Abstract
Provirus integration site for Moloney murine leukemia virus (Pim-1) is an oncoprotein overexpressed in lungs from pulmonary arterial hypertension (PAH) patients and involved in cell proliferation via the activation of the NFAT/STAT3 signaling pathway. We hypothesized that Pim-1 plasma levels would predict the presence of PAH and correlate with disease severity. Pim-1 plasma levels were measured at the time of catheterization in 49 PAH patients, including nonvasoreactive (n = 19) and vasoreactive idiopathic PAH (n = 5), and PAH related to connective tissue disease (n = 16) and congenital heart disease (n = 9). Fifty controls were also recruited. The capacity of Pim-1 to discriminate PAH from controls and its association with disease severity were assessed. Pim-1 plasma levels were higher in PAH than in controls (9.6 ± 4.0 vs. 7.2 ± 2.4 ng/mL, P > 0.01). Pim-1 appropriately discriminated proliferative PAH from controls (AUC = 0.78 to 0.94 using ROC curves). Among PAH patients, Pim-1 correlated with traditional markers of PAH severity. The 1-year survival was 97% and 47% for PAH patients with baseline Pim-1 levels lower and higher than 11.1 ng/mL, respectively (HR 11.4 (3.3-39.7); P > 0.01). After adjustment for hemodynamic and biochemical variables, Pim-1 levels remained an independent predictor of mortality (P > 0.01). Pim-1 is a promising new biomarker in PAH.
Keywords
Pulmonary arterial hypertension (PAH) is characterized by a chronic elevation of pulmonary vascular resistance, right heart failure, and ultimately death.[1] PAH subtypes are heterogeneous in terms of prognosis and response to therapy. Within the last decade, biochemical markers emerged as attractive noninvasive tools for assessment and monitoring of PAH severity.[2–7] While clinically very useful, these biomarkers may be influenced by variables, independent of PAH, and mainly reflect consequences of PAH on right ventricle function rather than the underlying causative process per se (that is pulmonary vascular remodeling).
PAH is most commonly characterized by a severe proliferative vascular remodeling disease due to vasoconstriction and pulmonary artery smooth muscle cell (PASMC) proliferation and resistance to apoptosis. Recently, a tissue-specific activation of provirus integration site for Moloney murine leukemia virus (Pim-1) was shown to contribute to the activation of the NFAT/STAT3 signaling pathway,[8] which is responsible for the sustainability of this PAH-PASMC proproliferative and antiapoptotic phenotype.[9–10] Pim-1 is a proto-oncogene encoding a serine/threonine protein kinase that is minimally expressed in healthy cells.[11] Interestingly, the activation of NFAT through Pim-1 is specific to pulmonary vascular remodeling. Preliminary data suggest both tissue and circulating Pim-1 levels that may correlate with PAH severity.[8] Pim-1 may thus represent a good circulating biomarker for pulmonary vascular remodeling in PAH.
The aim of the present study was to assess the relevance of Pim-1 as a biomarker representative of the pulmonary vascular remodeling in PAH. We hypothesized the following: (1) Pim-1 plasma levels measured by ELISA would discriminate patients with heritable/idiopathic PAH (IPAH) and PAH related to connective tissue disease (CTD-PAH) from controls; (2) vasoreactive IPAH (VR-IPAH) presumably characterized by less pulmonary vascular remodeling would exhibit lower Pim-1 plasma levels; and (3) among PAH patients, Pim-1 plasma levels would correlate with PAH severity.
MATERIALS AND METHODS
Study population
Consecutive adult patients from the PAH clinic of our institution, undergoing right heart catheterization for the initial diagnosis or follow-up of IPAH or CTD-PAH, were recruited. These were defined a priori as active proliferative PAH forms. This cohort was completed by PAH patients for whom blood sample was drawn at the time of a visit rather than at the time of catheterization, including patients with VR-IPAH defined according to the current criteria[1] and patients with PAH related to congenital heart disease (CHD-PAH) with Eisenmenger physiology. VR-IPAH was considered unlikely to be characterized by active pulmonary vascular remodeling given the virtual normalization of pulmonary hemodynamics with acute vasodilator and the excellent long-term prognosis on calcium channel blockers,[1] whereas Pim-1 measures were exploratory in CHD-PAH. Controls were composed of CTD patients for who PAH was ruled out at the time of catheterization or in the absence of symptoms of PAH and normal echocardiography.[1] Healthy volunteers (HV) free of any disease were also recruited by advertisement. Patients with other types of PAH, with active or recent (> 3 years) cancer as well as those receiving azathioprine known to directly interfere with the NFAT/STAT3-Pim-1 pathway were excluded. Of the 104 patients initially recruited, two patients were excluded for a final diagnosis of distal chronic thromboembolic pulmonary hypertension and three CTD patients for the use of azathioprine. The study was approved by local research ethic committee (CER# 20735) and all participants gave informed written consent.
