Abstract
Background:
Hypovolemia is common in colonoscopy due to fasting and bowel preparation, and propofol itself can reduce systemic vascular resistance, resulting in relative hypovolemia. Therefore, hypotension is not a rare event during propofol-based sedation for colonoscopy.
Objectives:
Our objective was to explore the efficacy of esketamine as a sedative adjuvant in reducing the incidence of hypotension during colonoscopy.
Design:
This was a prospective randomized trial. The trial was registered with the Chinese Clinical Trial Registry (ID: ChiCTR 2100047032).
Methods:
We included 100 eligible patients who planned to receive a colonoscopy and randomly divided them into 4 groups with 25 patients in each group, which were propofol 2 mg/kg (Group P), propofol 1 mg/kg with esketamine 0.2 mg/kg (Group E1), propofol 1 mg/kg with esketamine 0.3 mg/kg (Group E2), and propofol 1 mg/kg with esketamine 0.4 mg/kg (Group E3). The hemodynamic and respiratory parameters were documented at various times during the procedure, including the patient’s entry into the endoscopic room (T0), the induction of sedation (T1), the insertion of the colonoscope (T2), the removal of the colonoscope (T3), and the awakening of the patient (T4). The primary outcome was the incidence of hypotension. Secondary outcomes were cardiovascular side effects other than hypotension, incidence of hypoxia, cumulative changes in cardiovascular and respiratory parameters, total propofol dosage, anesthesia recovery time, and satisfactory levels of both patients and endoscopists.
Results:
The incidence of hypotension in Group E1 (16%), Group E2 (16%), and Group E3 (12%) was significantly lower than in Group P (60%), with p values 0.003, 0.003, and <0.001 respectively. The cumulative changes in diastolic blood pressure and mean arterial pressure in Groups E1, E2, and E3 were significantly higher than in Group P (p = 0.024, p < 0.001, p = 0.006, respectively). Cumulative changes in systolic blood pressure in Group E3 were significantly higher than those in Group P (p = 0.012). The respiratory-related parameters were not statistically significant.
Conclusions:
This study showed that the application of 0.4 mg/kg esketamine in propofol-based sedation reduced the incidence of hypotension during colonoscopy while providing satisfactory sedation.
Plain language summary
Background:
When people undergo colonoscopy and are sedated with propofol, about one-third of them experience low blood pressure. This study aims to see if adding esketamine to the propofol can help prevent or lessen these episodes of low blood pressure during colonoscopy.
Methods:
This study examined four groups who received different doses of esketamine along with propofol. It focuses on differences in the occurrence of low blood pressure, respiratory measures, the amount of anesthetic used, and levels of satisfaction. The study analyzed the relevant data for these parameters to understand the differences between the groups.
Results:
The results indicated that using esketamine along with propofol for sedation during colonoscopy led to several key outcomes, such as:
• Three different concentrations of esketamine effectively reduced the occurrence of low blood pressure in patients undergoing colonoscopy.
• Less propofol was needed in the groups that included esketamine.
• Esketamine at a dosage of 0.4 mg kg-1 showed the best respiratory parameters among the four groups
Conclusions:
During colonoscopy, using esketamine along with propofol for sedation not only achieves satisfactory sedation but also lowers the occurrence of hypotension. Specifically, employing 0.4 mg kg-1 esketamine along with propofol not only reduces hypotension occurrences but also enhances respiration.
Introduction
Colonoscopy is a common diagnostic and therapeutic tool for various colorectal diseases, such as polyps, tumors, and inflammation, as well as a primary screening test for colorectal cancer. 1 In the United States, United Kingdom, and China, almost all colonoscopies are performed under sedation. 2 Propofol-based sedation is widely employed for colonoscopy not only by anesthesiologists but also by a growing number of non-anesthesiologists. 3 It provides a favorable patient experience and operator satisfaction while facilitating efficient patient throughput. More than one-third of patients who underwent a colonoscopy with propofol sedation experienced episodes of hypotension that reached a level and duration typically associated with adverse effects in surgical patients. Extended duration of propofol sedation and larger doses of propofol were associated with more sustained and more severe hypotension due to the vasodilatory effects of propofol. 4 Furthermore, the administration of bowel preparation and fasting will lead to relative hypovolemic status for patients planned for colonoscopy. Other factors such as age and comorbidity would also predispose patients to hypotension.
