Abstract
Pulmonary arterial hypertension (PAH) is a progressive pulmonary vascular disease associated with dysfunction of pulmonary artery endothelial cells and pulmonary artery smooth muscle cells (PASMCs). To explore the potential mechanism of miR-193-3p in pulmonary arterial hypertension, human PASMCs and rats were respectively stimulated by hypoxia and monocrotaline to establish PAH model in vivo and in vitro. The expressions of miR-193-3p and p21-activated protein kinase 4 (PAK4) in the lung samples of PAH patients and paired healthy samples from the healthy subjects in PHA cells and rats were detected by quantitative reverse transcriptase-PCR. Morphological changes in lung tissues were determined using hematoxylin and eosin staining. Right ventricular systolic pressure (RVSP) and ratio of right ventricle to left ventricle plus septum (RV/LV p S) were measured. The binding relationship between miR-193-3p and PAK4 was analyzed by TargetScan and verified by luciferase reporter assay. Cell viability, apoptosis, and migration were detected by 3-(4, 5-Dimethylthiazol-2- yl)-2,5-diphenyltetrazolium bromide (MTT) flow cytometry, and wound-healing assays, respectively. The protein expressions of PAK4, proliferating cell nuclear antigen (PCNA), P21, p-AKT, and AKT in vivo or in vitro were determined by Western blot. In this study, we found that in pulmonary arterial hypertension, miR-193-3p expression was downregulated and PAK4 expression was up-regulated. MiR-193-3p directly targeted PAK4 and negatively regulated its expression. Hypoxia condition promoted cell proliferation, migration, and inhibited apoptosis accompanied with increased expressions of PCNA and p-AKT/AKT and decreased expression of P21 in PASMCs. MiR-193-3p overexpression attenuated the effects of hypoxia on PASMCs via downregulating PAK4. Monocrotaline treatment increased p-AKT/AKT and decreased P21 expression and caused pulmonary vascular remodeling in the model rats. MiR-193-3p overexpression attenuated pulmonary vascular remodeling, decreased p-AKT/AKT, and increased P21 levels via downregulating PAK4 in monocrotaline-induced rats. The results in this study demonstrated that upregulation of miR-193-3p reduced cell proliferation, migration, and apoptosis of PAH in vitro and pulmonary vascular remodeling in PAH in vivo through downregulating PAK4.
Introduction
Pulmonary arterial hypertension (PAH), which is a type of pulmonary hypertension that affects pulmonary vasculature primarily, 1 is a progressive pulmonary vascular disease related to high morbidity and mortality. 2 The application of novel targeted drugs (e.g. endothelin receptor blockers, prostaglandin analogs, phosphodiesterase-5 inhibitors) increased the five-year survival rate of PAH patients to 55% and even 70%, but long-term effect is still unfavorable.3,4 PAH is characterized by a persistent elevation of pulmonary arterial pressure, partially due to uncontrolled vascular remodeling. 5 Attenuating vascular remodeling has been considered as a strategy of managing PAH, but currently we still lack effective treatment targeting vascular remodeling. 6 The molecular mechanisms underlying vascular remodeling remains largely elusive, which require further investigation on the potential molecular mechanism of vascular remodeling in PAH and development of new effective treatments for PAH management.
MicroRNAs (miRNAs) are a series of small endogenous non-coding RNAs that serve as elemental modulators of numerous genes through binding to the 3′-untranslated regions (3′-UTR) of mRNAs, leading to the suppression of translation and degradation of the transcripts. 7 Dysregulation of miRNAs participates in the regulation of cardiovascular diseases such as neovascularization, vasoconstriction regulation, hypertension, and atherosclerosis. 8 It has been demonstrated that multiple miRNAs exhibit abnormal expressions in the development of PAH, and are involved in PAH progression and pulmonary arterial remodeling by regulating key factors in some downstream signaling pathways. 9 MiRNAs are reported to regulate pulmonary artery smooth muscle cells (PASMCs) phenotypes and pulmonary remodeling under hypoxia. 10 For example, upregulation of miRNA-17-5p facilitates the hypoxia-induced proliferation of human PASMCs by targeting p21 and phosphatase and tensin homologue (PTEN) 11 ; similarly, miR-34a facilitates the proliferation of human PASMCs through modulation of platelet-derived growth factor receptor alpha (PDGFRA) under hypoxia. 12 Upregulation of miR-214 contributes to the development of vascular remodeling in PAH caused by hypoxia via regulating cyclin L2 (CCNL2). 13 Downregulated miR-193 functions critically in the lungs from PAH rats. 14 Sharma et al. 15 suggested that miR-193-3p (miR-193) participates in the pathological process of PAH and attenuates pulmonary vascular remodeling; however, the potential molecular mechanism of miR-193-3p in pulmonary vascular remodeling of PAH remained to be fully explored.
In this study, we aimed to determine the expression of miR-193-3p and its corresponding downstream target gene as well as their interaction in pulmonary vascular remodeling of PAH in vitro and in vivo, hoping to provide new biomarkers for PAH therapy.
Materials and method
Ethics statement
All the patients had signed informed consent, and agreed that their tissues and plasma would be used for any experimental work involving humans. The clinical trial program had been reviewed and approved by the Ethics Committee of Tianjin Chest Hospital (TCH20180211015).
In compliance with the guidelines of the China Council on Animal Care and Use, all experiments for animals were performed in Tianjin Chest Hospital and approved by the Committee of Experimental Animals of Tianjin Chest Hospital (TCH20190325066). Efforts were devoted to minimize their sufferings.
Patients and clinical sample collection
Before operation, 10 lung tissue samples of PAH patients (six females and four males, average age of 36.1 ± 2.2 years, mean PAH was 86 ± 16 mm Hg) who received lung transplantation and 10 paired healthy control samples (seven females and three males, average age of 34.8 ± 2.0 years) from the unused donor control subjects were collected at Tianjin Chest Hospital from March 2018 to March 2019. All the PAH patients did not have a history of local or systemic treatment. The tissue samples were immediately transferred into chilled oxygenated Krebs solution (pH 7.4) containing 116.6 mM NaCl, 21.9 mM NaHCO3, 11 mM D-glucose, 1.2 mM MgSO4, 4.2 mM KCl, 1.6 mM NaH2PO4, and 2.5 mM CaCl2.
