Abstract
Background
Fospropofol is a phosphate-ester water soluble prodrug of propofol approved in the United States for the short-term sedation of adults undergoing diagnostic or therapeutic procedures with monitored anesthesia care. Fospropofol may be advantageous for procedural sedation because it is not a lipid formulation, it lacks pain on injection, it possesses a different pharmacokinetic profile due to in vivo conversion of fospropofol to propofol, and it offers rapid recovery from sedation.
Objective
To review published pharmacokinetic data, clinical trials, and the safety profile of fospropofol and provide comment on its use for sedation.
Clinical trials
Fospropofol has been studied for sedation in colonoscopy and bronchoscopy with fentanyl for analgesia. Results indicate that a 6.5 mg/kg bolus dose with repeated doses of 25% of the original bolus dose up to every 4 minutes produces an effective and safe level of sedation. Fospropofol comparisons to midazolam were not assessed. Patient satisfaction is high for those treated with the recommended dosage regimen. Most commonly reported adverse effects included pruritus and paresthesia. Sedation related side effects such as hypoxia and apnea were rarely reported and generally mild in scope.
Conclusion
Fospropofol represents an alternative sedative agent to midazolam and propofol for short-term sedation. However, the efficacy and safety of fospropofol compared to midazolam or propofol has not been assessed, therefore use will be based on practitioner preference. Restrictive package labelling requiring MAC poses medico-legal and cost-benefit issues thereby not conferring a benefit over propofol which caries similar labelling. Additional studies designed to compare fospropofol to propofol and/or midazolam for procedural sedation are needed.
Introduction
Fospropofol, an intravenous (IV) water soluble prodrug of propofol, is approved by the Food and Drug Administration (FDA) for the short-term sedation of adults undergoing diagnostic or therapeutic procedures under monitored anesthesia care (MAC). 1 Fospropofol is classified as a schedule four (C-IV) controlled substance by the FDA. The aqueous solubility of fospropofol provides an advantage over propofol which is available only as a lipid containing oil-water emulsion. Disadvantages of the omega-6 fatty acid based lipid emulsion of propofol include hypertriglyceridemia, risk of bacterial contamination and subsequent infection, immunomodulation thus reducing bacterial clearance, and injection site pain which is a frequent complaint from patients undergoing short-term sedation for procedures.2–4 Despite injection site pain, satisfaction scores of patients requiring short-term sedation for procedures are higher with the use of propofol compared to benzodiazepines, likely due to the quick onset and offset properties of propofol. 5 Fospropofol was designed to avoid the issues associated with the lipid formulation of propofol while providing a quick onset of sedation and a pharmacokinetic profile that limits the peak propofol serum concentrations associated with bolus doses of propofol that may lead to respiratory depression. This review summarizes the pharmacokinetic and pharmacodynamic data, published clinical trials, safety data, and the therapeutic applications of fospropofol.
A database search of PubMed (1980-March 2, 2010) was performed including search terms fospropofol, Aquavan, Lusedra, GPI 15715, monitored anesthesia-care, propofol, formaldehyde and formic acid. Reviewing bibliographies of articles found through PubMed revealed additional studies. Eisai Inc. was contacted for unpublished pharmacokinetic data. The following websites were accessed for additional data: www.fda.gov and clinicaltrials.gov.
Mechanism of Action, Metabolism and Pharmacokinetic/Dynamic Profile
Fospropofol's therapeutic action is based principally on its hydrolysis to propofol. Although the exact mechanism of propofol's sedative action is not fully described, it binds centrally to γ-aminobutyric acidA receptors potentiating the inhibitory effects of γ-aminobutyric acid at neurotransmitter synapses among other secondary actions. 6
Fospropofol's increased solubility is due to a methylphosphate linkage increasing its molecular weight 1.86 fold compared to propofol. Alkaline phosphatases in vivo completely convert the ester fospropofol to propofol (estimated bioavailability 54%), and formaldehyde and phosphate on a one to one molar basis. 1 A 560 mg dose of fospropofol (a standard bolus for an 86 kg patient) will release 301 mg of propofol, 50.5 mg of formaldehyde and 160 mg of phosphate (1.67 mmol). 7 Propofol is metabolized extensively by the liver to various inactive sulfate and glucuronide components. Formaldehyde is converted to formic acid by aldehyde dehydrogenase, which is further oxidized to carbon-dioxide and water by 10- formyltetrahydrofolate dehydrogenase in the presence of tetrahydrofolate. Extensive reviews of formaldehyde and formic acid kinetics and clinical consequences have been published elsewhere.8,9 Liberated phosphorus is renally eliminated or incorporated into normal metabolic pathways.
