Abstract
We report a case of epithelioid variant of pleomorphic liposarcoma (EPL) found in the the infrapatellar fat pad of Hoffa of a 31-year old male. Histologically, the predominant population was formed by epithelioid cells with eosinophilic or clear cytoplasm admixed with rare pleomorphic lipoblasts. The immunohistochemical panel was not helpful in the diagnosis. FISH analysis using the locus-specific indicator CHOP (12q13) dual color break apart was applied to representative formalin-fixed, paraffin-embedded tissue sections. The result of FISH indicated a rearranged CHOP (DDIT3) gene and confirmed the diagnosis of EPL. The EPL should be differentiated from a metastatic carcinoma or other type of sarcoma. In these cases a clinicopathological correlation and an exhaustive sampling of the specimen for demonstration of lipogenic areas or pleomorphic lipoblasts is always necessary. FISH with demonstration of CHOP gene rearrangement is useful in providing specific ancillary information for the difficult differential diagnosis of this case.
Keywords
Introduction
Pleomorphic liposarcoma (PL) is an uncommon subtype of liposarcoma recognized for the first time by Enzinger et al. in 1962 1 and characterized morphologically by the presence of pleomorphic lipoblasts admixed with a variety of nonlipogenic areas resembling other high-grade sarcomas or carcinoma. Epithelioid PL (EPL) represent a rare variant of PL composed mainly of carcinoma-like cells mimicking renal or adrenal cortical carcinoma.2–4 Molecular analysis showed the presence in epithelioid PL of a chimeric oncogene FUS-CHOP (FUS-DDIT3), generated by the most common chromosomal translocation t(12;16)(q13;p11) associated with myxoid/round liposarcomas. 5
The recent demonstration that EPL has a FUS-CHOP transcript may help in the diagnosis of this entity.5,6
Case Report
A 31-year-old Caucasian male was admitted to our Hospital for evaluation of soft tissue mass of right knee. The patient was an amateur tennis player who presented for right knee pain and difficulty walking for one-year duration. Physical examination and routine laboratory test results were within normal limits.
CT scan showed an ill defined soft tissue mass of the infrapatellar fat pad of the right knee without intraarticular extension, that measured 6,0 cm in the maximum dimension.
Axial and coronal T1-weighted magnetic resonance imaging (MRI) demonstrated an extra-articular, poorly circumscribed, solid mass of the infrapatellar fat pad of Hoffa with a prevalent component of high signal intensity on T1 and T2-weighted sequences. A preoperative FNA of mass was performed but the results were not diagnostic. Then, the patient underwent resection of the mass and of the fat pad of Hoffa with histological examination. Following the histological diagnosis, a total body CT scan was performed to exclude the presence of lesions in other sites. The postoperative course was uneventful and the patient was discharged after 5 days.
Gross examination: the surgical specimen measured 8,0 cm in its greatest diameter with a poorly circumscribed tumor measuring 6,0×3,0×2,0 cm in size surrounded by adipose tissue. The cut surfaces were whitish, with some fibrous septa resembling a soft tissue fibromatosis or fibrosarcoma. At low power field the lesion showed pushing but infiltrative margins. Microscopically, the tumor was composed of round to polygonal epithelioid cells generally arranged in large sheets (Figure 1a), with round nuclei, often prominent nucleoli, and variably eosinophilic to vacuolated cytoplasm (Figure 1b) resembling a metastasis from metastatic carcinoma. Scattered pleomorphic epithelioid cells were also seen.
