancer herbal medications may be aided by nanotechnology. Nanomedicine has emerged as the primary silver lining given the generally depressing reality of cancer. 28 Curcuma longa (turmeric) is a common spice, a source of the coloring compound curcumin, and a spice. 29 According to research, the constituents of Curcuma longa L. include sodium curcuminate, ar-turmerone, methylcurcumin, demethoxycurcumin, and bis-demethoxycurcumin. Curcumin is one of the most often utilized native compounds found in the essential fraction of Curcuma longa L. It has various defensive properties. 30 Our research concentrated on ball milling and nanoencapsulation method due to limited water solubility, low oral bioavailability, and quick curcumin metabolism. The potential for treating and preventing several human illnesses exists with nanocurcumin, a promising therapeutic candidate with advantageous medicinal qualities. 31
Our study mainly focuses on apoptosis and the hepatic cell cycle to comprehend the method of eliminating cancerous cells. The balance of apoptotic proteins strictly controls the intrinsic mitochondrial pathway and the extrinsic death receptor route, which are the two pathways most often involved in apoptosis. 32
The transmission electron microscopy picture reveals that the nanocurcumin particles were round and polydisperse, with an average diameter of around 15 ± 1.5 nm. TEM analysis confirmed that most of the nanoparticles were spherical, with a smooth surface, similar to the findings of Chuacharoen and Sabliov. 33 That was correlated with our images of spherical micelles—validated nanocapsules measuring between 7 and 42 nm in size. The free curcumin powder that Zhao et al. saw had an irregular shape and an average size of around 3.58 m. Because the size of the ball-milled curcumin powder is decreased by a factor of 100 compared to that of free curcumin powder, it can be delivered effectively through the body’s cell membrane. Previous studies indicated that curcumin’s aqueous solubility would be increased and maintained at physiological pH in vitro 34
Our flow cytometry measurement by PI/Annexin V also revealed that curcumin NPs led to total apoptosis of (12.26%) in treated cells. Forming curcumin NCs enhanced the early and total apoptosis in treated carcinoma cells within 24 h (>95%). These findings agreed with Mulik et al., 35 who revealed enhancement of in vitro anticancer efficacy by inducing apoptosis in solid lipid nanoparticles mediated by transferrin. Also, Li et al. 36 found that curcumin/TPP-CZL nanomicelles may help liver cancer cells undergo apoptosis. Adding curcumin nanoparticles to the drug’s composition enhanced its impact on cells.
Curcumin NPs and capsules are essential to decreasing cancer cell proliferation, including apoptosis and cell cycle arrest. The effects of curcumin NPs and nanoencapsulation were investigated to gain more evidence for cell proliferation inhibition. Curcumin nanocapsules might disrupt membrane structure and the cytoskeleton. That could imply that curcumin increased the expression while destroying the cytoskeleton. According to our research, Hep-G2 cells were vulnerable to the effects of curcumin regarding cell death. Cell cycle analysis showed that curcumin NPs and NCs caused cell cycle block at the G2/M phase, which agrees with Jiang et al., 37 who stated that numerous anticancer drugs that target the microtubules had extended mitotic arrest by triggering cell G2/M arrest.
Cell death in anticancer treatment often occurs by a toxicity-driven mechanism, such as apoptosis, oncosis necrosis, pyroptosis, or autophagy. 15 Mitochondrial stress, membrane abolish mediated cell death, autophagy, neutrophil apoptosis, and Golgi damage–induced adhesion loss are all mechanisms involved in nanoparticle-induced cell death. 29 Although CNPs have been shown to limit cancer cell proliferation and even trigger cell death, little emphasis has been devoted to the mechanisms underlying these effects, especially at the omics level. This parallels a recent publication stating CNPs triggered cell death via p53’s direct mediation. 8
As mentioned in the research, cancer develops by inhibiting cell death and uncontrolled cell proliferation. That is due to the resistance of tumor cells to apoptosis and to survive and metastasize. For these spots, therapeutic targets can initiate genetic modification and change apoptotic pathways in pathogenesis. Liu et al. 38 reached the diplomatic design pathways destroying cancer cells by activating apoptosis signaling selective pathways with no harmful effect on normal cells.