The initial proposed pharmacokinetic and pharmacodynamic model for fospropofol is multicompartmental; two compartments for fospropofol and three for propofol.10,11 Revised models using up to six compartments have since been published.11,12 However, recent retractions of both pharmacokinetic parameters and models have been published due to potentially erroneous laboratory measurements of liberated propofol. The assay inaccuracy is limited to propofol kinetics only, therefore previously published fospropofol kinetics are unaffected.13,14 Methods for validating measured propofol concentrations after fospropofol administration are currently underway but not yet published. The manufacturer, Eisai Inc. retains the original pharmacokinetic and pharmacodynamic studies which were not subject to propofol assay inaccuracies and will be discussed below. 15
Fospropofol has a low volume of distribution (Vd = 0.33 ± 0.069 L/kg), distributes quickly to the peripheral compartment and is quickly hydrolyzed (t1/2 = 7.2 ± 1.1 min) to propofol (Table 1). Fospropofol is ~98% protein bound, mostly to albumin. Renal excretion of fospropofol is negligible with less than 0.02% of the parent drug excreted unchanged in the urine. The mean fospropofol terminal phase half-life is 48 and 52 minutes in healthy individuals and patients who received 6.5 mg/kg doses during clinical studies, respectively.1,10,12,15 Mild to moderate renal insufficiency does not significantly alter the pharmacokinetics of fospropofol or propofol; therefore, dose adjustments are not required for patients with a creatinine clearance of ≥30 mL/min. Safety and efficacy data for patients with a creatinine clearance of <30 mL/min are limited. Similarly, fospropofol has not been adequately studied in patients with severe hepatic disease so caution is advised. 1 Propofol package labelling indicates that propofol pharmacokinetics are not significantly altered in patients with chronic cirrhosis. 16
See fospropofol dosing, dose adjustments and repeat dosing for MAC sedation.
Propofol dosing and repeat dosing may vary for MAC sedation.
Onset may be related to dose and/or infusion rate (for propofol), duration is dose dependent.
Short-term procedural sedation outside of MAC is not an approved FDA indication.
Propofol peak concentrations are slightly delayed after fospropofol injection compared to propofol injection. After a 6 mg/kg and 18 mg/kg bolus dose of fospropofol, the time to peak liberated propofol concentration was 12 minutes and 8 minutes, respectively. Peak liberated propofol concentrations were 1.08 ± 0.33 mcg/mL and 3.9 ± 0.822 mcg/mL following a 6 mg/kg and 18 mg/kg dose, respectively. Liberated propofol exhibits a lipophilic distribution (Vd = 5.8 L/kg), high protein binding (~98% mostly to albumin) and does not interfere with fospropofol protein binding. The mean terminal phase half-life of liberated propofol after receiving 6 mg/kg bolus of fospropofol was 2.06 ± 0.77 hours but does not reflect the duration of clinical effect due to rapid redistribution. Clinical onset of sedation after a recommended bolus dose of fospropofol is delayed by 4–8 minutes when compared to propofol (Table 1). Full recovery from sedative effects from fospropofol occurred at 20–45 minutes in healthy individuals following a single 10 mg/kg dose which is higher than the recommended dose.1,15
The FDA approved product labelling recommend that dosage regimens use actual body weight. In adults less than 65 years old who are otherwise healthy or have mild systemic disease (ASA P1 or P2), the dose is 6.5 mg/kg once with additional doses of 1.6 mg/kg (25% initial bolus) up to every 4 minutes as needed to reach adequate sedation (Table 2). Upper and lower weight limits of 60 kg and 90 kg are used when calculating doses. Therefore, doses should not exceed 577.5 mg or 16.5 mL as fospropofol is supplied as a 30 mL vial at 35 mg/mL concentration (Table 1). 1
Recommended dosing per package labeling. 1

ASA P1–4 = American Society of Anesthesiologist physical status classification system.