(a) Epithelioid component of tumor composed of large sheets of carcinoma-like cells (H–E 10×). (b) Epithelioid cells have abundant eosinophilic or clear cytoplasm (H–E 40×). (c) Lipogenic area composed of uni or multivacuolated lipoblasts with hyperchromatic, round to irregularly shaped nuclei (H–E 40×). (d) At high magnification some hyperchromatic and scalloped pleomorphic lipoblasts (H–E 40×).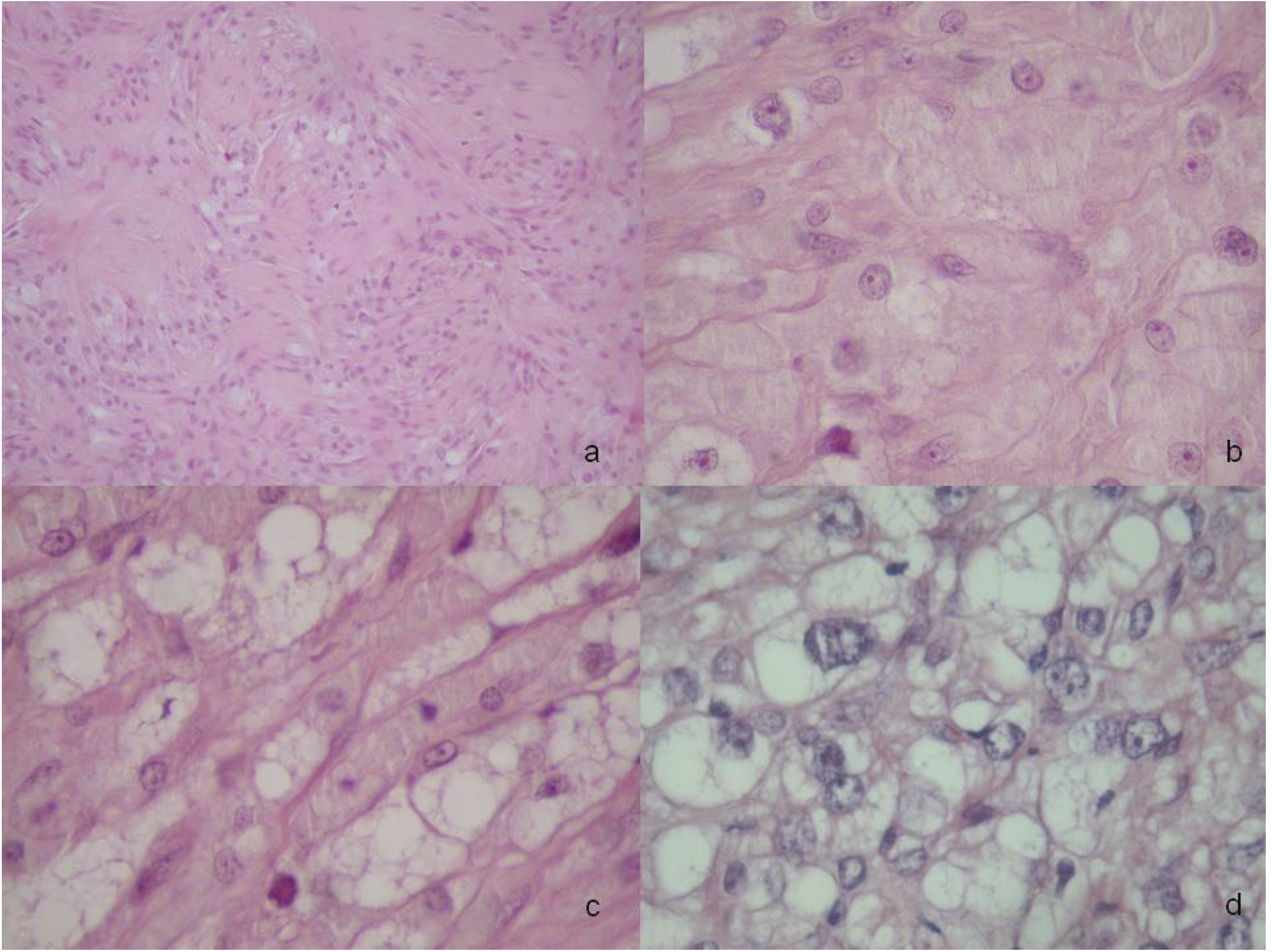
These epithelioid cells were histologically similar to epithelial cells of renal carcinoma. In other areas the tumor was composed of spindle cells in a fascicular arrangement with a variable degrees of fibrosis. No myxoid areas were noted in all sections examined. Necrosis was focal and mitotic activity moderate (mean 1–2 mitoses for 10 hpf). A large number of small and medium thick-wall vessels were also observed, some with hemangiopericytoma-like features. At periphery of tumor we have identified a small lipogenic area composed of uni-and multivacuolated lipoblasts (Figure 1c).