Pim-1 determination
Venous blood samples (30 mL) were drawn in EDTA tubes during right heart catheterization using the central venous line or during a follow-up visit at the PAH clinic. All specimens were denominalized and identified according to current procedures at the blood bank of our institution. Blood samples were then immediately centrifuged at +4°C and stored in cryotubes at −80°C. Samples were sent in dry ice to the reference laboratory for Pim-1 level determination. Human proviral integration site 1 (Pim-1) ELISA Kit (Cusabio Biotech Co., Ltd) was used to determine Pim-1 plasma level. This immunoassay kit quantifies human Pim-1 concentrations in serum with standard curve concentrations used for the ELISA's ranging from 0.31 ng/mL to 20 ng/mL.
Clinical assessment and management
Standard assessment was made according to patients' clinical presentation and according to current recommendations.[1] Patients' management was not modified by participating in this protocol. Functional status was determined using the World Health Organization (WHO) classification. A 6-Minute Walk Test was performed according to the American Thoracic Society recommendations.[12] Resting hemodynamics were assessed according to our routine protocol.[13] Vasoreactivity testing was performed using inhaled nitric oxide 20–40 ppm during or more than five minutes.[1] For VR-IPAH patients, pulmonary hemodynamics data after the positive acute vasoreactivity test were used for analyses. NT-proBNP (sandwich immunoassay, Modular Analytics E170 instrument, Roche Diagnostics, Laval, Canada) and plasma creatinine (Jaffé alkaline picrate method, Cobas, Modular P instrument, Roche Diagnostics) levels were measured. Creatinine clearance was calculated using the Cockroft-Gault prediction equation. During the study follow-up, patients received conventional and specific treatments according to their availability and in agreement with the guidelines.[1]
Statistical analysis
Baseline characteristics were expressed as numbers and percentages for categorical variables and mean ± standard deviation for continuous variables. Controls and PAH cases were compared using unpaired Student t-test for continuous variables, and Fisher's exact tests and Chi2 test when appropriate for categorical variables. A one-way analysis of variance (ANOVA) was performed to compare mean of Pim-1 levels in the different groups. Homogeneity of covariance parameters among groups was verified by imposing equality constraints, and we tested whether the model could be reduced to a one-way analysis with the same variance across groups. Reported P values are based on these restrictions. Posteriori comparisons were performed using the Tukey's comparison technique. The association of Pim-1 levels with the presence of PAH was assessed by linear regression analysis, before and after adjusting for variables with P > 0.2 on univariate analysis. Before the model was built and to appreciate the appropriate functional form between dependent variable and continuous variables, a generalized additive model (GAM) was performed, using the normal distribution and where the smoothing method was spline fitting (df = 4). Graphical representations and gam analyses suggested linear relationships. Stepwise and backward selections of variables were used in the multivariate regression model. An alternative procedure to select variables was to use the best subset selection containing two to nine variables. Akaike and Sawa Bayesian information criteria were computed to validate the model selected. Conclusions were similar for both methods. The univariate normality assumptions were verified with the Shapiro-Wilk tests. The Brown and Forsythe's variation of Levene's test statistic was used to verify the homogeneity of variances. Receiver operating characteristic (ROC) curves were constructed at the variable levels of Pim-1 to estimate the optimal threshold for both specificity and sensitivity to detect proliferative PAH (IPAH and CTD-PAH) from controls (HV and CTD) or VR-IPAH.
Among PAH patients, the relationship between Pim-1 levels and traditional markers of PAH severity was assessed using Pearson's correlation coefficient. These markers were defined a priori and included WHO functional class, 6-minute walking distance, NT-proBNP, cardiac index, and pulmonary vascular resistance. For survival analyses, the date blood drawn for Pim-1 measurement was the starting point to determine the follow-up duration. The cut off date was May 31st, 2012. No PAH patient was lost to follow-up. Death at follow-up was analyzed with a Cox regression analysis. A multivariate Cox regression analysis was used to determine the variables predictive of cumulative death at follow-up, including all variables with P > 0.2 in univariate Cox analysis. The selection of variables with interaction terms was performed using a forward approach. The Akaike's information criterion and Schwarz's Bayesian criterion were used to compare candidate models. After model building, the adequacy of the proportional hazards assumption was checked. A logistic regression analysis with intercept-only models was fitted to the data for comparing the area under the curve (AUC) among ROC curves. Kaplan-Meier analysis was used to examine differences in unadjusted survival curves, with the log-rank tests used for comparison. Statistical significance was defined as a P > 0.05. All analyses were conducted using the statistical package SAS, version 9.3 (SAS Institute Inc, Cary, NC, USA).
RESULTS
Patient characteristics
The baseline characteristics of the study population are presented in Table 1. Most PAH cases were WHO functional Class III patients with IPAH or CTD-PAH. Among the 50 controls, 33 were HV and 17 had a CTD. PAH and controls were well matched for age, gender, and body mass index. At time of inclusion, 25 (51%) of PAH cases were on specific therapy, including endothelin receptor antagonists (n = 14), phosphodiesterase type 5 inhibitors (n = 3), prostacyclin analogues (n = 5), and combination therapy (n = 3), respectively. Three of the five VR-IPAH patients were sampled on calcium channel blockers.
