Abstract
Three simple spectrophotometric and atomic absorption spectrometric methods are developed and validated for the determination of moxifloxacin HCl in pure form and in pharmaceutical formulations.
Introduction
Moxifloxacin [1-Cyclopropyl-b-fluoro-1,4-dihydro-8-methoxy-7-[(4aS,7aS)-octahydro-6H-pyrrolo [3,4-6] pyridine-6-yl]-4-oxo-3-quinoline carboxylic acid] 1 is a broad-spectrum antibiotic that is active against both Gram-positive and Gram-negative bacteria. 2 The bactericidal activity of the drug is mediated by the inhibition of DNA gyrase (topoisomerase II) and topoisomerase IV, essential enzymes involved in bacterial DNA replication, transcription, repair and recombination. Moxifloxacin is prescribed for the bacterial infections of the respiratory tract including sinusitis, community acquired pneumonia and acute exacerbations of chronic bronchitis. 3
HCl
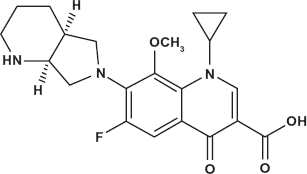
Few methods were reviewed in the literature for the analysis of moxifloxacin HCl. Spectrophotometric,4,5 spectrofluorimetric, 6 liquid chromatographic,7–10 TLC, 11 HPLC,12–16 Capillary electrophoresis,17,18 polarographic, 19 and voltammetric20,21 procedures were applied for its determination.
Still, there is a need for simple methods to compete with the new, advancement and automated ones. Therefore, the present study aims to use spectrophotometric and atomic absorption spectrometric (AAS) techniques for the determination of moxifloxacin HCl in pure form and pharmaceutical formulations. Methods A and B are based on the oxidation of the drug by Fe3+ in the presence of (o-phen) or (bipy) and then the tris-complex formed was measured at 510 and 520 nm respectively. While method C based on the precipitation of the ion pair of the drug with bismuth (III) tetraiodide and quantifying it via the formed precipitate of the metal ion present in the supernatant solution using (AAS). The proposed methods were successfully applied to the determination of moxifloxacin HCl in tablet dosage forms without interference of any additives or excipients.
Experimental Apparatus
A Shimadzu recording spectrometer UV-1800 equipped with 10 mm two matched quartz cells was used for spectrophotometric measurements. Atomic absorption measurements were carried out using Shimadzu atomic absorption spectrometric device model AA-640-13 at 223 nm analysis wavelength, lamp current 5 mA, slit width 0.38 nm, burner height 5 mm, burner slot, flame 10 cm air-C2H2, support gas flow 10 I min-1, fuel gas flow 2.6 I min-1 and absorption sensitivity of 0.6 ppm.
Materials and Reagents
All materials used were of analytical reagent grade, water was always doubly distilled. Pure sample moxifloxacin HCl was kindly provided by Sabaa International Company for pharmaceuticals and chemical industries S.A.E.
The Standard stock solution (0.8 mg ml-1) in water were prepared by dissolving 80 mg of pure drug in convenient amount of double distilled water in 100 ml volumetric flask followed by dilution to the mark with the same solvent, it is stable for at least 2 weeks if it was stored in a cool (<25 °C).
Pharmaceutical preparations: Moxifloxacin tablets (400 mg moxifloxacin HCl/tablet) were provided by Sabaa International Company for pharmaceuticals and chemical industries S.A.E. (Batch No. 09002).
Iron (III)-o-phenanthrolin 22 was prepared by mixing 0.198 g of 1,10 phenanthroline monohydrate (Aldrich Chem. Co. Miluwakee, USA), 2 ml 1 M HCl and 0. 16 g ferric ammonium sulphate dodecahydrate (Aldrich, Germany) before dilution with double distilled water to 100 ml in a calibrated flask. Iron (III) – bipyridyl 22 was prepared by mixing 0.16 g of 2,2’ bipyridyl (Sigma Chem. Co. Miluwakee, USA) with 2 ml 1 M HCl and 0.16 g ferric ammonium sulphate dodecahydrate, before dilution with double distilled water to 100 ml in a calibrated flask.