In several sections examined some unequivocal pleomorphic lipoblasts with hyperchromatic and scalloped nuclei were also found in some sections (Figure 1d). Margins of excision were tumor-free (less than 1 cm).
Additional paraffin-embedded sections were obtained for histochemical analysis using Alcian blue. For immunohistochemical study, slides were stained using an automated labeled streptavidin-biotin immunoperoxydase method according to the manufacturer's instructions (Ventana Medical Systems, Tucson, AZ). Primary antibodies vimentin, desmin, smooth-muscle actin, (SMA), CD34, HMB-45, EMA, CK7, CK AE1/AE3, CK8-18, CD10, S-100 protein, MIB-1, CD68 (Dako Cytomation, Copenhagen, Denmark) were applied. The neoplastic cells were positive for vimentin and CD10 (Figure 2a). A variable number of epithelioid cells showed membrane staining for CK-cocktail (Figure 2b) and CK8. The other markers were negative. A proliferative rate (MIB-1>30%) was seen and the nuclear p53 immunoreactivity was found in the majority of neoplastic cells. A final diagnosis in this case was difficult. The presence of epithelioid cells with eosinophilic or vacuolated cytoplasm requires at first a differential diagnosis with a metastasis from primary carcinoma of the kidney or the adrenal gland. A careful clinicoradiologic approach with the medical examination of the anatomic abnormalities or the clinical symptoms and the total body CT or MRI scan, is always necessary. In absence of clinical, radiological and operative evidence of tumor within other sites, this lesion could be considered a primary tumor of soft tissue. After the diagnosis, the radiotherapy was carried out post-operatively and the patient was followed up each the first 2 years by a chest CT scan and a knee MRI every 3 months. Actually the patient has no evidence of disease (follow-up 12 months).
(a) Epithelioid cells showing membrane staining for CD10 and for (b) CK (immunostaining, 20×).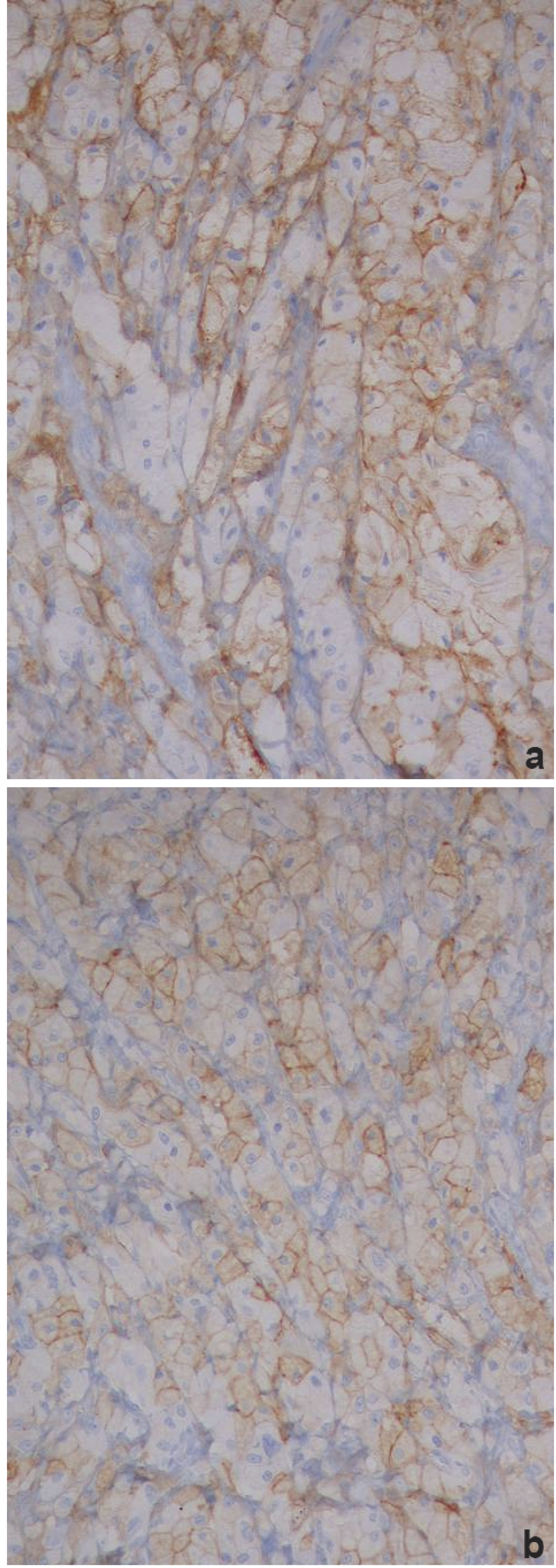
FISH analysis
Lipogenic and nonlipogenic areas of tumors were selected for FISH analysis using the locus-specific indicator CHOP (12q13) dual color, break apart probe. The CHOP FISH studies were performed using 4 µm-thick sections obtained from selected blocks of paraffin-embedded tissue and processed according to the manufacturer's instructions. (LSI CHOP Dual Color break-apart Rearrangement Probe supplied by the Vysis division of Abbott Laboratories, USA). The LSI CHOP Probe was composed of a mixture of two FISH DNA probes. The first probe was a spectrum orange-labeled probe that spans a 700-kb region just centromeric of the CHOP (DDIT3) gene; the second probe was a spectrum green-labeled probe that spans a 660-kb region just telomeric of the CHOP (DDIT3) gene. We counted 100 nuclei that showed at least two pairs of green and orange signals and the percentage of the split signals was calculated. The presence of two fusion signals per nucleus indicated an intact CHOP (DDIT3) gene. The presence of a single orange and single green signal indicated a rearranged CHOP (DDIT3) gene. Other signal configurations were observed and may be due to truncation of the nuclei secondary to sectioning. Some of these nuclei were not classifiable as rearranged or normal and were not included in the final calculations. Using FISH analysis the different histologic areas examined exhibited CHOP rearrangement in more than 50% of counted nuclei (Figure 3). This result combined with the demonstration of pleomorphic lipoblasts confirmed the diagnosis of EPL.