Many biological systems that interfere with one another make measuring the complex apoptotic pathway challenging. Many genes, including Bcl-2 and Bax, are involved in the intricate process of apoptosis, with a focus on the Bcl-2 family. According to previous research, the Bcl-2 family could negatively and favorably influence apoptosis and play various roles between apoptosis and proliferation.39,40 Bax (associated Bcl-2 X) is a death agonist because it speeds up apoptosis when cells overexpress it in response to death signals. On the other hand, death is suppressed when Bcl-2 (B-cell lymphoma 2) is produced because of its heterodimerization with Bax. 41
We looked into the apoptosis suppressors Bax and Bcl-2 to gauge the protection of hepatocytes to determine the effect of CNPs and CNCs. We looked into the gene expression associated with the apoptotic pathway to better understand how CNPs and NCC increase the apoptotic impact. Real-time PCR analysis showed that treatment with CNP and CNC results in a striking reduction in Bcl-2 expression levels, while Bax mRNA levels increased compared to individual exposure. Furthermore, the apoptotic index demonstrated a remarkable increase in the Bax in CNC-treated cells compared to that in CNPs-treated and control cells (Figure 4). There are some theories put out to describe why Bcl-2 can decrease apoptosis. In other words, increased Bax expression promotes sensitivity to apoptotic agents and inhibits tumor growth, according to Bargou et al. 42
The mitochondrial apoptotic pathway is the primary regulator of the ratio of the apoptotic proteins Bcl-2 and Bax. Bcl-2 acts as an apoptosis regulator and helps Bax launch the process by blocking the release of cytochrome C from mitochondria. A balance between the expression levels of the apoptosis-regulating genes Bcl-2 and Bax is essential. This happens because cytochrome C efflux from mitochondria is triggered, and the pathway is activated when the Bcl-2/Bax ratio is elevated. Thus, changes in Bcl-2 and Bax expression increase the probability of apoptosis in cells. Figure 4 shows that CNC treatment reduced the Bcl-2/Bax ratio and activated Caspase-3, which led to cell death. Our result agreed with Muangnoi et al., who found a larger value in untreated Hep-G2 cells than those treated with nanocapsules. 43 Bcl-2 gene expression can help cells survive without optimal cell culture concentration. Treatment with curcumin nanocapsules attracted the inhibition of the antiapoptotic/prosurvival members of the Bcl-2 family of proteins.
Killer caspase or Caspase-3 has been called with Caspase-6 and 7, and their cleavages have expressions with Bax and Bcl-2. In cancer cell lines, curcumin damages the mitochondria and triggers Caspase-dependent apoptosis. 44 This study showed that CNPs and nanocapsules treated Hep-G2 cell lines resulted in upregulation of Caspase-3 mRNA compared to control untreated Hep-G2 cell lines.
In the present study, the increase in Caspase-3 expression by CNC-treated cell lines is greater than that of those treated with CNPs compared to the untreated cell lines under control. Our findings concord with those reported by Hussar, 45 who stated that pro- and antiapoptotic components of Bcl-2 and its related proteins are combined with survival signals connected to the production of mitochondrial cytochrome C and the activation of caspases. Caspases are grouped into effectors (Caspases 3, 6, and 7) and initiators (Caspases 8 and 9) based on their function and the order in which they are turned on during apoptosis. In the present study, the tumor suppressor gene p53 increased with CNPs and CNC-treated cell lines compared to the control cell lines. Our study found that the p53 was raised with CNC-treated cell lines more than that treated with CNPs. This was correlated with Ting Liou et al., 46 who stated that p53 in Hep-G2 cell lines treated with CNPs exhibited a mild reduction.
Limitation
This study has a significant flaw in that a power analysis was not used to estimate the sample size.
Conclusion
Finally, we concluded the multiple health advantages of curcumin nanoparticles and capsules. It is the appropriate agent for further research to build more potent medicines against liver cancer because of its affordability and capacity to target numerous components of the disease. Increasing proapoptotic proteins (such as Caspase-3 and Bax) and decreasing antiapoptotic proteins (Bcl-2 and p53) inhibit the growth of liver cancer. Further research on curcumin as an addition to current anticancer treatments might be considered. Hep-G2 cells treated with CNPs and CNCs experience G2/M phase arrest, paraptosis, and apoptosis.
Footnotes
Acknowledgements
The authors thank Prof. Dr. Mahmoud M. Kamel for his support with the flow cytometry results.
Author’s contribution
M.M introduced the research idea. M.M and E.S did the experiment. G.N collected and interpreted the results. A.O did statistical analysis. All authors wrote the first draft. All of the authors approved the final revision.
Data and materials availability
The ability to send requested information, data, or materials is guaranteed.