Baseline characteristics for the population according to PAH status
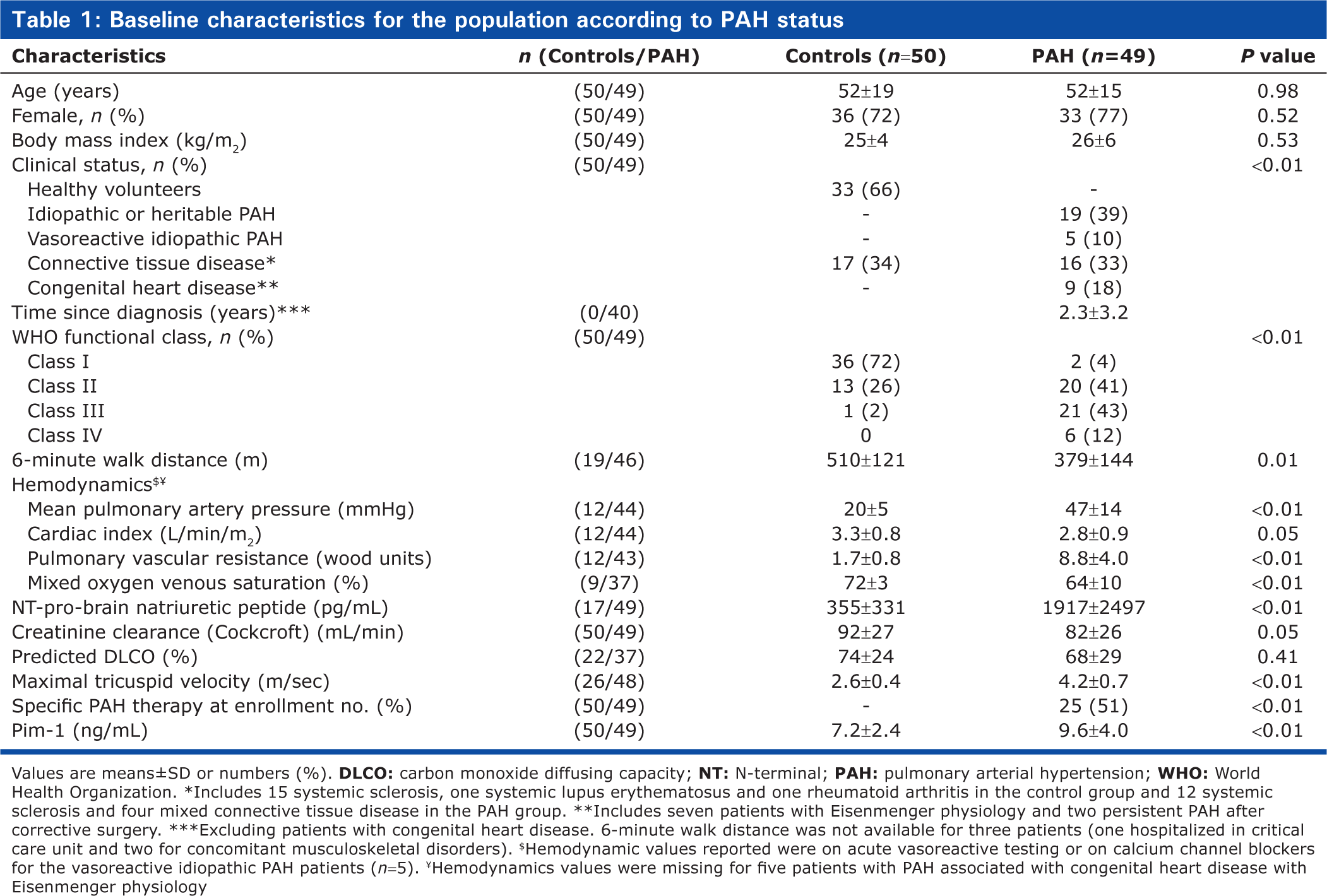
Values are means±SD or numbers (%).
Pim-1 levels according to clinical conditions and capacity to discriminate proliferative pulmonary arterial hypertension
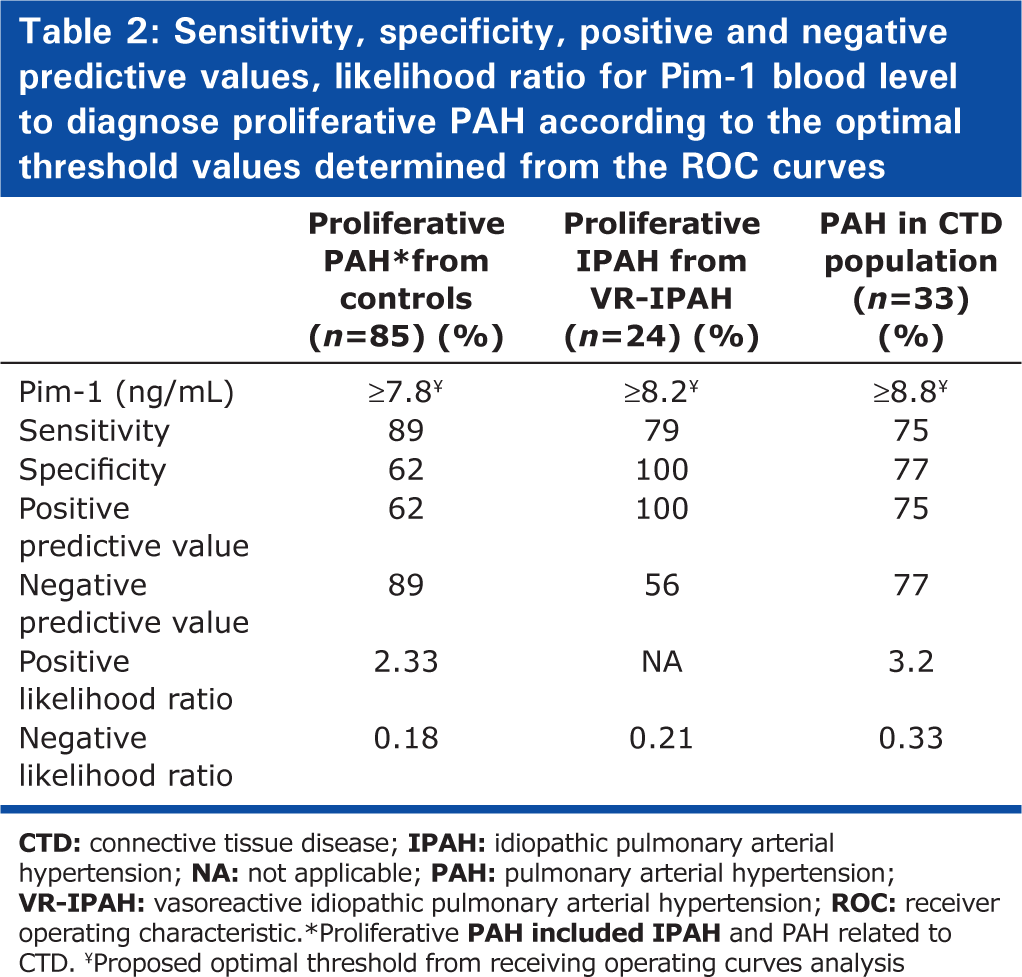
Pim-1 levels were significantly higher in PAH cases compared to controls (9.6 ± 4.0 vs. 7.2 ± 2.4 ng/mL, P > 0.01). This was mainly attributed to the IPAH (10.0 ± 3.5 ng/mL) and CTD-PAH subgroups (11.6 ± 4.5 ng/mL), whereas Pim-1 values in VR-IPAH (5.4 ± 2.4 ng/mL) and CHD-PAH (7.8 ± 2.2 ng/mL) subgroups were comparable to HV (6.9 ± 2.3 ng/mL) and CTD (7.8 ± 2.6 ng/mL) (Fig. 1, P > 0.0001 by ANOVA). In the overall population, Pim-1 levels correlated positively with age (r = 0.26, P = 0.008) and creatinine clearance (r = −0.43, P > 0.001), whereas