Patients weighing less than 60 kg or more than 90 kg should use 60 kg and 90 kg, respectively as their dosing weight. Rounding to a measurable dose is recommended.
Clinical Studies
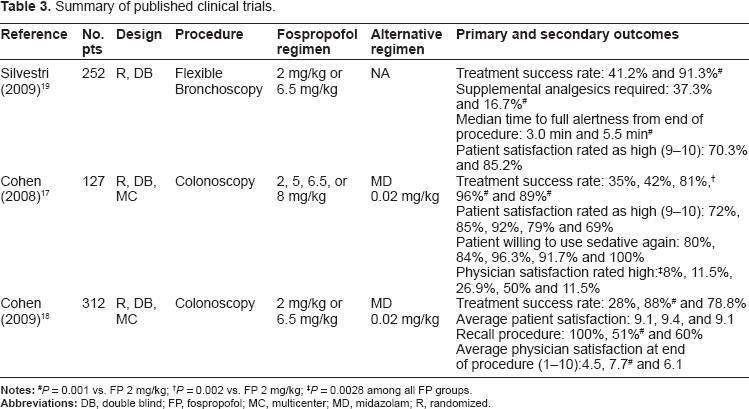
Three published clinical trials of fospropofol were published between 2008 and 2009, two for sedation in patients undergoing colonoscopy and one for sedation in patients undergoing bronchoscopy (Table 3).17–19 All trials used IV fentanyl five minutes prior to a bolus dose of fospropofol with repeated doses of 25% the original sedative dose as needed every 4 minutes to reach adequate sedation. Most procedures were completed within 30 minutes. No clinical trials to date have directly compared fospropofol to the standard of practice of either midazolam or propofol for sedation. Fospropofol is currently undergoing study for anesthesia during coronary artery surgery, 20 sedation during cardiac catheterization 21 and sedation during mechanical ventilation in the intensive care unit setting 22 but trial results and safety profiles are not yet available.
Summary of published clinical trials.
P = 0.001 vs. FP 2 mg/kg;
P = 0.002 vs. FP 2 mg/kg;
P = 0.0028 among all FP groups.
Sedation during colonoscopy
Fospropofol has been most studied for the provision of moderate sedation during colonoscopy. A multicenter double-blind trial evaluating 127 adults undergoing colonoscopy randomized patients to either fospropofol, dosed at 2, 5, 6.5, or 8 mg/kg, or midazolam dosed at 0.02 mg/kg. Supplemental doses of 25% of the initial fospropofol dose every four minutes or midazolam 1 mg every two minutes were permitted for sedation. Additional fentanyl could be given at 25 mcg every 10 minutes if needed for pain. Investigators used the Modified Observer's Assessment of Alertness/Sedation Scale (MOAA/S) to assess sedation (Table 4) and the Hopkins Verbal Learning Test-Revised (HVLT-R) to assess recovery from sedation. Sedation success was defined by three consecutive MOAA/S scores of ≤4 after administration of sedative medication, completion of the procedure without use of alternative sedative medication and no requirement for manual or mechanical ventilation. Treatment success was defined as completion of procedure without alternative sedative medications and no requirement of manual or mechanical ventilation. Patient satisfaction was assessed using a seven item survey at 24 hours and a nine item survey prior to discharge. Physician satisfaction was assessed using a 16 item survey. All surveys used a 1–10 scale ranging from unsatisfied (1) to highly satisfied (10). Safety outcomes such as apnea, hypoxemia, bradycardia and hypotension were recorded. 17
Sedation success was dose-dependent and occurred in 24, 35, 69, 96% of patients across the dosage regimens, respectively. Sedation success was significantly higher in the 6.5 and 8 mg/kg groups when compared to the 2 and 5 mg/kg groups (P < 0.05). Those that received midazolam had a sedation success rate of 81% which was also significantly higher than those that received 2 or 5 mg/kg of fospropofol (P < 0.05). A greater number of patients in the 8 mg/kg (25%) group had a MOAA/S score of 0 or 1 at any time during the procedure compared to the 2, 5, and 6.5 mg/kg (8, 4 and 4%, respectively, P value not reported). Memory retention, as assessed by the HVLT-R, was highest in the 6.5 mg/kg group, but not statistically so, indicating adequate sedation recovery 15 minutes after procedure completion in all groups including midazolam. Physician satisfaction at the end of the procedure was dose-dependent with fospropofol (overall P < 0.0001). 17
