Abstract
Spindle cell carcinoma (SpCC) is a unique variant of squamous cell carcinoma (SCC). SpCC confined to the nasal cavity is extremely rare, with only one case having been previously reported. We present a case report of nasal cavity SpCC and review the literature on this rare entity. A 29-year-old male presented with intermittent epistaxis from the left nasal cavity. On physical examination, the patient had an ulcerated mass in the left nasal vestibule and a biopsy showed a proliferation of spindle and epitheliod cells. The patient underwent wide local excision of the mass via a lateral alotomy approach and reconstruction with a composite conchal bowl skin and cartilage graft. Histologically, the mass had dyplastic squamous epithelium and spindle-shaped cells admixed with epitheliod cells. Immunohistochemistry was only positive for pancytokeratin AE1/AE3 and vimentin. Six months after surgery, the patient continues to have no evidence of disease. On literature review, only one previous case of SpCC confined to the nasal cavity was identified. We present a rare case of nasal cavity SpCC. No definite treatment protocol exists for this unique entity, but we believe that this tumor should primarily be treated with aggressive, wide local excision. Adjuvant radiation and/or chemotherapy have also been used anecdotally.
Introduction
Spindle cell carcinoma (SpCC) is a unique and challenging variant of squamous cell carcinoma (SCC). In the upper aerodigestive tract, similar to conventional SCC, SpCC occurs most commonly in the larynx. 1 Less common primary sites include the hypopharynx, oropharynx, sinuses, and nasal cavity. SpCC confined solely to the nasal cavity is extremely rare, with only one case having been previously reported. 2 Herein we present a case of SpCC confined to the nasal cavity.
Case Report
A 29-year-old previously healthy man presented to our clinic with a 2–3 week history of intermittent epistaxis from the left nasal cavity. He denied any dysphagia, odynophagia, change in voice, nasal obstruction, purulent rhinorrhea, anosmia, facial pain, or weight loss. On nasal endoscopy, the patient had an ulcerated, friable, well-circumscribed mass confined to the left nasal vestibule and attached to the cartilaginous septum. There was no palpable lymphadenopathy. Computed tomography (CT) of the face and neck with contrast showed the mass to be heterogeneously enhancing (Figure 1). Biopsy of the nasal mass showed a proliferation of spindle and epithelioid cells. The cells were immunoreactive for Cytokeratin 7 (CK7), vimentin, and pan keratin AE1/AE3. The tumor was negative for melanoma markers (HMB-45, Melan-A, S100), smooth muscle actin (SMA), CD34, and myoepithelial markers (CD10, Calponin, p63 and GFAP). The diagnostic impression was spindle cell neoplasm, favoring carcinoma.
Computed tomography scan with contrast showing the 1.3×0.9 cm left nasal cavity mass, contacting the cartilaginous septum. The mass showed heterogeneous enhancement.
The patient subsequently underwent wide local excision of the mass via a lateral alotomy approach. Grossly, the mass was well-circumscribed, ulcerated, and fixed to the medial nasal vestibule and cartilaginous septum (Figure 2). To achieve adequate margins, a full thickness portion of the left lower lateral alar cartilage was removed. This was reconstructed using a composite conchal bowl skin and cartilage graft.
Intraoperative photo of the left nasal cavity mass. The mass was fixed to the nasal vestibule medially and the cartilaginous septum.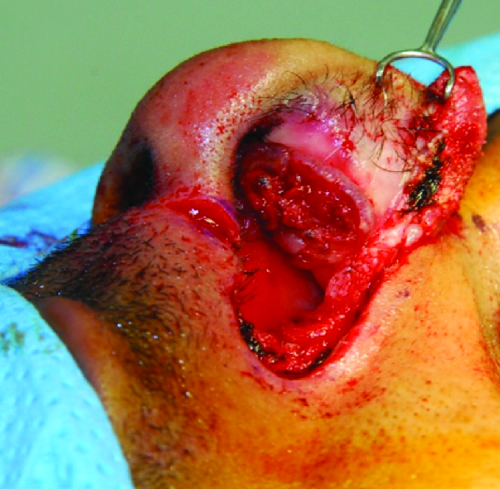
Histological examination showed the mass to be comprised of fasiculated and streaming spindle-shaped cells admixed with epithelioid cells. No keratin pearls were seen. The surface epithelium overlying the tumor was ulcerated, with squamous atypia (Figure 3). There was mild nuclear pleomorphism with a low mitotic count (8 mitoses/50 hpf). The proliferation index (assessed via Ki-67 immunostaining) was estimated to be 10–15%. Despite the low mitotic count and proliferation rate, focally vascular invasion, necrosis, and perineural invasion were seen. Alcian Blue/PAS stain for mucin was negative. On immunohistochemical staining, the tumor cells were reactive for AE1/AE3 and vimentin (Figure 4), but negative for CK5/6, CAM 5.2, epithelial membrane antigen (EMA), p63, SMA, myogenin, desmin, S100, Factor VIII, HMB-45, Melan A, CD10 and CD34. Ultrastructural examination revealed intermediate filaments in the spindle cells without desmosomes or other junctional complexes. With lack of mucin or myoepithelial cell differentiation, a diagnosis of spindle cell squamous carcinoma was made. Six months after surgery with no post-operative adjuvant therapy, the patient continues to have no evidence of disease. This report was exempt from review by the Manhattan VA Medical Center Institutional Review Board.