Cell culture and treatment
Human PASMCs were obtained from Cell Applications (352-05a, San Diego, CA, USA) and maintained in SmGM-2TM smooth muscle growth medium-2 (CC-3182, Lonza, USA, www.lonza.com/) with 5% fetal bovine serum (FBS). For normoxic treatment, the cells were cultured with 21% O2, 5% CO2, and balanced N2; for hypoxic induction, the cells were cultured with 2% O2, 5% CO2, and balanced N2 for the indicated time intervals, following a previous report. 16
Cell transfection
MiR-193-3p mimic (5′-AACUGGCCUACAAAGUCCCAGU-3′) and corresponding mimic control (5′-CGCAGGUCAAGUGGCCGACUUA-3′) were purchased from GenePharma. Modified pcDNA3.1 vector containing PAK4 sequence was commercially constructed by RiboBio (Guangzhou, China). Empty pcDNA3.1 vector (V79020, ThermoFisher, USA) was used as a negative control (NC). The PASMCs were transfected with miRNA mimics and pcDNAs using Lipofectamine 3000 Reagent (L3000015, Thermo Fisher Scientific, Inc.), according to the manufacturers’ instruction.
Luciferase reporter assay
TargetScan V7.2 (www.targetscan.org/vert_72/) was used to explore targets of miR-193-3p. The 3′-UTR segment of PAK4 (5′-
Cell viability
Cell viability of PASMCs was detected using MTT kit (11465007001, Sigma-Aldrich, Shanghai, China) following manufacturer’s protocol. In short, before the addition of MTT solution (5 mg/mL), the treated cells (1 × 103 cell/well) were inoculated into a 96-well plate for 24 h. Next, the cells were cultured for 4 h at 37°C. The absorbance at 490 nm was determined by a microplate reader (Tecan, Switzerland). After 4-h incubation at 37°C, the cells were further added with 150 μL of dimethylsulfoxide. Microplate reader (Tecan, Switzerland) was then employed to measure the absorbance at 570 nm.
Immunofluorescence assay
To determine the proliferation of the PASMCs, after transfected under hypoxic conditions for 48 h, the cells were fixed with 4% paraformaldehyde at room temperature for 15 min, and incubated overnight with anti-PCNA antibody at 4°C. Alexa Fluor 488 conjugated mouse IgG was used as a secondary antibody. Images were taken using a laser scanning confocal microscope (Leica, Heidelberg, Germany). 2-(4-Amidinophenyl)-6-indolecarbamidine dihydrochloride (DAPI) was used to counterstain nuclei and the nuclei of cell positive for PCNA was calculated.
Flow cytometry assay
To determine the apoptosis rate of the PASMCs, Annexin V-FITC and propidium iodide (PI) double staining was conducted using an Annexin V-FITC Apoptosis Detection Kit (CA1020, Solarbio, China). Briefly, the treated cells were collected, washed twice with phosphate buffer solution (PBS), and resuspended at 1 × 106 cells/mL in 100 μL of binding buffer. Then, the cells were treated with Annexin V-FITC and PI for 15 min at 37°C in the dark and blended with 400 μL of binding buffer. Flow cytometer Accuri™ C6 (BD Biosciences) was performed within 1 h. The results were analyzed by the Cell Quest software 3.3 (Becton-Dickinson).
Wounding-healing migration assay
Cell migration of PASMCs was determined by wound-healing assay. In short, the transfected cells (4 × 105 cells/well) were plated into in the complete medium in six-well plates. Following 8-h culture, the PASMCs were starved in SmGM-2 medium with 0.2% FBS overnight. Next, a scratch was performed on the cell monolayer using a 10 μL tip and the previous medium was replaced by complete medium. The images were photographed at 0 h under a 100 × inverted microscope (Ts2r-FL, Nikon, Japan). Then the cells were exposed to hypoxia or normoxia for 24 h, and images were captured by the same method. Cell migration rate was determined by measuring the reduced width of scratch on the PASMCs.
Animal model and groups
Thirty adult male Sprague Dawley rats (body weight 160–200 g) were purchased from the Slack Laboratory Animal based in Shanghai. A PAH rat model was induced by monocrotaline (MCT). The rats were randomly divided into five groups (
Recombinant adenovirus containing PAK4 (AdPAK4) or a green fluorescent protein (GFP)-only virus control (AdControl) was obtained from GenePharma (Shanghai, China). GFP-only virus control was a NC for AdPAK4. AgomiR-miR-193-3p and control agomiR (agomiR-NC) were purchased from RiboBio. To determine the effects of PAK4 and miR-193-3p in vivo, the PAH rats were nebulized with 50 μL agomiR-miR-193-3p (20 µmol/L) or/and 50 μL adenovirus (1.5 × 108 pfu) once a week for two weeks, as previously reported. 16
Hemodynamic measurements
Hemodynamic parameters were obtained: A heparinized pressure catheter (Millar Instruments, Houston, TX) was inserted into the right ventricle of rats through the right ventricle. Right ventricular systolic pressure (RVSP) was assessed using a physiological data acquisition system (LabChart 7.0; AD Instruments, Australia). The ratio of right ventricle to left ventricle plus septum (RV/LV + S) was used as an indicator of right ventricular hypertrophy (RVH). The pressure was recorded within 2 min and analyzed using Powerlab Pro software (AD Instruments, Germany).
Hematoxylin and eosin staining
Pulmonary artery tissues of rats were immediately collected after sacrifice and fixed with 10% paraformaldehyde for at least two days at room temperature, and then decalcified in 0.5 M EDTA (pH = 8.0) and embedded in paraffin. Sections were cut into 3-μm thick, mounted on glass slides, stained with an hematoxylin and eosin (H&E) staining kit (G1120, Solarbio, China) and visualized using a 100 × inverted microscope (Ts2r-FL, Nikon, Japan). To quantify the wall thickness (WT%) and the vessel area percentage (WA%) to evaluate the pulmonary vascular remodeling and the external and internal area: WT% = (external diameter – internal diameter)/external diameter × 100%. WA% = (external area – internal area)/external area × 100%.