In a double-blind, multicenter trial, 312 patients received either fospropofol 2 mg/kg or 6.5 mg/kg or midazolam 0.02 mg/kg randomized in a 2:3:1 fashion. The study procedure, assessment tools and end-points were similar to that outlined above. Sedation success was 87% in the fospropofol 6.5 mg/kg group compared to 26% in the 2 mg/kg group (P < 0.001) and 69% in the midazolam group. Patients were less likely to remember being awake, less likely to require more than two supplemental doses prior to procedure and less likely to require an alternative sedative at any time in the 6.5 mg/kg group compared to the 2 mg/kg group (51% vs. 100%, 24% vs. 88%, 12 vs. 72%, respectively, P < 0.001 for all). A statistical comparison for midazolam was not reported. Cognitive assessment using the HVLT-R 15 minutes after the procedure was completed was not statistically different between the 6.5 mg/kg group and the 2 mg/kg group (70% vs. 82%, respectively). Although direct comparisons were not made to midazolam in this trial, 33% required more than two supplemental doses prior to procedure, 19% required an alternative sedative, and memory retention was 41% 15 minutes post procedure. Overall patient satisfaction was high and not statistically different in any group. Physician satisfaction was higher in the fospropofol 6.5 mg/kg group compared to the 2 mg/kg group both at the end of sedation initiation and the end of the procedure (7.1 vs. 3.3 and 7.7 vs. 4.5, respectively, P < 0.001); the reference midazolam group scored 5.6 and 6.1, respectively. 18
Sedation during bronchoscopy
Fospropofol was also evaluated for use in flexible bronchoscopy in a randomized double-blind study of 252 patients. Patients were randomized to either 6.5 mg/kg or 2 mg/kg of fospropofol in a 3:2 ratio. Topical lidocaine and one additional 25 mcg dose of fentanyl were permitted. The primary endpoint was sedation success as defined above. Similarly, the MOAA/S score and HVLT-R performance were used to assess sedation and recovery, respectively. 19
Sedation success was significantly higher in the 6.5 mg/kg group compared to the 2 mg/kg group (88.7% vs. 27.5%, respectively, P < 0.001). Less than half (44%) of the patients in the 6.5 mg/kg group required a supplemental sedative dose, compared to 93% in the 2 mg/kg group (P value not reported). Only 8% of patients in the 6.5 mg/kg group required an alternative sedative (midazolam) compared to 58.8% in the 2 mg/kg (P value not reported). Overall mean supplemental doses of study sedative medication was less in the 6.5 mg/kg group compared to the 2 mg/kg group (1.7 vs. 2.9, respectively P < 0.001). The median time to sedation was shorter in the 6.5 mg/kg group compared to the 2 mg/kg group (4 vs. 18 minutes, respectively, P value not reported). Median time to readiness for hospital discharge from the end of the procedure was similar between fospropofol groups (8.5 vs. 8 minutes for 6.5 mg/kg and 2 mg/kg, respectively, P value not reported). Full alertness was reported at a median of 16 minutes after the initial 6.5 mg/kg dose. Fewer patients remember being awake during the procedure and more would agree to use the sedative again in the 6.5 mg/kg group compared to the 2 mg/kg group (16.7% vs. 44.6% and 94.6% vs. 78.2%, respectively, P < 0.001 for both). Mean physician satisfaction scores were numerically higher in the 6.5 mg/kg group compared to the 2 mg/kg group both at the end of sedation and at the end of procedure (8 vs. 3.9 and 8.3 vs. 5, respectively, P value not reported). 19
Together these studies suggest fospropofol with fentanyl is effective for moderate sedation during colonoscopy or bronchoscopy at recommended doses (Tables 2 and 3).17–19
Safety
Mild common adverse effects
The most common adverse events in studies were pruritus (16%–28%) and paresthesia (49%–74%), both more commonly reported when compared to midazolam. These events usually occur within 5 minutes of drug administration, last up to 2 minutes, are self limiting and are known adverse effects of other drugs containing phosphate esters such as fosphenytoin. Injection site pain was not commonly reported with fospropofol. 1
Sedation related
Safety concerns regarding the potential for deep sedation, loss of consciousness and respiratory depression, prompted FDA labelling of the drug to include use during MAC only. Disparate views exist between the position statements of the American Society of Anesthesiologists supporting the labelling and the American Society for Gastrointestinal Endoscopy challenging the labelling. It is noteworthy that all published trials to date were not conducted under MAC and patients were managed by the physician conducting the procedure.23–25 Larger trials and post marketing surveillance are needed to confirm safety results mentioned below.