Standard bismuth (III) solution, 0.01 M, was prepared by dissolving 0.1 g of Bi(NO3). 5H2O (Merck) in 2.5 ml of HNO3 and adding double distilled water to 25 ml and standardized complexometrically. 23 Potassium iodide solution, 0.5 M, was prepared by dissolving 8.28 g of KI (Merck) in 100 ml of double distilled water. 2% HNO3 solution was prepared in double distilled water. 0.8 × 10-3 M solutions of bismuth (III) nitrate and drug were prepared.
General Procedures
Methods A and B
(0.8-6) (0.8-4) μgml-1 aliquots of the standard solutions for methods A and B respectively were transferred to a series of 10 ml calibrated flasks. 4 ml of Fe3+-o-phen (method A) or 3.5 ml of Fe3+-bipy (method B) were added, then heating on a boiling water bath for 35, 30 minutes for methods A and B respectively. Mixture was cooled to room temperature (25 °C ± 1 °C), completed to volume with double distilled water. The coloured complexes formed were measured at 510 and 520 nm against a reagent blank treated similarly according to methods A and B respectively.
Procedures for the Kinetic Method
Aliquots of (0.8-6 μgml-1) of moxifloxacin HCl were assayed as in the general procedure for method A at different times (10, 25, 40, 60 minutes) in a boiling water bath.
Method C
Procedure 1
Spectral data for determination of moxifloxacin HCl using methods (A, B and C).
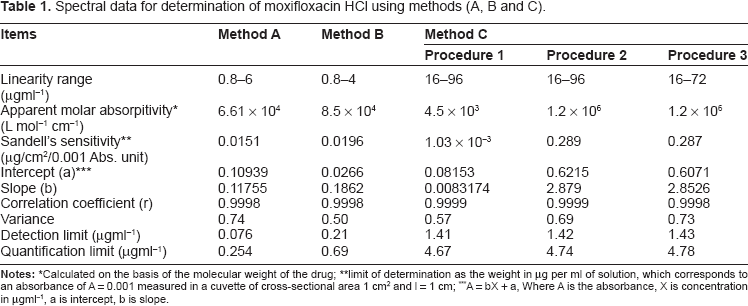
Calculated on the basis of the molecular weight of the drug
limit of determination as the weight in μg per ml of solution, which corresponds to an absorbance of A = 0.001 measured in a cuvette of cross-sectional area 1 cm2 and l = 1 cm
A = bX + a, Where A is the absorbance, X is concentration in μgml-1, a is intercept, b is slope.
Procedure 2
The precipitate in procedure 1 was quantitatively decomposed into 10 ml volumetric flasks using 1 ml concentrated HCl, the mixture was completed to 10 ml with double distilled water and aspirated directly in the atomic absorption spectrometer, absorption was measured at 2230 A° against an appropriate blank prepared simultaneously. Concentration of the consumed bismuth was calculated from a calibration graph of standard Bi(NO3) solution or using regression equation.
Procedure 3
The filtrate of procedure 1 were transferred to 25 ml volumetric flask, diluted to volume with double distilled water, absorbance was then measured using atomic absorption spectrometer at 2230 A° against an appropriate blank prepared simultaneously, excess concentration was then determined from a calibration graph of standard Bi(NO3) solution or using regression equation. The concentration of the drug was then calculated where 15.27 μgml-1 Bi (III) = 16 μgml-1 drug.
Procedure for Dosage forms
An accurately weighed quantity of the pulverized tablets equivalent to 80 mg of the studied drug was extracted with double distilled water. Mixture was filtered through a filter paper and washed with water, the filtrate and washing were collected in a 100 ml standard flask and diluted to volume with double distilled water. Aliquots of this solution were transferred in a series of 10 ml volumetric flasks, and the analysis was completed as previously mentioned using methods A, B and C by using standard addition technique.
Results and Discussion
Methods A and B
1,10 (o-phen) and 2,2'(bipy) are common reagents for ferrous ion as their red color chelate [Fe(phen)3]2+, [Fe(bip)3]2+ complexes remained stable for weeks. These methods were based on the reducing properties of the drug, it reduces Fe(III) to Fe(II) which was converted rapidly to the corresponding stable coloured reagent Fe(II) complex. (Fig. 1).