Western blot
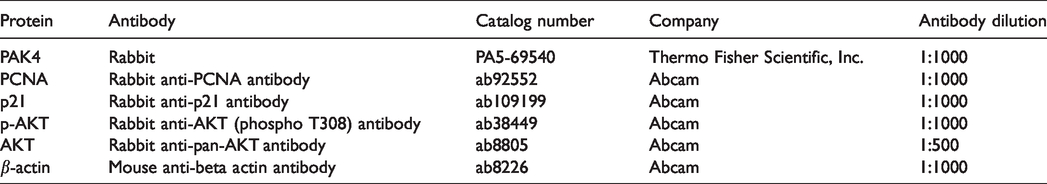
Total protein was isolated from the cultured PASMCs and collected from pulmonary artery tissues using a radio immunoprecipitation assay buffer (RIPA) buffer (P0013, Beyotime, China), and the content was quantified by BCA protein assay kit (PC0020, Solarbio). The proteins were separated using 10% SDS-PAGE and blotted onto polyvinylidene difluoride (PVDF) membranes (FFP32, Beyotime). Protein markers (PR1910 (11-180kDa) and PR1920 (11-245KD)) were purchased from Beijing Solarbio Science & Technology Co., Ltd. After blocking the membranes using 5% bovine serum albumin (BSA) for 2 h 37°C, the membranes were first treated with primary antibodies (presented in Table 1) overnight at 4°C and then treated with corresponding secondary antibodies (ab205719 or ab205718, Abcam, USA). Finally, the membranes were subjected to ECL solution (P0018FS, Beyotime), and specific bands were visualized by a Quantity One 1-D Analysis Software (Bio-Rad). The protein expression was normalized to that of β-actin. Primary antibodies used for western blots were listed in Table 1.
List of primary antibodies used for western blots.
Quantitative reverse transcription polymerase chain reaction
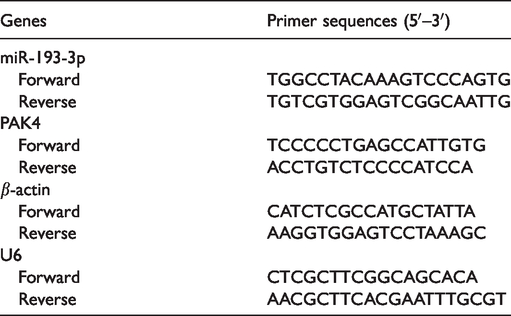