Apnea appears to be uncommon (<1%) when using recommended doses. In dose ranging studies using higher than recommended doses, apnea was reported in up to 3% of patients. Hypoxemia has been reported in 4% of all patients treated at recommended doses. In one clinical trial using fospropofol sedation for bronchoscopy, doses of 6.5 mg/kg had a higher percentage of patients experiencing hypoxemia, defined as an oxygen saturation of less than 90% for greater than 30 seconds, or requiring any type of airway assistance compared to the 2 mg/kg group (15.4% vs. 12.6% and 21.5% vs. 13.6%, respectively, P value not reported). The most common intervention was increasing oxygen flow for both groups. Two patients experienced severe hypoxemia in the 6.5 mg/kg group. One had significant comorbidities and required manual assisted ventilation while the other required repositioning, suction, increased oxygen flow and verbal stimulation; both recovered without incident. In patients given higher than recommended doses, hypoxemia was much more common (27%). Due to respiratory depressive effects, all patients receiving fospropofol should be appropriately positioned and given supplemental oxygen. Clinicians must be prepared to perform airway assistance maneuvers as needed. Over sedation (defined as a MOAA/S score of 0 or 1) during colonoscopy was uncommon with 4% of patients experiencing deep sedation 2–16 minutes in duration. In patients undergoing bronchoscopy 16% of patients became deeply sedated ranging 2–20 minutes in duration.1,19
Cardiovascular related
Hypotension has been reported in 4% of all patients treated at recommended doses. During colonoscopy, 2% of patients experienced hypotension compared to 5.4%–7% in those undergoing bronchoscopy. When fospropofol was used at higher than recommended doses hypotension occurred in 6% of patients. Patients with compromised myocardial function, decreased vascular tone or reduced intravascular volume may be at higher risk for hypotension. Bradycardia, a well known side-effect of propofol, has not been frequently reported with fospropofol in published trials. 1 To the contrary, increased heart rate by an average 5% was reported, most notably early during sedative use. During escalating bolus doses of fospropofol for target Bispectral Index suppression in healthy individuals, transient resolving tachycardia followed by a drop in mean arterial pressure was noted (mean percent change from baseline 25% and 20%–25%, respectively). 26
Propofol related infusion syndrome (PRIS), a serious condition characterized by refractory bradycardia often with metabolic acidosis and rhabdomyolysis, has not been reported with fospropofol. PRIS was first described with propofol doses greater than 4 mg.kg-1.h-1 with infusions lasting greater than 48 hours. More recently PRIS has been reported in lower doses and during shorter administration times. Although the exact mechanism of toxicity is unknown, it may in part be attributed to the excess lipid load with high, prolonged doses of propofol in highly stressed individuals and might be less likely to occur with fospropofol. However, because propofol impairs intracellular oxygen utilization, inhibits electron flow in cardiomyocytes, inhibits mitochondrial oxidative phosphorylation, interferes with mitochondrial long-chain free fatty acid transport, reduces beta-adrenergic responsiveness, and inhibits beta-oxidation which all contribute to PRIS, this toxicity may occur with fospropofol, especially in patients with excessive metabolic demand. 3 The likelihood of PRIS occurring during short-term sedation with fospropofol is exceedingly low; however, additional investigation requiring post approval monitoring by clinicians will be needed to estimate whether a true concern is warranted.
Other concerns
Formic acid concentrations are not significantly increased from baseline in subjects undergoing short-term infusions or bolus injections when compared to propofol. Many experts believe the concern for formic acid accumulation is unwarranted in the use of phosphate-ester prodrugs that release formaldehyde. 7 When used as a bolus injection or for short-term sedation, accumulation of formic acid to a toxic level is extremely unlikely.10,26,27 However, if infusion time duration and/or doses increase, toxicity due to formic acid accumulation may become a concern in susceptible patients. Studies have indicated increases in phosphate serum concentrations outside the normal range of 2.5–4.5 mg/dL during short-term infusions and boluses of high-dose fospropofol in healthy subjects. These increases did not reach toxic ranges (mean 4.8 and 4.7 mg/dL during 20 and 60 minute infusions, respectively) and resolved within 24 hours.26,28 Phosphate may accumulate, however, in patients with severe renal dysfunction despite the low amount of phosphate released from fospropofol. Fospropofol is contraindicated in patients with an allergy to fospropofol or the chemical moiety of propofol.