a) Low power image showing the tumor with overlying ulcerated mucosa, as well as adjacent normal squamous epithelium, (Haematoxylin & Eosin x40); b) at higher magnification the lesion is composed of spindled cells arranged in fascicles admixed with more epithelioid appearing cells. Mild nuclear atypia is appreciable. (Haematoxylin & Eosin x400).
a) Tumor cells display focal immunoreactivity for pancytokeratin marker AE1/AE3 (400x); b) tumor cells display diffuse immunoreactivity for vimentin (400x).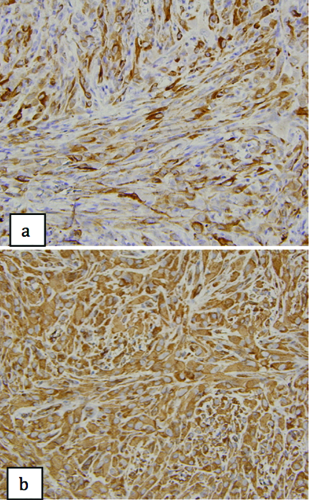
Discussion
SpCC is a variant of SCC that histologically exhibits spindled or pleomorphic tumor cells. SpCC has similar demographics to that of conventional SCC. It occurs most often in the fifth and sixth decades, is strongly associated with smoking and alcohol use, and shows a strong male preponderance. 3 The larynx (glottis) is the most common primary site, followed by the oral cavity (tongue, floor of mouth, gingivae). Less common sites include the hypopharynx, oropharynx, sinuses, and nasal cavity.4–6 SpCC has a unique, macroscopic growth pattern. 1 In the larynx, greater than 90% of the tumors present as polypoid, ulcerated, exophytic masses.3,5,7
Histologically, SpCC are often biphasic, showing areas of conventional SCC mixed with areas of spindle and/or pleomorphic tumor cells. However, it has been reported that in up to 28% of cases, a classical SCC component cannot be identified.8–9 While the clonal proliferation of atypical spindle cells may simulate a sarcoma of mesenchymal origin, recent studies have shown head and neck sarcomatoid carcinomas to arise from transformation of more conventional epithelial components.10–13 While recent molecular studies have lent credence to this argument, clinically this can be shown through the use of immunohistochemistry - most of these tumors stain with both epithelial and meschymal markers. The most common epithelial stain, AE1/AE3 (pancytokeratin), is positive in anywhere from 26% to 62% of cases.3,5,14 Some cases have also been shown to express other common epithelial markers including EMA and p63.3,5,6,14 In regards to mesenchymal-type markers, almost 100% of cases are positive for vimentin.3,5 Some SpCC lesions have also been shown to express markers of muscle differentiation including SMA and muscle specific actin.
3
Ultrastructurally, the presence of cytokeratin type intermediate filaments, cadherins, desmosomes, tight junctions or adherens junctions also supports an epithelial origin of these tumors.
10
Using a PubMed search (search terms:
Another case of a nasal cavity SpCC, this time extending into the maxillary sinus, has been recently reported. 15 The patient was a 75-year-old man who presented with nasal obstruction. Nasal endoscopy showed a 3×3 cm polypoid mass, extending from the nasal septum into the maxillary sinus. A biopsy showed malignant spindle cells with hyperchromatic nuclei. Immunohistochemistry showed positivity for pancytokeratins, high molecular weight cytokeratin, CK 5/6, CK18, CK19, p63, and vimentin. Thus, a diagnosis of SpCC was made. The patient underwent resection of the right maxilla followed by chemotherapy (5-fluorouracil) and radiation at another hospital. At the time of the report, the patient was alive without metastasis 5 years after initial presentation.
Conclusions
Our patient's tumor had a similar immunohistochemical profile to that of the two previously published cases of sinonasal SpCC. The tumor was positive for pancytokeratin marker AE1/AE3, as well as vimentin. Our patient presented with no evidence of lymph node metastasis, and we were able to achieve negative surgical margins. No definite treatment protocol exists for this unique entity, but we believe that this tumor should primarily be treated with aggressive, wide local excision. Adjuvant radiation and/or chemotherapy have also been used anecdotally.