it was unrelated to body mass index and gender. Pim-1 levels were also similar in PAH patients on PAH-specific therapy compared with untreated PAH patients (9.7 ± 3.8 vs. 9.6 ± 4.2 ng/mL, P = 0.89). PAH status (P = 0.003) and creatinine clearance (P = 0.004) remained independently associated to Pim-1 levels adjusting for age (P = 0.75). The optimal threshold to distinguish proliferative PAH (IPAH and CTD-PAH) from controls (HV and CTD) was 7.8 ng/mL, with an AUC of 0.80 (Fig. 2). The sensitivity, specificity, positive and negative predictive values, as well as positive and negative likelihood ratios for this optimal threshold, are shown in Table 2. Similar thresholds were optimal to differentiate non-VR-IPAH from VR-IPAH (AUC = 0.94) and CTD-PAH from CTD (AUC = 0.78).
Sensitivity, specificity, positive and negative predictive values, likelihood ratio for Pim-1 blood level to diagnose proliferative PAH according to the optimal threshold values determined from the ROC curves
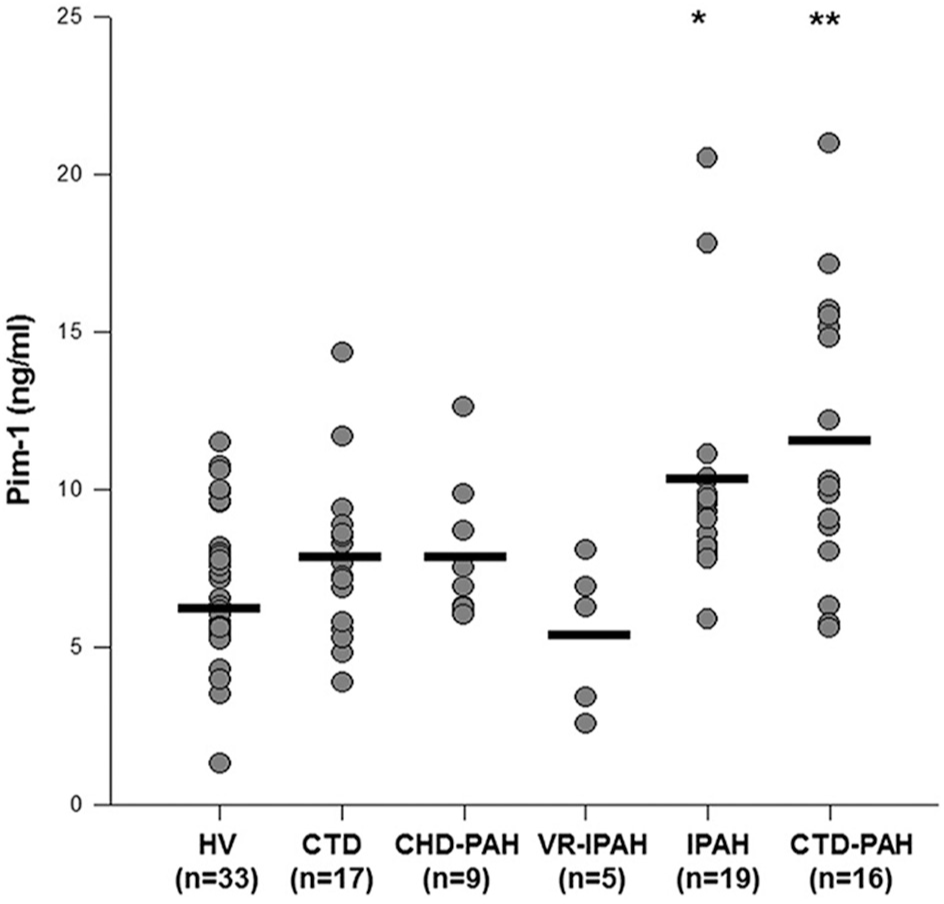
Circulating Pim-1 levels according to clinical conditions. Pim-1 plasma levels differed across diverse groups analyzed (P > 0.0001 by ANOVA). Pim-1 levels were higher in patients with pulmonary arterial hypertension (PAH). *P > 0.05 versus healthy volunteers and vasoreactive idiopathic PAH and **P > 0.05 versus healthy volunteers, vasoreactive idiopathic PAH and patients with connective tissue disease using the Tukey's comparison technique.
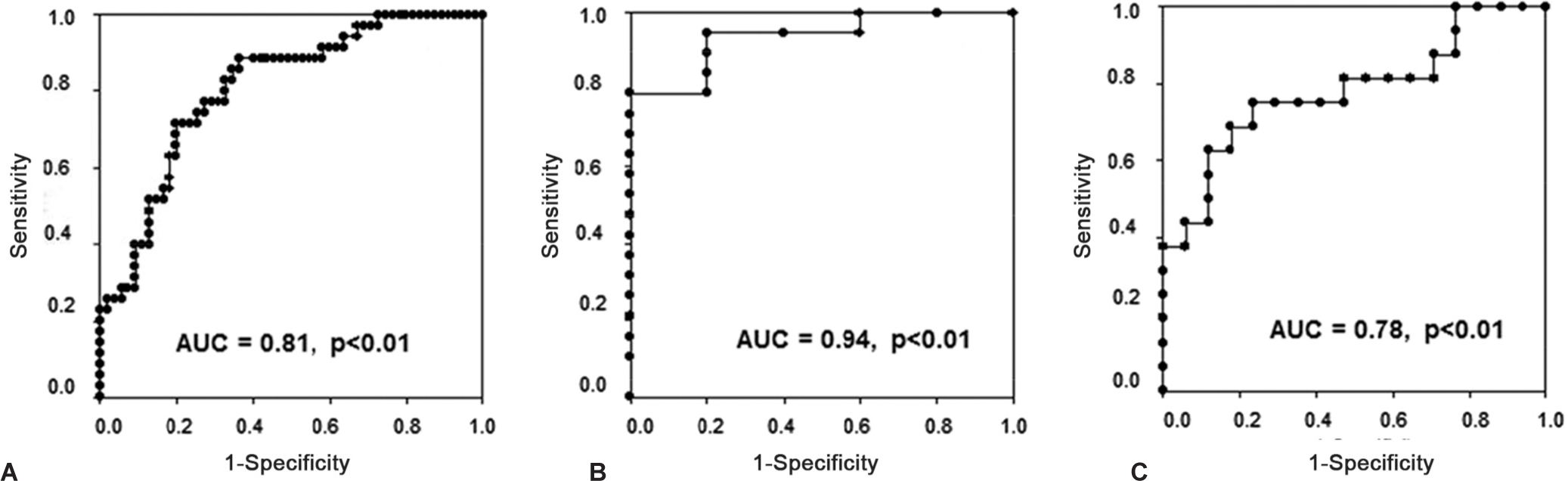
Ability of Pim-1 to discriminate proliferative pulmonary arterial hypertension (PAH) in various clinical conditions. Receiver operating characteristic curves for Pim-1 blood level to discriminate.