Total RNA was separated from the lung samples and cultured cells by applying TRIzol® (Invitrogen; Thermo Fisher Scientific, Inc.), and then quantified using a NanoDrop spectrophotometer (Thermo Fisher Scientific, Inc.). TaqMan™ MicroRNA Reverse Transcription Kit (4366596, Applied Biosystems; Thermo Fisher Scientific, Inc.) and TaqMan MicroRNA assay (4427975, Applied Biosystems) were applied to perform quantitative reverse transcription polymerase chain reaction (qRT-PCR) for determining miR-193-3p expression. U6 was an endogenous control. Reverse transcription-PCR kit (RR036B, Takara) and TB Green PCR reagent (RR430A, Takara) were used to perform qRT-PCR for determining PAK4 expression in an Applied Biosystems 7300 Real-Time PCR system. β-actin was an internal control for PAK4. The relative expression of PAK4 was calculated by the 2−ΔΔCt method. 17 The primers (shown in Table 2) were obtained from Sangon Biotech (Shanghai, China).
Primer sequences used for quantitative reverse transcription polymerase chain reaction (qRT-PCR).
Data analysis
Data were presented as the means ± SD. Statistical analyses were conducted with SPSS version 13.0 software (SPSS, Chicago, IL, USA). Two-group comparison was performed with the Student’s
Results
The expressions of miR-193-3p and PAK4 in the lung tissues from PAH patients and their binding relationship
The expression of miR-193-3p was observed to be markedly decreased in PAH patients compared with healthy groups (Fig. 1a,