Reactions of moxifloxacin HCl with 1,10 (o-phen) and 2, 2'(bipy).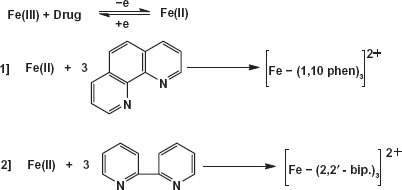
Absorption spectra were measured at 510 and 520 nm for 1,10 (o-phen) and 2, 2'(bipy) respectively (Figs. 2 and 3).
Absorption spectra of the complex formed through reaction of 4.8 μgml-1 Moxifloxacin HCl with 1,10 phenanthrolin and FeCl3. Absorption spectra of the complex formed through reaction of 1.8 μgml-1 Moxifloxacin HCl with 2,2’ bipyridyl and FeCl3.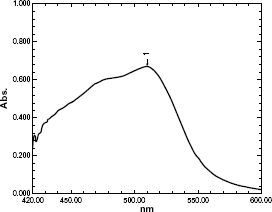
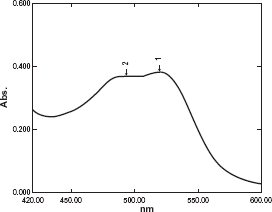
Optimum conditions affecting the reaction were studied:
Effect of reagent volume: It was found that 4 and 3.5 ml of 1,10 (o-phen) and 2,2'(bipy) respectively were suitable to give optimum results with moxifloxacin HCl. Effect of temperature and heating time: At ambient temperature (25 °C ± 2 0C), the reaction was very slow, when temperature increases, the reaction was faster until it reach the maximum absorbance in a boiling water bath (100 ° C). Heating for 35-30 minute in a boiling water bath gave the maximum absorbance for methods A and B respectively. Effect of solvent: Different solvents such as water, ethanol and isopropyl alcohol were tried in dilution, water was found to be the most suitable solvent.
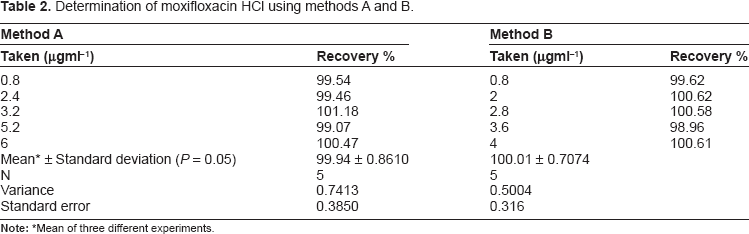
Determination of moxifloxacin HCl using methods A and B.
Mean of three different experiments.
Results and Discussion of Kinetic Spectrophotometric Procedure for Determination of Moxifloxacin with 1,10 (O-Phen) and FeCl3
The rate of the reaction was found to be dependent on the drug concentration, the rate was followed at 100 0C with various concentrations of the studied drug in the range of (0.8-6) μgml-1 (Fig. 4).
Absorbance versus time graphs for the reaction between moxifloxacin HCl and 1,10 phenanthrolin showing the dependance of the reaction on drug concentration (1) 1.826 × 10-6; (2) 5.48 × 10-6; (3) 7.307 × 10-6; (4) 1.187 × 10-5; (5) 1.370 × 10-5.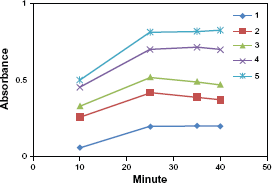
It is clear that the rate increases as the studied drug concentration increases, indicating that the reaction rate obeys the following equation:
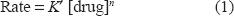
where K' is the pseudo-order constant of the reaction and n is the order of the reaction. The rate of the reaction may be estimated by the variable-time method measurement 25 as ΔA/Δt, where A is the absorbance and t is the time in seconds. Taking logarithms of rates and concentration (Table 3) equation (1) is transformed into:
Logarithms of the rates for different concentrations of moxifloxacin hydrochloride at constant concentration of 1,10 (o-phen) and FeCl3.