Using an adjunct medication during propofol sedation reduced the consumption of propofol consumption and associated hypotension. 5 Esketamine is an antagonist of N-methyl-D-aspartate (NMDA) receptors, employed as an adjunct in propofol-based sedation for endoscopic procedures. 6 It has been used as an anesthetic and analgesic agent, inducing less hypotension than other anesthetics due to its sympathomimetic effect. 7 Esketamine, the S-enantiomer of ketamine, manifests potent sedative and analgesic effects along with reduced psychotropic side effects compared to racemic ketamine. 8 A low dose of esketamine reduced the need for propofol and maintained cardiorespiratory stability in endoscopic retrograde cholangiopancreatography 9 and esophageal variceal ligation. 10 But there is limited evidence for colonoscopy.
In this randomized clinical study, we aim to explore the use of esketamine as an adjunct to propofol during colonoscopy procedures. We hypothesized that by the inclusion of esketamine, we could potentially reduce the occurrence and severity of hypotension during propofol sedation for colonoscopy.
Methods
Study design
This prospective randomized trial is registered prior to the enrollment of patients and is strictly conducted according to the CONSORT guidelines. 11 This study protocol was approved by the Hospital Ethics Committee of the Second Affiliated Hospital, Chongqing Medical University (Approval ID: 2021-044), and all patients provided their written informed consent prior to the study. The study was registered in the Chinese Clinical Trial Registry (ID: ChiCTR 2100047032).
We included patients undergoing colonoscopy in our center from July 2021 to September 2021 with inclusion criteria as follows: age more than 18 years old, body mass index 18.5–28, American Society of Anaesthesiologists (ASA) physical status I–III, and provided informed consent statement. Exclusion criteria included a documented drug allergy to propofol and esketamine, uncontrolled hypertension, history of severe cardiovascular disease, cognitive dysfunction, glaucoma, and participation in other drug trials within the preceding 3 months. We prepared 100 opaque envelopes, used computers to generate random numbers, and randomly assigned patients to four groups. The random numbers were encapsulated in sequentially numbered envelopes. After the patients were enrolled, an independent investigator (YL) used envelopes to assign them to various groups. The envelopes were subsequently resealed and kept at the investigation site until the conclusion of the study. The group assignments were concealed from both participants and researchers involved in the colonoscopy procedure. The anesthesiologists in charge of administering drugs were not involved in collecting, entering, or analyzing data.
Endoscopic procedure and monitoring
Upon entering the endoscopic suite, patients were equipped with standard monitors, including pulse oximetry (SpO2), electrocardiogram, noninvasive blood pressure, and heart rate (HR). Patients were positioned on their left side with a slight backward tilt of the head and were given 3 L/min of oxygen through a nasal cannula. Cardiovascular and respiratory parameters were monitored as described in our previous research. 10 The level of sedation was evaluated using the Observer’s Assessment of Alertness and Sedation Scale (OAA/S), which delineates the spectrum of responsiveness grading from 0 to 5. 12 A score of 0 denotes an absence of response to painful stimuli, whereas a score of 5 denotes an alert patient capable of promptly responding to their name in a normal tone.
Patients were allocated into four sedation groups, which were Group P (propofol 2 mg/kg), Group E1 (esketamine 0.2 mg/kg with propofol 1 mg/kg), Group E2 (esketamine 0.3 mg/kg with propofol 1 mg/kg), Group E3 (esketamine 0.4 mg/kg with propofol 1 mg/kg). The dosage of propofol was determined under the Chinese endoscopic guideline 12 and our previous research. 10 For the esketamine groups, esketamine was meticulously prepared based on body weight with normal saline to achieve the appropriate concentration in a 10 mL syringe by an independent researcher (XC).