Abuse potential
Propofol is not currently a controlled substance as classified by the FDA. The rewarding and reinforcing effects of propofol in the animal self-administration model is comparable to midazolam and other C-IV benzodiazepines. Propofol can also produce euphoria in humans. It is now widely recognized that propofol is a drug of abuse, most commonly among healthcare providers.29,30 Since propofol is the active component of fospropofol it follows that fospropofol may exhibit a similar potential for abuse. Fospropofol may lead to limited physical or psychological dependence and is classified as a C-IV medication. 31 Because of limited clinical use, reports of diversion, abuse, addiction, dependence and tolerance have not yet been reported with fospropofol. Also concerning is the delay in peak effect after injection which may lead abusers to use repeated doses leading to respiratory depression and death. Both the potential for abuse and serious consequences of overdose justify its classification as a C-IV medication. 30
Place in Therapy
For procedural sedation, fospropofol is effective and may be advantageous to propofol due to the lack of injection site pain and its aqueous solubility which avoids safety issues related to lipid-containing formulations. However, high occurrence rates of pruritus and paresthesia may limit its clinical application. Theoretically the conversion of fospropofol to propofol in vivo provides a safer pharmacokinetic profile with bolus dosing compared to propofol due to flatter peaks and minimal requirements for repeat dosing. Clinically lower peak liberated propofol concentrations are observed with fospropofol when compared to a 2 mg/kg bolus of propofol (Table 1).15,32 High peak propofol concentrations can lead to more sedation related side effects. In practice lower boluses of propofol (0.5–1 mg/kg) may be used, however, repeated doses may be needed during the procedure. 33 To date, there are no published trials comparing fospropofol to propofol and it is unclear if any theoretical advantage of fospropofol will translate into clinical benefit or safety advantage in practice. Physician and patient satisfaction with sedation has not been compared between propofol and fospropofol.
The theoretical advantage of avoiding a lipid emulsion and decreased bacterial contamination risk should be considered but may be less clinically significant if single use vials and aseptic technique are used appropriately. In 1990 the Centers for Disease Control traced multiple post-operative infections back to the use of preservative free propofol and lack of aseptic technique during medication preparation. Throughout the 1990s infections were linked to extrinsically contaminated propofol vials.16,34 Formulations now contain additives to inhibit microbial growth in the case of extrinsic contamination but care must be taken as it is not considered a preserved product. Practices such as aseptic technique, using vials within six hours of opening, and changing propofol vials and intravenous lines every 12 hours if used as an infusion are recommended.16,34 Recent reports of propofol infusions causing fevers and chills have been investigated by the FDA prompting recalls in 2007 due to elevated endotoxin levels in the product. Finally, a recall occurred in 2009 due to particular matter contamination. 35 Particular matter, which is easily seen in an aqueous product, is difficult to detect in a lipid emulsion. Patients with a documented allergy to the lipid emulsion of propofol (eggs, soybean, soy products, etc) could be candidates for fospropofol. These differences should be weighed against the difference in drug cost between fospropofol and propofol when selecting a procedural sedative.
FDA labelling for sedation with MAC is similar for both fospropofol and propofol making potential cost savings unlikely unless more effective sedation and/or fewer adverse events can be shown with fospropofol.1,24,25 The similar labelling makes a randomized trial comparing fospropofol and propofol crucial to define the role fospropofol will play in procedural sedation. Fospropofol average wholesale price is $43.20 (US) per 30 mL vial equating to roughly $16–24 (US) for a 6.5 mg/kg bolus dose and weight limits of 60–90 kg. Propofol is generic with an average wholesale price of roughly $3.44 (US) per 200 mg vial and a total procedural dose of 2 mg/kg using 60–90 kg range equating to $2.00–3.10 (US). 36 Actual costs to a given institution will likely be less based on wholesale contracts or other buying group discounts.
