Abstract
Cefixime trihydrate is a broad spectrum cephalosporin antibiotic, effective against gram-positive and gram-negative bacterial infections. Simple and rapid method has been developed for the determination of cefixime trihydrate in bulk and pharmaceutical formulations. This method was based on the formation of bluish-green ion-pair complex of cefixime trihydrate with bromophenol blue in dimethyl sulfoxide (DMSO)–acetonitrile medium. Different parameters were studied and optimized. A 2:1 complex was formed between the drug and reagent almost instantaneously at room temperature which has δmax of 610 nm. Under optimum conditions, calibration curve was found to be linear over the range of 10-130 µg mL-1. The method was subjected to analytical quality control. The limit of detection was found to be 1.08 µg mL-1. Recovery studies and interference studies were carried out. The proposed method was successfully applied to the determination of cefixime trihydrate in bulk and pharmaceutical formulations with high precision and accuracy.
Introduction
Cefixime trihydrate is broad spectrum, third generation, cephalosporin class of antibiotic. It is one of the essential medicines according to World Health Organisation. 1 It is Indian, British, United States, European, Japanese, and Martindale pharmacopeia recommended antibiotic.2–7 It is active against gram-positive and gram-negative bacterial infections and used to treat otitis media, pharyngitis, bronchitis, and urinary-tract infections. Bromophenol blue (BPB) is a triphenylmethane dye and is commonly used as indicator and spectrophotometric reagent. The structures of cefixime trihydrate and bromophenol blue are shown in Figure 1.
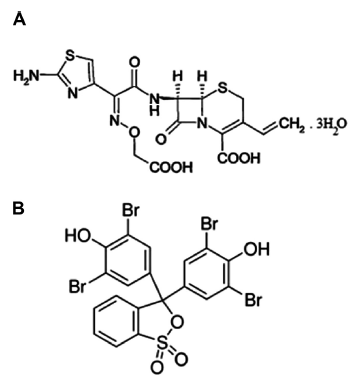
Structure of Cefixime trihydrate (A) and bromophenol blue (B).
Several methods have been reported in the literature for the analysis of cefixime trihydrate such as HPLC, 8 spectrofluorometry, 9 capillary electrophoresis, 10 voltammetry, 11 flow injection technique, 12 mass spectroscopy, 13 and so on. Spectrophotometric methods are generally based on the formation of complex between drug and reagent which can be estimated using visible spectrophotometer. The complex between drug and reagent is either an ion-pair type or charge-transfer type.
In ion-pair complex formation, ions of opposite electric charge held together in solution by Coulomb attraction to form a distinct chemical entity. It behaves as a single unit. Ion-pair formation, initially investigated by the physical chemists, has been found extremely interesting for the chemical analysis, including pharmaceutical analysis. 14 Reported spectrophotometric methods for cefixime trihydrate include complex formation with 7,7,8,8-tetracyanoquinodimethane (TCNQ) and iodine, 15 methyl orange and methylene blue, 16 8-hydroxy-1,3, 6-pyrenetrisulfonic acid trisodium salt (HPTS), 17 4-chloro-7-nitrobenzo-2-oxa-1,3-diazole (NBD-Cl), 18 Congo red, 19 etc. However, most of these methods suffer from narrow dynamic absorbance–concentration linearity; some of them involve complicated sample treatment and extraction procedures.
The proposed method is based on formation of ion-pair complex of cefixime trihydrate with bromophenol blue. Bromophenol blue has been used for the first time with significantly low detection limit, high sensitivity, and wider dynamic range. An important feature of this method is that no extraction is required and it is feasible at room temperature. This method could be applied to the analysis of pharmaceutical formulations.
Methods
Materials and reagents
All chemicals and reagents used were of analytical grade. Cefixime trihydrate was obtained from ZIM laboratories. Bromophenol blue, HPLC grade DMSO, and acetonitrile were obtained from LOBA Chemie. All the chemicals were used as received without further purification.
Instruments
Spectrophotometric studies were carried out with Spectronic 20D+ (Thermo-Spectronic) visible spectrophotometer. A Mettler balance H-51AR (Ner-Parma Instrument Corp., LC = 0.01 mg) was used for weighing purpose.
Standard stock solutions preparation
Stock solutions of cefixime trihydrate and bromophenol blue were prepared as 0.01 M in DMSO and acetonitrile, respectively. The solutions were further diluted as per requirement.
Procedure for calibration curve
Suitable aliquots of cefixime trihydrate were transferred into 10 mL volumetric flasks. To it, 1.6 mL of 8 × 10-5 M bromophenol blue was added and volume was made up to 10 mL with 1:1 DMSO–acetonitrile solvent. This made the final concentration of bromophenol blue as 12.8 µM. After 10 minutes of complex formation, the absorbance of bluish-green solution was measured at 610 nm against the appropriate reagent blank in a cell of path length 10 mm.