Colonoscopy started when the patient reached a level of sedation corresponding to an OAA/S score of 2. An additional 0.5 mg/kg propofol was added to maintain the patient at the required sedation level. Once pulse oxygen saturation drops below 90%, jaw thrust maneuverer will be performed to open the airway. If saturation further deteriorates, we will increase the oxygen in the nasal cannula to 6 L/min and face mask ventilation will be considered when saturation is continuously below 80%. 13 The systolic pressure or the mean arterial pressure (MAP) continues to be lower than 40% of baseline, dopamine infusion will be given to treat the hypotension.
Outcome measurements
Hemodynamic and respiratory parameters were systematically recorded at distinct time points during the study, including the patient’s entry into the endoscopic room (T0), the induction of sedation (T1), the insertion of the colonoscope (T2), the removal of the colonoscope (T3), and the awakening of the patient (T4). The primary outcome of this trial was the incidence of hypotension, defined as a reduction of ⩾20% from the baseline blood pressure. 14 Secondary outcomes included additional cardiovascular adverse events, namely bradycardia (HR ⩽ 55 bpm or decrease ⩾20% from baseline), tachycardia (HR ⩾ 100 bpm or increase ⩾20% from baseline), hypertension (defined as systolic blood pressure ⩾160 mmHg or increase ⩾20% from baseline), 15 and desaturation (defined as pulse oxygen saturation reading ⩽90%) that occurs during the procedure.
Furthermore, to assess the comprehensive effects of various therapeutic approaches on the cardiovascular and respiratory systems, cumulative alterations of cardiovascular and respiratory parameters at various time points (T1–T4) were calculated relative to the baseline (T0). Total propofol dosage, duration of recovery from anesthesia, and satisfactory rating of patients and colonoscopists were recorded.
Statistical analysis
The calculation of the sample size was based on the pilot study. Using PASS software 11.0 (NCSS, LLC), we calculated a sample size of 80 based on an effect size of 0.422, derived from sedation group incidences, to achieve 90% power with a χ2 test (three degrees of freedom) at a 0.05 significance level. Considering an estimated 20% loss to follow-up, the overall required sample size was adjusted to 100 patients, evenly distributed with 25 patients assigned to each group.
In our study, data analysis followed the principle of intention-to-treat using the SPSS software (version 22.0; Chicago, IL, USA). For normal distribution data, continuous variables are represented by mean and standard deviation, while the median with quartile range is used for non-normal distribution data. Qualitative variables are represented by numbers and percentages. The incidence of hypotension was subjected to analysis using the Chi-square test or Fisher’s exact test when necessary. Following Bonferroni correction, the unadjusted p-value was adjusted by multiplication with three.
To compare the incidence of hypotension in three esketamine groups against Group P, relative risks (RRs) with 95% confidence intervals (CIs) were calculated. Comparative analysis of continuous data with normal distribution between the esketamine groups and propofol group employed the Dunnett test. Conversely, for non-normally distributed data, comparisons were conducted using the Kruskal-Wallis analysis, followed by the Bonferroni post hoc test. Significance was established at a two-sided p-value <0.05.
Results
As illustrated in Figure 1, the final analysis included a total of 100 patients, with 25 individuals allocated to each group. A comprehensive exposition of the demographic and baseline characteristics of patients across the diverse groups is detailed in Table 1, with no statistically significant differences observed among the four groups in terms of these parameters.

Flowchart of the study.
Baseline parameters for patients in different groups.

Data are expressed as means standard deviations, medians (interquartile range), or n (%).
BMI, body mass index; DBP, diastolic blood pressure; MAP, mean arterial pressure; OAA/S, Observer’s Assessment of Alertness and Sedation Scale; SBP, systolic blood pressure; SpO2, pulse oximetry.