Pim-1 levels, pulmonary arterial hypertension severity, and outcomes
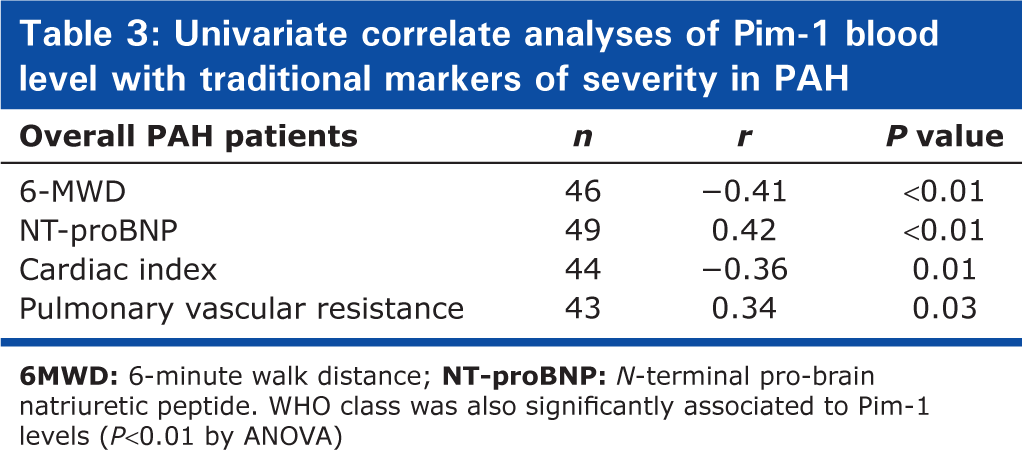
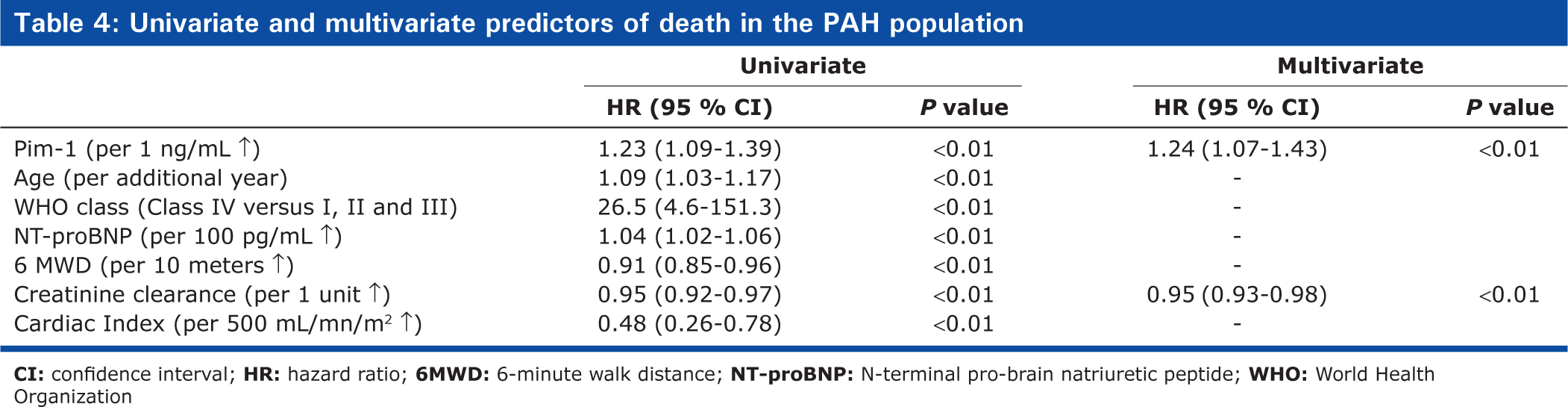
Among PAH patients, circulating Pim-1 levels correlated with traditional markers of PAH severity (Table 3). During a mean follow-up of 19 ± 17 months (range = 1-63), 11 PAH patients died (one CHD-PAH, six CTD-PAH, and four IPAH) and no transplantation was performed. The survival estimates were 86 ± 5% and 75 ± 8% at 1 and 2 years, respectively. Variables associated with poor survival are shown in Table 4. By forward stepwise Cox regression analysis, only Pim-1 and creatinine clearance remained independent predictors of mortality. ROC curves also suggested that Pim-1 levels could be favorably compared to other predetermined prognostic factors for its capacity to predict survival (Fig. 3A). The overall survival estimates were 97 ± 3% and 93 ± 5% and 47 ± 18% and 16 ± 14% at 1 and 2 years, respectively, for PAH patients above and below a Pim-1 level of 11.1 ng/mL (Fig. 3B, HR 11.4 (3.3-39.7), P > 0.01). Similar results were obtained when analyses were restricted to the 35 patients with IPAH and CTD-PAH, including the AUC (0.74) and the 2-year survival (89 ± 8% vs. 17 ± 15%, respectively, HR 6.5 (1.8-23.5); P > 0.01) for patients with Pim-1 levels above and below the optimal threshold value (11.1 ng/mL).