When compared to midazolam, fospropofol may provide procedural sedation that lends to quicker hospital or clinic discharge and a higher throughput in terms of ability to perform a greater number of procedures as seen in cases of propofol verses midazolam. However, direct comparisons have not been conducted and need to be validated before justifying use based on cost. In one study modelled as a provider cost economic analysis, break even costs for rapid recovery agents (propofol and fospropofol), assuming a one to one operating room to recovery room ratio for colonoscopies, were $71.53 and $61.48 (US) for hospital outpatient clinics and ambulatory surgical centers, respectively. These data suggest that cost savings could be realized when comparing propofol or fospropofol alone to meperidine and midazolam. Of note, MAC was not accounted for in any of the groups which increases operating room cost. Also, since this study was modelled using historical readiness to discharge times and not actual comparative data for fospropofol, caution should be exercised in interpreting results. 37 Further research directly comparing provider costs with fospropofol in a randomized trial to standard midazolam and/or propofol with and without adjustments for MAC should be conducted before economic benefit is inferred.
Fentanyl use was consistent in all three published clinical procedure studies. Like propofol, fospropofol can provide sedation and amnesia for procedures but whether its action is adequate for procedures without additional analgesia remains to be seen.
Studies comparing fospropofol with propofol for anesthesia during coronary artery surgery and with midazolam for sedation during cardiac catheterization are underway and could provide another niche for use. Fospropofol is of interest in this population because it possesses a sedation profile of short onset and fast clearance with moderate hemodynamic effects and complete avoidance of the lipid formulation. Fospropofol may also be of interest in short-term sedation of patients in the intensive care unit requiring mechanical ventilation, especially those requiring frequent daily awakenings for neurological assessments. A phase II study is currently underway but the results are not yet available. Therefore efficacy and safety in these patient populations cannot be assessed and fospropofol should not be used for these indications. Close attention should be paid to fospropofol pharmacokinetics and “clinical off-set” when it is used as a continuous infusion since delays in drug conversion and/or drug accumulation may occur resulting in longer sedative effects. In addition, formic acid concentrations may need to be monitored during and after infusions to provide stronger evidence against accumulation in critically ill patients, especially those with liver disease and/or concurrent renal disease. During short-term infusions and compared to propofol, there was no indication of increasing formic acid concentrations from baseline (range, of <15–49 mcg/mL). 28 It is unlikely that formic acid concentrations will exceed toxic levels in patients with normal stores of tetrahydrofolate and 10-formyltetrahydrofolate dehydrogenase activity with short-term use, however the potential for toxicity in prolonged intravenous use in critically ill patients does exist. Package labelling for fospropofol describes a patient with multiple comorbidities including renal and hepatic insufficiency who experienced an increase in plasma formic acid concentrations from 66 mcg/mL at baseline to 212 mcg/mL after a 12 hour infusion. 1 Given that the half-life of formic acid is 1–6 hours (up to 20 hours) in humans and visual dysfunction may occur at a concentrations of 200–300 mcg/mL, it is prudent to characterize formic acid accumulation and sequela in critically ill patients prior to standard clinical use.8,9,38
Conclusion
Overall, fospropofol provides another option to clinicians for short-term sedation during procedures. Future randomized controlled trials comparing fospropofol to standard of practice are needed to define what role fospropofol should play in procedural sedation. Studies designed to compare fospropofol to midazolam or propofol for sedation are lacking making efficacy, safety and cost-effectiveness analysis indeterminate. Drug formulation, onset of action, recovery time from sedation, lack of injection site pain and pharmacokinetic profile distinguish fospropofol from other commonly used sedatives. However, the clinical benefit of these differences compared to what is commonly used remains to be seen. Meaningful differences in physician and patient satisfaction, efficacy and safety over other sedatives, specifically propofol, need to be studied head to head before utility is inferred. Use for anesthesia and sedation during longer procedures or for mechanical ventilation is presently under investigation and therefore cannot be currently recommended. At this time use is for short-term procedural or diagnostic sedation and should be based on physician or institution preference.
Disclosures
This manuscript has been read and approved by all authors. This paper is unique and is not under consideration by any other publication and has not been published elsewhere. The authors and peer reviewers of this paper report no conflicts of interest. The authors confirm that they have permission to reproduce any copyrighted material.