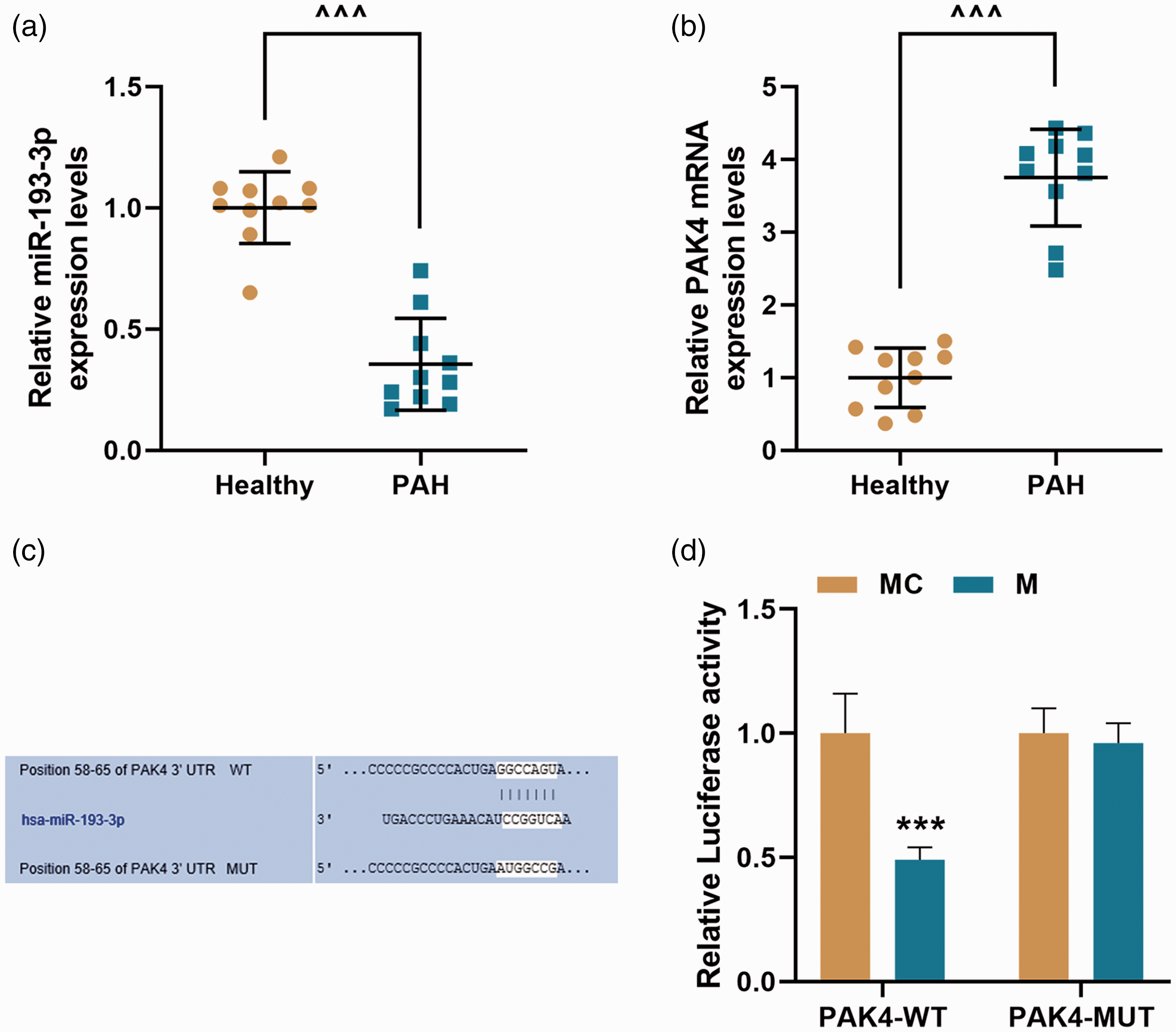
The expressions of miR-193-3p and PAK4 in the lung tissues from human with PAH and their binding relationship (
The expressions of miR-193-3p and PAK4 and their interaction in hypoxia-induced PASMCs
The HPASMCs were subjected to hypoxia for 0, 3, 6, 12, 24, and 48 h. The results from qRT-PCR showed that compared with normal group (normoxia treatment), miR-193-3p expression was markedly decreased, especially at 24 h (Fig. 2a,
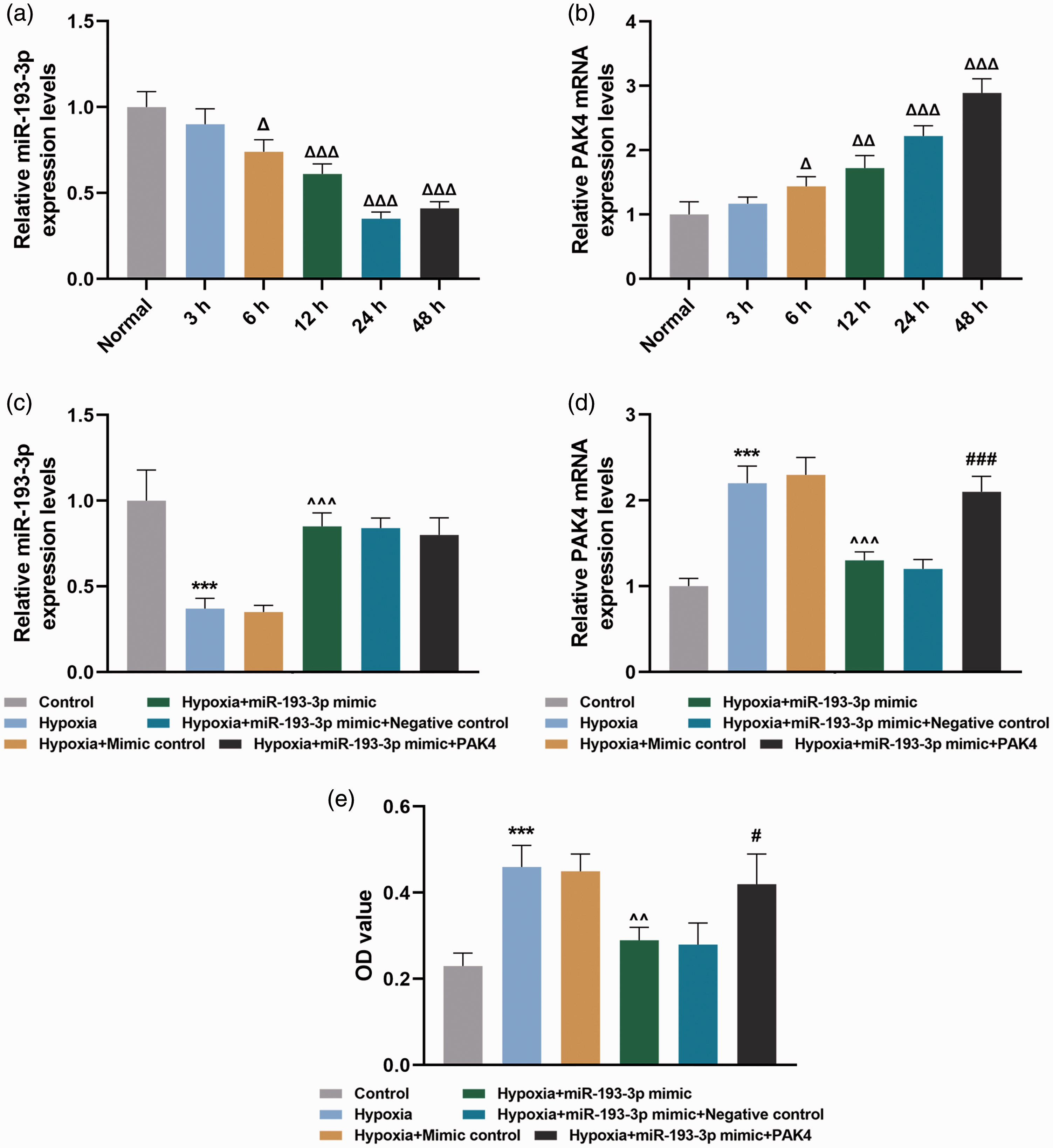
The expressions of miR-193-3p and PAK4 and their interaction in hypoxia-induced PASMCs. (a) and (b) PASMCs were exposed to hypoxia for different times (0, 3, 6, 12, 24, and 48 h), followed by qRT-PCR assay on miR-193-3p expression (a) and PAK4 mRNA expression (b). (c)–(e) PASMCs were divided into control, hypoxia, hypoxia + mimic control, hypoxia + miR-193-3p mimic, hypoxia + miR-193-3p mimic + negative control, hypoxia + miR-193-3p mimic + PAK4 groups, followed by the detection of miR-193-3p expression by qRT-PCR analysis (a), PAK4 mRNA expression by qRT-PCR analysis (d), and cell proliferation ability by MTT assay (e). The experiment was repeated three times. △
MiR-193-3p overexpression attenuated hypoxia-induced phenotypes and related-molecule expressions in the PASMCs through inhibiting PAK4 expression
The viability of PASMCs (Fig. 2e,
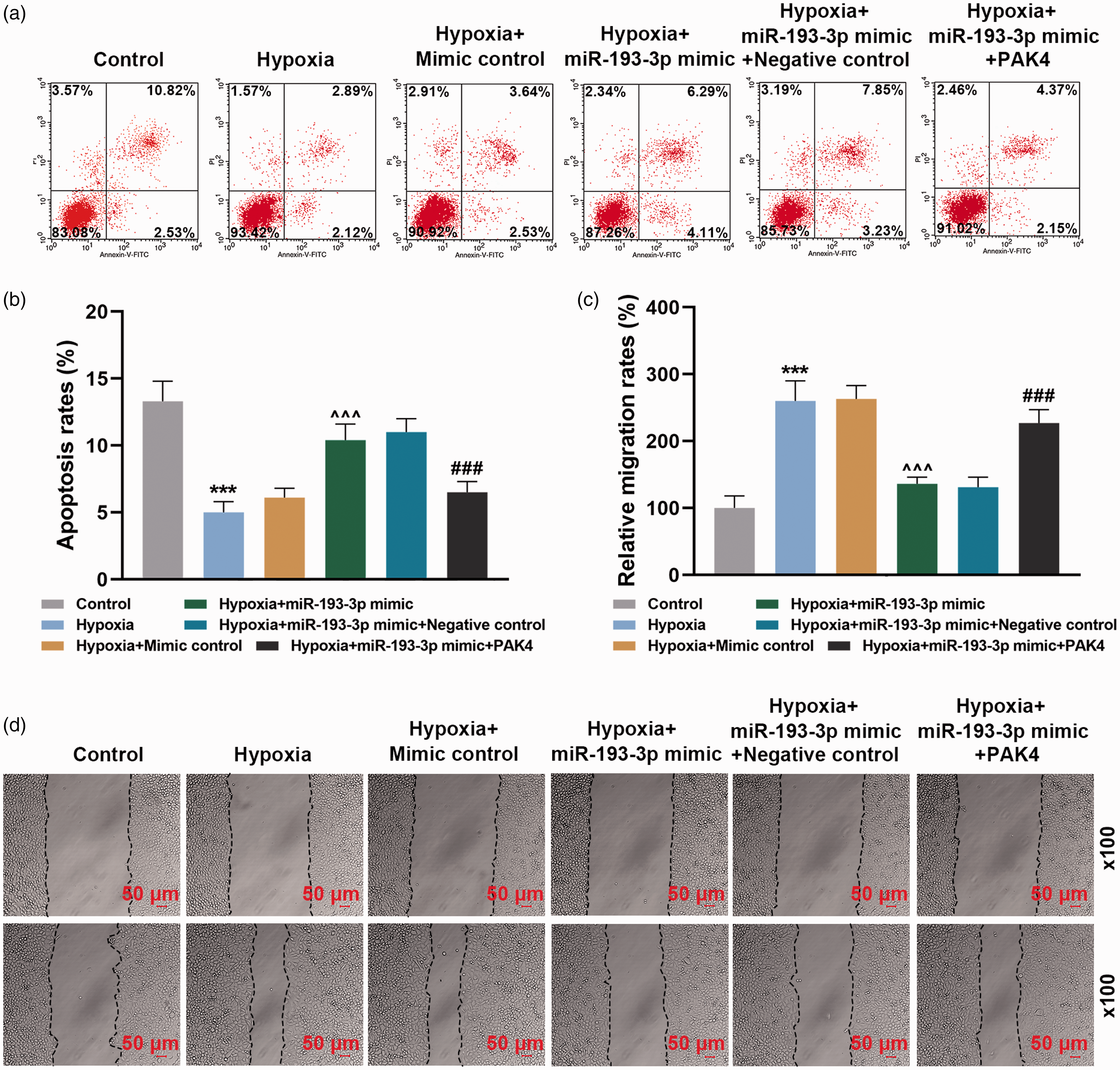