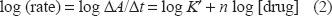
Regression of log (rate) versus log (drug) gave the regression equation:
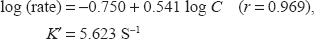
Hence the reaction is first order (n ≈ 1) with respect to drug concentration.
Evaluation of the Kinetic Methods
The quantitative of the studied drug under the optimized experimental conditions outlined above, would result in a pseudo-first order reaction with respect to its concentration. However, the rate will be directly proportional to drug concentration in a pseudo-first order rate equation as follows:
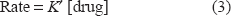
Rate-Constant Method
Values of K' calculated from slope of log A vs. t graphs multiplied by -2.303 for different concentrations of moxifloxacin hydrochloride and constant concentration of reagent.

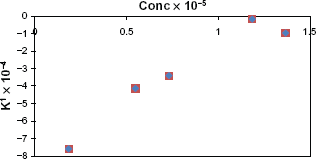
Values of K' calculated from slopes of different log A vs. t graph multiplied by -2.303 for different concentrations of drug at constant concentration of reagent.
Regression of (C) versus K' gave the equation:
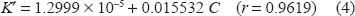
The value (r) indicates poor linearity, which is probably due to inconsistency of K' as a result of slight changes due to the elevated temperature of the reaction.
Fixed-Concentration Method
Values of reciprocal of time taken at fixed absorbance for different rates of variable concentration of moxifloxacin hydrochloride.

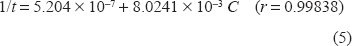
The range of the concentration of the studied drug giving the most acceptable calibration graph with the above equations was very limited, which could be disadvantage.
Fixed Time Method
Calibration equations for moxifloxacin hydrochloride at different fixed time over the range of 1.826 × 10-6 -1.370 × 10-5 M in presence of constant concentration of reagent.
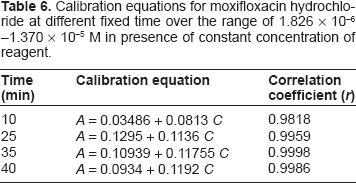
After optimizing the reaction conditions, the fixed time method was applied to the determination of moxifloxacin hydrochloride in pure form and in pharmaceutical formulations over the concentration range of (0.8-6 μgml-1). Analysis of the date gives the following regression equation:
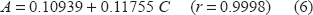
Method C
Formation of ion- pairs between many nitrogenous drugs and metal complexes found wide applications in the field of drug analysis applying colourimetric and (AAS) methods as well as ion-selective electrodes.28–37 In this work the investigated drug react with bismuth (III) tetraiodide to form stable ion- pair complex, this interaction and subsequent formation of the ion- pair occur in acidic medium via the two centers of tertiary amino group of the drug, one of them was formed due to the addition of acid) and two molecules of bismuth (III) tetraiodide. Being insoluble in aqueous solution, this ion- pair complex might be possibly isolated by direct filtration or extraction into organic solvents and assayed accurately without interference from excess unreacted metal complex.
Bismuth (III) tetraiodide complex was used as a reagent for the determination of some nitrogenous compounds.35–37 On mixing aqueous solutions of bismuth (III) tetraiodide complex and drug in acidic medium, a reddish orange precipitate instantaneously appeared that is attributed to the ion- pair formed in the reaction. This precipitate was filtered off and the residual unreacted bismuth (III) tetraiodide complex in the filtrate was analyzed using atomic absorption spectrometric technique. On the other hand the precipitate could be dissolved in acetone and analyzed spectrophotometrically at its peak absorption maximum at 462 nm (Fig. 6) or dissolved in HCl for atomic absorption spectrometric estimation.
Absorption spectra of Bi(III)-88 μgml-1 moxifloxacin ion pair (—) versus reagent blank (—).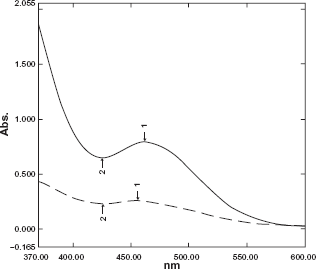
Extraction of the formed ion- pair with different solvents was also studied, low polarity solvents such as chloroform and dichloromethane were inefficient due to insolubility of the ion- pair in such solvents, solvents with increase polarity such as n-butanol, acetone, and isobutyl methyl ketone lacked selectivity and did not differentiate between the ion- pair formed and the residual unreacted bismuth (III) tetraiodide in the aqueous phase, therefore filtration was necessary to separate the formed ion- pair.