There are significant differences in the incidence of hypotension, as shown in Table 2. The incidence of hypotension in Groups E1 (p = 0.003), E2 (p = 0.003), and E3 (p < 0.001) was significantly lower than in Group P. Compared to Group P, the RRs of hypotension in Groups E1, E2, and E3 were 0.44 (95% CI 0.15–0.73), 0.44 (95% CI 0.15–0.73), and 0.48 (95% CI 0.19–0.77) respectively. The cumulative change in systolic blood pressure after induction of sedation only in Group E3 (p = 0.012) was significantly higher than in Group P. Cumulative changes in diastolic blood pressure (DBP) after induction of sedation in Group E1 (p = 0.024), E2 (p < 0.001), and E3 (p = 0.006) were significantly higher than those of Group P. Cumulative changes in MAP after induction of sedation in Groups E1 (p < 0.001), E2 (p < 0.001), and E3 (p < 0.001) were all significantly higher than those of Group P, as shown in Figure 2. There was no significant difference in the cumulative changes in HR between the four groups. At different time points, the time of cardiovascular parameters such as systolic blood pressure, DBP, MAP, and HR in four groups and the interaction between groups are shown in Supplemental Table 1. Dunnett t-test showed that at T3 and T4, DBP at T3 and T4, in Group E2 was significantly higher than that in other groups at T3 (p = 0.037) and T4 (p = 0.038). MAP at T4, for Groups E1 and E2, was statistically higher than other groups, p = 0.046 and 0.049, respectively.
Comparison of adverse events in cardiovascular and respiratory systems among different groups.

Data are expressed as n (%).
Statistical difference compared to group P with p < 0.01.
Statistical difference compared to group P with p < 0.001.
SpO2, pulse oximetry.

Cardiovascular data, including the cumulative change from baseline value for (a) systolic pressure, (b) diastolic pressure, (c) MAP, and (d) HR after drug administration compared to baseline data. P-values after Bonferroni corrections for comparisons between groups E1, E2, and E3 and groups P are shown (*P < 0.05, **P < 0.01, ***P < 0.001).
The respiratory data of four groups at different time points, including tidal volume, respiratory frequency, ventilation per minute, and SpO2, have no significant statistical significance, as shown in Figure 3. The tidal volume in Group E3 was higher than other groups (p = 0.017). However, the minute ventilation showed no difference between the four groups. Table 2 summarizes the cardiovascular and respiratory adverse events among the different groups.

Respiratory data, including the cumulative change from baseline value for (a) tidal volume, (b) respiratory rate, (c) minute ventilation, and (d) SpO2 after drug administration compared to baseline data.
Additional related data during and after colonoscopy are summarized in Table 3. The total propofol dose in Groups E1 (p = 0.003), E2 (p < 0.001), and E3 (p < 0.001) were all significantly higher than in Group P. There is no significant difference in other data among the four groups at different time points.