Procedure for pharmaceutical formulations
For analysis of tablets, five tablets were weighed and average weight of one tablet was determined. They were powdered and 0.05 g was exactly weighed and shaken with 30 mL of DMSO for 30 minutes. This solution was filtered with Whatman filter paper no. 40 and volume made up to 50 mL with DMSO. The same procedure was applied for oral suspension of drug using 1 mL suspension. Suitable aliquots were analyzed using general procedure.
Results
Effect of solvent
Various organic solvents have been tried to achieve maximum sensitivity and product stability. DMSO for cefixime trihydrate and acetonitrile for bromophenol blue were found to be most suitable solvents.
Absorption spectra
Cefixime trihydrate form bluish-green colored ion-pair reaction product with bromophenol blue having absorbance maximum 610 nm (Fig. 2).
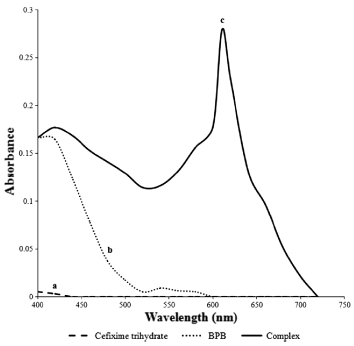
Absorption spectra of (a) Cefixime trihydrate (508 µg mL-1), (b) bromophenol blue (8 µM), and (c) their complex.
Stoichiometric relationship and stability studies
Composition and stability constant of the complex was established by applying Job's method of continuous variation. The results indicated that the complex was formed in the ratio of 2:1 (D:R) (Fig. 3). Mechanism of formation of such complex with composition (DH)+2(R)2- has been discussed by Gainza and Konyeaso. 20 The mechanism has been depicted in Figure 4.
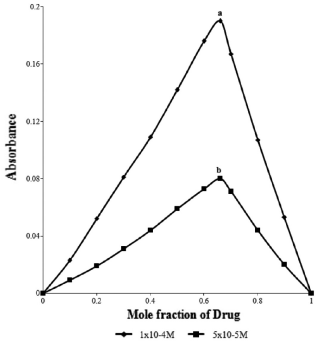
Continuous variation plots for the ion-pair complex of cefixime trihydrate (a) 1 × 10-4 and (b) 5 × 10-5 with bromophenol blue.
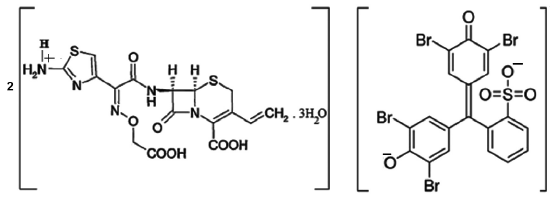
Proposed structure of ion-pair complex of cefixime trihydrate and bromophenol blue.
Effect of time
Mixtures of drug and reagent were prepared; the optimum reaction time was determined by recording the absorbance of the formed complex at different time intervals. The variation has been shown in Figure 5.
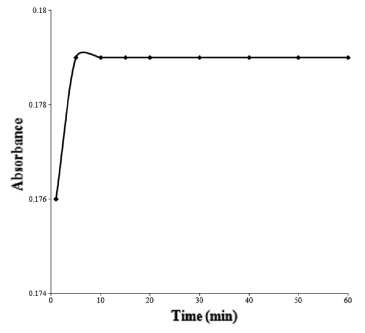
Effect of time on the absorbance of ion-pair complex of cefixime trihydrate with bromophenol blue.
Effect of reagent concentration
The optimum concentration of bromophenol blue was determined by adding various concentrations of bromophenol blue to two different concentrations (100 µg mL-1 and 200 µg mL-1) of cefixime trihydrate. The absorbance was found to be maximum at 12.8 µM bromophenol blue concentration (Fig. 6).
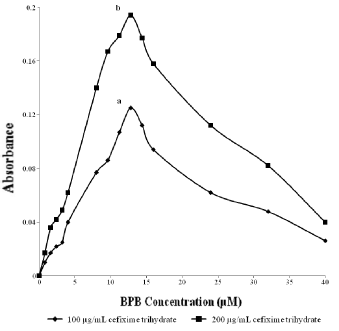
Effect of bromophenol blue concentration on ion-pair complex of cefixime trihydrate (a) 100 µg ml-1 and (b) 200 µg ml-1.