Determination of the residual non-consumed bismuth (III) tetraiodide complex in the filtrate had the advantages of being rapid, simpler, precise than the direct dissolution of the isolated ion- pair precipitate owing to possible errors during isolation steps, automation of the methods enhance the overall analytical progress of the proposed procedure, making them more suitable for routine quality control analysis of the studied drug.
The different experimental parameters affecting the formation of the ion- pair complex were studied to determine the optimum conditions for the assay procedure:
Effect of reagents volume: It was found that 0.7 ml of 0.01 M bismuth nitrate and 0.8 ml of 0.5 M potassium iodide were required to obtain the maximum precipitation of the drug as its ion- pair. Effect of acid concentration: The choice of a suitable pH value at which the ion associate exhibit the lowest solubility (at 25 °C) is of prime importance in the use of such compounds in quantitative analysis. To determine this pH value, 0.05 M of KOH or 2% HNO3 was used to adjust the pH of the solution, from the obtained results, it was observed that at pH > 3, there is a decrease in the precipitation yield with increase in pH, probably because of precipitation of bismuth as a hydroxospecies. The absorbance was maximum at pH 1.7 which was achieved by using 0.7 ml 2% HNO3.
Determination of moxifloxacin HCl using method C (Procedures 1, 2 and 3).
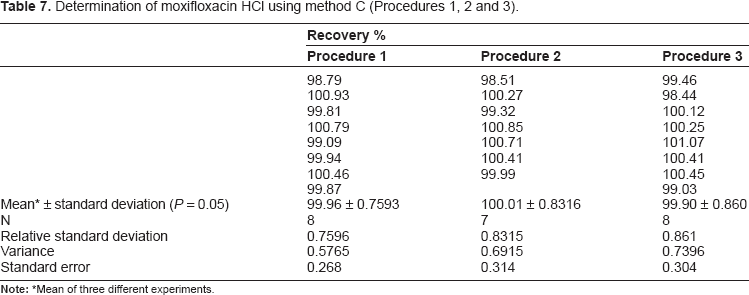
Mean of three different experiments.
Composition of the Ion- Pair Associates
The composition of the ion- pair associates was established by molar ratio method. 38 using equimolar solutions of the drug and reagent (0.8 × 10-3), the results obtained indicate that the composition of the associates was (1:2) drug to reagent. According to this ratio it was found that 15.27 μgml 1 Bi (III) = 16 μgml 1 moxifloxacin HCl.
Methods of Validation
Under the experimental conditions described above the optical characteristics such as Beer's law limits, Sandell's sensitivity and molar absorptivity 39 were calculated for the proposed methods and the results are summarized in (Table 1). Regression equations, intercepts, slopes and correlation coefficients for the calibration data are presented also in the same table while standard deviation, relative standard deviation and standard error are summarized in (Tables 2 and 7).
Sensitivity
The detection limit (LOD) for the proposed methods were calculated using the following equation according to definition
40
:

The limits of quantitation, LOQ, is defined as;

Accuracy and Precision
In order to determine the accuracy and precision of the proposed methods, solutions containing 3 different concentrations of drug were prepared and analyzed in six replicate. The relative standard deviation as precision percentage relative error (Er %) as accuracy of the suggested methods were calculated at 95% confidence levels and can be considered satisfactory. Precision was carried out by six determinations at three different concentrations, the percentage relative error was calculated according to the following equation:
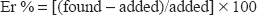
The inter-day precision and accuracy data for moxifloxacin HCl obtained by the proposed methods.