Comparison of perioperative data among different groups.
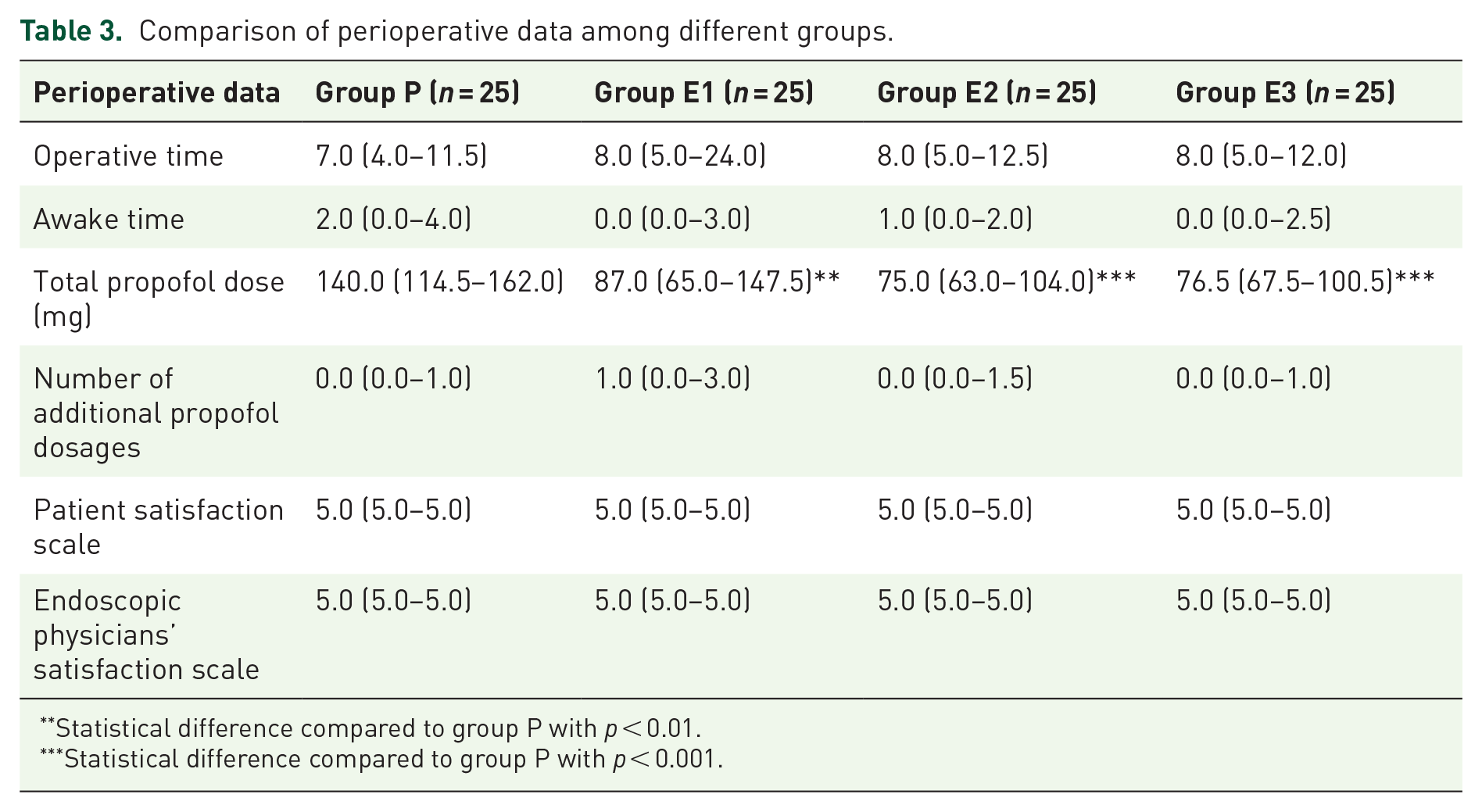
Statistical difference compared to group P with p < 0.01.
Statistical difference compared to group P with p < 0.001.
Discussion
In our study, we found that all three varying concentrations of esketamine effectively reduced the incidence of hypotension in patients undergoing colonoscopy, demonstrating notably more stable hemodynamics compared to the propofol group. No significant difference was found in respiratory parameters. In addition, esketamine was associated with a reduction in the required dosage of propofol.
The stable hemodynamics resulting from esketamine is consistent with previous studies.10,16 Among the four groups, patients in Group E3 had the most stable hemodynamics. It may be related to the sympathomimetic effect of esketamine, which could cause releasing of catecholamine and increase norepinephrine in circulation. 17 On the other hand, the primary impact of propofol on hemodynamics is a notable reduction in sympathetic activity, causing blood vessels to dilate and leading to decreased total peripheral resistance, which results in a drop in MAP. In our study, the total dosage of propofol in the esketamine groups was lower than that in the pure propofol group, due to the sedative and analgesic effects of esketamine by antagonizing NMDA receptors.18,19 The decreased dosage of propofol may make an additional contribution to the stable hemodynamics in the three esketamine groups. Age plays an important role in circulatory stability. Elder patients are generally more susceptible to propofol-induced hypotension. In our study, we recorded the ages of patients in four groups and found no statistically significant differences.
In our study, the heart rate in four groups had no significant difference. Only looking at heart rate does not reveal changes in the activity of the autonomic nervous system. Changes in autonomic nervous activity during colonoscopy play an important role in precipitating cardiovascular events such as arrhythmias, hypotension, hypertension, and myocardial infarction.20,21 The sympathetic and vagal components of the autonomic nervous system could be influenced by stimulation of the bowel during colonoscopy. Through the analysis of heart rate variability (HRV), a noninvasive technique for evaluating sympathovagal balance, it was observed that the dominant sympathetic component of the autonomic nervous system is enhanced in patients undergoing colonoscopy during midazolam or propofol-based sedation.22,23 Increased sympathetic modulation is also observed in moderate hypotension, a common cardiovascular event during colonoscopy under midazolam or propofol-based sedation, while esketamine decreased the incidence of hypotension.24,25 Further study about the HRV analysis is needed to investigate autonomic nervous balance fluctuation under esketamine sedation for colonoscopy.