MiR-193-3p overexpression attenuated hypoxia-induced phenotypes in PASMCs through inhibiting PAK4 expression. (a)–(d) PASMCs were divided into control, hypoxia, hypoxia + mimic control, hypoxia + miR-193-3p mimic, hypoxia + miR-193-3p mimic + negative control, hypoxia + miR-193-3p mimic + PAK4 groups, followed by the detection of apoptosis by flow cytometry analysis (a) and (b), and cell migration by wound-healing migration assay (c) and (d). The experiment was repeated three times. ***
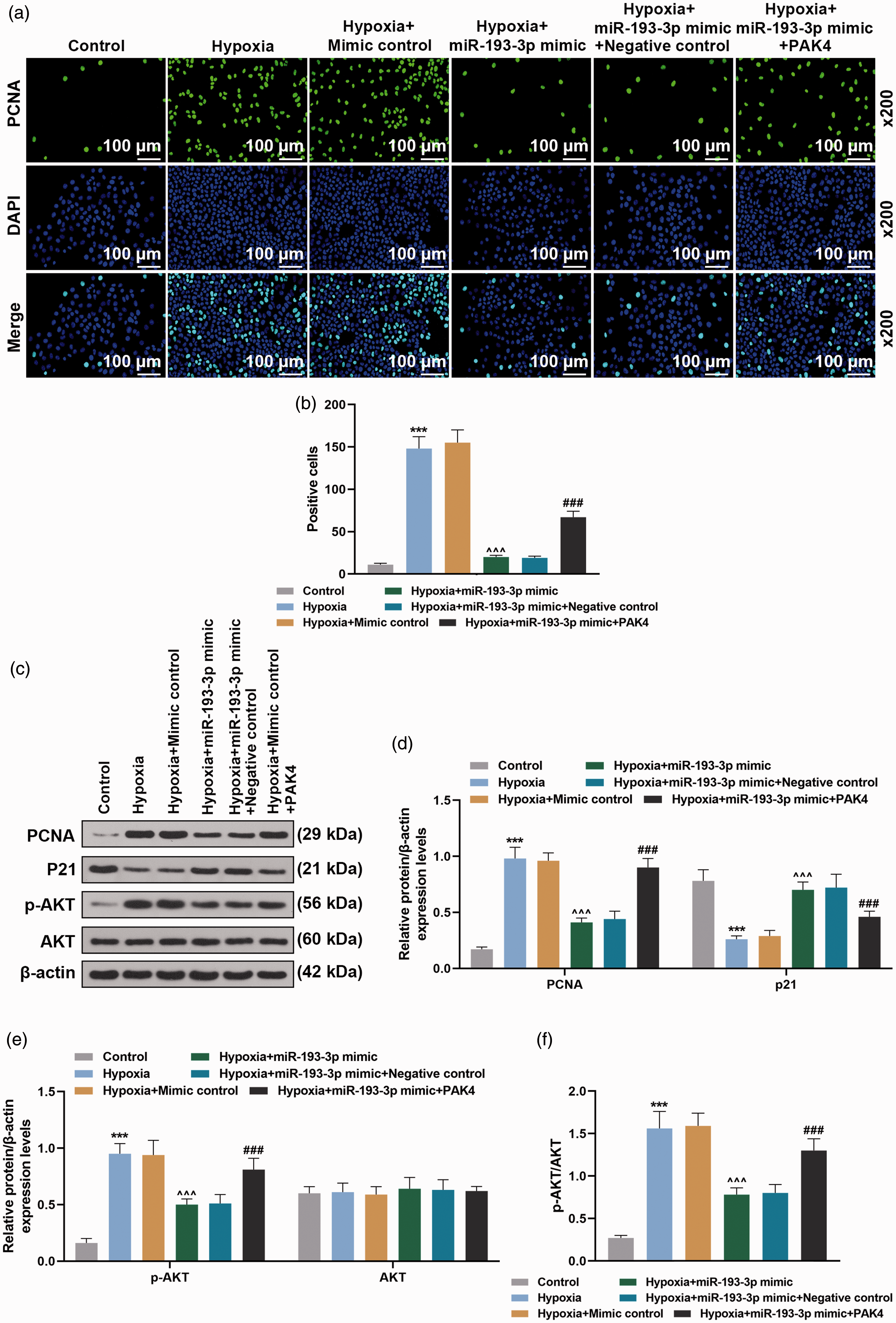
MiR-193-3p overexpression regulated molecular expressions involved in hypoxia-induced PAH in PASMCs through inhibiting PAK4 expression. (a) and (b) PASMCs were divided into control, hypoxia, hypoxia + mimic control, hypoxia + miR-193-3p mimic, hypoxia + miR-193-3p mimic + negative control, hypoxia + miR-193-3p mimic + PAK4 groups, followed by the detection of PCNA by immunofluorescence, PCNA positive cells were counted. (c)–(e) The detection of protein expressions of p-AKT, AKT, PCNA, and p21by Western blot. (f) A ratio of p-AKT to total AKT protein (p-AKT/AKT). The experiment was repeated three times. β-actin was used as internal control. ***
MiR-193-3p attenuated pulmonary vascular remodeling in the PAH rats through inhibiting PAK4 expression
MCT treatment significantly inhibited the expression of miR-193-3p (Fig. 5a,