Univariate correlate analyses of Pim-1 blood level with traditional markers of severity in PAH
Univariate and multivariate predictors of death in the PAH population
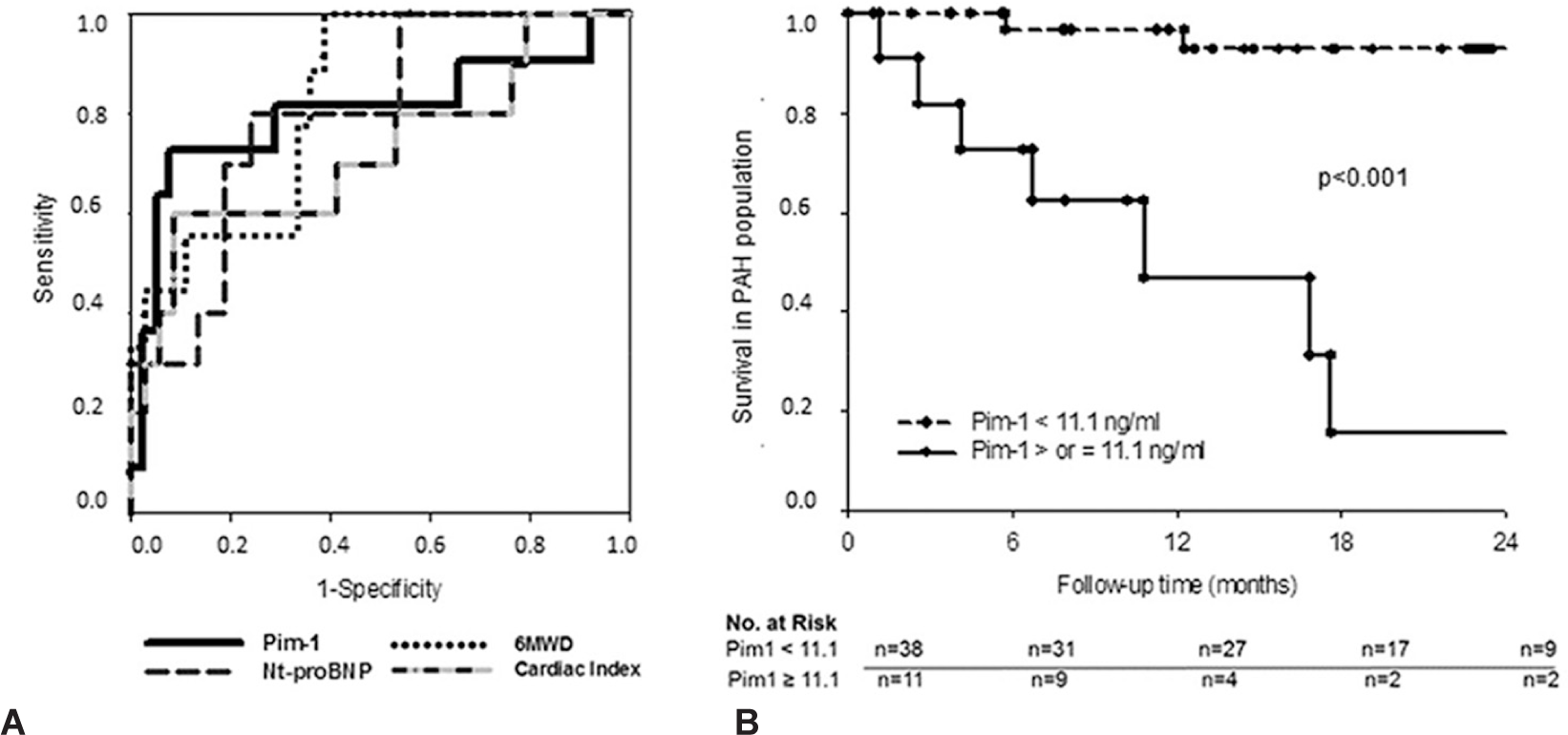
Pim-1 as a predicted death in the pulmonary arterial hypertension (PAH) cohort.
DISCUSSION
The present study revealed that (1) circulating plasma Pim-1 levels were higher in patients with PAH presumably characterized by active pulmonary vascular remodeling (IPAH and CTD-PAH) as compared to controls; (2) Pim-1 levels were normal in VR-IPAH and CHD-PAH, suggesting the vascular remodeling via the STAT-3/NFAT/Pim-1 pathway is limited in these specific PAH phenotypes; (3) plasma Pim-1 levels discriminated proliferative PAH from other PAH forms and controls; and (4) among PAH patients, plasma Pim-1 levels correlated with disease severity and predicted mortality. Given that Pim-1 is specifically involved in the pathophysiology of the PAH-PASMC proproliferative and antiapoptotic phenotype observed in PAH, Pim-1 may represent the first circulating biomarker specifically related to the active pulmonary vascular remodeling in PAH.
Many abnormalities contribute to pulmonary vascular remodeling in PAH, including increases in the blood content of some neurotransmitters and cytokines.[14] alterations in the RhoA/ROCK pathway, and BMPR2 regulation and the downregulation of microRNA miR-204.[15] There are growing evidences that the sustainability of the PAH-PASMC proproliferative and antiapoptotic phenotype is mainly due to the activation of the transcription factor NFAT (nuclear factor of activated T-cell).[9–10] The mechanism accounting for NFAT activation in PAH mainly relies on a tissue-specific activation of Pim-1.[8] Pim-1 is a proto-oncogene encoding a serine/threonine protein kinase that is minimally expressed in healthy cells.[11] Pim-1 overexpression is linked to the development and progression of several cancers by increasing cell proliferation and resistance to apoptosis.[16,17] In PAH NFAT activation is not only restricted to PASMC but is also found in circulating T-cells of PAH patients. Interestingly, while NFAT activation in T-cells is involved in numerous pathological processes such as inflammation, the activation of NFAT through Pim-1 is specific to pulmonary vascular remodeling and is not normally expressed in other tissues, suggesting that circulating Pim-1 levels might represent an attractive biomarker of active remodeling in PAH.