Split signals (inset) in a lipogenic area of pleomorphic liposarcoma (original magnification ×1000).
Discussion
Pleomorphic liposarcoma (PL) is a rare variant of liposarcoma that tends to occur on the extremities of elderly patients (>50 years).1–4 Histologically, this entity is typified by the presence of lipogenic component with pleomorphic lipoblasts and nonlipogenic areas with features of malignant fibrous histiocytoma-like, round cell liposarcoma-like, and/or epithelioid/carcinoma-like.1–3
Prevalent epithelial-like features and immunoreactivity for some keratins represent the hallmark of the epithelioid PL (EPL).2–4 This rare variant is still under recognized and may closely simulate other epithelial neoplasms, mainly adrenal cortical carcinoma. Because of its rarity and challenging diagnosis there are limited studies with long-term follow-up information.2–4 From the genetic point of view, molecular analysis have demonstrated the presence of a reciprocal translocation of two genes. FUS-CHOP also named FUS-DDIT3 is a chimeric oncogene generated by chromosomal translocation t(12;16)(q13;p11). This traslocation is present in more than 90% of myxoid and round cell liposarcoma (MLS/RC). The presence of CHOP rearrangement in this case may be useful for the final diagnosis of EPL and demonstrated that this variant of liposarcoma is cytogenetically related to MLS/RC. 5 We described a new case of EPL in a young man with a of right knee mass and characterized from the histological point of view by prevalent carcinoma-like features. In fact, the non-lipogenic areas were composed of a large number of atypical epithelioid cells whereas the lipogenic foci were nor prominent. Usually unequivocal pleomorphic lipoblasts were always required for a diagnosis of PL. In our case lipogenic areas were composed of frank lipoblasts and vacuolated lipid-rich cells resembling xantoma cells. Only after a serial examination of surgical specimen scattered pleomorphic lipoblasts were evident. Because these carcinoma-like features may be predominant, a diagnosis of PL may be obscured by the suspect of metastatic carcinoma or confused with a non-lipogenic epithelioid sarcoma. In this case, a metastasis from renal carcinoma should be always excluded, especially in tumor that expressed CK (often focally) and CD10.2–4 Further some lipoblasts can be confused with clear cells in hematoxylin-eosin sections. In these cases the clinical history of patient should be carefully examined. Histologically the recognition of frank lipoblasts and the presence of CK expression only in few cells or small areas were useful finding for the diagnosis of EPL. The differential diagnosis should also include a dedifferentiated liposarcoma and myxofibrosarcoma (MFS). 