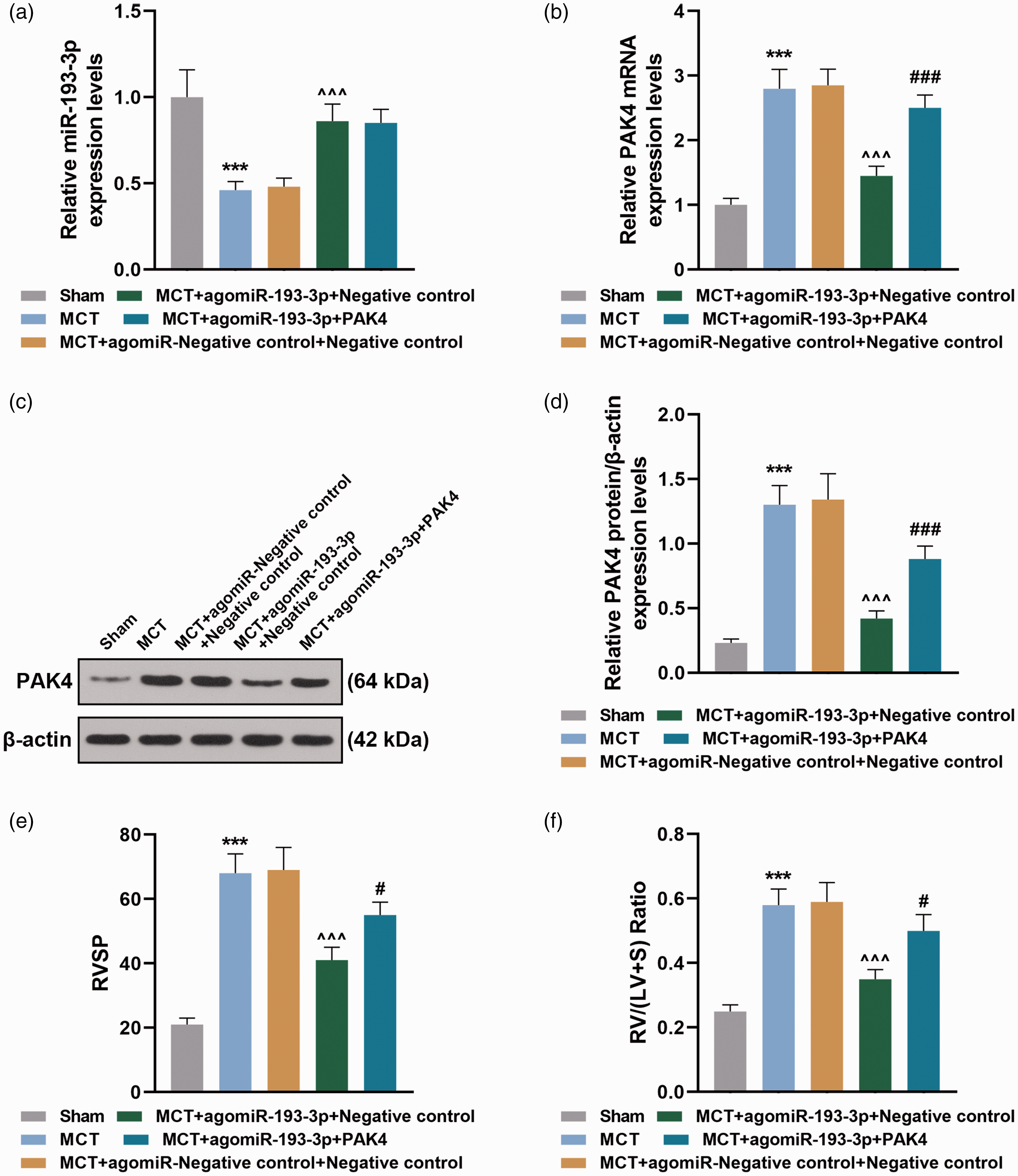
MiR-193-3p overexpression inhibited PAK4 expression in PAH rats (
As shown in Fig. 6a, H&E staining showed severe pulmonary hemorrhage, pulmonary vascular remodeling, and pulmonary fibrosis in the MCT group as compared with Sham group, but such conditions in the MCT group were noticeably attenuated by miR-193-3p overexpression. However, the positive effect of miR-193-3p overexpression was effectively reversed by PAK4 overexpression. WT% and WA% of the pulmonary arteries were significantly higher in MCT group, miR-193-3p overexpression significantly reversed this trend, and PAK4 overexpression markedly reversed the effect of agomiR-miR-193-3p (Fig. 6b and c,
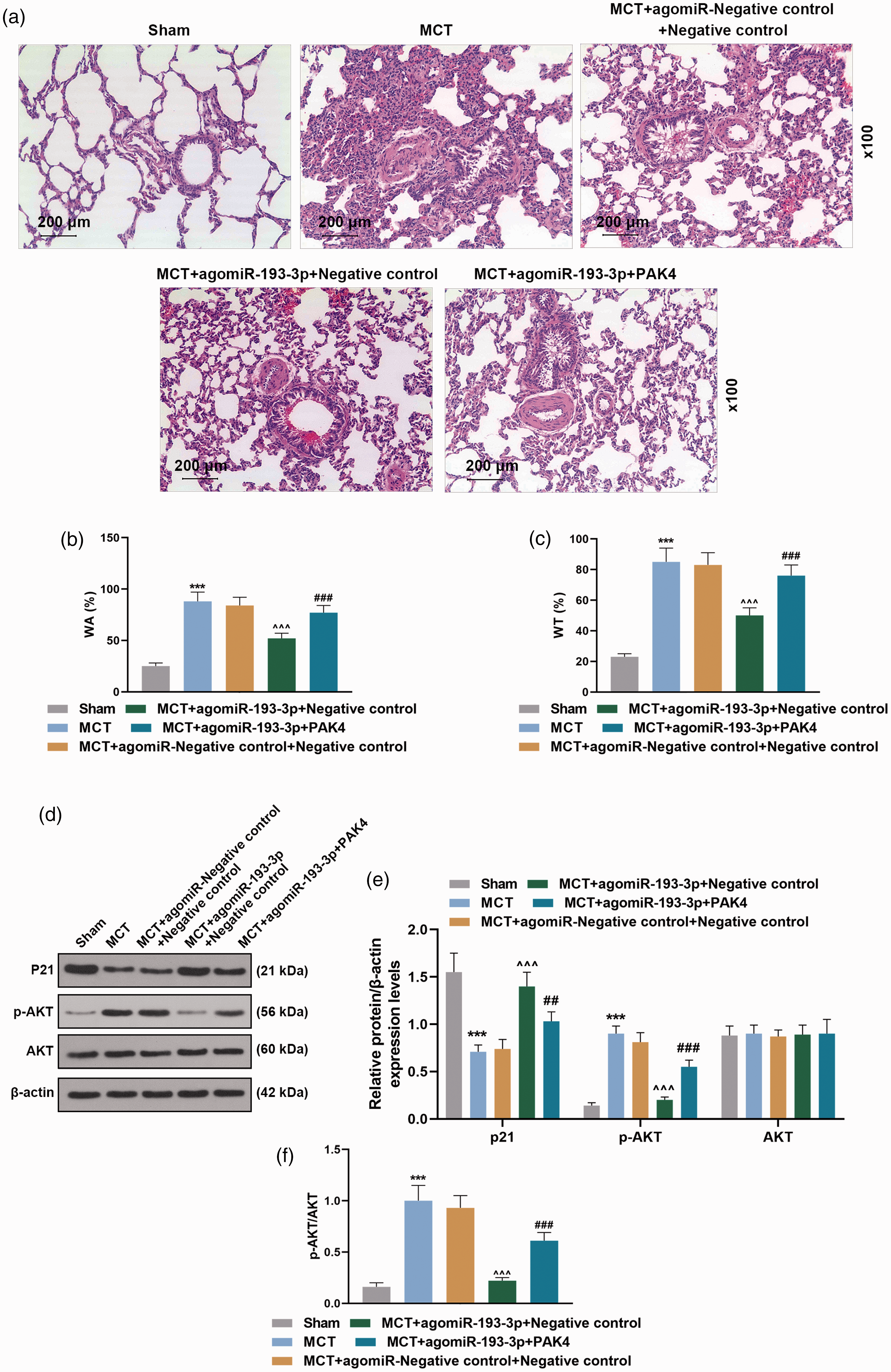
MiR-193-3p attenuated pulmonary vascular remodeling in PAH rats through inhibiting PAK4 expression (
Discussion
PAK4 is normally low-expressed in multiple normal adult tissues, 18 and its overexpression has been found in multiple cancers.19–21 Jian et al. 22 reported that miR-193-3p is significantly overexpressed in both gastric cell lines and human gastric tumors. Similarly, the present study found that miR-193-3p was downregulated in lung tissues of PAH patients, hypoxia-induced PASMCs, and the lung tissues of MCT-induced rats, whereas PAK4 resulted in opposite results. Thus, remarkably downregulated miR-193-3p in PAH should be further explored.