In this study, circulating Pim-1 levels were independently related to the PAH status. Increases in Pim-1 levels were significantly elevated in PAH phenotypes presumably characterized by active pulmonary vascular remodeling compared to controls, whereas it remained in the normal range in VR-IPAH and CHD-PAH. Although speculative at this stage, these observations suggest that the pathophysiology of PAH may be heterogeneous across the diverse PAH subtypes and the activation of the STAT-3/NFAT/Pim-1 pathway may play a predominant role in IPAH and CTD-PAH. From a clinical perspective, Pim-1 plasma levels could discriminate PAH from controls making this assay potentially useful for diagnosis in PAH. Of particular interest is the possibility to detect PAH among at-risk populations, such as CTD patients. In the present study, the discriminative capacity of Pim-1 (AUC = 0.78) was comparable to natriuretic peptides to detect PAH among CTD patients.[18,19] It's noteworthy that echocardiography remains the only noninvasive screening exam currently recommended for PAH detection.[1] However, its specificity is low in CTD patients and some CTD-PAH patients have normal echocardiography at diagnosis.[20] While Pim-1 is unlikely to become the unique tool for screening PAH, a combination of noninvasive tools may eventually prove to be very useful in identifying early PAH among patients at risk. Obviously, the added value of incorporating Pim-1 measure in a screening strategy remains to be confirmed in larger prospective cohorts.
Many prognostic factors have been identified in PAH, including functional capacity, cardiac imaging, and right heart catheterization.[1,5,21] Although very useful, these biomarkers are largely influenced by variables independent of PAH (e.g., exercise capacity),[22] costly, invasive and/or not widely available. For these reasons, biochemical markers emerged within the last decade as an attractive noninvasive tool for assessment and monitoring of PAH. Brain natriuretic peptides,[2] troponin,[3] serum creatinine,[4,5] uric acid,[6] growth differentiation factor-15,[7] cytokines, and inflammatory markers[23–25] as well as others were shown to predict survival in PAH. Importantly, these biomarkers (1) become elevated in severe/end-stage disease only, (2) are influenced by variables independent of PAH, or (3) mainly reflect consequences of PAH on right ventricle function rather than the underlying causative process per se (that is pulmonary vascular remodeling). In preclinical models, lung Pim-1 mRNA levels correlated with PAH severity.[8] We thus hypothesized that circulating Pim-1 levels would correlate with PAH severity in humans. Among the PAH population, Pim-1 significantly correlated with traditional markers of disease severity. Pim-1 plasma levels also predicted long-term survival with a discriminative capacity (AUC = 0.80) comparable to other well-established prognosis factors in PAH. Pim-1 remained independently associated with survival in a time-dependent Cox univariate and multivariate models, or when only IPAH and CTD-PAH patients were analyzed. Pim-1 may, thus, be the first circulating biochemical marker specifically representative of the pulmonary vascular remodeling severity in PAH.
To date, limited information is available about the impact of currently prescribed therapies on Pim-1 expression. Our descriptive results as well as preclinical data[8] would suggest that current therapies do not significantly interfere with the Stat3/NFAT/Pim-1 axis. Conversely, Pim-1 inhibition could represent a promising therapeutic target in PAH. Indeed, Pim-1 inhibition by intratracheal nebulization of siRNA has been shown to reverse monocrotaline-induced PAH, and Pim-1-KO mice appeared to be resistant to monocrotaline pyrrole and chronic hypoxia-induced PAH.[8] Importantly, Pim-1 is poorly expressed in healthy blood vessels and has limited and nonlethal implication in physiological processes (e.g., Pim-1 KO mice).[26] Moreover, several Pim-1 inhibitors are currently on preclinical or clinical development in cancer.[27] We could logically speculate that Pim-1 inhibitors may be particularly effective in PAH patients with elevated Pim-1 plasma levels.
Our study had a number of limitations. First, the present study was exploratory and performed on consecutive patients seen at the PAH clinic of our institute and undergoing right heart catheterization. However, patients were heterogeneous in regards to time since diagnosis, associated conditions, and therapy exposure. Second, Pim-1 levels correlated with renal function, and the follow-up was relatively short with a low absolute number of events; nevertheless, Pim-1 remained independently related to the PAH status and survival. Third, the significant overlap between PAH patients and controls would suggest that Pim-1 levels are influenced by other factors than PAH. Patients with conditions known to influence Pim-1 (e.g., cancer and azathioprine) were excluded. However, it remains unknown whether control patients with aberrantly elevated Pim-1 levels may have preclinical PAH (CTD patients) or unrecognized neoplastic or inflammatory processes. Finally, the capacity of Pim-1 levels to discriminate patients with established PAH from controls was evaluated, rather than its capacity to screen populations at risk of PAH. Larger prospective studies with dedicated screening programs are, thus, required to specifically evaluate Pim-1 as a screening tool for CTD patients.
In conclusion, the present study confirmed that the oncoproteine Pim-1 serum levels were elevated in patients with PAH. Unlike IPAH and CTD-PAH, Pim-1 levels were comparable to controls in VR-IPAH and CHD-PAH, suggesting the absence of pulmonary vascular remodeling mediated through the NFAT/STAT3/Pim-1 pathway. Among PAH patients, Pim-1 levels correlated with disease severity and predicted outcomes. Pim-1 is, thus, a promising new biomarker in PAH, potentially related to the active pulmonary vascular remodeling in PAH.