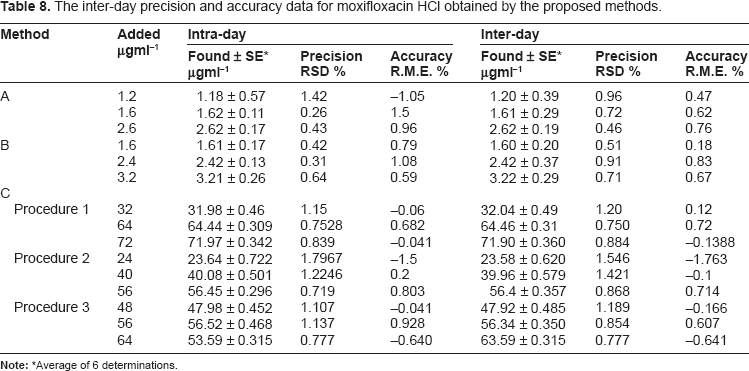
Average of 6 determinations.
Robustness and Ruggedness
Results of evaluation of the ruggedness of the proposed spectrophotometric methods for determination of moxifloxacin HCl.

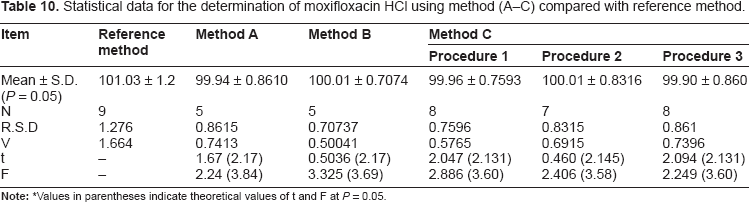
Statistical data for the determination of moxifloxacin HCl using method (A-C) compared with reference method.
Application
Application of standard addition technique for the determination of moxifloxacin HCl in pharmaceutical preparations using methods A and B.
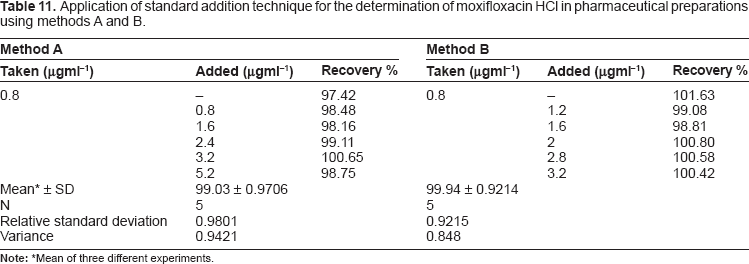
Mean of three different experiments.
Application of standard addition technique for the determination of moxifloxacin HCl in pharmaceutical preparations using method C (Procedures 1, 2 and 3).
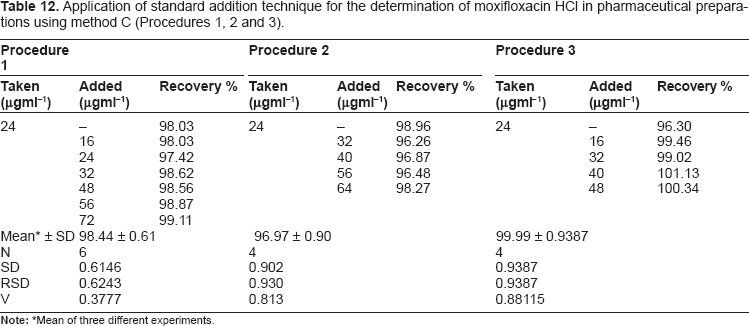
Mean of three different experiments.
Conclusion
The proposed methods described in this paper are simple, economic, sensitive, don't require expensive reagents and sophisticated instruments. These methods are applicable for routine analysis of the studied drug in raw materials and pharmaceutical formulations over wide concentration range without interference from common excipients. The methods can use both spectrophotometric and (AAS) techniques for the final measurement step, moreover, they also have the advantages that no extraction is needed to separate the ion- pair formed and so avoiding the hazards of the organic solvents being simpler and more convenient. The statistical parameters indicate the reproducibility and accuracy of the methods.
Footnotes
Author(s) have provided signed confirmations to the publisher of their compliance with all applicable legal and ethical obligations in respect to declaration of conflicts of interest, funding, authorship and contributorship, and compliance with ethical requirements in respect to treatment of human and animal test subjects. If this article contains identifiable human subject(s) author(s) were required to supply signed patient consent prior to publication. Author(s) have confirmed that the published article is unique and not under consideration nor published by any other publication and that they have consent to reproduce any copyrighted material. The peer reviewers declared no conflicts of interest.