A new cancer-like concept has been proposed for PAH because the pathogenesis of PAH is similar to cancers, such as excessive cell proliferation and apoptosis inhibition. 23 Increasing reports demonstrated that multiple miRNAs, such as miR-485 24 and miR-342, 25 regulate the development of cancer cells through targeting PAK4. The present study reported the association between miR-193-3p and PAK4 in PAH. We found that miR-193-3p carried a binding site with APK4 using TargetScan V7.2, and the prediction was confirmed by dual-luciferase reporter assay. Moreover, miR-193-3p overexpression negatively regulated PAK4 expression under hypoxia. However, the regulatory roles of miR-193-3p and PAK4 in PAH phenotype remained unclear. It has been demonstrated that miRNAs play key parts in PAH pathogenesis, and can act as biomarkers and new therapeutic targets for PAH. 26 Tang et al. 27 demonstrated that miR-143-5p facilitates proliferation and migration of PASMCs in hypoxia by regulating Hypoxia-inducible factor-1α (HIF-1α). Zhao et al. 28 indicated that upregulation of miR-593-5p greatly suppresses the proliferation and migration of PASMCs through regulating polo-like kinase 1 (PLK1). Yue et al. 29 showed that miR-143 and miR-145 facilitate proliferation and migration of PASMCs via modulating ABCA1 expression under hypoxic condition. Similarly, we found that miR-193-3p overexpression inhibited cell proliferation and migration, provoked apoptosis of PASMCs in hypoxia through inhibiting PAK4 expression, pointing to the regulatory role of miR-193-3p and PAK4 in PAH phenotype. We then further explored the roles of miR-193-3p and PAK4 in PAH and the potential molecular mechanisms.
P21 is a cyclin-dependent kinase inhibitor that facilitates cell cycle arrest in response to various stimuli. 30 AKT plays a key role in modulating tumor cell survival and cell cycle progression. 31 Previous study revealed that PAK4 plays a critical role in the initiation of cell cycle by modulating p21. 32 Investigation by Yuan et al. showed that upregulation of PAK4 in vascular smooth muscle cells significantly suppresses p21 expression, enhances AKT activation, and further regulates cell cycle development and cell proliferation. 33 A previous report indicated that PAK4 is modulated by miRNA-433 and subsequently inhibits AKT signaling, thereby regulating the proliferation of hepatocellular carcinoma cells. 34 Moreover, the upregulation of miR-342 has been found to inhibit PAK4 expression, subsequently inactivates the AKT and ERK pathways in glioma, thus inhibiting the cell proliferation, invasion, and apoptosis resistance of glioma. 25 Similarly, in this study, we observed that miR-193-3p overexpression suppressed the expression of proliferation marker PCNA, AKT activation, and promoted p21 expression, which were all partially reversed by PAK4 overexpression in PASMCs under hypoxic condition. The present data suggested that miR-193-3p inhibited the proliferation of PASMCs by mediating cell cycle progression through targeting PAK4 via regulating p21 expression and AKT activation, and such a process was accompanied with decreased expression of PCNA. However, further research is required to study whether the effects of AKT activation and the expressions of p21 and PCNA on the miR-193-3p targeting PAK4 in affecting the proliferation, migration, and vascular remodeling of PAH PASMCs is independent. The data from PAH rats caused by MCT also showed the same results to in vitro results in this study. In addition, the results from H&E staining assay showed that miR-193-3p overexpression attenuated lung vascular remodeling in rats injected with MCT through inhibiting PAK4 expression. It is known that the primary cellular mechanism underlying vascular remodeling is reflected by excessive proliferation and migration of PASMCs. 23 Thus, the current findings suggested that miR-193-3p attenuated the vascular remodeling in PAH by inhibiting PAK4 expression. Other researchers 11 have shown that miR-17-5p plays a part in PAH and the proliferation of PASMCs by regulating p21. A latest report 35 indicated that miR-182-3p can regulate pulmonary hypertension vascular remodeling and proliferation of PASMCs through KLF4/p21-dependent mechanism, which is consistent with our findings. However, based on the complex occurrence and development of diseases, whether there is an interaction network between miR-193-3p and other miRNAs such as miR-182-3p and miR-17-5p in PAH vascular remodeling should be further explored by future studies.
Taken together, in PAH, miR-193-3p was downregulated and PAK4 was up-regulated. MiR-193-3p suppressed PAK4 expression in PAH in vitro and in vivo. MiR-193-3p attenuated the proliferation, migration, and apoptosis resistance of PASMCs induced by hypoxia and vascular remodeling in the rats induced by MCT through targeting PAK4 to regulate Akt signaling, the p21 and PCNA. These findings demonstrated that miR-193-3p attenuated vascular remodeling in PAH by targeting PAK4 and may serve as a promising therapeutic target for the therapy of PAH. However, miRNAs-based treatments should confirmed by conducting clinical experiments.
Footnotes
Author contributions
Substantial contributions to conception and design: Z.W. and J.G.
Data acquisition, data analysis, and interpretation: Y.Q., J.L., Y.B., and Z.G.
Drafting the article or critically revising it for important intellectual content: Z.W. and J.G.
Final approval of the version to be published: all authors.
Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of the work are appropriately investigated and resolved: all authors.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Ethics approval and consent to participate
All procedures performed in this study involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fund of Tianjin Chest Hospital (2018XKZ16).