Validation
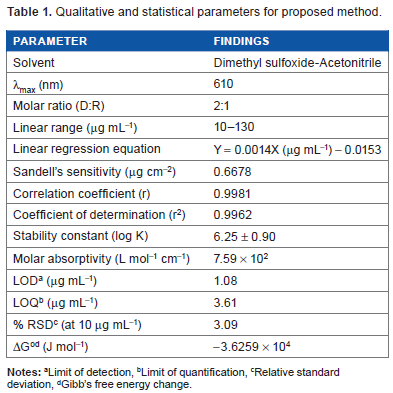
The proposed method was validated according to ICH guidelines 21 with respect to certain parameters such as linearity, Sandell's sensitivity, limit of detection, limit of quantitation, correlation coefficient, coefficient of determination, stability constant, Gibb's free energy change, accuracy, precision, and specificity.
Discussion
Various solvents like methanol, ethanol, acetone, dichloromethane, dichloroethane, DMSO, chloroform, and acetonitrile were used to check the solubility and complex formation and also to achieve maximum sensitivity and product stability, it was found that DMSO for cefixime trihydrate and acetonitrile for bromophenol blue were the best solvent with regard to absorptivity and stability. This is probably because of high dielectric constant of solvents which stabilizes the formed complex.
Solution of cefixime trihydrate in dimethyl sulfoxide and bromophenol blue in acetonitrile were prepared. Absorption spectra of these solutions were recorded individually. When cefixime trihydrate solution was mixed with bromophenol blue solution, bluish-green complex was formed with absorption maxima at 610 nm. Under experimental conditions, the reagent as well as the drug showed negligible absorbance while the complex showed maximum absorbance at this wavelength. Hence, it was concluded that the studies for quantitative analysis could be carried out at this wavelength.
Equimolar solutions of the drug and the reagent were mixed in various proportions and absorbances of mixtures were recorded by applying Job's method of continuous variation. This method indicated that the complex was formed in the ratio of 2:1 (D:R). The absorbance of the complex was used to calculate stability constant and Gibb's free energy. The stability constant (log K) was found to be 6.25 ± 0.90 showing high stability and Gibb's free energy change for complex formation was found to be -3.6259 × 10 4 J mol-1 showing spontaneity of the process.
The optimum reaction time was determined by recording the absorbance of the formed complex at different time intervals. It was found that the complex was formed instantaneously at room temperature but the absorbance was steady after 10 minutes of complex formation. Hence, in order to remove time effect, all observations were made after 10 minutes of complex formation.
Various concentrations of bromophenol blue were added to cefixime trihydrate solution. The color intensity was found to increase with addition of bromophenol blue up to 12.8 µM and then decreases, which shows that 12.8 µM bromophenol blue was sufficient to complex all cefixime trihydrate present in the medium. Therefore, this concentration was used to prepare calibration curve.
Analytical parameters
Calibration curve for cefixime trihydrate was plotted between absorbance and concentration. A linear absorbance–concentration correlation was found to be 10-130 µg mL-1 with correlation coefficient 0.9981. The molar absorptivity value was found to be 7.59 × 10 2 L mol-1 cm-1. The limit of detection and limit of quantitation were calculated in accordance with equations,
LOD = 3σ/S
LOQ= 10σ/S
The σ is the standard deviation of the response and S is the slope of calibration graph. The limit of detection and limit of quantitation were found to be 1.08 µg mL-1 and 3.61 µg mL-1, respectively, for 10 observations. Sandell's sensitivity was determined and it was found to be 0.6678 µg cm-2. 22 Under optimum conditions, various analytical parameters were obtained as presented in Table 1. The value of correlation coefficient indicates good linearity for the system. Value of molar absorptivity and Sandell's sensitivity reflect high sensitivity of the method. The linear range was found to be 10 µg mL-1 (A = 0.002) to 130 µg mL-1 (A = 0.179).
Qualitative and statistical parameters for proposed method.
Limit of detection
Limit of quantification
Relative standard deviation
Gibb's free energy change.
Recovery studies
Recovery studies were carried out for cefixime trihydrate using calibration curve at three different concentrations over the linear range. The recoveries were found to be 100.3 ± 2.4% (Table 2).
Recovery studies of cefixime trihydrate.
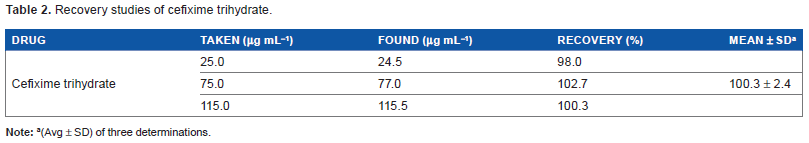
(Avg ± SD) of three determinations.
Accuracy and precision
Accuracy expresses the closeness between the reference value and the found value. It was evaluated as percentage relative error between the measured concentrations and taken concentrations of cefixime trihydrate. The precision of this method was calculated in terms of intermediate precision (intra-day and inter-day). Three concentrations of cefixime trihydrate were analyzed in three replicates during same day (intra-day precision) and three consecutive days (inter-day precision). RSD (%) values of intra-day and inter-day studies show high degree of precision (Table 3).