7 Rarely MFS may show multifocal epithelioid areas in an abundant myxoid matrix. The presence of conventional areas of MFS and absence of atypical lipoblasts are useful for the final diagnosis. Dedifferentiated liposarcoma may show overlap with EPL in non lipogenic sarcomatous component. The evidence of transition from areas of well differentiated liposarcoma to non-lipogenic sarcoma is the histologic hallmark of this tumor in addition to mdm2 and/or cdk4 nuclear expression and MDM2 and/or CDK4 amplification. Generally, the immunohistochemistry have a limited value in differential diagnosis of EPL. S-100 protein immunoreactivity is focal in lipogenic areas or often absent.1–4 A positive focal stain for keratins and particularly for cytokeratin 8, uncommon in liposarcoma is relatively frequent in epithelioid cells of PL.2,5 Another peculiar feature of this case was the diffuse expression of CD10 antibody in a large number of neoplastic cells. 4 These findings in addition to its histological appearance may represent a potential diagnostic pitfall with a metastatic renal carcinoma.
The recently studied gene expression in epithelioid PL showing the presence of FUS-CHOP transcript also named FUS-DDIT3, a translocation characteristic of myxoid/round cell liposarcoma may give aid to correct diagnosis. 5 In fact the distinct chromosomal translocations t(12;16)(q13;p11) FUS-DDIT3 may be identified with fluorescence in situ hybridization (FISH) probes spanning the genomic regions of DDIT3 (12q13), and FUS (16p11) evaluated in formalin-fixed, paraffin-embedded tissues as in our case. Many kinds of FISH probes may be used for the differential diagnosis of soft tissue tumor but the interpretation of the cutoff value of the positive signal differs probe by probe. Using the same probe, a CHOP gene rearrangement was described in 100% of MLS/RC previously analyzed. Whereas, other subtype of liposarcoma, as pleomorphic liposarcoma, showed only few CHOP split signals. 6 So, the presence of CHOP gene rearrangement is correlated with a diagnosis of MLS/RC. The specificity of this FISH probes to identify a characteristic translocation present in MLS/RC and EPL indicate that: i) EPL appear to correlate cytogenetically to MLS/RC rather than PL; ii) the demonstration of CHOP split signals with FISH analysis may be used for differential diagnosis with other epithelioid tumors. Despite molecular similarity with MLS/RC, EPL could be considered an high grade sarcoma because usually it is associated with an aggressive clinical course as others subtype of PL.8–13 In this case a wide surgical excision of mass with adequate margins (more than a 2 cm) should be performed for the high risk of recurrences and distant metastases. In patients with margin-positive sarcoma adjuvant radiotherapy is necessary for the management of these tumors. It is important to bear in mind that the differential diagnosis of EPL is fairly broad and that thorough sampling and searching for lipoblasts may be rewarding in tumours firstly presenting histologically as one of the mimics (metastatic carcinoma or melanoma).
In difficult cases FISH is useful of providing specific ancillary information for the difficult differential diagnosis of this case.
We think that it is important to be aware of these uncommon soft tissue lesions and the pitfalls of epithelioid variant of PL that often cause diagnostic problems. The aim of this report is to help to avoid misdiagnoses of malignant mesenchymal or epithelial tumors with serious consequences, including an incorrect therapeutic management of patient.