Our research showed that there was no significant significance in the incidence of hypoxia or respiratory parameters. The effect of esketamine on respiration is not completely understood, which may be dose related. Studies have proved that esketamine can stimulate breathing at sub-anesthetic doses, 26 and higher doses may inhibit breathing. 27 Previous studies suggested that esketamine counteracted the effect of respiratory depression caused by opioids. 28 In our study, we did not observe any respiratory benefits from esketamine. This could be attributed to several factors. First, we did not administer opioids, and the respiratory depression induced by propofol is typically transient.29,30 Second, the impact of propofol on respiratory measures varies with dosage. 31 Our propofol dosage was relatively low, thereby exerting a relatively minimal effect on respiration. 32 Lastly, unlike gastroscopy, colonoscopy does not involve airway sharing and exerts less influence on breathing.
The recovery time in the four groups was of no difference. Both the total dosage of propofol and the use of esketamine may affect the recovery time. Although esketamine may delay the anesthetic recovery, the use of esketamine decreased the total dosage of the propofol. In addition, we found that compared with the propofol group, satisfactory levels of both patients and endoscopists were not affected in the three esketamine groups.
Limitations
Our study had some limitations. First, we use propofol by bolus rather than continuous infusion to keep all patients at a similar level of sedation. Continuous infusion offers greater hemodynamic stability compared to a bolus, yet achieving the desired sedation level takes a longer time. In addition, it requires more equipment, such as infusion pumps and associated electrical facilities, which can be challenging in an endoscopy suite with limited space. Second, we did not adjust the doses of propofol and esketamine based on age, as older patients may be more prone to have hypotension during propofol sedation, despite there being no significant age differences among the four groups. Further research is needed to clarify the role of esketamine in maintaining hemodynamic stability during colonoscopy in elderly patients. Finally, although we excluded patients with unstable cardiovascular disease, our study did include some stable patients classified as ASA III with good effort tolerance and well-managed underlying medical conditions.
Conclusion
In conclusion, this prospective randomized trial indicated that esketamine, across three varied concentrations, reduces the incidence of hypotension during propofol-based sedation for colonoscopy. Notably, the 0.4 mg/kg esketamine concentration demonstrated the most consistent hemodynamics among the three concentrations. Meanwhile, propofol dosage was decreased in the esketamine groups. However, further trials investigating esketamine are necessary to enhance our understanding of its clinical application.
Supplemental Material
sj-doc-2-taw-10.1177_20420986241278499 – Supplemental material for Impact of different doses of esketamine on the incidence of hypotension in propofol-based sedation for colonoscopy: a randomized controlled trial
Supplemental material, sj-doc-2-taw-10.1177_20420986241278499 for Impact of different doses of esketamine on the incidence of hypotension in propofol-based sedation for colonoscopy: a randomized controlled trial by Mengyue Fu, Bo Sheng, Rui Liu, Yongjie Li, Guizhen Chen, Hai Chen, Xuehan Chen, Guangyou Duan, He Huang, Jie Chen and Yuanjing Chen in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-1-taw-10.1177_20420986241278499 – Supplemental material for Impact of different doses of esketamine on the incidence of hypotension in propofol-based sedation for colonoscopy: a randomized controlled trial
Supplemental material, sj-docx-1-taw-10.1177_20420986241278499 for Impact of different doses of esketamine on the incidence of hypotension in propofol-based sedation for colonoscopy: a randomized controlled trial by Mengyue Fu, Bo Sheng, Rui Liu, Yongjie Li, Guizhen Chen, Hai Chen, Xuehan Chen, Guangyou Duan, He Huang, Jie Chen and Yuanjing Chen in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
Thanks to Hongtao Shi, the genius senior endoscopist in our medical center, who gave us a lot of support during the colonoscopy procedure. Thanks to Bin Shu, one of our attending anesthetists who gave us a lot of help with statistical analysis.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