Evaluation of inter-day and intra-day precision and accuracy.
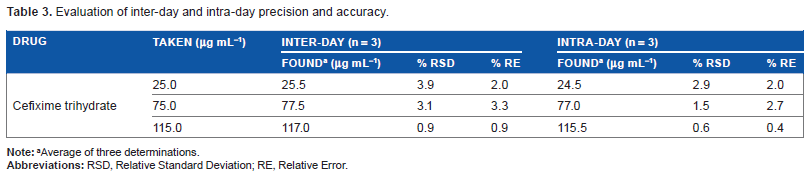
Average of three determinations.
Robustness
The robustness of the system was evaluated by making small incremental changes in time (15 ± 5 minutes) and the effect of the change of absorbance was studied on ion-pair complex (Table 4).
Evaluation of robustness.

Reaction time (15 ± 5 min).
Interference studies
The effect of common excipients and other additives were tested for possible interferences in the assay. Various amount of excipients such as lactose, dextrose, cellulose, magnesium stearate, talc, starch, and gelatin were added to known amount of cefixime trihydrate and were examined using developed procedure. It was found that they did not interfere in the determination of cefixime trihydrate even when present 100 times in excess. To evaluate the selectivity of the proposed method of analysis in pharmaceutical formulations, placebo blank was compared with synthetic mixture and the results were incorporated as % RSD (Table 5).
Determination of cefixime trihydrate in presence of excipients.
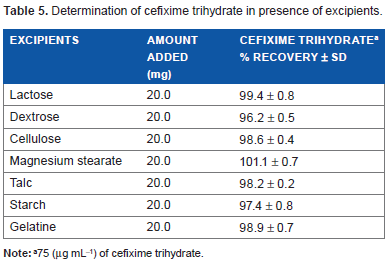
75 (µg mL-1) of cefixime trihydrate.
Applications
The proposed method has been successfully applied to the determination of cefixime trihydrate in pharmaceutical formulation such as tablets and oral suspension. Calibration curve method and standard addition method were adopted for quantitative analysis (Table 6).
Analysis of pharmaceutical formulations.
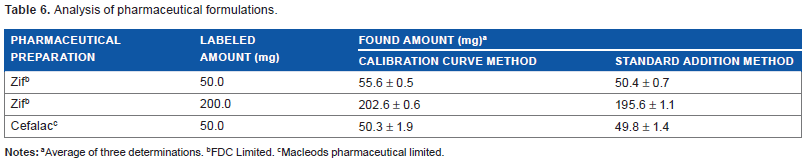
Average of three determinations.
FDC Limited.
Macleods pharmaceutical limited.
Conclusion
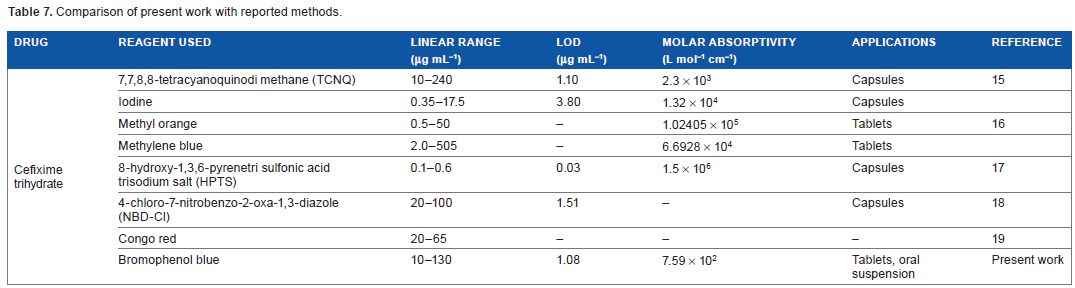
The developed method was found to be versatile and has many advantages over the previously reported methods. A comparison of this method with reported methods has been presented in Table 7. It has been observed that bromophenol blue method has wider linear range as compared to most of the reported methods.15–17,19 The detection limit is lower than that of the reported method.15,18
Comparison of present work with reported methods.
This method requires only dye and solvent, which are comparatively cheaper and readily available. This method is simple as it does not involve adjustment of critical conditions like temperature, pH, or tedious sample preparation. This method has many advantages over other analytical methods due to its simplicity, sensitivity, rapidity, low-cost instrumentation, accuracy, free from interference by common additives, and excipients. Due to these advantages, this method can be used for quality control and routine analysis.
Author Contributions
Conceived and designed the experiments: RJ, MK. Analyzed the data: MK, RJ. Wrote the first draft of the manuscript: MK. Contributed to the writing of the manuscript: RJ. Agree with manuscript results and conclusions: MK, RJ. Jointly developed the structure and arguments for the paper: MK, RJ. Made critical revisions and approved final version: MK, RJ. All authors reviewed and approved of the final manuscript.
